Abstract
Objective
Hypertension is a major risk factor for myocardial infarction. However, the association between the dietary oxidative balance score and myocardial infarction risk among individuals with hypertension remains unclear. This study aimed to investigate the relationship between dietary oxidative balance score and the risk of myocardial infarction in hypertensive adults.
Methods
In this cross-sectional study, we analyzed data from 14,137 hypertensive participants in the National Health and Nutrition Examination Survey (NHANES) from 1999 to 2018. Dietary oxidative balance score was calculated based on the intake of 16 dietary components, including 14 antioxidants and two pro-oxidants. Logistic regression models were used to assess the association between dietary oxidative balance score and myocardial infarction, and restricted cubic splines were applied to explore potential nonlinear relationships.
Results
A significant inverse association was observed between dietary oxidative balance score and myocardial infarction risk. In the fully adjusted model, each one-point increase in dietary oxidative balance score was associated with a 2.4% reduction in the odds of myocardial infarction (OR: 0.976; 95% CI: 0.966–0.985). Compared to the lowest tertile, participants in the highest dietary oxidative balance score tertile had a 28.9% lower risk of myocardial infarction (OR: 0.711; 95% CI: 0.609–0.831). Although nonlinearity was not statistically significant, a threshold effect was observed, with the most pronounced risk reduction at lower dietary oxidative balance score levels.
Conclusion
Higher dietary oxidative balance score values were associated with a reduced risk of myocardial infarction in hypertensive patients. These findings suggest that dietary patterns emphasizing oxidative balance may serve as an effective strategy for myocardial infarction prevention in high-risk populations.
Introduction
Myocardial infarction (MI) is characterized by an elevation and/or reduction in cardiac troponin levels, with a minimum of one measurement surpassing the 99th percentile upper reference limit, alongside indications of myocardial ischemia. This evidence comprises (1) manifestations of ischemia (e.g. chest pain or dyspnea); (2) novel ischemic electrocardiogram alterations (e.g. ST-segment elevation/depression, new left bundle branch block, or pathological Q waves); (3) imaging documentation of recent myocardial necrosis or regional wall motion irregularity (e.g. echocardiography or cardiac magnetic resonance imaging); or (4) detection of intracoronary thrombus via angiography or autopsy.1–3 It is a major contributor to the morbidity and mortality worldwide. In 2019, an estimated 19 million individuals globally succumbed to cardiovascular disease (CVD), with a large proportion of these deaths attributable to coronary heart disease. 4 Hypertension is highly prevalent in adults and is a common precursor of CVD, posing an imminent risk of MI. 5 Traditional risk factors for MI, including smoking, obesity, and physical inactivity, are widely acknowledged, 6 increasing evidence suggests that a dietary imbalance between pro-oxidant and antioxidant nutrients may be a key modifiable option to reduce the risk of MI among patients with hypertension. 7
The dietary oxidative balance score (DOBS) is a summary score that evaluates the balance between dietary prooxidants and antioxidants, and influences the overall oxidative stress of a person.8–11 Antioxidants (vitamins C and E, carotenoids, and flavonoids) counteract reactive oxygen species (ROS) and diminish oxidative stress. Oxidative stress is a pivotal factor in the pathogenesis of atherosclerosis (AS) and acute MI.12,13 In contrast, prooxidants, such as saturated fats, iron, and red meat, generate peroxides that promote oxidative stress, leading to breach of endothelial integrity and plaque formation. A higher DOBS concentration indicates a diet with a stronger antioxidant potential, potentially lowering oxidative stress and mitigating the risk of MI. Prior research indicates that DOBS correlates with a diminished risk of specific chronic diseases. A negative correlation has been identified between DOBS and the risk of ischemic heart disease.14,15 A higher DOBS, indicating greater antioxidant exposure, was associated with a lower risk of incident CKD after adjusting for confounding factors. 16 An elevated DOBS correlates with reduced all-cause and cardiovascular mortality in diabetic patients. 17
However, whether DOBS is associated with MI in hypertensive patients remains unknown. We utilized data from the National Health and Nutrition Examination Survey (NHANES, 1999–2018) to evaluate the correlation between DOBS and MI in hypertensive patients. We hypothesized that a higher DOBS, reflecting a larger intake of antioxidants, would be inversely associated with the risk of MI in this population. These results may offer new perspectives for dietary interventions to reduce MI risk in populations at high risk of hypertension, and could help develop dietary guidelines aimed at achieving better cardiovascular outcomes.
