Abstract
Background
Although abnormalities in circulating lipids and lipoproteins are associated with increased cancer risk, their specific impact on lung cancer progression and prognosis is still unclear. This study retrospectively assessed the influence of preoperative lipid and lipoprotein levels on non-small cell lung cancer progression and prognosis, stratified by age.
Methods
In this retrospective study, we analyzed 849 patients to investigate the association between lipid markers and lung cancer progression, and examined postoperative prognosis in a subset of 222 patients. Data was analyzed using restricted cubic spline curves, Kaplan–Meier survival analysis, and Cox proportional hazards models.
Results
A significant nonlinear relationship was observed between total cholesterol (TC), high-density lipoprotein (HDL), ApoB, ApoAI, ApoE, and baseline tumor diameter (BSLD) (PTC = 0.025; PHDL < 0.001; PApoB = 0.037; PApoAI =0.001; PApoE < 0.001). In contrast, Lp(a) showed a significant linear relationship with BSLD (P = 0.002). The Cox regression analysis revealed that triglyceride (TG) (hazard ratio (HR) = 0.50, 95% confidence interval (CI): 0.28–0.92, P = 0.025) was significantly negatively associated with lung cancer mortality in patients under 58 years. For patients over 58 years, higher ApoB levels were linked to a reduced risk of lung cancer death (HR = 0.59, 95% CI: 0.36–0.97, P = 0.038).
Conclusion
This study reveals a significant negative correlation between ApoAI and HDL levels with BSLD, while Lp(a) shows a positive correlation. In terms of long-term prognosis, high-serum ApoB are associated with a lower mortality risk in all lung cancer patients, and high-serum TG levels associated with reduced mortality risk in patients aged under 58 while high-serum TC levels associated with reduced mortality risk in patients over 58, with high Lp(a) levels indicating a greater risk of mortality in older patients.
Introduction
Lung cancer is one of the most prevalent and lethal cancers worldwide, with mortality rates continuing to rise annually.1,2 Global cancer statistics present a bleak outlook, with a 5-year survival rate of only 25% for lung cancer patients. 3 By 2050, the global cancer burden is projected to double, with lung cancer anticipated to be a major contributor to this increase. Non-small cell lung cancer (NSCLC) accounts for approximately 80% of all lung cancer cases. 4 The widespread adoption of computed tomography scans has significantly enhanced early detection rates, leading to a 20% reduction in lung cancer mortality. 5 Although liquid biopsies have improved lung cancer detection, identifying reliable biomarkers for early-stage detection remains a persistent challenge. 6 This highlights the urgent need for more effective early detection techniques and enhanced screening strategies for high-risk individuals in clinical practice.
Dyslipidemia, traditionally linked to cardiovascular disease, has also been increasingly associated with cancer through its impact on lipid metabolism.7–9 Advances in plasma lipidomics indicate that specific lipidomic signatures can assist in the diagnosis of early-stage lung cancer. For instance, Wang et al. demonstrated that glycerophospholipid and triglyceride (TG) dysregulation in tumor cells and immune microenvironments could distinguish early-stage NSCLC patients from healthy controls, underscoring the potential of lipidomics in clinical stratification. 5 TGs, a major component of blood lipids, promote fatty acid oxidation and are directly associated with cell proliferation and tumor growth.10,11 Total cholesterol (TC) modulates the anti-cancer immune response in tumor-associated macrophages of human lung adenocarcinoma via the cholesterol metabolic pathway.12,13 High-density lipoprotein (HDL) may exert anti-tumor effects, thanks to its antioxidant and anti-inflammatory properties. 14 Furthermore, research indicates that HDL levels exhibit a negative correlation with the size of lung cancer tumors. 15 Low TC levels have also been linked to increased cancer mortality across several cancer types.16,17 While prospective studies have examined the roles of apolipoprotein A-I (ApoAI), apolipoprotein B (ApoB), and lipoprotein(a) [Lp(a)], their associations with cancer mortality remain inconsistent.18–20
Lipids play a key role in both health and aging. There is evidence that lipid metabolism is closely related to cellular senescence, and the lipid content and metabolism of senescent cells undergo significant changes.21,22 Moreover, the accumulation of senescent cells increases with age, while the immune system's capacity to clear these cells diminishes over time. Additionally, research indicates that aging adversely affects the prognosis of lung cancer patients, significantly elevating their mortality risk.23,24 Therefore, investigating age-specific lipid markers and their prognostic implications could provide valuable insights. Lipidomic profiling in NSCLC has emerged as a critical tool for predicting therapeutic responses across chemotherapy, immunotherapy, and targeted therapies. Specific phospholipids and cholesterol esters in serum correlate with chemotherapeutic efficacy, 25 while elevated free fatty acid (FFA) predict improved immunotherapy outcomes by modulating immune membrane dynamics. 26 Notably, EGFR-mutant tumors display unique lipid signatures, linking oncogenic signaling to lipid remodeling. 27 Additionally, pretreatment cholesterol metabolism (low TC/LDL levels) independently predicts survival benefits in anti-angiogenic therapy recipients. 28 These findings underscore lipid as dual biomarkers for treatment stratification and EGFR-driven metabolic adaptation in NSCLC.
Few studies have investigated the relationship between blood lipids, lipoproteins, and lung cancer progression and prognosis, especially in terms of how age modulates the influence of lipid levels on lung cancer outcomes. The anti-cancer potential of lipids and lipoproteins remains a topic of debate, with their underlying mechanisms still largely unexplored. This study seeks to retrospectively assess the role of preoperative lipid and lipoprotein levels in NSCLC progression and prognosis across various age groups.
Method
Study population
This single-center, retrospective study included 849 lung cancer patients diagnosed at Jinling Hospital between January 2017 and December 2023 to investigate the correlation between routine lipid markers and baseline tumor diameter (BSLD). A subset cohort of 222 patients who reached a 5-year follow-up were selected for post-operative survival analysis from 849. Inclusion criteria included: (i) the age of 18 years or older, (ii) a confirmed diagnosis of NSCLC, and (iii) the absence of concurrent cancers. Exclusion criteria were (i) incomplete lipid data, (ii) medical conditions related to elevated blood lipids (e.g. diabetes, hyperlipidemia, or metabolic syndrome) or prior treatment for hyperlipidemia, or (iii) used lipid-lowering medications before admission (Figure 1).

The flowchart for data collection in the retrospective study.
Data collection
Basic demographic data, such as age, sex, BMI, adjusted Charlson Comorbidity Index (aCCI), and BSLD, were collected for all participants. Preoperative fasting blood samples were analyzed for lipid markers, including TC, TG, HDL, LDL, ApoAI, ApoB, apolipoprotein E (ApoE), Lp(a), and FFAs, which were measured preoperatively after fasting. The aCCI was calculated based on each patient's medical history and age. Smoking status was defined as consuming one or more cigarettes per day for over 6 months or quitting within the past 6 months. Alcohol consumption was classified as drinking at least once per week for over 6 months or having abstained for less than 6 months. Death was defined based on confirmation from patient medical records, hospital records, and follow-up data. Censor was defined as failure to adhere to the follow-up protocol outlined in the eighth edition of the lung cancer guidelines. All 849 patients were undergoing radical lung cancer resection in line with international guidelines (the NCCN Guidelines).
Follow-up
Follow-up assessments were conducted according to the eighth edition of lung cancer criteria (the AJCC criteria). Following discharge, patients were monitored every 3 months during the first 2 years, and every 6 months thereafter, until 5 years post-surgery or death, with the final follow-up occurring on 1 December 2023.
Statistical analysis
Qualitative variables were expressed as percentages, with group comparisons conducted using the chi-squared test. Quantitative variables with a normal distribution were expressed as mean ± standard deviation and analyzed using T-tests. For non-normally distributed data, the median and interquartile range were calculated, and group differences were analyzed using non-parametric tests, such as the Mann–Whitney U test. Additionally, outliers were addressed by correcting entry errors and imputing missing values with the mean.
