Abstract
Background:
Detecting distant metastases when staging lung cancer is critical to avoid unnecessary surgery and provide appropriate multidisciplinary treatment. However, it is controversial as to whether staging studies should be performed routinely for patients with early-stage lung cancer who have no evidence of distant metastasis. Thus, this study aimed to examine the need for extrathoracic metastasis screening in patients with clinical stage IA non-small cell lung cancer, understand the association between extrathoracic metastasis and other clinical features, and evaluate the diagnostic efficiency of imaging screening for preoperative extrathoracic metastasis in patients with early-stage lung cancer.
Methods:
From 2010 to 2019, 510 patients diagnosed with clinical T1N0 lung cancer, excluding contralateral lung metastases, pleural dissemination, malignant pleural effusion, and malignant pericardial effusion, were treated for primary lung cancer. Patients were divided into two groups, and their clinicopathological characteristics were investigated.
Results:
Five patients (1.0%) had extrathoracic metastases. The histological types were adenocarcinoma in three of the cases, and squamous cell carcinoma and large cell neuroendocrine carcinoma in the other two cases. The T factor was T1b in one case and T1c in four cases. Four patients had solid tumors and one had a solid predominant tumor with an average tumor diameter of 23.0 ± 2.9 mm. The size of solid tumors with extrathoracic metastases was larger than their counterparts.
Conclusion:
When evaluating stage IA non-small cell lung cancer with a solid component diameter < 22 mm, or clinical T1mi-1bN0 in computed tomography evaluation, screening for preoperative extrathoracic metastasis may be omitted.
Introduction
Lung cancer is the most common and lethal type of cancer worldwide 1 with a high mortality rate owing to multiorgan metastasis. Extrathoracic metastases are commonly observed, and approximately 40% of cases are reported during diagnosis. 2 The brain, liver, adrenal glands, and skeletal system are the most likely sites of metastasis in patients with lung cancer. 2 Occult extrathoracic metastasis may result in early postoperative recurrence and poor prognosis. Thus, detecting distant metastases when staging lung cancer is critical to avoid unnecessary surgery and provide appropriate multidisciplinary treatment. 3 However, it is controversial as to whether staging studies should be performed routinely for patients with early-stage lung cancer who have no evidence of distant metastasis. In fact, routine head screening for lung cancer with a clinical stage ≤ IA is not recommended.4,5 Thus, this study aimed to determine the incidence of extrathoracic metastasis in patients with clinical stage IA non-small cell lung cancer (NSCLC), understand the association between extrathoracic metastasis and other clinical features, and evaluate the diagnostic efficiency of imaging screening for preoperative extrathoracic metastasis in patients with early-stage lung cancer.
Methods
Study design and patients
Between January 2010 and December 2019, 1344 patients were diagnosed with lung cancer and treated at our hospital. The patient records were extracted from a hospital-based cancer registry. The inclusion criteria were as follows: patients had computed tomography (CT) images acquired before receiving treatment, and clinical T1 was categorized based on CT imaging findings according to the eighth edition of the Union for International Cancer Control tumor-node-metastasis (TNM) classification of malignant tumors. 6 The N category reported on CT, complementarily evaluated using whole-body positron emission tomography (PET)/CT scan using 2-deoxy-2-(18F)-fluorodeoxyglucose (FDG) examination, was based on axial images of the enlarged (≥10 mm) short axis of the lymph node. The exclusion criteria were metastasis other than extrathoracic metastasis, identified by FDG-PET/CT and head CT or magnetic resonance imaging (MRI) examination. A total of 539 patients were clinically diagnosed with stage IA (T1N0) lung cancer using chest CT and whole-body FDG-PET/CT. Brain metastasis was evaluated using contrast-enhanced CT in 60 patients, plain MRI in 82, and contrast-enhanced MRI in 373. Nineteen patients who did not undergo brain metastasis screening and 10 patients who were diagnosed with small cell lung cancer were excluded. Extrathoracic metastasis was confirmed in five cases (Figure 1). A definitive pathological diagnosis was obtained by surgical biopsy under general anesthesia in 378 cases, transbronchial lung biopsy or cytology in 126 cases, and CT-guided needle biopsy in six cases. Post-diagnosis treatment included surgical resection in 475 patients (lobectomy in 273, segmentectomy in 58, and partial resection in 143), radiotherapy such as stereotactic body radiation therapy in 23, best supportive care in 8, and systemic chemotherapy in 4.

Patient selection flowchart.