Methods
NHANES
NHANES (National Health and Nutrition Examination Survey) is a continuous national health study in the United States that evaluates the health and nutritional status of both adults and children. 18 It was led by the Centers for Disease Control and Prevention (CDC), which developed a questionnaire with support from the National Center for Health Statistics. Professionals conducting the NHANES collect a wide range of data primarily through personal interviews, physical examinations, and laboratory tests. This information is essential for determining the incidence of diseases and health risk factors in the US population, as well as other public health aspects. This information serves as the cornerstone for many types of national health programs, with a particular emphasis on chronic diseases such as obesity and hypertension. 18 The NHANES program employs a multistage, stratified, probability sampling design to obtain a nationally representative sample of the non-institutionalized US population. This design ensures representativeness across age, sex, race/ethnicity, and socioeconomic status.
Ethical considerations and study design
This study employed a retrospective cross-sectional design based on data from the NHANES collected between 1999 and 2018. As a publicly available and de-identified dataset, NHANES has been approved by the Ethics Review Board of the National Center for Health Statistics (NCHS), and all participants provided written informed consent. The analytical procedures of this study complied with the ethical principles of the Declaration of Helsinki 19 and received exemption from further review by the Ethics Review Board of the NCHS, USA. All patient data were fully de-identified prior to analysis to ensure confidentiality. The reporting of this study conforms to the Strengthening the STROBE guidelines. 20
Study population
This study included 14,137 participants with hypertension from ten cycles of NHANES data collection (1999–2018). Hypertension was defined as self-reported physician diagnosis based on the response to the enquiry “Has a doctor or other health professional ever informed you that you have hypertension?,” use of antihypertensive medication, or measured systolic blood pressure ≥ 130 mmHg and/or diastolic blood pressure ≥ 80 mmHg during the physical examination exam of the NHANES. The exclusion criteria were delineated as follows: (i) age < 18 years, (ii) no hypertension or missing data on hypertension, (iii) missing data on MI, and (iv) missing data on DOBS. See Figure 1 for details.
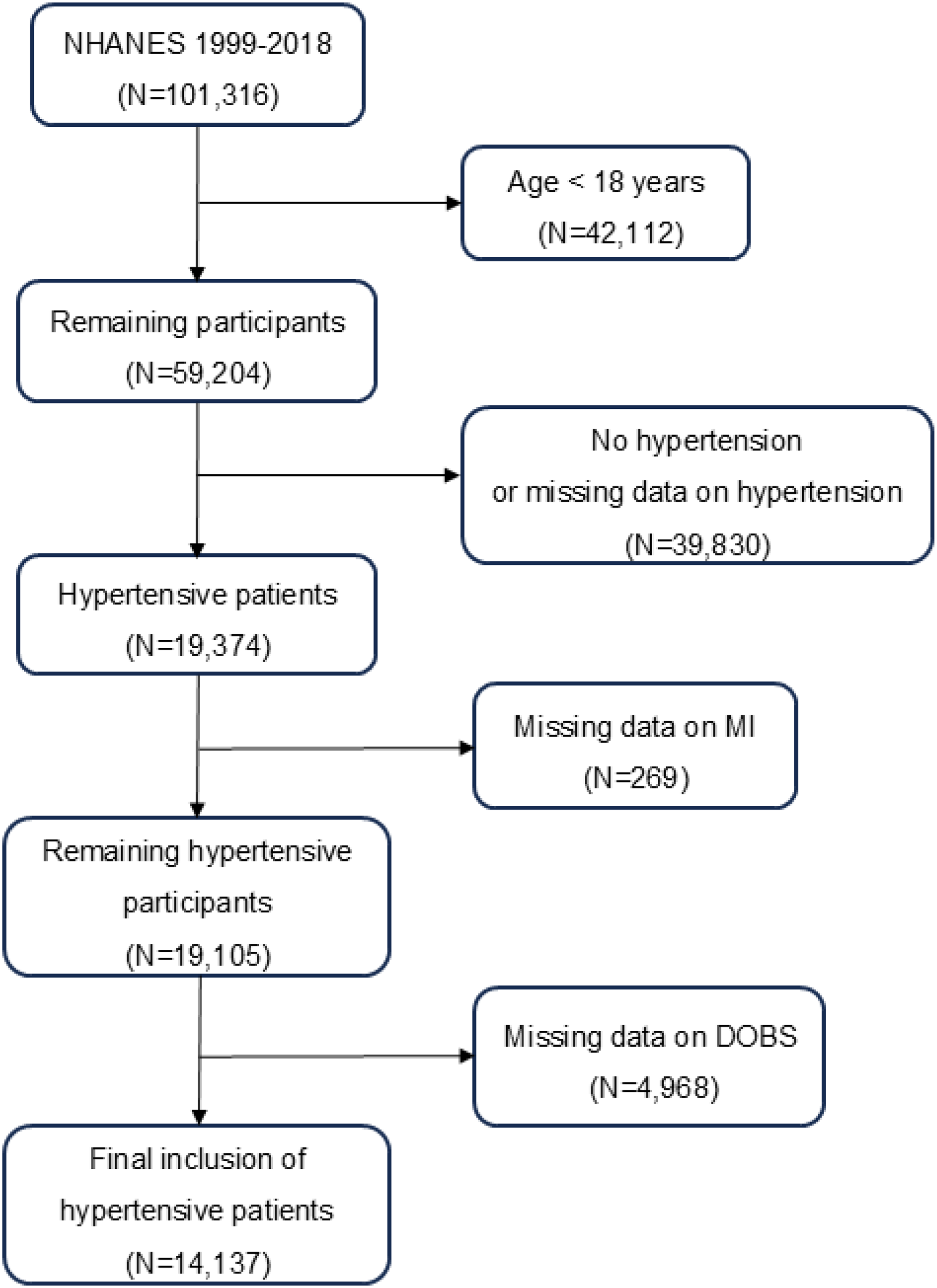
Flowchart of study population inclusion.
