Abstract
Lung cancer is among the most common types of malignancies, with small-cell lung cancer (SCLC) being a particularly aggressive subtype. The prognosis for SCLC is typically poor once it recurs and metastasizes after first-line treatment. Although numerous studies have investigated second-line therapies for SCLC, the outcomes have been suboptimal. In this case report, we examined the effectiveness of anlotinib combined with palliative chemoradiotherapy in a patient with second-line treatment of SCLC. The patient achieved a long progression-free survival of 31 months with anlotinib combined with palliative chemoradiotherapy. Anlotinib combined with palliative chemoradiotherapy may offer significant therapeutic benefits as a second-line recurrent or metastatic SCLC.
Keywords
Introduction
Lung cancer remains the leading cause of cancer-related morbidity and mortality worldwide as of 2022, 1 and small-cell lung cancer (SCLC) accounts for approximately 13%–17% of all lung cancers. 2 Owing to the aggressive nature of SCLC, it often leads to distant metastasis early in the disease course; most patients diagnosed at an advanced stage, resulting in a poor prognosis. Limited-stage SCLC is commonly managed with chemoradiotherapy, although surgery may be considered in very early-stage cases. For extensive-stage SCLC (ES-SCLC), the primary treatment includes chemotherapy and immunotherapy, often in combination with radiotherapy in selected cases. Once SCLC recurs and progresses after first-line therapy, options such as chemotherapy or targeted therapy are recommended3–5; however, the prognosis is very poor. Despite ongoing research into new treatments, such as lurbinectedin, camrelizumab plus apatinib, tarlatamab, and antibody-drug conjugates,5–8 effective treatments for recurrent or metastatic SCLC are still lacking.
The efficacy of anlotinib, a multitarget, small-molecule tyrosine kinase inhibitor, in the treatment of SCLC has been extensively explored. Anlotinib targets receptors such as fibroblast growth factor receptor, vascular endothelial growth factor (VEGF) receptor, platelet-derived growth factor receptor, and c-kit, inhibiting angiogenesis and tumor growth. 9 Anlotinib has demonstrated survival benefits as third- or later-line treatments for SCLC 10 and has also shown survival benefits in combination therapy as both the first-line and later-line treatments of SCLC.10–12 However, the specific lines of therapy for which anlotinib will be most beneficial and its best combination strategies have not been thoroughly investigated. This report presents a case of ES-SCLC that achieved long-term progression-free survival (PFS) upon treatment with anlotinib combined with palliative chemoradiotherapy as second-line treatment.
Case presentation
A 58-year-old male patient was admitted to People's Hospital of Leshan with a 1-month history of cough in November 2020. He had no family history of cancer nor any history of smoking. A routine physical examination did not reveal any abnormalities. Laboratory examinations indicated the following findings: carcino-embryonic antigen, 10.09 ng/mL; neuron-specific enolase, 74.80 ng/mL; cytokeratin 19 fragment, 4.34 ng/mL; and gastrin-releasing peptide precursor, 470.00 pg/mL. His squamous cell carcinoma antigen level was within normal limits. Cervicothoracic computed tomography (CT) revealed a mass in the right lung hilus, along with enlarged lymph nodes at the root of the right side of the neck and mediastinum. Head magnetic resonance imaging (MRI) showed abnormal signals in the right frontal skull with unclear features, although no intracranial abnormalities were evident. Abdominal CT showed no metastasis, and whole-body bone imaging revealed no significant abnormalities. The patient underwent a bronchoscopic biopsy of the right middle lobe. Histopathological and immunohistochemical analyses confirmed small-cell carcinoma (Figure 1). Immunohistochemical staining results were as follows: PCK (+; spot), CD56(+), CgA(+), Syna(±), TTF-1(+), P63(-), CK8/18(+; spot), and Ki67 (+; 80%). Then the patient's specialized pathological consultation for difficult cases at the West China Hospital, Sichuan University still indicated SCLC. He was diagnosed with ES-SCLC(T3N3M1).

Hematoxylin and eosin stain × 400.
