Abstract
While Raman spectroscopy itself stands on the principle of inelastic scattering of light, surface-enhanced Raman spectroscopy (SERS) amplifies those normally rather weak Raman signals by way of interactions between molecules and nanostructured metal surfaces. The technique has rapidly evolved into a very powerful analytical tool with enormous potential in combating cancer and COVID-19. SERS is a useful tool for diagnostics and treatment monitoring because of its remarkable sensitivity and ability to detect low-abundance molecules; nevertheless, standardizing techniques, guaranteeing reproducibility across several platforms, and overcoming problems related to signal enhancement and sensitivity under different experimental conditions remain challenges. SERS is also being explored about the COVID-19 pandemic, where its high sensitivity and specificity hold a promise in diagnostics, treatment monitoring, and even environmental tracing of the virus. When it comes to treatment, SERS-based theragnostic applications offer a two-pronged approach by combining therapeutic interventions with diagnostic capabilities that would make different therapies more accurate and effective. Approaches to SERS-guided drug delivery systems are discussed that would allow the drug to reach exactly where the antiviral agent is wanted, hence reducing side effects and enhancing treatment outcomes. Other approaches examined, including nanoparticle-based SERS for targeted therapy and the design of SERS tags, allow therapy and develop new ways of treatment against this virus. Finally, potential future developments of SERS technology and its wider applications in cancer and virology are discussed, with a specific view on the impact SERS might have on how infectious diseases are treated. In addition to discussing its present and potential uses, this narrative review emphasizes the critical role that SERS plays in developing and tracking cancer and COVID-19 treatments.
Introduction
Raman spectroscopy is a powerful analytical technique based on the so-called Raman effect named after the pioneer scientist Sir Chandrasekhara Venkata Raman on inelastic scattering of light by a molecule. While most of the photons are elastically scattered, there is no change in the energy of the scattered photons as compared to the incident photons (Rayleigh scattering); only a small portion of light gets inelastically scattered: it contains photons whose energy has changed from incident light. This discrepancy in energy amounts resonates with the vibratory modes of the said molecule forming a signature of sorts that assists in recognizing and learning more about the molecular composition. 1 Surface-enhanced Raman spectroscopy (SERS) is a modernized version of Raman spectroscopy to improve the weak Raman signals of molecules attached to selectively design metal surfaces or nanoparticles. SERS although well based on very weak Raman signal, can amplify the signals to many folds. The Raman effect does indeed represent a kind of molecular fingerprint; the problem is that the obtained signal is normally too weak to detect low concentrations of molecules. SERS has taken advantage of the interaction of molecules with nanostructured metal surfaces, usually of gold, silver, or copper metals, owing to their strong plasmonic features in the visible to near-infrared range. When the molecule has been adsorbed onto a roughened metal surface or close to metal nanoparticles, this happens with an enhancement of Raman signal by factors of up to 106 to 1010 and more in favorable cases.2,3
The enhancement of the Raman signal in SERS is primarily driven by two mechanisms: electromagnetic and chemical enhancement. The more dominant electromagnetic enhancement is due to localized surface plasmon resonance, where conduction electrons on the surface of metal nanoparticles collectively oscillate in response to incident light. 4 This oscillation creates intense, localized electromagnetic fields, especially at “hotspots” such as gaps between adjacent nanoparticles or sharp features on their surfaces. 5 Molecules in these regions experience a significantly amplified electromagnetic field, which greatly increases the intensity of their Raman scattering signal. The Raman signal intensity in SERS is proportional to the fourth power of the field enhancement, allowing for the detection of even single molecules under ideal conditions. In addition to this, a smaller chemical enhancement occurs due to charge transfer between the metal surface and the adsorbed molecules. 6 This interaction modifies the electronic properties of the molecules, increasing their polarizability and further boosting the Raman signal. The effectiveness of the SERS effect depends strongly on the nature of the metal surface or nanoparticles (e.g. their size, shape, and distribution), which needs to be stringently controlled in substrate fabrication for reproducibility and high signal enhancement. The choice of a metal surface or nanoparticles and their preparation are the keys to achieving a strong SERS effect. To optimize the localized surface plasmon resonance effect, specific shapes of nanoparticles or roughened surfaces are used, such as spheres, rods, and stars. Nanostructures should be dimensioned in a manner that would be able to match the wavelength of the incident light, usually between 10 and 100 nm, to fine-tune the resonance conditions. The metals used most commonly for SERS are gold and silver because their plasmon resonances in the visible to near-infrared fall within the spectral range from which Raman spectroscopy is often performed. Copper may also be used though less frequently due to its tendency to oxidize. It could be functionalized with particular chemical groups or ligands capable of binding the target molecules to the analyte of interest on the metal surface. 7
SERS enables the detection of extremely low concentrations of molecules, thereby allowing breakthroughs in, though not limited to, early disease diagnosis, identifications of pollutants, and characterizations of complex materials. So far, the fabrication of substrates by accurately controlling nanoparticle size, shape, and distribution remains a very challenging task, and this often results in variations in SERS performance. Besides, the interaction between analyte molecules and the metal surface usually results in complex spectra; interpretation of these spectra can be done with yet another layer of complexity. These enhanced signals can, at times, be from several contributing molecular species or due to subtle changes in the vibrational modes of molecules, making analyses difficult and needing sophisticated data processing techniques to deconvolute the spectra. However, even with these challenges, continued advances are being reported in nanofabrication techniques that provide increasingly uniform and reproducible SERS substrates. In addition, the development of portable SERS devices enables sensitive on-site analysis in real time, which broadens the practical applications of SERS in various industries. These are not only technologies enhancing the reliability and versatility of SERS but also forcefully thrust SERS as an indispensable tool in scientific research and practical analysis. With the further development of these technologies, the possible influence of SERS may be extended from medical diagnostics to environmental studies by offering new insights into the detection, analysis, and understanding at a molecular scale.
Over the past decades, cancer remains as one of the most prevalent health challenges worldwide. It demands innovative strategies in both diagnosis and treatment. Traditional cancer therapies, including surgery, chemotherapy, and radiation, have various side effects and limited specificity. Recent approaches in cancer therapy have focused on precision medicine approaches like targeted therapies, immunotherapy, and so on. These personalized treatments aim to target specific molecular pathways or immune responses that drive cancer growth, leading to more effective and less toxic therapies. Also, improvements in technology like next-generation sequencing and liquid biopsies have made it easier to find genetic mutations and biomarkers that can help doctors decide what treatment to use. As a result, the landscape of cancer therapy is rapidly evolving, offering new hope for patients with previously untreatable cancers. The personalized drug delivery systems that aim to increase treatment efficacy thereby minimizing the side effects toward healthy cells. However, with these advancements, there is a growing need for precise, real-time monitoring tools that can track therapeutic progress and detect early signs of treatment response or resistance. The development of new imaging technologies capable of seeing tumor development and responses to treatment at a cellular level presents one exciting way to meet this demand. Researchers can keep an eye on these things by using advanced imaging technologies, such as positron emission tomography scans and magnetic resonance imaging, to track the size, metabolism, and blood flow of tumors in real time. Such monitoring enables the early identification of therapeutic response or resistance, permitting clinicians to modify therapy regimens as needed. Also, these imaging methods might make it easier to find any side effects related to treatment, which would allow for proactive management and the avoidance of consequences. All things considered, including cutting-edge imaging technologies into cancer treatment plans will transform our monitoring and management of cancer treatment, enhancing patient outcomes. In the context of cancer, SERS has shown potential to monitor drug delivery, detect circulating tumor cells, and identify treatment response biomarkers at the molecular level, offering real-time insights into a patient's therapeutic journey. The degree of accuracy and detail in monitoring cancer treatment may promote the customization of therapeutic strategies and enhance patient care. Through the application of SERS technology, healthcare providers can make informed choices about therapy modifications, early interventions, and supportive care strategies. Being able to understand how well a therapy is working in real time can help find ways and help doctors choose other medicines that might work better, which can improve patient outcomes and quality of life. Because SERS imaging is noninvasive, it can be used over and over again. This enables doctors get a full picture of how the patient is responding to therapy and quickly make changes to their care plan. Through its ability to capture detailed molecular information noninvasively, SERS provides specific fingerprint patterns to clinicians and researchers for evaluating treatment efficacy. As cancer therapies continue to evolve toward more personalized approaches, SERS holds tremendous potential to bridge the gap between diagnosis and real-time treatment monitoring, making it a valuable tool in the future landscape of oncology. Given its exciting contribution to oncology, SERS also has great promise in the field of virology, providing cutting-edge diagnosis and treatment options for viral infections including those brought on by the great variety of viruses that present continuous public health concerns.