DOBS definitions
DOBS was evaluated by analyzing the consumption of 16 dietary nutrients, comprising 14 antioxidants (total dietary fiber, riboflavin, niacin, vitamin B6, total folate, vitamins B12, C, E, carotene, calcium, magnesium, zinc, copper, and selenium) and two prooxidants (total fat and iron). Dietary data were collected using the 24-hour dietary recall method. The DOBS of each participant was calculated using established dietary scoring methods. 11 Nutrient intake was categorized as low, medium, and high based on tertiles, with specific oxidative stress scores assigned to each category. The overall DOBS was obtained by summing the individual scores, with higher scores indicating a higher dietary intake of antioxidants. Detailed scoring criteria are provided in Table 1.
Assignment scheme for DOBSs.

DOBS: dietary oxidative balance score; A: antioxidant; P: pro-oxidant.
Covariate definitions
To control for potential confounding factors, the following covariates were considered: age; sex; ethnicity; education level; marital status; poverty income ratio (PIR); sedentary time; pulse; body mass index (BMI); drinking status; smoking status; and history of hypertension, hyperlipidemia, and CKD. The PIR represents the ratio of a household's total income to the federal poverty threshold, with higher values indicating a more favorable economic status. Sedentary time was evaluated based on participants' self-reported average daily sitting duration. This encompassed time spent seated at school, at home, or during transportation, as well as time spent reading, using a computer, or watching television, while excluding sleep. The total duration was initially recorded in minutes and subsequently converted to hours for analysis. The BMI was calculated based on body mass (kg) and height (m2). Respondents were categorized according to their drinking status as “never drinker” (no more than 12 alcoholic drinks in a lifetime), “former drinker” (more than 12 alcoholic drinks but not within the last year), or “current drinker” (more than 12 alcoholic drinks, of which some were within the previous year). Smoking status was categorized into three distinct classifications: “never smoker” (individuals who have never smoked more than 100 cigarettes), “former smoker” (individuals who have smoked at least 100 cigarettes in their lifetime but do not currently smoke), and “current smoker” and “smoker” (individuals who have smoked a minimum of 100 cigarettes in their lifetime and continue to smoke). Data on hypertension, hyperlipidemia, and CKD were obtained through self-reported questions.