In December 2020, treatment commenced with etoposide (100 mg/m2, intravenously, days 1–3) and cisplatin (25 mg/m2, intravenously, days 1–3) administered every 3 weeks for six cycles, with signed consent to treatment obtained from the patient. However, the patient declined immunotherapy owing to its cost. After two cycles of chemotherapy, the tumor lesions and lymph nodes shrank and could be contained within one radiation field. Concurrent chemoradiotherapy began in January 2021, after two cycles of chemotherapy. The radiotherapy regimen included a clinical target volume (CTV) of 60 Gy in 30 fractions for the right lung tumors, mediastinum, and right neck root. The patient declined prophylactic cranial irradiation. CT showed no residual tumor after the first-line treatment, and the patient remained stable with regular follow-ups.
In December 2021, a CT scan revealed brain metastasis (Figure 2(a)) and right adrenal metastasis (Figure 2(b)), with no recurrence in the right lung. Since the patient did not receive immunotherapy in the first-line treatment, we recommended palliative radiotherapy and chemotherapy combined with immunotherapy as the second-line treatment. The patient declined immunotherapy again owing to its cost. We estimated that it would be difficult to achieve complete control of the tumor after the patient undergoes palliative radiotherapy and chemotherapy. Thus, we suggested him to choose anlotinib combined with palliative radiotherapy and chemotherapy, as well as maintenance treatment. Although anlotinib had been recommended by the Chinese Society of Clinical Oncology guidelines as a third-line treatment for SCLC, its use was off-label for second-line treatment of SCLC. The patient consented to anlotinib (12 mg orally, days 1–14) every 3 weeks and whole brain radiotherapy, with a CTV of 30 Gy in 10 fractions. The patient did not experience any adverse reactions related to radiotherapy. In December 2021, the patient commenced treatment with irinotecan (65 mg/m2, intravenously, days 1 and 8) and carboplatin (area under the curve, 5; intravenous, day 1) every 3 weeks for two cycles. He again declined immunotherapy owing to its cost after that. MRI showed significant shrinkage of the intracranial lesions (Figure 2(c)), with a partial response (PR), according to the Response Evaluation Criteria in Solid Tumors 1.1 (RECIST 1.1). CT also showed mild shrinkage of the right adrenal lesion (Figure 2(d)), which was evaluated as a stable disease (SD) according to RECIST 1.1. The patient completed three additional cycles of the initial chemotherapy regimen and continued targeted therapy with anlotinib.
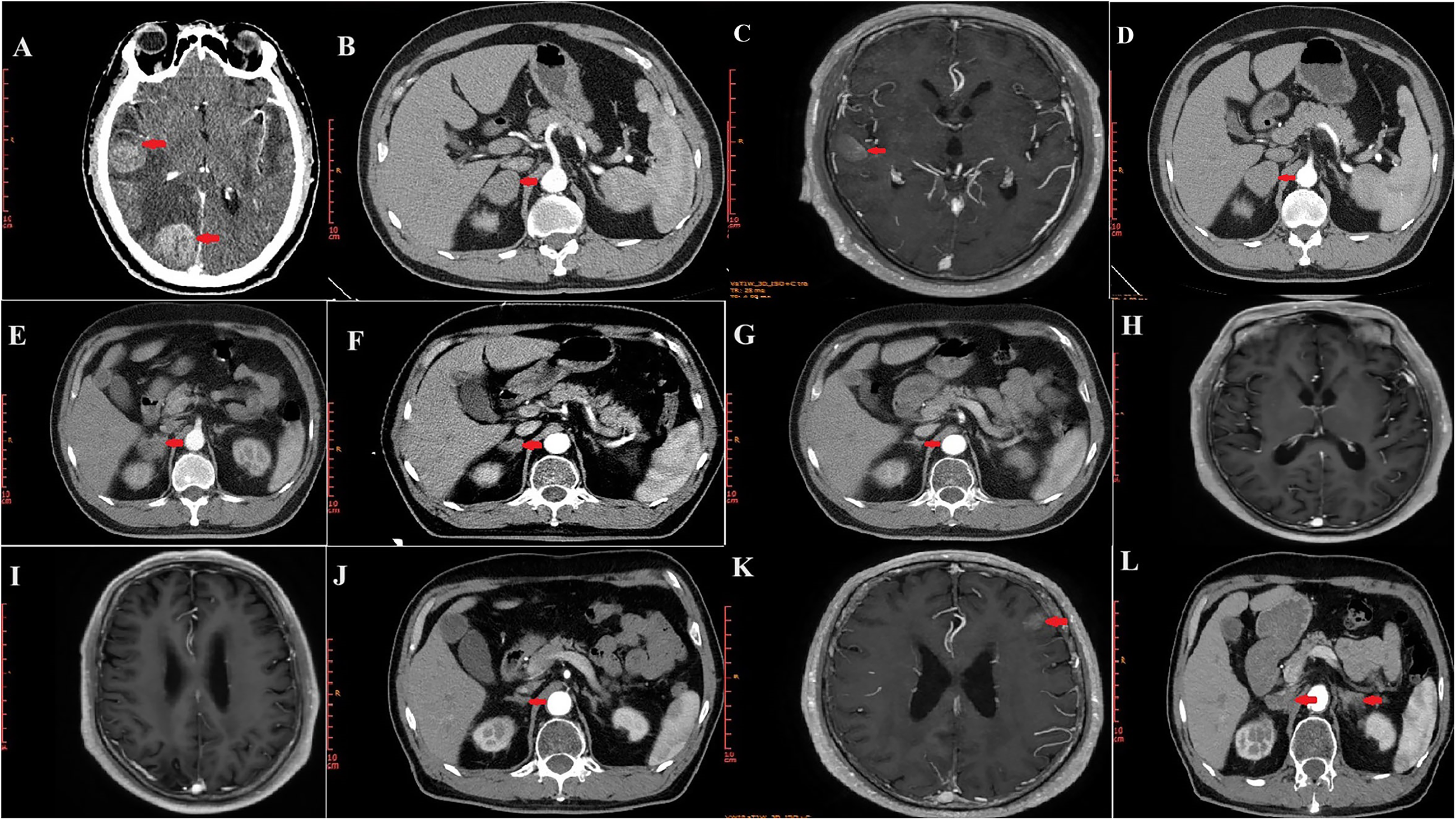