Viruses are the most abundant life forms on earth, composed of an enormous and diverse library of nucleic acids infective for each form of life. They have also been a major contributor to public health concerns, responsible for about one-third of the deaths caused by infectious diseases. Among these, Covid Viruses (CoVs) constitute an important group since alpha and beta genera are most commonly found responsible for respiratory infections. At the beginning of the 21st century, several zoonotic viruses capable of infecting humans and causing serious illness emerged. Such viruses include Middle East respiratory syndrome (MERS-CoV) and severe acute respiratory syndrome coronavirus 2 (SARS-CoV), both of bat origin. The most recent coronavirus outbreak in 2019, that of 2019-nCoV in Wuhan, China, caused by SARS-CoV-2, was declared by the World Health Organization (WHO) to be a pandemic. 8 SARS-CoV-2, purportedly of bat origin via an intermediate host, became the seventh coronavirus known to cause human disease. The outbreak itself in its initial stages had infected more than 10 million people and caused over 500,000 deaths worldwide due to this virus. Per WHO reports to date, there are over 875 million infections reported worldwide. With the start of 2023, a new critical phase was attained when the Omicron subvariant XBB.1.5, considered the most transmissible so far, with a set of mutations that improve binding to the angiotensin-converting enzyme 2 (ACE2) receptor and further promote immune evasion, received worldwide circulation. 9
The COVID-19 pandemic put unprecedented pressure on healthcare systems worldwide, and a need for novel diagnostic and therapeutic approaches became very timely. SERS is developing to face such challenges. It advances Raman scattering signals of molecules adsorbed onto nanostructured surfaces, thus allowing the very sensitive and specific detection of low-abundance analytes. This capability makes SERS a double-edged sword in both COVID-19 treatment and its monitoring. In this narrative review, we shall look into the various ways in which SERS finds applications in the context of COVID-19, focusing on aspects related to treatment and disease monitoring. About the aspect of treatment, there are SERS-based theragnostic applications that could represent a unique approach toward integrating therapeutic and diagnostic functions. This function duality enables the tracking of therapeutic efficacy and distribution in real time, thus allowing treatment modalities that are more appropriate and effective. Moreover, targeted therapy with SERS based on nanoparticles and fabrication of SERS tags for improved therapy is an important development in the fight against COVID-19. The monitoring of the progress and effect of COVID-19 is just as important in managing the disease. SERS technology has shown its usefulness in the detection of COVID-19 biomarkers, monitoring immune responses, and viral presence tracing in the environment. Of special value for the tracking of disease progression and transmission routes, with further use in public health interventions and containment strategies, is the real-time high-resolution capability of SERS. Despite the tremendous potential of SERS, many factors are working against the widespread translation of SERS into COVID-19 management. While highly powerful, the full exploitation of SERS in various applications requires solving the technical limitations, logistics constraints, and regulatory barriers. Further research and innovations are vital in these aspects to surmount the obstacles and enhance the application of SERS in virology. Finally, the challenges and future perspectives of SERS technology in the context of COVID-19 will be examined, highlighting its potential to transform infectious disease management. 10 Overall, this narrative review aims to deeply look into how SERS can be used to diagnose COVID-19 and cancer, focusing on its diagnostic value, treatment options, cutting-edge technology, and potential future. It examines the function of SERS in identifying biomarkers, monitoring disease advancement, and enhancing therapy accuracy. It also examines the prospective advancement of SERS in oncological research and the therapy of infectious diseases.
SERS in cancer and COVID-19 treatment
Nanotechnology aims to study drug interactions for potential treatments, particularly using SERS to investigate antiviral drug–protein connections. SERS-active nanoparticles in drug delivery show promise for targeted therapy and monitoring drug release in both cancer and COVID-19. Furthermore, the use of nanotechnology in drug delivery has revolutionized the way medications are administered, allowing for more precise targeting of diseased cells while minimizing side effects on healthy tissues. By encapsulating drugs in nanocarriers, researchers have been able to enhance drug stability, improve bioavailability, and prolong drug circulation in the body. These advancements have led to more effective treatments with lower doses of medication, ultimately improving patient outcomes and quality of life. These technologies improve therapeutic approaches for several diseases, including cancers, not only for viral infections. These applications, not exclusive to COVID-19, may advance therapeutic strategies for viral infections. Ongoing research examines combining SERS-active nanoparticles with therapeutic agents to enhance treatment outcomes. The reliability of SERS in cancer therapy allows real-time assessment of drug effectiveness, offering insights into the interplay between therapeutic agents and tumor cells. In the context of COVID-19, SERS-active nanoparticles offer possibilities for improved drug targeting, hence enhancing the delivery and effectiveness of antiviral procedures. Current research focuses on the integration of SERS-active nanoparticles with therapeutic medications for improving treatment efficacy, offering a potential solution for both communicable diseases, such as COVID-19, and noncommunicable diseases, such as cancer. Furthermore, SERS is being explored for photothermal treatment, a method in which nanoparticles produce heat upon light exposure to eliminate infected cells. This method demonstrates the adaptability of SERS in therapeutic applications, broadening its use beyond cancer to respiratory diseases such as COVID-19, where precise and effective treatments are essential for enhancing patient outcomes. Despite the potential, further investigation is needed for the direct application of this approach to communicative and noncommunicative disease therapy. Additionally, SERS is explored for photothermal therapy, showcasing its versatility in potential interventions for various diseases, including respiratory infections.11,12
Theragnostic applications of SERS
Theragnostic integrates diagnostic and therapeutic functions using SERS effectively. In diagnostics, SERS enables highly sensitive molecular detection by binding strongly to biomolecules, detecting disease markers at low concentrations in complex samples. Live imaging with SERS provides real-time insights into molecular processes, aiding in tracking drug distribution and understanding treatment responses. SERS-based cellular imaging visualizes complex cellular processes for understanding disease mechanisms and assessing treatment efficacy. In therapeutics, SERS plays a crucial role in drug delivery and real-time monitoring of drug response. Engineered SERS-active nanoparticles act as precise carriers for therapeutic agents, enhancing effects while minimizing off-target impact. SERS substrates also enable photothermal therapy, generating heat for localized damage to specific cells or tissues, showing potential for treating various diseases, including cancers.13,14 While SERS substrates may have potential for photothermal therapy, there are still many challenges to overcome before they can be effectively utilized in clinical settings for treating diseases like cancer. Additionally, the long-term effects and safety of using SERS substrates for this purpose are not yet fully understood.
SERS enhances gene therapy and gene delivery platforms, employing customized substrates for precise nucleic acid transportation, allowing targeted gene delivery and modulation for therapeutic purposes.15,16 It enables real-time monitoring of molecular changes during treatment, facilitating dynamic tracking and adjustment of treatment plans based on patient responses, promoting personalized treatment processes. A recent theragnostic nanosystem, utilizing gold nanoparticles (AuNPs), was proposed for precise cancer diagnosis and treatment. This system employs micro ribonucleic acid (miRNA) modified with AuNPs along specially designed Y-motifs (AuNP-Ys) and double-stranded DNA (dsDNA) modified AuNPs called AuNP-Ds for sequential SERS imaging of intracellular MiRNA-106a (miR-106a) using DNAzyme for cancer cells. This nanosystem combines SERS imaging and DNAzyme-based therapy, improving specificity, sensitivity, and efficacy, showing promise for accurate cancer diagnosis and treatment. 17 SERS-based theragnostic have applications in various biomedical fields, such as personalized cancer diagnostics and targeted therapy in oncology, rapid detection methods for infectious diseases, and insights into molecular mechanisms in neurology. Integrating SERS into nanomedicine enhances impact, with SERS-active nanoparticles serving as unified theragnostic agents for imaging, diagnostics, and therapy.18,19
SERS-guided drug delivery
SERS-guided drug delivery combines molecular sensing with precise drug targeting. SERS-active nanoparticles act as carriers for therapeutic agents, allowing dynamic monitoring and controlled drug release at specific body locations. For instance, Au and AgNPs with Raman-active molecules and drugs can be precisely designed to accumulate in target tissues, forming a theragnostic platform. 20 In cancer therapy, this approach minimizes off-target side effects by ensuring drug delivery specifically to cancer cells. A recent investigation introduced a targeted drug-delivery system (small molecular targeted drug-delivery conjugate, SMTDDC) using cyclic arginylglycylaspartic acid and cathepsin B-specific peptides on an octaguanidine sorbitol scaffold. SMTDDC disrupted glioblastoma cell microtubule networks through controlled paclitaxel (PTX) release, monitored by Cath B, high-performance liquid chromatography, and SERS. Time-dependent SERS analysis demonstrated rapid and accurate PTX release, aligning with cytotoxicity, apoptotic events, and microtubule alterations in U-87 MG cells. This study presents a promising SERS-guided peptidomimetic sorbitol transporter for enhanced PTX delivery in glioblastoma treatment. 21 The sorbitol transporter exhibited significant selectivity for tumor cells, facilitating the targeted delivery of PTX directly to malignant tissue while reducing off-target effects. In preclinical experiments, the SERS-guided peptidomimetic sorbitol transporter shows substantial enhancements in PTX accumulation in glioblastoma tumors, resulting in improved treatment efficacy and less systemic toxicity. The results indicate that this innovative medication delivery method may transform the treatment of glioblastoma and other challenging malignancies. In a different investigation, a melanoma-targeted theragnostic nanoenvelope was created, consisting of a bovine serum albumin-stabilized gold nanocluster on a gold nanorod. The designed nanosystem provides a unique platform for SERS-based imaging and therapy in cancer treatment, so enabling both improved detection of melanoma cells and precise drug delivery for targeted therapeutic interventions. This all-in-one theragnostic system integrates photothermal therapy, photodynamic therapy, and chemotherapy, along with a SERS detection technique. 22
Enhanced Raman signals accurately track drug distribution, enabling precise monitoring of drug release and therapeutic response. This has the potential to improve chemotherapy efficacy with reduced toxicity. In infectious diseases, SERS-guided drug delivery by guiding nanoparticles loaded with antibiotics to infection sites is used. SERS signals monitor drug release kinetics, enhancing precision and minimizing side effect.23–25 In neurology, SERS-active nanoparticles with blood–brain barrier-crossing ligands are investigated for delivering therapeutic agents to specific brain regions. Real-time SERS signal monitoring proves valuable for optimizing drug delivery in neurological disorders.26,27 Integrating SERS into drug delivery involves expertise in nanotechnology, chemistry, and biomedical engineering. Ongoing research focuses on refining the design and functionality of SERS-active nanoparticles for improved drug delivery. The versatile nature of SERS-guided drug delivery holds significant potential for advancing therapeutics, offering a targeted and real-time monitoring approach aligned with precision medicine principles.
Monitoring therapeutic efficacy using SERS
SERS is a highly sensitive method for monitoring therapeutic effectiveness, providing real-time insights into molecular changes from external interventions. Its ability to detect molecules at low concentrations makes it distinctive for assessing treatment efficacy. In drug therapies, it observes pharmacokinetics and pharmacodynamics by incorporating Raman-active molecules into drugs or using SERS-active nanoparticles as carriers. This enables dynamic evaluation of drug distribution, metabolism, and clearance in vivo. SERS is crucial for evaluating the impact on the molecular characteristics of target tissues, like analyzing Raman spectra changes in cancer cells postchemotherapy or radiation therapy. These changes provide information on treatment responses and potential resistance mechanisms. SERS's real-time nature allows continuous monitoring, facilitating timely adjustments to therapeutic regimens based on individual patient responses.28,29 In a recent study, SERS nanoprobes and machine learning (ML) were employed to image early effects of immune checkpoint blockade in tumor-bearing mice. SERS multiplexed images, spectrally resolved, were segmented into superpixels based on unmixed signals. ML models trained on these superpixels successfully, and classified mice into different groups, identify tumor regions with diverse treatment responses. 30 SERS plays a crucial role in monitoring diverse therapeutic modalities, encompassing drug therapies, photothermal therapy, and gene therapy.31–34 In photothermal therapy, SERS-active nanoparticles that generate heat provide insights into treatment efficacy by tracking temperature-induced molecular changes in targeted tissues (Figure 1). In a recent study, a comprehensive method using SERS nanoprobes and ML was employed to image the early effects of immune checkpoint blockade in tumor-bearing mice. Antibody-functionalized SERS nanoprobes simultaneously visualized immunotherapy-related targets. Multiplexed images, spectrally resolved, were segmented into superpixels based on unmixed signals. ML models trained on these superpixels successfully classified mice into different groups, identifying tumor regions with diverse treatment responses. 35 After looking more closely at the data, we found important patterns in the locations of certain molecular markers in the tumor microenvironment. These findings provide significant insights into the molecular mechanisms behind the varied treatment responses noted in the mice. The incorporation of multiomics data provided a more profound understanding of the molecular pathways implicated in the development of cancer and therapeutic response. In gene therapy, SERS can track the delivery and expression of therapeutic genes using nucleic acid-functionalized substrates. It also assesses treatment-induced changes in molecular disease profiles, aiding in the identification of potential biomarkers for therapeutic response. This information is crucial for personalized treatment development and optimization. SERS, capable of providing both qualitative and quantitative data, enhances its utility in monitoring therapeutic efficacy across medical interventions. The ongoing translation of SERS for clinical monitoring is advancing in applications such as oncology, infectious diseases, and neurology. Ongoing developments, including portable and noninvasive systems, further boost its potential for routine clinical use. SERS-based monitoring stands at the forefront of precision medicine, offering a real-time, comprehensive approach to tailor treatments based on individual patient responses, ultimately improving overall therapeutic effectiveness.36–38
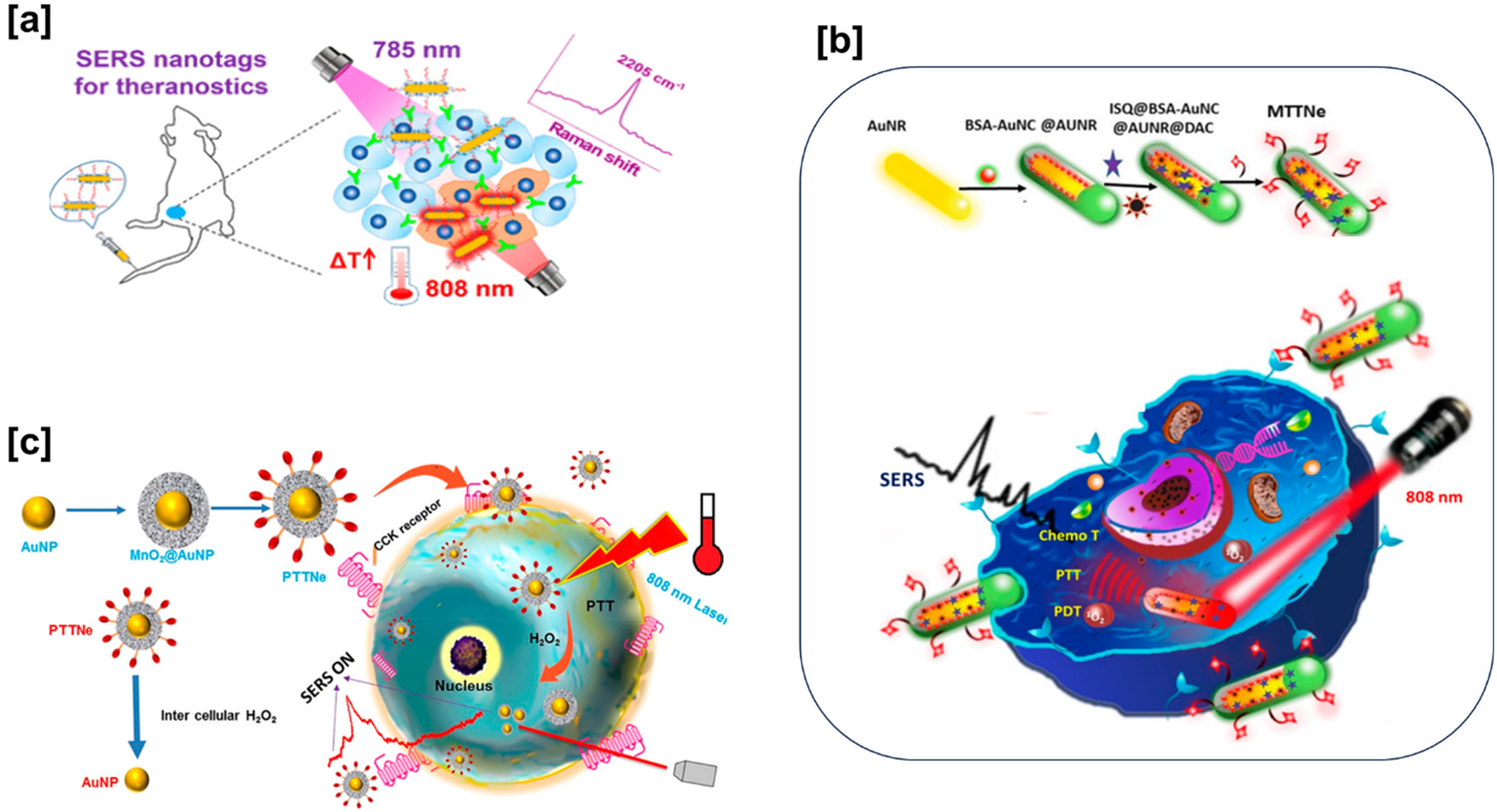
(a) A bioorthogonal Raman reporter and aptamer-functionalized SERS nanotags with a strong Raman signal at 2205 cm–1, adapted with permission. 32 Copyright 2020, American chemical society. (b) A melanoma-targeted theranostic nanoenvelope (MTTNe: BSA-AuNC@AuNR) integrating photothermal, photodynamic, and chemotherapy with SERS detection, adapted with permission. 33 Copyright 2019, American Chemical Society. (c) A photothermal theranostic nanoenvelope for real-time monitoring of therapy in pancreatic cancer cells through SERS, featuring MnO2 overlaid gold nanoparticles and a distinctive Raman peak at 569 cm–1, adapted with permission. 34 Copyright 2021, American Chemical Society.
Nanoparticle-based SERS for targeted therapy
Au NP or AgNP produce strong Raman signals for sensitive molecular detection, offering a versatile platform for targeted therapy when combined with therapeutic agents. This approach provides tunable properties for precise control, optimizing interactions with biological targets. Functionalization with targeting ligands ensures specific recognition and binding to diseased cells, ensuring high selectivity. In cancer therapy, utilizing nanoparticles conjugated with anticancer drugs enhances bioavailability and targeted delivery to tumor sites. Engineered surfaces recognize overexpressed receptors on cancer cells, enabling selective binding and internalization.39,40 SERS real-time monitoring tracks drug release kinetics and therapeutic responses, offering crucial insights into treatment efficacy. This nanoparticle-based versatility extends to infectious diseases, providing targeted therapy with minimal off-target effects. Functionalized SERS-active nanoparticles specifically recognize pathogens, allowing direct delivery of antimicrobial agents to infection sites, potentially reducing drug resistance development. In a recent study, stable Janus nanoparticles (JNPs) were formed using click chemistry, incorporating polyethylene glycol (PEG) and two DNA types. These JNPs enable in situ SERS detection and cancer cell imaging by creating hotspots in the nanogap between AuNP dimers. Monitoring adenosine triphosphate or acidity in cancer cells enhances diagnostic accuracy. Loaded drugs and photosensitizers on the opposite side of JNPs demonstrated a synergistic antitumor effect. 41 In neurology, SERS shows promise for precise brain drug delivery by overcoming challenges posed by the blood–brain barrier. Raman-active nanoparticles, equipped with suitable ligands, navigate this barrier to deliver therapeutic agents to specific brain regions. Real-time monitoring of drug distribution is vital in neurological disorders, where accurate targeting is crucial for therapeutic success. The clinical use of nanoparticle-based SERS for targeted therapy is a dynamic field, with ongoing efforts to address challenges like biocompatibility, stability, and scalability. Recent advancements involve creating multifunctional nanoparticles that combine imaging, diagnostics, and therapy in a single platform for better cancer treatment.42,43
SERS tag design for enhanced cancer treatment outcomes
Enhancing treatment outcomes with SERS tag design require skillful integration of Raman-active tags with therapeutic agents, enabling sensitive molecular detection, targeted drug delivery, and real-time treatment monitoring. Typically made of noble metals like Au or Ag, these tags produce robust Raman signals, serving as a potent tool for molecular sensing. In therapeutic contexts, optimizing SERS tag design is crucial for effectiveness and impact on treatment efficacy.44,45 An innovative approach involves embedding these tags into nanocarriers for drug delivery. For instance, researchers have developed SERS-active nanoparticles loaded with therapeutic agents for cancer treatment. The unique optical properties of SERS tags facilitate monitoring nanoparticle distribution in vivo, guiding their accumulation at tumor sites. This focused drug delivery strategy improves treatment outcomes by precisely concentrating therapeutic agents, reducing systemic side effects, and enhancing overall efficacy. SERS tag design includes surface functionalization with targeting ligands, such as antibodies or peptides, for specificity to diseased cells. In cancer therapy, tags can recognize cancer cell surface receptors, enabling selective delivery of therapeutic agents. Combining SERS tags with imaging agents and therapeutic compounds creates multifunctional platforms for simultaneous molecular imaging and treatment. Real-time monitoring of SERS tags visualizes treatment responses, allowing adjustments to treatment protocols based on individual patient dynamics.46,47
A study explores enhancing gold nanostars with anticancer drug doxorubicin molecules and an immunomimetic thiolated PEG coating for effective SERS tag imaging probes. The resulting doxorubicin-conjugate AuNPs exhibit strong Raman signal enhancement, physiological stability, and low cytotoxicity. Laser irradiation generates a robust SERS signal, allowing nanoparticle localization within cells. Prolonged irradiation demonstrates thermally induced drug delivery, highlighting the potential of combining SERS with laser scanning confocal microscopy for real-time drug release analysis in living cells. 48 The versatility of designing Raman active tags is evident in infectious disease research, where functionalizing tags with molecules recognizes specific pathogens. SERS tags act as sensitive probes for detecting viral or bacterial infections. Integrated into antiviral or antibacterial drug delivery systems, these tags facilitate targeted therapy by precisely delivering agents to infection sites, offering a potential strategy against drug-resistant strains.
In a study, researchers developed a sensitive SERS-based lateral flow immunoassay strip to simultaneously detect influenza A H1N1 virus and human adenovirus. Magnetic SERS nanotags offer specific recognition and magnetic enrichment of target viruses. Dual-layer Raman dye molecules and target virus-capture antibodies enable direct SERS detection on the strip for real biological samples without prior treatment. The proposed strip is deemed suitable for high throughput, showing promise for early virus infection detection. 49 In neurology, SERS tag design explores targeted drug delivery to the brain. SERS-active nanoparticles, with ligands traversing the blood–brain barrier, can deliver therapeutic agents to specific brain regions, crucial for neurological disorders requiring precise drug delivery. 50 Recent advancements include “smart” SERS tags responding to specific stimuli for controlled drug release. Innovations in tag design hold promise for advancing precision medicine, tailoring therapeutic interventions based on molecular insights for improved clinical outcomes. Ongoing research shapes personalized medicine and innovative disease treatment approaches.
SERS as a tool for monitoring drug distribution in vivo
SERS is valuable for monitoring in vivo drug distribution, providing detailed insights into pharmacokinetics and therapeutic agent biodistribution. SERS-active nanoparticles act as drug carriers, enabling real-time tracking of their journey within the organism. Their ability to generate strong, distinctive Raman signals allows precise identification and quantification of drugs, offering a noninvasive and highly sensitive approach to studying drug distribution dynamics. An exemplary application involves crafting SERS-active nanoparticles loaded with anticancer agents (Figure 2(a) and (b)). This capability helps researchers comprehend the spatiotemporal dynamics of drug delivery, ensuring effective targeting of therapeutic agents and facilitating the optimization of treatment regimens.51–54

(a) Illustration of up conversion fluorescence (UCF)-SERS dual mode tags for live-cell and in vivo imaging, showcasing their potential for medical diagnostics and therapy, adapted with permission. 53 Copyright 2014, American chemical society. (b) Schematic illustration of a new nanomaterial probe consisting of superparamagnetic iron oxide nanoparticles complexed with gold nanostructures, serving as a dual-function T2-weighted MRI contrast agent and SERS substrate for a dye molecule. The synthesized probe demonstrates SERS activity in both aqueous solution and in vivo, adapted with permission. 54 Copyright 2021, American Chemical Society. (c) The rapid progress of SERS through integration with various tools for versatile microbial characterization and diagnostics, adapted with permission. 56 Copyright 2014, American Chemical Society. (d) A hybrid SERS substrate with plasmonic Au@Ag@mSiO2 nanorattles enabling optophysiological monitoring of microbial extracellular metabolism, ensuring robust and sensitive measurements, adapted with permission. 57 Copyright 2021, American Chemical Society.
Current technologies cannot identify macrophage phenotypes in living organisms. However, a recent investigation has introduced AH1, a SERS nanoprobe capable of accurately determining physiological pH, thereby revealing macrophage repolarization in brain lesions after drug intervention. This innovation provides a dynamic tool for monitoring disease-associated immune microenvironments and evaluating the effectiveness of immune therapeutics in vivo. 55 SERS serves as a versatile tool for examining drug distribution across various diseases, including infectious ones (Figure 2(c) and (d)).56,57 In antiviral therapies, Raman-active nanoparticles coupled with antiviral drugs can be monitored in vivo to assess biodistribution postadministration. This aids in evaluating the drug delivery system's targeting efficiency, enabling the development of more efficient antiviral treatments with reduced side effects. In a novel approach, AgNP, combined with photosensitizers (Chlorin e6) and a Raman reporter (4-Mercaptobenzonitrile), demonstrated significantly improved antibacterial activity against multidrug-resistant bacterial infections. In methicillin-resistant Staphylococcus aureus- and carbapenem-resistant Pseudomonas aeruginosa-infected mouse models, in vivo antibacterial effectiveness reached 96.8% and 93.6%, respectively. The SERS signal at the wound site returned to normal tissue levels within 24 h, indicating complete metabolism of the nanoagents from the infected area. 58 Customizing SERS-active nanoparticles with targeting ligands improves specificity for tissues or cells, ensuring precise drug delivery and reducing off-target effects. Monitoring Raman spectra changes offers insights into therapeutic agent fate, guiding optimal drug formulation and dosing regimens. SERS, with its ability to monitor drug delivery dynamics, enhance targeting, and understand drug metabolism, is invaluable for developing and improving drug delivery systems.59,60 Ongoing research explores leveraging SERS capabilities for a deeper understanding of drug behavior, contributing to personalized and effective therapeutic interventions.
SERS in COVID-19 monitoring
SERS is a valuable tool for monitoring and understanding COVID-19, providing sensitive virus detection and innovative diagnostic approaches. SERS-based biosensors are developed for COVID-19 monitoring, using substrates with complementary DNA sequences to selectively capture specific regions of the SARS-CoV-2 genome, allowing rapid virus detection as an alternative to traditional polymerase chain reaction methods. SERS is also explored for detecting viral proteins associated with COVID-19. Nanoparticles with antibodies or aptamers recognizing viral antigens enable sensitive protein detection, crucial for diagnosing infections and monitoring disease progression. The multiplexing ability of SERS allows simultaneous detection of multiple biomarkers, providing a comprehensive diagnostic profile. SERS is employed to monitor the immune response to COVID-19, detecting antibodies from SARS-CoV-2 infection and capturing and quantifying antibodies in patient samples. This aids in diagnosing past infections and provides insights into immune response dynamics. SERS shows promise in monitoring COVID-19 treatment efficacy, delivering antiviral drugs to infected cells, with real-time monitoring, visualizing drug distribution, and assessing treatment responses for more effective interventions.61,62 SERS aids in environmental monitoring of SARS-CoV-2. This wastewater-based epidemiology acts as a community-level early warning system, enhancing public health surveillance. In COVID-19 monitoring, SERS versatility extends to breath analysis, offering noninvasive viral particle detection. SERS-active substrates capture volatile organic compounds (VOC) linked to viral infections in exhaled breath, providing a rapid screening method in various settings.63,64 SERS plays a multifaceted role in COVID-19, including diagnostics, immune response profiling, treatment efficacy assessment, and environmental surveillance. Ongoing research explores new applications, showcasing SERS as a potent tool in the pandemic fight.
SERS in monitoring COVID-19 disease progression
SERS is crucial for tracking COVID-19 progression, offering sensitive molecular insights. It identifies viral RNA with specialized biosensors throughout disease stages. SERS observes molecular profile changes in biological fluids, revealing COVID-19-related biomarkers. Using Raman-active nanoparticles, researchers detect variations in protein, lipid, or metabolite levels, potentially indicating disease severity and progression for prognostic marker development. Immunoassays monitor the COVID-19 immune response, detecting antibodies. SERS substrates with viral antigen capture and quantify antibodies from SARS-CoV-2 infection. Monitoring antibody kinetics informs immune response dynamics and assesses immunity progression in COVID-19 recovery. SERS aids therapeutic monitoring in COVID-19 treatment; nanoparticles deliver antiviral drugs to infected cells (Figure 3(a) and (b)). Real-time SERS monitoring visualizes drug distribution, assesses treatment responses, enhances understanding, and optimizes COVID-19 treatment regimens.65–67 A highly sensitive COVID-19 biosensor was introduced in a recent study, utilizing SERS to detect the SARS-CoV-2 virus in saliva. The SERS-active substrate includes two layers of uniform AuNP films for reproducibility and sensitivity in the SERS immunoassay. The detection process with the designed SERS biosensor demonstrates exceptional specificity and sensitivity for SARS-CoV-2, offering a potential avenue for early COVID-19 diagnosis. 68 SERS holds a unique potential for monitoring the respiratory system damages in COVID-19. Breath analysis using SERS-active substrates capturing VOC linked to respiratory infections offers a noninvasive method for assessing disease severity. Another study introduced an innovative approach to detect SARS-CoV-2 in respiratory aerosols. 69 Raman substrates with viral RNA affinity can detect and quantify SARS-CoV-2 in wastewater, serving as an early warning system for viral spread and aiding in tracking virus prevalence in different areas. SERS plays a multifaceted role in COVID-19 monitoring, covering diagnostics, immune response profiling, therapeutic monitoring, respiratory analysis, and environmental surveillance.

(a) Scheme illustrates a highly sensitive SERS aptasensor platform for quantifying severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) lysates. Utilizing a spike protein DNA aptamer as the receptor and a self-grown Au nano popcorn surface for enhanced sensitivity, adapted with permission. 66 Copyright 2023, American Chemical Society. (b) Schematic illustration of a sensitive SERS aptasensor platform to quantify SARS-CoV-2 lysates. The receptor employed was a spike protein DNA aptamer, and detection utilized a self-grown Au nano popcorn surface for enhanced sensitivity, adapted with permission. 67 Copyright 2021, American Chemical Society.
COVID-19 biomarker detection using SERS
SERS is crucial for COVID-19 biomarker detection, playing a vital role in diagnostics. It identifies viral RNA using SERS-based biosensors with substrates designed to selectively bind to specific regions of the SARS-CoV-2 genome, ensuring swift and accurate confirmation of virus presence in patient samples. SERS also detects protein biomarkers through immunoassays, utilizing nanoparticles with antibodies specific to viral antigens. This enables selective capture and identification of viral proteins like the spike or nucleocapsid protein, serving as a diagnostic tool for active infections and monitoring disease progression. Additionally, SERS aids in identifying host response biomarkers reflective of the immune response to COVID-19. Researchers use SERS-active substrates with an affinity for host-derived biomolecules to discern changes in protein, lipid, or metabolite levels in patient samples. These changes have potential as biomarkers for gauging disease severity, contributing to prognosis and treatment decisions.70,71
In an interesting study, researchers evaluated the effectiveness of gold nanocavities as SERS-active substrates for detecting SARS-CoV-2 pseudovirus and vesicular stomatitis virus G pseudotype lentivirus. The optimized substrates underwent validation for measurement repeatability, reproducibility, and detection limit. The Raman spectra of live and inactivated SARS-CoV-2 S and vesicular stomatitis virus G pseudoviruses exhibited varying characteristic peak positions due to differences in substrate geometry and composition. 72 The flexibility of SERS enables the simultaneous identification of various biomarkers in a single sample. 73 In COVID-19 diagnostics, SERS breath analysis targets virus-associated VOCs, offering a swift, noninvasive approach. SERS-active substrates capturing exhaled breath VOCs may enable early detection via point-of-care tests. Besides clinical use, SERS aids in monitoring SARS-CoV-2 in wastewater, acting as an early warning system for community-level viral spread and guiding public health interventions. A recent portable breathalyzer, utilizing SERS, achieved >95% sensitivity and specificity in under 5 min across 501 participants, regardless of the symptoms. This system utilizes unique vibrational patterns from breath metabolites and molecular receptors, forming a robust model for high-throughput classifications via partial least-squares discriminant analysis, validated through experimental and in silico methods. 74 SERS proves a versatile platform for COVID-19 diagnostics, detecting viral RNA, proteins, host response biomarkers, or breath-based VOCs (Figure 4(a) and (b)).75,76

(a) A rapid detection method for SARS-CoV-2 RNA in human nasopharyngeal swab specimens by combining a SERS sensor with a deep learning algorithm. The SERS sensor, made from a silver nanorod array substrate, utilizes DNA probes to capture the virus RNA, adapted with permission. 75 Copyright 2022, American Chemical Society. (b) SERS sensor for SARS-CoV-2 using ACE2-mimetic peptides. Spike protein interaction with peptide-modified surface enables quantification via a multivariate calibration model. This study paves the way for SERS-based SARS-CoV-2 assay and future biosensors for other viruses, adapted with permission. 76 Copyright 2021, American Chemical Society. (c) Schematic of a single-step SERS immunoassay utilizing plasmonic coupling enhancement (pink corona) through sandwiched antibody–antigen assembly, adapted with permission. 77 Copyright 2013, American Chemical Society.
SERS in real-time monitoring of immune response during COVID-19 infection
SERS serves as a vital tool for monitoring the immune response in COVID-19, offering insights into antibody dynamics and aiding in assessing disease progression. SERS-based immunoassays quantify SARS-CoV-2 antibodies using functionalized substrates with viral antigens, identifying antibodies in patient samples. This monitors antibody production kinetics in real time, crucial for understanding the temporal aspects of the immune response and making informed decisions about vaccination strategies, booster doses, and evaluating immunity effectiveness and duration. 77 SERS profiles immune response diversity by analyzing antibody-associated spectra and identifying variations among COVID-19 patients. This personalized approach informs treatment decisions and enables tailored interventions based on unique immune profiles. SERS distinguishes between antibody isotypes, such as immunoglobulins M type (IgM) and immunoglobulins G type (IgG), indicating the immune response stage, with IgM being early and IgG more sustained. Monitoring isotype ratio and evolution with SERS characterizes individuals’ immune status in different COVID-19 phases. SERS versatility allows multiplexed detection of various antibodies in a single assay, providing a comprehensive view of the immune response. Simultaneously monitoring antibodies targeting different viral antigens or specific isotypes enhance SERS diagnostic and prognostic capabilities in assessing COVID-19 immune responses (Figure 4(c)).
A group of researchers proposed a multiplex SERS micro assay for detecting SARS-CoV-2's spike and nucleocapsid structural proteins. This SERS microassay aids early COVID-19 detection, reducing transmission rates and facilitating appropriate treatments for severe cases. 78 SERS can detect cytokines, signaling molecules crucial in the immune response.79,80 Using specific capture molecules for cytokines, researchers can monitor changes in cytokine levels in response to viral infection. This information aids in understanding the inflammatory processes related to COVID-19 and may guide therapeutic interventions to modulate the immune response. Real-time tracking of these immune parameters enhances comprehension of COVID-19 pathogenesis, informs treatment decisions, and contributes to personalized strategies for disease management. Ongoing research aims to refine and expand SERS applications in monitoring the immune response to COVID-19, providing valuable insights for infectious disease diagnostics and therapeutics.
SERS in environmental monitoring for tracing SARS-CoV-2 viral presence
SERS is a potent tool for environmental monitoring, particularly in detecting SARS-CoV-2 in various environments. Notably, it detects viral RNA in wastewater, offering a sensitive indicator of community-level viral spread. SERS-active substrates with specific capture molecules for SARS-CoV-2 RNA allow virus detection and quantification in wastewater samples. 81 Monitoring viral RNA levels serves as a noninvasive strategy for effective surveillance, providing early warnings and contributing to public health interventions. Researchers developed SERS-based biosensors for SARS-CoV-2 detection in environmental samples. These biosensors use SERS-active nanoparticles with viral antigens or aptamers recognizing the virus. Deployed in the air or on surfaces, these biosensors selectively capture and generate Raman signals in the presence of SARS-CoV-2, enabling rapid and sensitive detection (Figure 5(a) and (b)). This approach is relevant for monitoring high-traffic areas, healthcare facilities, or public spaces with elevated viral transmission risk.82–85

(a) Schematic of the COVID-19 FET sensor process, featuring graphene as the sensing material. SARS-CoV-2 spike antibody conjugation onto the graphene sheet is facilitated by 1-pyrenebutyric acid N-hydroxy succinimide ester as a probe linker, adapted with permission. 84 Copyright 2020, American Chemical Society. (b) Illustration of a spectroscopic assay using silver nanorod array substrates and SERS for swift and highly sensitive trace virus detection, adapted with permission. 85 Copyright 2006, American Chemical Society. (c) SERS-based system for rapid and highly sensitive SARS-CoV-2 antigen detection within 20 min, utilizing a stable SERS substrate and a supervised deep learning algorithm, adapted with permission. 87 Copyright 2023, American Chemical Society.
SERS in environmental surveillance extends to the analysis of indoor air quality. 86 SERS substrates capture aerosolized viral particles in indoor spaces, tracking alterations in spectra linked to SARS-CoV-2 presence. This evaluates airborne transmission risk, crucial in ventilated environments. SERS versatility extends to surface detection; substrates on frequently touched surfaces capture and identify SARS-CoV-2 particles, guiding cleaning practices to minimize fomite transmission. A recent SERS platform, combining a highly effective substrate with a deep learning algorithm, achieves rapid (<20 min) and highly sensitive SARS-CoV-2 antigen detection (Figure 5(c)). The V-shaped resonant cavity array substrate, designed for resonance coupling, enables direct and consistent detection of the virus, identifying variants and accurately detecting it in clinical and environmental samples. 87 Furthermore, SERS is being explored for monitoring bioaerosols containing viral particles. Integration into air sampling devices enables SERS-active substrates to capture and detect aerosolized SARS-CoV-2 particles in real time. 88
Surface and air sampling using SERS in COVID-19 monitoring
SERS is crucial for COVID-19 surveillance, swiftly detecting the virus through surface and air sampling in diverse environments. In surface sampling, SERS substrates capture SARS-CoV-2 particles on various surfaces. Functionalized nanoparticles, acting as SERS-based biosensors, selectively bind and detect the virus on high-traffic surfaces, facilitating real-time, noninvasive monitoring. 89 SERS is vital in air sampling, seamlessly integrated into devices to detect aerosolized viral particles, improving monitoring of airborne viral presence indoors and outdoors. Employed in healthcare facilities, public transport, and enclosed spaces, SERS-enabled air sampling addresses concerns related to airborne transmission risk. SERS real-time capabilities enable continuous monitoring, providing valuable insights into the dynamics and persistence of viral aerosol distribution.90–92
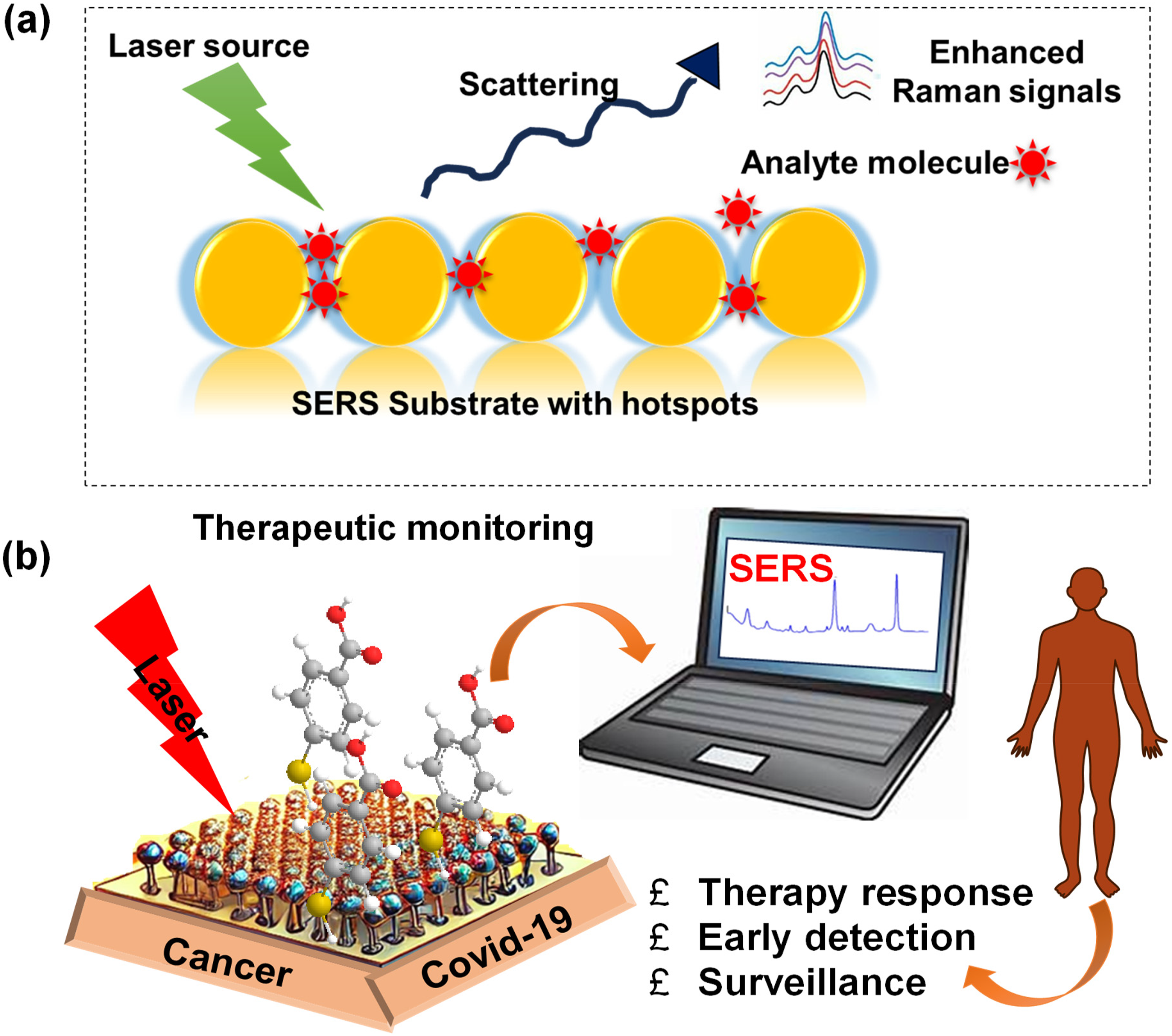
(a) Mechanism of SERS signal generation. It works by increasing Raman scattering signals when molecules interact with metal surfaces, usually gold or silver. These surfaces create localized electromagnetic fields that boost the scattered light, which lets scientists find small amounts of analytes. (b) Schematic representation of overview of the SERS applications in theragnostic.
Portable Raman devices employ substrates or sensors for on-site air sampling, capturing, and identifying viral particles in real time. Their portability allows rapid deployment in diverse environments, enabling comprehensive monitoring of SARS-CoV-2 presence in the air. Particularly beneficial in areas of high human activity or critical air quality maintenance, this capability assesses the risk of viral transmission. 93 Researchers explore SERS-based multiplexed assays identifying various respiratory viruses, including SARS-CoV-2, in a single air sampling event, enhancing monitoring efficiency. Integrated into environmental monitoring networks, air sampling strategically deploys sensors in urban areas or potential viral hotspots, ensuring continuous monitoring. Data from these networks contribute to public health strategies, offering early warnings of viral spread and supporting preventive measures. The real-time, noninvasive, and multiplexed nature of SERS-based methods enhances surveillance efficiency, providing crucial information for risk assessment, infection control, and public health decision making. 94
SERS for monitoring SARS-CoV-2 viral transmission routes
SERS is crucial for monitoring SARS-CoV-2 transmission in various environments. It efficiently detects viral RNA on surfaces, providing insights into fomite transmission. Customized SERS substrates with SARS-CoV-2 RNA-specific molecules enable sensitive detection in high-traffic areas, healthcare facilities, or locations prone to surface contamination, guiding effective cleaning strategies. 95 This is vital in crowded or enclosed spaces where respiratory droplets significantly contribute to viral spread. 96 SERS biosensors for respiratory droplets, using functionalized nanoparticles, are vital in crowded spaces where droplets contribute to viral spread. Real-time SERS monitoring aids in risk assessment and informs preventive measures against airborne transmission. In air sampling, SERS monitors aerosolized viral particles for transmission risk, providing insights into airborne spread dynamics in healthcare, public transport, and enclosed spaces. SERS also aids wastewater monitoring, capturing viral RNA for early community-level viral spread detection, complementing other transmission assessments. 97 A recent study introduced a highly sensitive SERS biosensor using an ACE2-functionalized gold nanostructure, termed as virus traps. This biosensor selectively captures and rapidly detects S-protein-expressed coronaviruses, including the current SARS-CoV-2. This real-time monitoring can establish identification standards for future unknown coronaviruses, enabling extremely sensitive and rapid detection. 98 SERS facilitates multiplexed detection across transmission routes, with assays simultaneously identifying viral particles on surfaces, in respiratory droplets, in the air, and wastewater. This streamlined monitoring offers a comprehensive view of potential transmission routes, aiding targeted interventions and enhancing understanding of factors influencing viral spread. The multiplexed capabilities of SERS contribute to risk assessment, inform preventive measures, and provide a comprehensive understanding of intricate virus spread pathways. 99
Challenges and future perspectives
SERS in COVID-19 management brings challenges and opportunities. Standardizing results and establishing protocols are essential for clinical applications. Optimization of SERS substrates involve material, size, and shape considerations, with ongoing efforts for reproducibility, sensitivity, and stability. Real-world use faces challenges in creating portable devices without compromising performance. Integrating SERS into multiplexed platforms for molecular profiling presents both challenges and exciting prospects. Achieving simultaneous detection of biomarkers enhances diagnostics, but addressing signal cross-talk is necessary for successful multiplexing. Future perspectives include developing advanced SERS assays with increased multiplexing capacity for comprehensive profiling of COVID-19-related molecular signatures. 100 Successfully managing COVID-19 with SERS requires addressing challenges related to complex sample matrices like clinical or environmental samples. Matrix effects, sample interference, and diverse compositions can impact SERS accuracy and specificity. Researchers are actively overcoming these challenges through improved sample preparation, signal processing, and advanced data analysis, enhancing the reliability of real-world SERS-based diagnostics. Despite current obstacles, SERS holds promising prospects in COVID-19 management. Ongoing research on nanomaterials, substrates, and engineering will bolster SERS capabilities for viral detection. Integration with artificial intelligence and microfluidics will yield systems with enhanced sensitivity and user-friendliness. SERS platforms may play a pivotal role in future pandemics, demonstrating their adaptability for swift and accurate diagnostics. Continuous research and advancements are expected to address challenges, enabling widespread deployment of SERS in clinical diagnostics and environmental monitoring and contributing to effective pandemic management.101,102
Current challenges in implementing SERS for COVID-19 management
SERS faces challenges in COVID-19 management, notably in standardization and reproducibility due to varying experimental conditions and instrumentation. Establishing standardized protocols and benchmarks is crucial for its integration into routine COVID-19 management. Another hurdle involves optimizing SERS substrates, requiring enhanced sensitivity, stability, and reproducibility. Addressing substrate-related challenges, including tailoring substrates for specific purposes, will strengthen the reliability of SERS-based assays in the context of COVID-19. Instrumentation challenges hinder widespread SERS adoption; while lab setups provide high sensitivity, miniaturizing SERS instruments for point-of-care use and field monitoring is essential. This requires ensuring performance, ease of use, and accessibility. Integrating SERS into multiplexed platforms poses challenges, particularly in mitigating signal interference and cross-talk for simultaneous biomarker detection. Overcoming these challenges will significantly improve SERS diagnostic capabilities in COVID-19 management. 103 Implementing SERS in COVID-19 diagnostics faces challenges, particularly in complex sample matrices like clinical or environmental specimens. Overcoming these hurdles involves efficient sample preparation, refined signal processing, and advanced data analysis. Ensuring SERS specificity and accuracy in real-world samples is crucial for successful deployment across applications. Pressing concerns include accessibility and affordability. Despite SERS potency, cost, complexity, and expertise hurdles limit widespread use, especially in resource-limited settings. Enhancing SERS cost-effectiveness, user-friendliness, and adaptability is vital for broader COVID-19 applications. Challenges for COVID-19 management encompass measurement standardization, substrate optimization, instrumentation, multiplexed platform development, addressing matrix effects, and ensuring accessibility.
Innovations and ongoing research in SERS technology for COVID-19 management
Continued SERS research for COVID-19 management showcases its dynamic evolution, addressing challenges with innovations. Plasmonic nanoparticles, such as gold or silver nanostars, enhance Raman signals, optimizing substrates for reliable viral detection. A notable advancement is the integration with microfluidics, enabling compact on-site diagnostic devices. Microfluidic SERS platforms offer benefits like reduced sample volumes, automation, and potential multiplexed detection, making them promising for point-of-care use. This integration tackles instrumentation challenges and enhances SERS accessibility in healthcare settings. Ongoing research emphasizes multiplexed SERS assays, crucial for simultaneously detecting various COVID-19 biomarkers, enabling comprehensive molecular profiling. Illustrative assays identify viral components, antibodies, and cytokines in a single test, providing a holistic patient status view for more accurate COVID-19 diagnostics.104,105
To overcome the challenges in complex sample matrices, sophisticated algorithms and ML enhance SERS measurement specificity and reliability. These innovations ensure accurate SERS-based diagnostics across diverse environments. 106 Exploring breath analysis using SERS introduces a novel approach in COVID-19 management. 107 SERS substrates capture respiratory infection-related VOCs, utilizing SERS's distinctive fingerprinting for COVID-19 breath signatures. This noninvasive method holds potential for quick screening, facilitating early detection and monitoring in diverse settings. Improved affinity SERS substrates allow sensitive and selective detection in complex environments. Incorporating SERS into monitoring networks enables ongoing surveillance, offering crucial data for public health interventions and early alerts. Advances in SERS technology for COVID-19 management encompass nanomaterials, integrated technologies, multiplexed assays, signal processing, breath analysis, and environmental monitoring.
Potential future developments and applications of SERS in virology and cancer
Ongoing research promises exciting future developments and applications of SERS in virology. Researchers explore advanced nanomaterials like graphene, metal–organic frameworks, and hybrid nanomaterials to enhance SERS sensitivity and broaden viral analysis capabilities. These innovations aim to optimize SERS performance in virological applications. The integration of SERS with emerging technologies, such as microfluidics, lab-on-a-chip systems, and biosensors, is a key focus for future developments. This combination offers the potential for miniaturized, portable devices with improved capabilities. SERS-based microfluidic platforms, for instance, enable rapid on-site virus detection, supporting point-of-care diagnostics and field surveillance. The integration of SERS with other technologies paves the way for versatile tools in virological studies and applications. 108 In viral diagnostics, future developments target enhanced early detection and strain differentiation. Multiplexed SERS assays, currently in research, aim to simultaneously detect various viruses or strains, offering comprehensive virological profiling. 109 This advancement revolutionizes viral diagnostics, providing insights into prevalence, coinfections, and genetic variations. In antiviral drug development, SERS shows promise for molecular-level monitoring of virus–drug interactions. Research explores its use in studying drug efficacy, assessing delivery mechanisms, and understanding drug–virus dynamics. For example, SERS monitors vibrational spectra changes in viral proteins or nucleic acids postdrug treatment, supplying valuable insights for drug development and optimization. 110
Future applications of SERS in virology may include advancements in imaging and tracking. Researchers are exploring SERS-based techniques to visualize viral particles at the single-particle level, enabling detailed studies of viral processes. 111 SERS tracking of viral particles in complex biological environments offers insights into spatial and temporal dynamics. These developments deepen our understanding of virology and present new perspectives for antiviral strategies. SERS's potential future application in environmental monitoring involves designing substrates for selective detection of viral components in air, water, or surfaces. This capability aids in tracking virus prevalence in various settings, serving as an early warning system for potential outbreaks. Ongoing research spans advanced materials, integrated technologies, multiplexed diagnostics, drug development, imaging, and environmental monitoring, promising transformative contributions to virological studies. 112
SERS faces several challenges such as reproducibility issues, high costs, and practical application issues. We need comprehensive solutions to address these issues, enabling more people to utilize SERS. Hotspots often form unevenly on SERS substrates, leading to signal variability and making replication more challenging. This issue can be resolved by using more uniform nanomaterials and standardizing the production process. Exploring more cost-effective materials like copper or carbon-based alternative substrates may alleviate the high costs associated with SERS equipment and substrates. Problems with using SERS technology, like the need for special training and regular maintenance on the equipment, show how important it is to make solutions that are simple for people to use and can be easily added to existing processes. Confronting these issues is essential to fully realize the potential of SERS across various applications, including biological diagnostics and environmental monitoring. Nanomaterials play a crucial role in the development of compact, portable gadgets. To guarantee practical applicability, SERS systems must be adaptable to varying environmental conditions; this can be achieved by incorporating environmental controls and advanced data processing algorithms to mitigate sample influence.
Alongside these technological issues, it is imperative to emphasize user training and assistance to guarantee the successful adoption of SERS technology. Intuitive interfaces and comprehensive documentation will be essential for promoting widespread adoption and use of these technologies. Moreover, engagement with industry stakeholders and regulatory bodies would be essential in formulating standards and guidelines for the development and application of SERS technology across diverse sectors. By addressing these problems and working toward a more unified and user-centered approach, SERS technology can fully realize its potential to change many scientific fields and sectors. Selectivity can also be improved in complex samples by changing the surface of the substrate or by using SERS with other analytical methods together, which increases both sensitivity and specificity. By overcoming these challenges, SERS can develop into a more accessible, reliable, and versatile tool in fields such as environmental monitoring, medical diagnostics, and food safety. Integrating SERS with other analytical methods may enhance sensitivity and specificity, but it also introduces complexity and possible sources of error into the analytical process. The expenses and technical proficiency necessary for substrate surface modification may hinder the extensive implementation of SERS technology across many sectors and scientific domains. Researchers are investigating innovative methods to improve the efficacy of SERS and facilitate its application. A viable approach is the creation of portable and user-friendly SERS devices that necessitate minimum sample preparation and technical proficiency. These new developments could make SERS much more useful in real life by making it easier to quickly and accurately find trace analytes in different samples. Additionally, ongoing efforts to improve substrate materials and production methods aim to lower costs and boost the accuracy of SERS measurements, making this method easier for a larger group of people to access. The ongoing invention and enhancement of SERS technology possess significant promise to transform analytical chemistry and propel scientific research across all disciplines (Table 1).
SERS: surface-enhanced Raman spectroscopy; PCR: polymerase chain reaction; ELISA: enzyme-linked immunosorbent assay; CT: computed tomography; MRI: magnetic resonance imaging; PET: positron emission tomography.
Advancements in Raman spectrometers for SERS and clinical applications
Raman spectrometers are essential for SERS investigations. Usually, these tests need high-quality, lab-grade instruments that are exceedingly sensitive and clear. This equipment is sometimes unwieldy and inappropriate for field or in vivo applications where portability is crucial. Recent advancements have led to the development of tiny, portable Raman spectrometers that provide continuous functionality across diverse environments. Confocal Raman microscopes from manufacturers such as WITec, Renishaw, and Bruker have superior imaging capabilities compared to conventional Raman spectrometers. They provide high-resolution chemical mapping and depth probing within a specimen. These systems utilize advanced optics and detectors to provide intricate measurements, particularly advantageous for research and clinical applications. Furthermore, handheld Raman spectrometers with diverse excitation wavelengths, including those from Bruker (BRAVO), Anton Paar (Cora 100), and Axiom Optics (IndiGo), are progressively utilized in clinical environments owing to their compact size and versatility. 115
These portable devices present promising opportunities in medical diagnostics and field applications, where rapid, noninvasive sample analysis is crucial. Because these devices can be set up to work with different excitation wavelengths (488 nm, 532 nm, 633 nm, 785 nm, etc.), they can be used for a wide range of tasks, such as analyzing tissues and identifying materials. Contemporary portable Raman spectrometers have attained considerable miniaturization, with certain devices measuring as compactly as 10 cm×10 cm×5 cm and weighing under 1 kg. These instruments are engineered to accommodate several sampling attachments, hence augmenting their adaptability for field applications. Centimeter-scale Raman spectrometers that use nonstabilized laser diodes and small optics have shown performance similar to that of bigger, more expensive systems (spectroscopyonline.com). Researchers have developed handheld SERS analyzers to enhance the mobility and use of these instruments. These analyzers have high-performance spectrometers and are engineered for user-friendliness in diverse settings (rta.biz). Portable Raman spectrometers can now sample more accurately thanks to new technologies like Coded-Aperture. This makes them useful for testing materials that are not all the same and have high signal-to-noise ratios (thorlabs.com). Notwithstanding these developments, attaining the optimal equilibrium between mobility and performance continues to pose a difficulty. In order to make spectrometers that are light and can take high-resolution measurements, future progress must focus on improving optical parts, using small but effective detectors, and coming up with new ways to reduce their size. Taking these things into account can help portable Raman spectrometers become more useful and effective tools for SERS use in the field and inside living things. While improving optical components and downsizing techniques may enhance portability, it is important to also consider the potential trade-offs in performance and accuracy that may result from such miniaturization efforts. Additionally, the cost-effectiveness of implementing these advancements must be carefully evaluated to ensure practicality for widespread use.
Conclusion
SERS is a promising tool for COVID-19 management and cancer research, enhancing diagnostics, monitoring, and surveillance. It enables the detection of SARS-CoV-2, cancer biomarkers, and early outbreak detection. SERS also supports proactive pandemic responses and precision medicine in oncology. Its versatility allows for monitoring immune responses, studying disease progression, and evaluating therapeutic interventions for personalized treatment strategies. SERS-based assays give a full picture of how COVID-19 works by letting researchers find viral parts, host biomarkers, and therapeutic agents all at the same time. This approach guides tailored treatment strategies and supports personalized medicine for infectious diseases. SERS-based breath tests have the potential to diversify diagnostics and create user-friendly tools for various settings. Advancements in SERS technology are valuable assets for future infectious disease outbreaks due to its adaptability, sensitivity, and multiplexing capabilities. The continuous evolution of SERS-based technologies establishes a robust infrastructure for proactive monitoring and response to emerging viral threats.
Footnotes
Acknowledgements
Jyothi B. Nair thanks European Union Horizon PF research and innovation program under the Marie Skłodowska-Curie (Grant Agreement 101151173). Manu M. Joseph thanks the CHRIST University for the research grant through Seed Money Scheme (CU-ORS-SM-24/04).
Ethical considerations
This study does not involve human participants, animals, or sensitive data requiring ethical approval. Therefore, ethical clearance was not required for this work.
Author contributions
Manu M. Joseph contributed to data collection, manuscript writing, proofreading, and redrafting. Sanoop P was responsible for data collection. Jayadev S. Arya handled reference arrangement and data collection. Jyothi B. Nair contributed to data collection, editing, fine arrangement, figure making, and overall article arrangement as the corresponding author.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the European Union Horizon PF, Seed Money Scheme, (grant number 101151173, CU-ORS-SM-24/04).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study are available upon reasonable request from the corresponding author.
Consent
All authors confirm that they have read and approved the final version of the manuscript for submission. The authors affirm that the work is original, has not been published previously, and is not under consideration for publication elsewhere. Additionally, all authors agree to be accountable for the accuracy and integrity of the work.