Informed consent was obtained from all patients before treatment for later access to their clinical records. Additionally, the ethics committee of our university permitted access to and use of patient data for this study in April 2020 (approval numbers: I160 and I449).
Statistical analyzes
Statistical analyzes were conducted using JMP software (version 14.0; SAS Institute Inc., Cary, North Carolina, USA). Two-category comparisons were performed using the chi-square test A receiver operating characteristic (ROC) curve was constructed according to the solid size of the tumor, consolidation/tumor (C/T) ratio, and cutoff values for the existence of extrathoracic metastasis. Differences were considered at a p-value < 0.05.
Results
The clinicopathological characteristics of patients with stage IA NSCLC (T1N0) are presented in Table 1, and a summary of characteristics of patients with extrathoracic metastasis (M1b and 1c) is presented in Table 2. We assessed 510 patients (195 women and 316 men) with an average age of 69.8 ± 8.9 years (range, 37–92 years). There were 559 patients without extrathoracic metastasis (M0) and six patients with extrathoracic metastasis (M1b or M1c), excluding M1a factors. The clinical T factor was T1mi in 42 patients (8.2%), T1a in 84 (16.5%), T1b in 228 (44.7%), and T1c in 156 (30.6%). The clinical M factor was M1b in four patients (0.8%) and M1c in one patient (0.2%). The clinical stage, not accounting for extrathoracic metastases, was stage IA1 in 126 patients (24.7%), stage IA2 in 228 (44.7%), and stage IA3 in 156 (30.6%). The mean total size and solid size of the tumor were 19.5 ± 6.5 mm (range, 6–52 mm) and 16.0 ± 7.2 mm (range, 2–30 mm), respectively. The mean C/T ratio was 70.5 ± 33.3% (0%–100%). Twenty-one patients opted to receive supportive care because of their old age or received treatment from another hospital. Histological examination revealed adenocarcinoma in 389 patients (76.3%); squamous cell carcinoma (Sq) in 92 (18.0%); neuroendocrine tumors, such as carcinoid tumors and large cell neuroendocrine carcinoma (LCNEC), in 13 (2.5%); and other types such as pleomorphic carcinoma and adenosquamous carcinoma in 16 (3.1%). Patients with extrathoracic metastasis had a significantly larger solid size (p = 0.027).
Clinicopathological features of clinical T1N0 cases with or without extrathoracic metastasis.

Cases of clinical T0-1N0M1b-c.

M: male, F: female, LCNEC: large cell neuroendcrine carcinoma, SCLC: small cell lung carcinoma, Sq: squamous cell carcinoma, Ad: adenocarcinoma, BRA: brain metastasis, OSS: osseous metastasis, HEP: hepatic metastasis, SRT: stereotactic radiation therapy.
Extrathoracic metastasis was found in five patients (three men and two women). The T factor was T1b in one case and T1c in four cases. The histological types were adenocarcinoma in three of the cases, and LCNEC and Sq in one case each. Four patients had solid tumors with a C/T ratio of 100%, and one had a solid dominant tumor with a C/T ratio of 85.2%. Four patients received anticancer chemotherapy, and two with brain metastases received stereotactic radiotherapy or a γ knife. Three patients died, and two remained alive. The mean observation interval was 26.5 ± 15.5 months (range 4–52 months).
The risk factors for extrathoracic metastasis were extracted from these results. The cut-off value for the solid size calculated from the ROC curve was 22 mm (Figure 2). Furthermore, tumors containing solid components of 22 mm or more (p = 0.006) and having a clinical stage of ΙA3 (p = 0.016) showed significantly more extrathoracic metastasis than their counterparts (Table 3).