Images of intracranial and adrenal lesions during second-line treatment. (a, b) Images before second-line treatment. (c, d) Images after two cycles of chemotherapy. (e) Image after five cycles of chemotherapy. (f) Image of right adrenal lesion after radiotherapy. (g–j) Images obtained during anlotinib maintenance therapy. (k, l) Images of second recurrence and metastasis.
In May 2022, CT confirmed SD in the right adrenal lesion (Figure 2(
In July 2022, CT revealed PR in the right adrenal lesion (Figure 2(f)) according to RECIST 1.1. By October 2022, CT showed that the right adrenal lesion had continued to shrink (Figure 2(g)). In June 2023, MRI showed further shrinkage of the intracranial lesions (Figure 2(h) and (i)), with a PR according to RECIST 1.1, while the right adrenal lesion remained SD (Figure 2(j)). No recurrence or new lesions were observed elsewhere in the body. However, by July 2024, MRI revealed new metastatic lesions in the left cerebellar hemisphere, left temporal lobe, and left frontal lobe (Figure 2(k)). CT also showed left adrenal metastasis and right adrenal enlargement (Figure 2(l)), with the overall evaluation being progressive disease according to RECIST 1.1.
After disease progression, the patient refused to continue chemotherapy due to concerns about the treatment's adverse reactions. He also refused other treatments, such as immunotherapy, lurbinectedin, and Tarlatamab, due to the cost. The patient requested to continue treatment with anlotinib. The disease remained stable following second-line treatment with anlotinib and palliative chemoradiotherapy for 31 months. No adverse drug reactions were reported during anlotinib treatment. The reporting of this study conforms to CARE guidelines. 13 Written informed consent was obtained from the patient for publication of this article and any accompanying images. We had de-identified all patient details.
Discussion
SCLC is highly malignant, and the prognosis is particularly poor following recurrence and metastasis. While SCLC often occurs in smokers, the patient in the present case denied any history of smoking, although we do not know the status of environmental pollution around the patient. Routine genomic testing is not recommended in SCLC; however, the possibility of oncogenic driver mutations in never-smokers is higher than that in typical SCLC populations. In the current study, the patient refused genomic testing due to cost. Pathological examinations from two medical institutions indicated SCLC, avoiding any potential misdiagnosis. Patients with recurrent SCLC typically have a median overall survival (OS) of only 4–5 months after further chemotherapy.3,4 Although new treatments for recurrent and metastatic SCLC are under investigation, outcomes remain suboptimal.
Lurbinectedin, a selective inhibitor of oncogenic transcription, has shown effectiveness as a second-line treatment for SCLC, with an objective response rate (ORR) of 35.2%, PFS of 3.5 months, and OS of 9.3 months. 5
Antibody-drug conjugates are novel biopharmaceutical compounds that combine monoclonal antibodies with cytotoxic drugs. They represent a promising therapeutic approach for SCLC. 8 Tarlatamab has shown promising results in previously treated SCLC patients in the DeLLphi-300 and DeLLphi-301 trials. It is a bispecific T-cell engager. The overall response rate was 42.9% in SCLC patients based on real-world data. 14 While its marketing application was accepted by China's National Medical Products Administration in July 2025 under priority review, it has not yet been approved for commercial use in the country.
Immunotherapy is also widely used, with the KEYNOTE-028/158 clinical trial demonstrating that pembrolizumab achieves an ORR of 19.3%, PFS of 2.0 months, and OS of 7.7 months in patients treated with three or more lines of SCLC therapy. 15 However, single-agent immunotherapy in later lines has shown limited benefit. Notably, chemotherapy combined with immunotherapy has shown good antitumor activity and is now the standard first-line treatment in ES-SCLC. In IMpower133, the median OS and PFS were longer with Atezolizumab plus chemotherapy compared with chemotherapy alone (12.3 months vs 10.3 months, p = 0.007, and 5.2 months vs 4.3 months, p = 0.02, respectively). 16 In the present case, the patient declined immunotherapy owing to its cost. 17 As an alternative, a newer therapeutic model combining anti-angiogenic therapy with immunotherapy has shown synergistic antitumor effects. 18 The PASSION clinical trial demonstrated that apatinib combined with camrelizumab as second-line treatment for ES-SCLC, achieved an ORR of 34.0%, PFS of 3.6 months, and OS of 8.4 months, 6 highlighting the potential role of anti-angiogenic therapy in SCLC.
Anlotinib, an antitumor angiogenic drug targeting multiple pathways, has shown efficacy, particularly in third-line or later treatments for SCLC, and has been approved for use in SCLC in China. It has not been approved by the FDA and European Medicines Agency. In a randomized, double-blind, placebo-controlled phase 2 study at 11 institutions in China, a total of 82 and 38 patients were randomly assigned to receive anlotinib and placebo, the median PFS was significantly longer in the anlotinib group (n = 82) compared with the placebo group (n = 38) (4.1 months [95% confidence interval (CI), 2.8–4.2] vs 0.7 months [95% CI, 0.7–0.8]; hazard ratio [HR] 0.19 [95% CI, 0.12–0.32], p < 0.0001), and the OS was also significantly longer with anlotinib (7.3 months [95% CI, 6.1–10.3] vs 4.9 months [95% CI, 2.7–6.0]; HR 0.53 [95% CI, 0.34–0.81], p = 0.0029). The incidence of any adverse events was 100% in the anlotinib group and 92.3% in the placebo group (p = 0.033), and the incidence of grade 3 or worse adverse events was 51.9% in the anlotinib group and 43.6% in the placebo group (p = 0.440). Treatment-related adverse events (TRAEs) were 87.7% with anlotinib and 74.4% with placebo (p = 0.114). The grade 3 or worse TRAEs were 35.8% with anlotinib and 15.4% with placebo (p = 0.031). The most common grade 3 or worse TRAEs were hypertension (13.6 vs 2.6%), γ-glutamyltransferase elevation (4.9 vs 7.7%), hand and foot skin reaction (4.9 vs 0%), lipase elevation (3.7 vs 0%), hypophosphatemia (3.7 vs 0%), hyponatremia (3.7 vs 0%), and triglyceride increase (3.7 vs 0%). 10 In a single-arm, phase II study, anlotinib plus etoposide and cisplatin/carboplatin as first-line therapy for ES-SCLC achieved a median PFS of 10.3 months (95% CI: 6.0–14.5) and median OS of 17.1 months (95% CI: 11.1–19.3); it also showed acceptable tolerability. 12
Immunochemotherapy is now standard first-line treatment in ES-SCLC, and combined anti-angiogenesis may improve efficacy. In a multicenter, double-blind, randomized, placebo-controlled phase 3 trial, the median OS was significantly longer in the benmelstobart and anlotinib plus etoposide/carboplatin (EC) group compared with the EC alone group (19.3 months [95% CI 14.2 to not estimable] versus 11.9 months [95% CI 10.7–13.4]; HR, 0.61 [95% CI 0.47–0.79]; p = 0.0002). The anlotinib plus EC group had no significant OS benefit compared with the EC alone group (13.3 months [95% CI 11.1–15.1] versus 11.9 months [95% CI 10.7–13.4]; HR, 0.86 [95% CI 0.67–1.10]; p = 0.1723). Compared with the EC alone group, median PFS was significantly longer in both the benmelstobart and anlotinib plus EC group (6.9 months [95% CI 6.2–8.3] versus 4.2 months [95% CI 4.17–4.24]; HR 0.32 [95% CI 0.26–0.41]; p < 0.0001) and in the anlotinib plus EC group (5.6 months [95% CI 5.6–6.8] versus 4.2 months [95% CI 4.17–4.24]; HR 0.44 [95% CI 0.36–0.55]; p < 0.0001). 11 Despite these advancements, the overall prognosis for SCLC remains poor, underscoring the need for additional treatment options to improve patient survival.
In this case, the patient developed intracranial and right adrenal metastases after first-line treatment. He declined immunotherapy owing to its cost but consented to anlotinib combined with palliative chemoradiotherapy as a second-line treatment strategy. The right adrenal metastases were well controlled, and the intracranial lesions reduced significantly. Preclinical studies have not directly demonstrated that anlotinib crosses the blood–brain–barrier. However, in the process of tumor progression, the integrity of the blood–brain–barrier is destroyed, and the blood-tumor barrier is formed. The blood-tumor barrier has heterogeneous permeability and perfusion with small and large molecules. Notably, VEGF regulates blood–brain–barrier permeability, 19 and anlotinib regulates VEGF function by binding to VEGF receptor. 9 In addition, the survival benefit of anti-angiogenic therapy in patients with glioblastoma has been established.20,21 Many factors affect the ability of drugs to cross the blood–brain–barrier, including lipid solubility, molecular weight, tertiary structure, charge, and the degree of protein binding. 22 The ability of anlotinib to control intracranial tumor growth across the blood–brain–barrier may be because it is a hydrophobic small molecule with a molecular weight of 480.36 Da under neutral conditions.
The benefits of anlotinib in patients with SCLC and non-SCLC having brain metastases have been further confirmed in recent studies.23,24 Radiotherapy not only kills tumor cells but also damages the blood–brain–barrier 19 ; consequently, it increases the permeability of anlotinib to the brain tissue. Radiotherapy combined with anlotinib effectively controls intracranial lesions. Although anlotinib has shown benefits in both the first-line and later-line treatments of SCLC, the optimal treatment approach still requires further exploration. The differences in efficacy and safety between anlotinib and other drugs, such as lurbinectedin and Tarlatamab, still require further exploration through more research.
Conclusion
We present a case of ES-SCLC that achieved long-term PFS of 31 months upon treatment with anlotinib combined with palliative chemoradiotherapy as second-line treatment, thereby opening up more options for follow-up treatment. The recurrence interval is closely related to patient survival, and this outcome suggests that anlotinib combined with palliative chemoradiotherapy may offer therapeutic value as a second-line treatment for SCLC. However, further research is needed to determine whether this treatment strategy can significantly improve survival in patients with SCLC. Studies are also required to explore the mechanism of action of anlotinib combined with palliative chemoradiotherapy in controlling intracranial lesions.
Footnotes
Acknowledgments
The authors express their gratitude to the patient for consenting to share this information. The authors have read the CARE Checklist(2016), and the manuscript was prepared and revised according to the CARE Checklist(2016).
Ethical considerations
The study protocol was approved by the Ethics Committee of The People's Hospital of Leshan, Leshan, China (LYLL[2024]KY 140).
Consent to participate
We obtained signed consent to treatment from the patient.
Consent for publication
Written informed consent was obtained from the patient for publication of this article and any accompanying images.
Author contributions
NZ wrote the paper and collected the case data; DY prepared the photos; XZ collected the case data; ZL guided the article writing and proofread the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by Beijing Health Alliance Charitable Foundation.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data are available from the authors upon reasonable request.