Statistical analysis
Data analysis was performed utilizing Stata 17.0 software. All analyses accounted for the complex NHANES sampling design and applied sample weights to yield nationally representative results. Missing data were managed through multiple imputations using a chained equations approach,21,22 assuming data were missing at random. Baseline characteristics were compared between the MI and non-MI groups. Standard deviations (SD), percentages, and means were calculated for each study variable. Student's unpaired t-tests and chi-square tests were used to evaluate the variations between the groups. After adjusting for confounding variables using stepwise multivariate logistic regression models, we assessed the relationship between DOBS score and MI in patients with hypertension. Three distinct models were developed: Model 1 (unadjusted), Model 2 (adjusted for age, sex, and ethnicity), and Model 3 (additionally adjusted for education level, marital status, PIR, sedentary time, pulse, BMI, alcohol consumption, smoking, hypertension, hyperlipidemia, and CKD). The nonlinear association between DOBS and MI was evaluated utilizing restricted cubic splines (RCSs). Subgroup analyses were conducted according to sex, age, BMI, smoking, drinking, hyperlipidemia, and CKD status to further explore the effects of heterogeneity across different populations. To ensure the robustness of the results, a sensitivity analysis was performed on patients with hypertension from the 1999 to 2008 cycle for internal validation. All statistical analyses employed two-sided tests, with p < 0.05 deemed statistically significant.
Results
Baseline characteristics
A total of 14,137 participants were initially included in the potential analyses during the 1999–2018 NHANES survey cycle, consisting of 6062 men and 7332 women. Table 2 summarizes the baseline characteristics of the participants. Among the 12,868 participants without MI and 1269 with MI, those with MI were older (67.61 ± 11.50 vs. 59.28 ± 15.17 years, p < 0.001) and had a higher prevalence in men (65.0% vs. 46.5%, p < 0.001). Participants with MI had lower PIRs and education levels. Participants with MI had a slightly lower BMI (30.70 ± 7.14 vs. 31.14 ± 7.31 kg/m², p = 0.041). Former smokers and drinkers were also significantly more common in the MI group (p < 0.001), as were comorbidities, including diabetes (38.8% vs. 24.4%), hyperlipidemia (71.9% vs. 55.2%), and CKD (15.1% vs. 5.7%, p < 0.001). In addition, the DOBS was significantly lower in patients with MI (15.65 ± 6.96 vs. 16.76 ± 6.77, p < 0.001).
Baseline characteristics stratified by MI and non-MI.

Note: Continuous and categorical variables are presented as means ± SD or percentages, respectively. MI: myocardial infarction; PIR: poverty income ratio; BMI: body mass index; CKD: chronic kidney disease; DOBS: dietary oxidative balance score.
Association between DOBS and MI
As shown in Table 3, a higher DOBS was significantly associated with a lower risk of MI. In the unadjusted model (Model 1), each one-point increase in DOBS was associated with a 2.4% reduction in MI risk (OR: 0.976; 95% CI: 0.968–0.984). When categorized into tertiles, participants in the middle (Q2) and highest (Q3) tertiles had 26.2% (OR: 0.738; 95% CI: 0.641–0.849) and 28.2% (OR: 0.718; 95% CI: 0.625–0.825) lower odds of MI, respectively, compared to the lowest tertile (Q1).
ORs and 95% CIs for the associations between DOBS and MI in hypertensive patients.
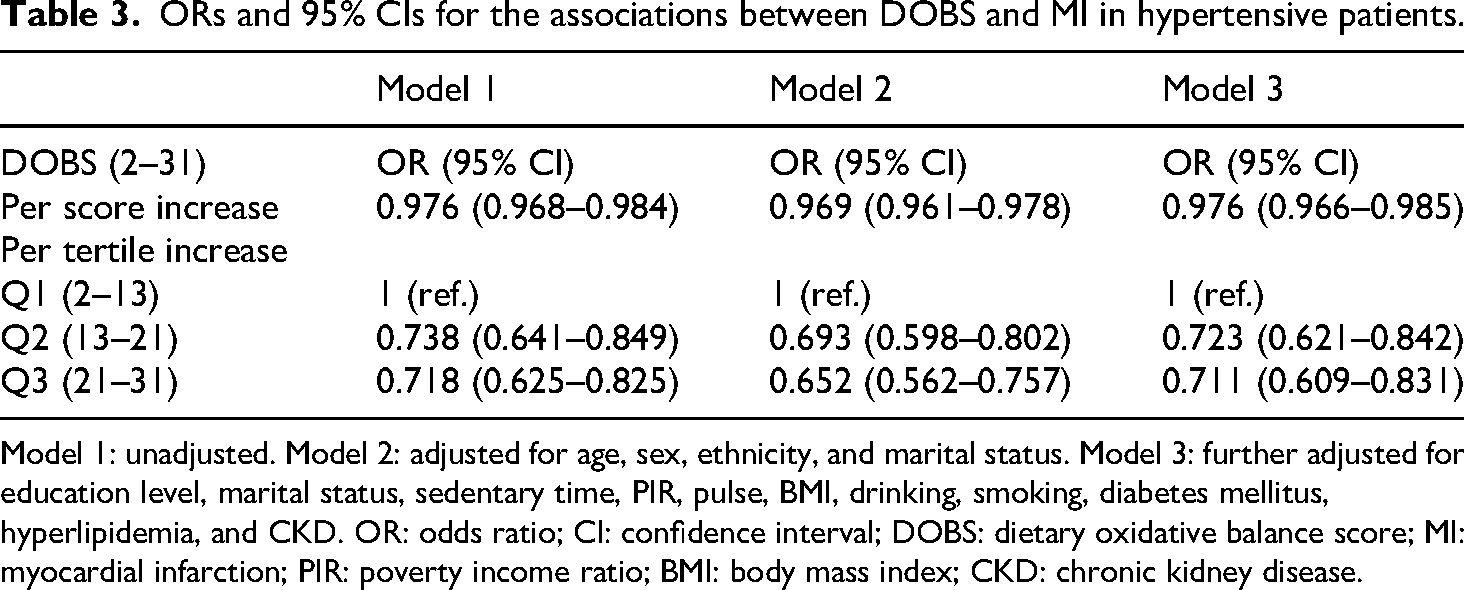
Model 1: unadjusted. Model 2: adjusted for age, sex, ethnicity, and marital status. Model 3: further adjusted for education level, marital status, sedentary time, PIR, pulse, BMI, drinking, smoking, diabetes mellitus, hyperlipidemia, and CKD. OR: odds ratio; CI: confidence interval; DOBS: dietary oxidative balance score; MI: myocardial infarction; PIR: poverty income ratio; BMI: body mass index; CKD: chronic kidney disease.
This inverse association remained robust after adjustment for potential confounders. In the fully adjusted model (Model 3), each additional point in DOBS was still associated with a 2.4% reduction in MI risk (OR: 0.976; 95% CI: 0.966–0.985). Compared to Q1, the Q2 and Q3 groups had 27.7% (OR: 0.723; 95% CI: 0.621–0.842) and 28.9% (OR: 0.711; 95% CI: 0.609–0.831) lower odds of MI, respectively. These findings confirm a consistent and independent inverse relationship between DOBS and MI risk across all models.
Nonlinear relationship
As illustrated in Figure 2, RCS analysis did not reveal a statistically significant nonlinear association between DOBS and MI risk (all p for nonlinearity > 0.95). However, the dose–response curve exhibited a steep decline in MI risk at lower DOBS levels, followed by a plateau as DOBS increased.
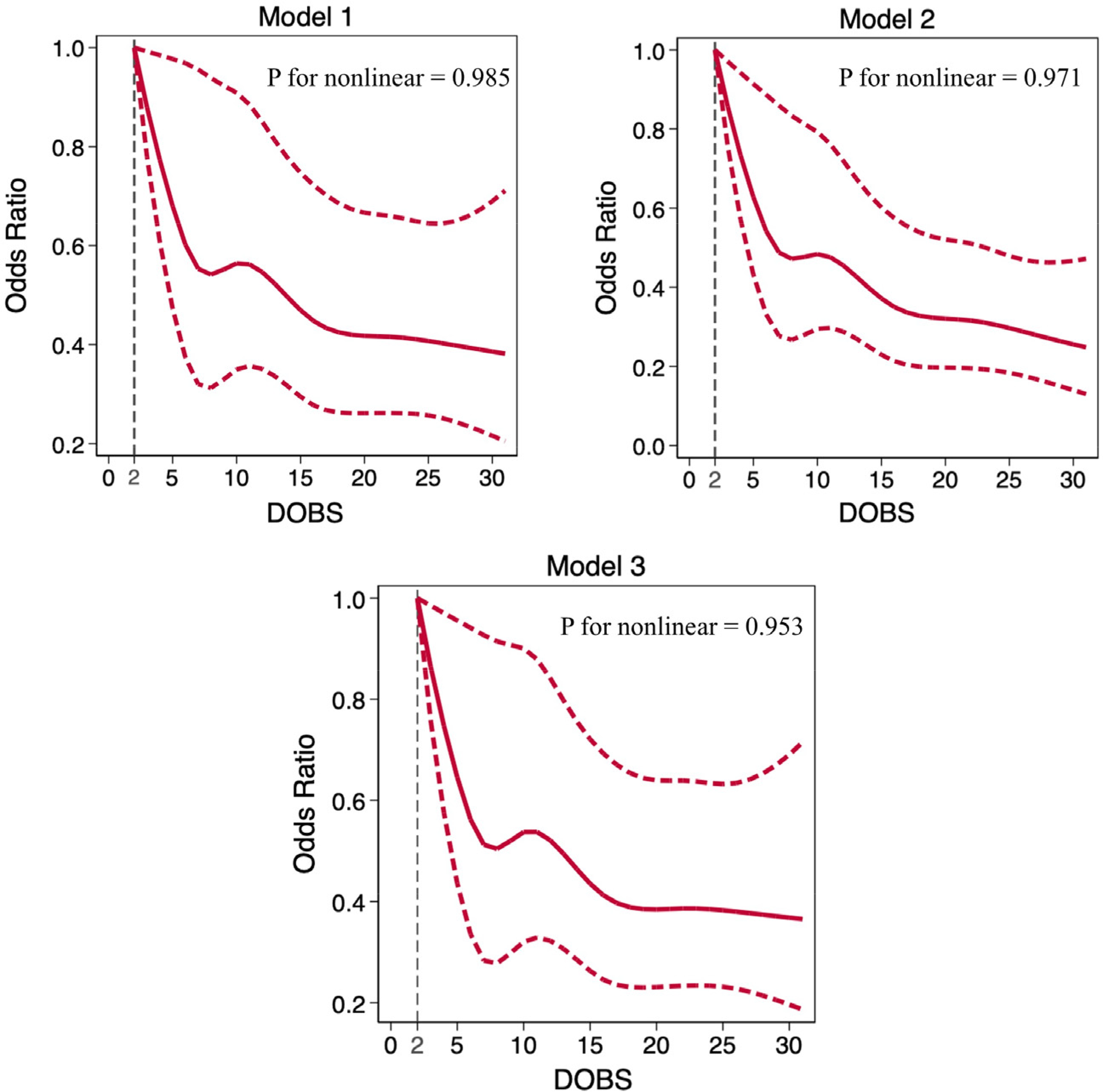
RCS curves for the association between DOBS and MI. Same models as Table 3.
Subgroup and sensitivity analysis
Subgroup analyses indicated that the inverse association between DOBS and MI persisted across multiple population strata, including sex, age, body mass index, smoking status, drinking status, DM, hyperlipidemia, and CKD (Figure 3). No significant interactions were detected between DOBS and any of the stratifying variables (p for interaction > 0.05), suggesting that the protective effect of higher DOBS was generally consistent across diverse subpopulations.

Subgroup analyses for the association between DOBS and MI. All models were adjusted as in Model 3 in Table 3. DOBS: dietary oxidative balance score; MI: myocardial infarction; OR: odds ratio; CI: confidence interval; P-int: P-interaction.
To determine whether the inverse association between DOBS and MI remained consistent across time, we conducted sensitivity analyses stratified by NHANES survey cycles (1999–2004, 2005–2010, and 2011–2018). As shown in Table 4, the negative association between DOBS and MI was observed across all three cycles, although the strength and statistical significance of the association varied.
ORs and 95% CIs for the associations between DOBS and MI in hypertensive patients in different cycles.

Model 1: unadjusted. Model 2: adjusted for age, sex, ethnicity, and marital status. Model 3: further adjusted for education level, marital status, sedentary time, PIR, pulse, BMI, drinking, smoking, diabetes mellitus, hyperlipidemia, and CKD. OR: odds ratio; CI: confidence interval; DOBS: dietary oxidative balance score; MI: myocardial infarction; PIR: poverty income ratio; BMI: body mass index; CKD: chronic kidney disease.
Discussion
This study is the first to examine the association between the DOBS and MI risk specifically in hypertensive adults. Based on fully adjusted models using NHANES 1999–2018 data, each one-point increase in DOBS was associated with a 2.4% reduction in MI risk (OR = 0.976; 95% CI: 0.966–0.985). Compared to the lowest tertile, participants in the highest tertile had 28.9% lower odds of MI (OR = 0.711; 95% CI: 0.609–0.831). Although the test for nonlinearity was not statistically significant, a threshold-like pattern was observed, with the most substantial risk reduction occurring at lower DOBS levels. Subgroup analyses confirmed that the inverse association was consistent across strata of age, sex, BMI, lifestyle, and comorbidities, and no significant interactions were identified. Sensitivity analyses across survey cycles further supported the temporal stability of the findings. These results suggest that maintaining a favorable oxidative dietary profile may offer a protective effect against MI in hypertensive individuals.
Previous studies have shown that DOBS components have antioxidant properties. For instance, vitamin C exerts its antioxidant activity by inhibiting tumor progression. 23 Dietary fiber and carotenoids have been shown to exhibit antioxidant effects both in vivo and in vitro. 24 A higher intake of vitamin E—primarily found in nuts, seeds, and vegetable oils—has been consistently associated with a reduced incidence of MI. 25 Epidemiological studies have shown that higher dietary intake of carotenoids reduces the risk of MI. 26 Our findings are partly supported by Zheng et al., 27 who reported an inverse association between oxidative balance score (OBS) and resistant hypertension and arterial stiffness. Although their OBS included both dietary and lifestyle factors, and their outcomes focused on intermediate vascular phenotypes, both studies highlight the protective role of oxidative balance in hypertensive individuals. Unlike their study, ours specifically targeted dietary components (DOBS) and focused on MI as a hard endpoint, thus extending the evidence to more severe cardiovascular outcomes. Jin et al. 28 reported that higher OBSs were significantly associated with lower 10-year atherosclerotic CVD risk (ASCVD) risk and reduced cardiovascular mortality, especially among individuals at high risk. Their findings further support the protective role of oxidative balance in CVD prevention. While their outcomes focused on long-term risk prediction and mortality, our study centered on clinically diagnosed MI, providing complementary evidence that oxidative balance is relevant not only for future risk estimation but also for actual cardiovascular events in hypertensive populations. Consistent with our results, Zhu et al. 29 found that higher OBSs were significantly associated with lower prevalence of chronic kidney disease (CKD) in individuals with metabolic syndrome. Their findings highlight the broad relevance of oxidative balance in preventing target organ damage across multiple disease contexts. Although their study focused on kidney outcomes and a different high-risk population, the shared inverse relationship supports the notion that maintaining oxidative balance may be a common protective mechanism against both renal and cardiovascular complications in metabolically vulnerable groups.
Hypertension is a major and prevalent risk factor for CVD and plays a critical role in the pathogenesis of MI.30,31 Sustained elevations in blood pressure lead to vascular wall injury, promote inflammatory responses, and accelerate the development of AS.32,33 This process contributes to plaque formation and progressive luminal narrowing, which can restrict coronary blood flow. Plaque rupture may subsequently trigger thrombus formation and acute coronary artery occlusion. 34 In addition, chronic hypertension induces myocardial hypertrophy as a compensatory response, but over time, this structural remodeling impairs systolic and diastolic function, increasing susceptibility to ischemic events. 35 Hypertension also disrupts endothelial integrity and impairs vascular tone, further compromising myocardial perfusion. 36 These multifactorial effects highlight the importance of investigating MI risk in hypertensive populations.
ROSs are critical for the progression of MI. Their levels and effects varied across the three different MI stages. Most antioxidants fail to effectively adapt to these changes, limiting their therapeutic potential in the treatment of MI. 37 ROS derived from p22phox-containing NADPH oxidase (NOX) plays a vital role in the pathogenesis of hypoxic diseases. 38 NOX1, NOX2, and NOX4 play essential roles in ROS production. 39 Moreover, ROS can affect MI through the p53 pathway. ROS-induced DNA damage leads to p53 activation, which induces apoptosis. 40 Another critical factor is the PI3 K/Akt signaling pathway. PI3 K/Akt promotes cell survival and protects cells from oxidative stress. However, when MI occurs, ROS disrupt PI3 K/Akt signaling, exacerbating cell damage and apoptosis. 41 These mechanistic insights are supported by both experimental and clinical evidence. For example, in diabetic models, elevated ROS disrupt antioxidant enzyme activity and activate NF-κB signaling, promoting inflammatory cytokine release and myocardial damage; inhibition of CB1R by AM251 ameliorated oxidative stress and myocardial necrosis. 42 Similarly, empagliflozin mitigated MI by attenuating MDA levels and enhancing STAT3 signaling, suggesting protective roles of mitochondrial redox regulation. 43 Furthermore, ischemic injury has been shown to induce maladaptive chromatin remodeling that suppresses PGC1α-mediated mitochondrial biogenesis, aggravating oxidative stress and myocardial dysfunction. 44 Collectively, these findings support a central role of redox imbalance in MI pathogenesis and underscore the potential of antioxidant-focused interventions such as DOBS in reducing MI risk.