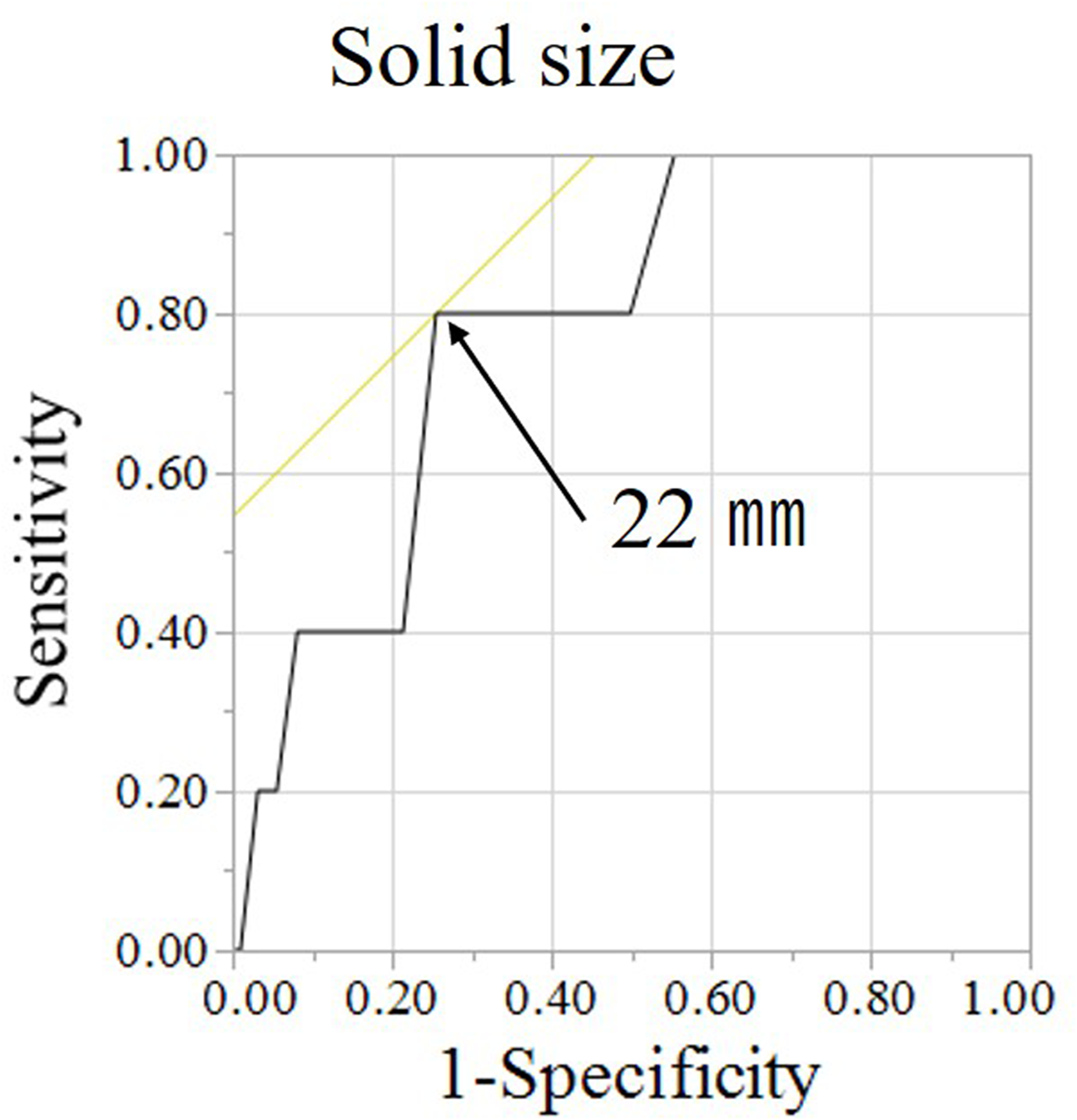
Receiver operating characteristic (ROC) curve.
Risk factors for extrathoracic metastasis of clinical T1N0.

Discussion
For the Union for International Cancer Control-TNM classification for primary lung cancer, the new 8th edition revised edition was applied from 2017. Along with this, factor T was evaluated by the total tumor diameter and the solid component diameter. Carcinoma in situ (Tis) and microinvasive cancer (T1mi) were defined. Furthermore, tumors with solid component diameters of 1 cm or less, 2 cm or less, and 3 cm or less were classified as T1a, T1b, and T1c, respectively, and the stage was also subdivided accordingly. The usefulness of the evaluation of extrathoracic metastasis of early-stage lung cancer in the new stage classification is controversial, and there are few reports. This study aimed to determine the incidence of extrathoracic metastasis in patients with clinical stage IA NSCLC, understand the association between extrathoracic metastasis and other clinical features, and evaluate the diagnostic efficiency of imaging screening for preoperative extrathoracic metastasis in patients with early-stage lung cancer. Especially for brain metastases, screening is recommended for patients with stage ≥2 lung cancer or neurological symptoms; it is not recommended for cases with stage ≤Ιin American College of Chest Physicians and National Comprehensive Cancer network guidelines.7,8 Contrastingly, the Japanese Lung Cancer Society guidelines recommend screening for brain metastasis in all patients, and the European Society for Medical Oncology guideline notes that screening for brain metastasis using MRI might be useful for curative therapy.9,10 The frequency of brain metastases in clinical stage I lung cancer was approximately 0.57%–4.49%, 11 and the prevalence of brain metastases in clinical stage IA lung cancer at the time of presentation was approximately 0.5%–0.7%.3,11 In contrast, the frequency of asymptomatic brain metastases from lung cancer has been reported to be 7.5% and brain metastases in 19% of patients with N0 lung cancer. 12 It has also been reported that epidermal growth factor receptor mutations are more likely to cause brain metastases even in cases of T1-2aN0. 13 Therefore, brain metastasis screening should be conducted for patients with stage I lung cancer.