Spearman correlation coefficients (r) were used to assess correlations between variables. A single-factor regression model and restricted cubic spline (RCS) curve with five knots were applied to illustrate nonlinear relationships between lipid markers and BSLD. Linear regression was performed to analyze linear relationships, and results were presented in scatter plots.
The receiver operating characteristic curve with Youden index was used to calculate the best cut-off value for age; the best cut-off value for age was 57.5 at which the maximum Youden index is 0.11, and the age group was rounded to 58. Age in the prognostic cohort was rounded to <58 years and ≥58 years, and subgroup analyses were conducted based on the median lipid index, using the lower median category as the reference. The hazard ratio (HR) and its 95% confidence interval (CI) were calculated using a Cox proportional hazard regression model. Univariate and multi-factor Cox regression analysis was performed first. An adjusted model was then developed based on univariate regression, incorporating demographic characteristics, lifestyle factors, and medical history as covariates to reduce potential confounding effects. Forest plots were used to visualize the results. Kaplan–Meier curves were also generated to compare survival differences across lipid groups. Statistical analyses were conducted using IBM SPSS version 29.0 and R version 4.4.2, with a p-value <0.05 considered statistically significant.
Ethical statement
This study adhered to medical ethics standards and was conducted in accordance with the Declaration of Helsinki. It received approval from the Jinling hospital's Ethics Committee and obtained a waiver for informed consent. To ensure data privacy, we implemented encryption, anonymization and access control measures to ensure the security and confidentiality of patient information. We have identified the detailed information of the patients to ensure that they will not be identified in any way. The reporting of this retrospective study conforms to STROBE guidelines. 29
Results
Baseline characteristics
Table 1 provides a summary of the baseline characteristics of the study population and explores the association between lipid markers and BSLD (continuous variable). The study included 849 patients, with a median age of 58 years, and 57.8% of the cohort were male. Approximately 19.9% of the patients had a history of smoking, while 86.5% reported no history of alcohol consumption. In the analysis of patients with 5 years of follow-up, it was observed that preoperative neoadjuvant therapy was administered to 1.6% of patients, with adenocarcinoma being the predominant pathology in 92.5% of cases.
Demographic and patient characteristics of the study population on BSLD correlation.

Note. aCCI: adjusted Charlson Comorbidity Index; TG: triglyceride; TC: total cholesterol; HDL: high-density lipoprotein; BSLD: baseline tumor diameter; FFA: free fatty acid; IQR: interquartile range.
Table 2 presents the baseline characteristics for the analysis of lipid markers and mortality. Based on the optimal cut-off for age, 222 patients were divided into two groups: < 58 years (n = 105) and ≥58 years (n = 117). The <58 age group had a higher proportion of males (63.8% vs. 47.9%, p = 0.017). The median aCCI score was significantly lower in the younger group [3.0 (2.0–3.0)] compared to the older group [4.0 (4.0–5.0)] (p < 0.001). The percentage of smokers was lower in the younger group (17.1%) than in the older group (33.3%, p = 0.006). Additionally, the incidence of lung adenocarcinoma, compared with the >58 group, was significantly higher in the <58 group (92.5% vs. 7.5%, p = 0.002).
Demographic and patient characteristics of the 5-year mortality study population.

Note. P-values <0.05 are marked with *. TG: triglyceride; TC: total cholesterol; HDL: high-density lipoprotein; BSLD: baseline tumor diameter; FFA: free fatty acid; IQR: interquartile range.
Correlation analysis of variables
Positive correlations were observed between TC and LDL, ApoB, and ApoE (r = 0.83, r = 0.74, r = 0.41, respectively). TG showed a negative correlation with HDL (r = -0.25) but was positively correlated with ApoB (r = 0.57) and ApoE (r = 0.54). Additionally, a strong positive correlation was noted between LDL and ApoB (r = 0.69), with a similar relationship between ApoB and ApoE (r = 0.43).
Concerning BSLD, negative correlations were identified with HDL (r = -0.07) and ApoAI (r = -0.13), while Lp(a) showed a positive correlation (r = 0.10). A strong positive correlation was observed between age and the aCCI score (r = 0.85) (Figure 2).

Correlation Heatmap between various factors and BSLD.
Correlation between lipid markers and baseline tumor diameter
Analysis of both linear and nonlinear relationships between lipid markers and BSLD, using a single-factor RCS curve, revealed significant nonlinear associations for TC (Pnonlinear = 0.025), HDL (Pnonlinear < 0.001), ApoB (Pnonlinear = 0.037), ApoAI (Pnonlinear = 0.001), and ApoE (Pnonlinear < 0.001). Conversely, Lp(a) demonstrated a significant linear relationship with BSLD (Plinear = 0.002) (Figure 3). However, the relationships between LDL, TG, and FFA with BSLD were not significant, with p-values exceeding 0.05 (supplement 1).

Linear and nonlinear relationship between various factors and BSLD.
Relationship between lipid indexes and mortality
Patients were divided into two age groups: < 58 and ≥58 years, followed by univariate and multivariate regression analyses. In the <58 group, univariate analysis identified gender (HR = 0.56, P = 0.040), TG (HR = 0.59, P = 0.047), and ApoB (HR = 0.53, P = 0.021) as significant factors influencing lung cancer mortality risk. For the ≥58 group, TC (HR = 0.62, P = 0.042), ApoB (HR = 0.60, P = 0.030), and Lp(a) (HR = 1.69, P = 0.026) were identified as significant predictors of lung cancer mortality. However, none of the lipid markers remained statistically significant in the multivariate regression analysis (supplement 2).
An adjusted model, based on single-factor regression results, revealed that in patients under 58 years old, TG (HR = 0.52, 95% CI: 0.29–0.93, P = 0.028) and ApoB (HR = 0.56, 95% CI: 0.33–0.97, P = 0.039) were significantly associated with reduced lung cancer mortality. In Model 2, TG levels were also significantly associated with reduced mortality risk (HR = 0.50, 95% CI: 0.28–0.92, P = 0.025). For the ≥58 group, Model 1 indicated a positive correlation between Lp(a) and mortality risk (HR = 1.63, 95% CI: 1.01–2.63, P = 0.044), while Model 2 found that higher ApoB levels were associated with a reduced risk of lung cancer death (HR = 0.59, 95% CI: 0.36–0.97, P = 0.038). No other lipid markers were found to be statistically significant (Figure 4).

The forest map of the model adjusted for age groups.
Kaplan–Meier survival curve analysis revealed that in lung cancer patients under the age of 58, the median survival time in the high TG group was significantly longer than that in the low TG group (P = 0.042). Similarly, patients with high ApoB levels had a significantly higher survival rate compared to those with low ApoB levels (P = 0.018). In patients over 58, the median survival time was significantly higher in the high TC group (P = 0.037) and high ApoB group (P = 0.026) compared to the low TC and low ApoB groups. In contrast, the lung cancer survival rate was significantly lower in the high Lp(a) group compared to the low Lp(a) group (P = 0.022) (Figure 5).
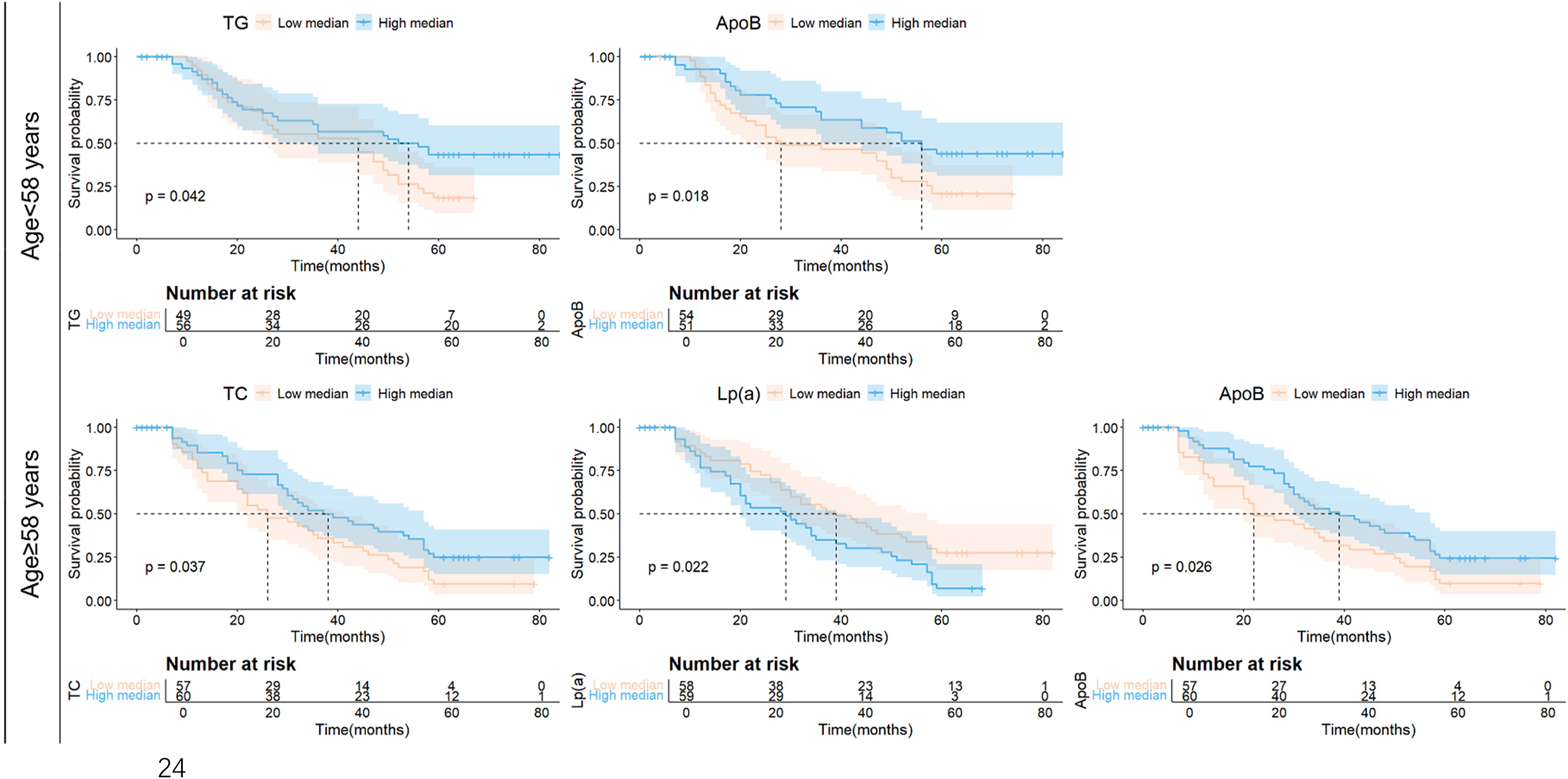
Kaplan–Meier survival curves between lipid indicators and NSCLC. NSCLC: non-small cell lung cancer.
Discussion
Lipids play a pivotal role in the development and progression of malignant tumors, with numerous studies highlighting associations between circulating lipids, lipoproteins, and cancer onset or prognosis.9,30–33 In this retrospective study, we investigated these relationships and their implications for long-term cancer prognosis. Our findings align with previous studies, showing a negative correlation between HDL levels and tumor sizes.15,34 ApoAI and HDL levels exhibited a protective effect against BSLD within a defined range. To our knowledge, this study highlights a positive linear relationship between Lp(a) and BSLD, which has not been widely reported.
For long-term prognosis, we found that high-serum ApoB are associated with a lower mortality risk in all lung cancer patients, and high-serum TG levels associated with reduced mortality risk in patients aged under 58 while high-serum TC levels associated with reduced mortality risk in patients over 58. Higher Lp(a) levels increased mortality risk in older patients. This finding is consistent with previous studies showing a linear relationship between Lp(a) and cancer mortality. 35 Studies have shown that TG and HDL are independent prognostic factors for NSCLC.36,37 Low TC levels are linked to increased cancer mortality.17,19 A Mendelian randomized study has identified a negative link between ApoAI and cardiovascular mortality. 38 However, sample deviation or age factors may have influenced our results, limiting comparable findings in multi-factor analysis.
Lipid metabolism disorders are increasingly associated with age in recent studies,23,39 for example, one study on the relationship between lipids and age in women showed that lipid markers increase with age, 40 age-related differences were observed in lipidomic profiles, including lipid levels, subclasses, and specific lipid species. 41 Aging has also been identified as a significant predictor of lung cancer survival, with advancing age notably increasing the risk of lung cancer mortality. 42 Xia, Meng et al. 43 found that the abundance of gut microbiota in elderly patients with hyperlipidemia was correlated with serum lipid and apolipoprotein levels. Thus, age-related changes in gut microbiota may disrupt lipid metabolism, which could, in turn, impact cancer prognosis to some extent.44–46
While our adjusted model associates lipid biomarkers with cancer prognosis, the exact biological mechanisms underlying these relationships remain unclear. Zablocka-Slowinska, K et al. 47 noted a complex relationship between oxidative stress and lipid dysregulation in lung cancer patients, with the overall oxidative state being modulated by fluctuations in lipid levels. Lipid metabolism products, such as excess reactive oxygen species (ROS), may influence tumor initiation and progression through oxidative stress mechanisms. For example, ROS can activate the MAPK/ERK pathway, 48 and MAPK-induced phosphorylation and subcellular translocation of PDHE1αpromote tumor immune evasion. 49 Consequently, lipid metabolism disorders can influence the onset and progression of various cancer types.50,51 Another study showed that in early lung cancer, the metabolic pathways of glycerophospholipids and TGs in tumor cells as well as T cells, B cells, NK cells, and fibroblasts are dysregulated, 5 The negative correlation between HDL and oxidative stress may mainly stem from the antioxidant properties of HDL, which affect the hydrolysis of lipid peroxides, and thus affect the metabolic pathways of glycerophospholipids and TGs in the development of lung cancer. Additionally, studies have shown that lipid metabolism impacts cellular siderosis through the formation of iron-dependent lipid hydroperoxides. 52
In this study, some of the relevant biologic mechanisms mentioned may be suggestive of it. Previous researchers have found that ApoB levels may be positively associated with the risk of colorectal cancer and head and neck squamous cell carcinoma.18,53 Exome sequencing studies show that ApoB, which is influenced by lipid metabolism, strongly correlates with LDL levels. 54 As an important structural component of LDL, ApoB levels might potentially be related to the malignancy of tumors and the overall prognosis of tumor patients. 55 We hypothesize that ApoB may influence NSCLC progression by modulating cholesterol transport and downstream cholesterol metabolites. Regarding TC, based on existing knowledge, it is possible that it can affect the immune system and anti-tumor immune response through cholesterol metabolism. 12 In addition, the mechanism for Lp(a) may be to increase cancer risk by promoting oxidative stress and inflammatory responses, and the oxidized form of Lp(a) may trigger chronic inflammation, further promoting the development and deterioration of cancer. 56 TG is thought to be involved in the development of oxidative stress and the formation of ROS. These processes play a role in normal cell proliferation and might also promote the proliferation and anti-apoptotic ability of cancer cells.11,57 In Model 1, we observed that high ApoB and high TG levels did not show a protective effect on mortality risk in patients aged ≥58. This may be due to the decline in immune function with aging, which impairs the ability of immune cells to surveil and eliminate tumor cells. Elevated ApoB levels may promote chronic inflammation, hindering tumor cell clearance and worsening outcomes in elderly lung cancer patients. 58 However, the exact interaction mechanism remains unclear and needs further investigation. Some studies suggest that high Lp(a) is more likely to induce the formation of a fibrin network and thrombus, which might potentially promote the adhesion, invasion, and metastasis of cancer cells. 59 But this is also a hypothesis that requires experimental verification.
In recent years, the global burden of dyslipidemia has increased, undeniably increasing the potential risk of various cancers.60,61 Our findings build on previous reports examining the relationship between lipid profiles and the development and prognosis of lung cancer. We modeled the effects of aging in lung cancer patients through age groups to more accurately predict their long-term prognosis in the real world. The inclusion of lipid indicators that are now more widely used provides new evidence for the relationship between lipid predictors and lung cancer, highlighting the need for further research into the biological mechanisms behind these relationships and the potential use of these biomarkers in clinical practice. Integrating the findings of these biomarkers into clinical decision-making allows physicians to develop more precise treatment strategies and provide personalized treatment options. For high-risk patients, such as elderly individuals with elevated Lp(a) levels, more aggressive therapeutic interventions may be required to enhance survival rates. Future studies should cover a wider range of lipid components and delve into more detailed subtypes of lung cancer to further shed light on this key topic.
Limitation
First of all, the sample size included in our study is still relatively small, and there may be some sample bias in the single-center study. For the prognostic cohort, age grouping may be subject to error due to sample limitations and the classification method here needs to be optimized differently for different sample distribution characteristics and is not universal. In addition, there found no significant results consistent with α=0.025 levels after supplementing the Bonferroni correction, which may be due to important confounding factors, such as dietary habits, metabolic status, physical activity, and genetic predisposition, may have been overlooked in the analysis. Inadequate control for these factors may have contributed to the loss of statistical significance of the observed effects. At present, preclinical studies of various targeted drugs for lipid metabolism have been reported,62,63 exploring lipid levels as a potential biomarker for predicting lung cancer risk. In the future, it is hoped to further expand the multi-center research on lipid metabolism in lung cancer staging, so as to provide new evidence for early diagnosis and personalized treatment of lung cancer.
Conclusion
In summary, in this retrospective study, we found that ApoAI and HDL were negatively correlated with BSLD. While Lp(a) we found a positive linear relationship with BSLD. Moreover, in the long-term prognosis, we found that high-serum ApoB are associated with a lower mortality risk in all lung cancer patients, and high-serum TG levels associated with reduced mortality risk in patients aged under 58 while high-serum TC levels associated with reduced mortality risk in patients over 58. The combination of circulating lipid levels and age shows potential as a method for predicting lung cancer prognosis. This study provides new avenues for adjuvant treatment options and highlights the critical intersection of aging, lipid metabolism, and cancer as public health priorities.
Supplemental Material
sj-docx-1-sci-10.1177_00368504251352375 - Supplemental material for Preoperative lipid levels and prognosis in non-small cell lung cancer: A retrospective age-stratified study
Supplemental material, sj-docx-1-sci-10.1177_00368504251352375 for Preoperative lipid levels and prognosis in non-small cell lung cancer: A retrospective age-stratified study by Yv-Xuan Liu, Jing Chen, Chen Liu, Maierhaba Maitiyasen, Xinyv Hou, Jingfeng Li and Jun Yi in Science Progress
Supplemental Material
sj-docx-2-sci-10.1177_00368504251352375 - Supplemental material for Preoperative lipid levels and prognosis in non-small cell lung cancer: A retrospective age-stratified study
Supplemental material, sj-docx-2-sci-10.1177_00368504251352375 for Preoperative lipid levels and prognosis in non-small cell lung cancer: A retrospective age-stratified study by Yv-Xuan Liu, Jing Chen, Chen Liu, Maierhaba Maitiyasen, Xinyv Hou, Jingfeng Li and Jun Yi in Science Progress
Footnotes
Acknowledgments
The authors are grateful to Dr Jun Yi for help with the preparation of figures in this paper.
Author contributions
YL and JC contributed equally to this work. YXL and JC conceived and designed the study. CL and MM collected the data. XH and JL performed data analysis and the figures. JY contributed to data interpretation and manuscript revision. YL and JC drafted the manuscript, and all authors reviewed and approved the final version.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The datasets generated during the current study are not publicly available due to protecting the privacy of individuals that participated in the study but are available from the corresponding author on reasonable requests.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