Dietary patterns have profound impacts on cardiovascular health, especially in the context of MI. Emerging evidence highlights the role of gut-heart axis disturbances, mitochondrial dysfunction, and metabolic dysregulation in the pathogenesis of MI. For instance, gut dysbiosis and increased gut-derived pro-inflammatory metabolites like TMAO and LPS have been shown to exacerbate systemic oxidative stress and accelerate MI progression. 45 Dietary interventions such as ketogenic regimens or ketone body supplementation (e.g. β-hydroxybutyrate) can improve cardiac mitochondrial function, attenuate oxidative injury, and reduce infarct size. 46 Studies have shown that mitochondrial DNA content, regulated in part by diet and insulin resistance, predicts cardiovascular outcomes, supporting the view that dietary modulation of oxidative metabolism plays a critical role in MI development. 46 Notably, adherence to healthy food sources, such as Mediterranean diets supported by short food supply chains, is associated with reduced metabolic syndrome prevalence and lower cardiovascular risk. 47 These findings underscore the centrality of dietary oxidative balance in influencing MI risk and support the value of composite indices such as DOBS to quantify diet-related redox status.
Strengths and limitations
This study has several strengths. To our knowledge, it is the first large-scale cross-sectional analysis to explore the association between DOBS and MI specifically in hypertensive adults. This underscores the potential clinical value of promoting antioxidant-rich dietary patterns as part of cardiovascular risk management strategies in this population. By utilizing data from 14,137 participants across ten NHANES cycles, the study provides nationally representative evidence with a robust sample size and generalizability. The comprehensive adjustment for a wide range of demographic, lifestyle, and clinical variables, along with subgroup and sensitivity analyses, further enhances the validity of the findings.
However, several limitations should be acknowledged. First, due to its cross-sectional design, causal inference between dietary oxidative balance and MI cannot be established. Second, dietary intake was assessed using a single 24-hour recall, which may introduce recall bias and fail to reflect long-term dietary habits. Third, the OBS, based on self-reported data, does not account for individual differences in nutrient metabolism or potential measurement errors, and residual confounding may persist due to data limitations. Fourth, the findings may not be generalizable to non-hypertensive or non-US populations. Lastly, although the scoring system was constructed using established methods, further prospective studies are needed to validate these findings.
Threshold effect
Although no significant non-linear association was observed between DOBS and MI risk, a potential threshold effect cannot be ruled out. It is plausible that once a certain level of dietary antioxidant intake is achieved, the body's oxidative balance reaches a physiological plateau, beyond which additional intake yields minimal benefit. This may be due to saturation of antioxidant transport mechanisms, metabolic feedback regulation, or limited bioavailability at higher doses. Thus, even in the absence of statistical non-linearity, biological mechanisms may still underlie a threshold-like behavior.
Conclusion
Higher DOBSs are associated with a lower prevalence of MI in hypertensive patients, which may provide a useful reference for exploring dietary strategies in the prevention and management of MI in this high-risk population.
Footnotes
Abbreviations
Acknowledgements
We would like to thank the NHANES administration for providing publicly available data and reports, which enabled us to prepare this manuscript. The authors used ChatGPT (OpenAI, San Francisco, CA) to assist with language refinement and editing during manuscript preparation. The authors reviewed and approved all content.
Ethical considerations and consent to participate
This study used publicly available, de-identified data from the National Health and Nutrition Examination Survey (NHANES), which is conducted by the National Center for Health Statistics (NCHS). NHANES protocols were approved by the NCHS Research Ethics Review Board, and written informed consent was obtained from all participants. Therefore, no additional ethical approval was required for this secondary data analysis.
Authors’ contributions
Zehao Jin and Xu Li wrote the manuscript. Ziyang Huang and Ya Lin assisted with data extraction and language editing, Xiaoyi Zhan and Jialin Liu assisted with data analysis, whereas Kangwei Wang provided statistical guidance and was responsible for final revisions.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