In our study, five of the 510 patients diagnosed with stage IA NSCLC using CT and PET-CT were identified as having preoperative extrathoracic metastases. A total of six sites of extrathoracic metastasis were identified: three were in the brain, two were in bones, and one in the liver. Five primary lesions with extrathoracic metastases were observed in four patients with solid tumors and a C/T ratio of 100%, as well as in one patient with solid dominant tumor and a C/T ratio is 85.2%. From the analysis of the ROC curve, the risk of extrathoracic metastasis of tumors with a solid component diameter of < 22 mm may be low. Stage I solid predominant adenocarcinoma has been reported to be a predictor of extrathoracic metastasis in the early postoperative period.14,15 This suggests that solid-predominant tumors have a high potential for distant metastases and should be screened for distant metastases preoperatively. In contrast, some adenocarcinomas exhibit areas of ground-grass opacity (GGO) on high-resolution CT (HRCT), reflecting a lepidic growth pattern of tumor cells microscopically. 16 Therefore, lung tumors that display areas of focal GGO on HRCT contain components of histological lepidic tumor growth. 17 Defined as in situ lesions, these tumors exhibit a lepidic growth pattern and are less invasive. 18 Patients with GGO-dominant lung adenocarcinoma have been reported to have a good prognosis.19–23 Furthermore, patients with a ground-glass nodule and a primary lesion of ≤2 cm and a solid component diameter of ≤25% have almost no distant metastasis. 24 In such cases, staging using FDG-PET/CT, or contrast-enhanced MRI or CT is not essential. 8
Our study has some limitations. This was a single-center retrospective study, and the number of cases was limited. Therefore, a case analysis at multiple centers will be required in the future. Additionally, our findings suggest that preoperative metastasis screening may be useful for at least stage ΙA3 NSCLC.
Conclusion
We examined preoperative metastasis screening for stage IA NSCLC. Preoperative extrathoracic metastasis screening may be omitted in the cases involving tumors with a solid size of < 22 mm or clinical stage IA 1 or 1A2 NSCLC. Conversely, extrathoracic metastasis screening is beneficial in at least clinical stage IA3 NSCLC.
Footnotes
Acknowledgments
We would like to express our gratitude to all patients who participated in this study.
Declarations
Authors’ contributions
YI performed data analysis and wrote the first draft of the manuscript. All authors contributed equally to this work.
Consent for publication
All authors provided consent to the publication of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
The access and use of patient data were approved by the ethics committee of Kanazawa Medical University. (approval numbers: I160 and I449).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Informed concent was obtained from patients preoperatively and included access to their clinical records.
Author biographies
Yoshihito Iijima M.D., Ph.D. is a General Thoracic and Pulmonary Surgeon in Kanazawa Medical University. His research interests include Oncology of lung cancer, metastatic lung disease. Email:
Masahito Ishikawa M.D. is a General Thoracic and Pulmonary Surgeon in Kanazawa Medical University. His research interests include Infectious lung disease. Email:
Shun Iwai M.D. is a General Thoracic and Pulmonary Surgeon in Kanazawa Medical University. His research interests include Respiratory physiology. Email:
Nozomu Motono M.D., Ph.D. is a General Thoracic and Pulmonary Surgeon in Kanazawa Medical University. His research interests include Oncology, Surgery. Email:
Shigeki Yamagishi M.D., Ph.D. is a General Thoracic and Pulmonary Surgeon in Aidu Chuo Hospital now. His research interests include general thoracic surgery. Email:
Kiyoshi Koizumi M.D., Ph.D. is a General Thoracic and Pulmonary Surgeon in Aidu Chuo Hospital now. His research interests include general thoracic surgery. Email:
Hidetaka Uramoto M.D., Ph.D. is a General Thoracic and Pulmonary Surgeon in Kanazawa Medical University. His research interests include Oncology, Thoracic Surger, Molecular biology of lung cancer. Email:
