Abstract
Objective
Curcumin is a polyphenol extracted from the roots of Curcuma longa L. It is a multifunctional biological substance that has notable antioxidant, anti-inflammatory, anti-cancer, and antibacterial properties, as reported in numerous previous papers. However, curcumin has limited bioavailability due to its low water solubility. Furthermore, pH, temperature, and light all have the potential to decrease curcumin's biopharmaceutical activities. Silver nanoparticles have excellent antibacterial characteristics and are used in various applications. The main objective of this work was to synthesize the gelatin microgel-stabilized silver nanoparticles loaded with curcumin (GelCurAg complex) to increase the biopharmaceutical activities of the complex.
Methods
Numerous techniques, including UV-vis spectroscopy, FTIR spectroscopy, Zetasizer Nano instruments, TEM images, and XDR patterns, were used for investigating the characterization of the GelCurAg complex. The various methods, such as the DPPH antioxidant assay, cytotoxicity analysis, and the agar-well diffusion method, were used for the analysis of the biopharmaceutical activities of the GelCurAg complex.
Results
We successfully synthesized and characterized the GelCurAg complex, consisting of gelatin, curcumin, and silver nanoparticle biofilms using polyvinylpyrrolidone. The average nanoparticle size was 50 nm. The GelCurAg complex improved water solubility and dispersion compared with the single curcumin component. The IC50 value for antioxidant activity was 4.87 μg/mL, and for antiproliferation on SK-MEL-28, it was 3.98 µg/mL. GelCurAg solution provided remarkable antibacterial activities against four bacterial strains: Bacillus cereus, Escherichia coli, Pseudomonas aeruginosa, and Staphylococcus aureus. The GelCurAg complex showed better antibacterial activities than the single curcumin component, with the antibacterial inhibition zone in the range of 0.9–1.6 cm.
Conclusions
The GelCurAg complex enhanced the solubility and dispersion of curcumin in water while increasing its antibacterial activity compared to curcumin. The GelCurAg complex has excellent biopharmaceutical properties to suggest its various applications in the future.
Keywords
Introduction
Curcumin is the most important pharmacologically active compound isolated from Curcuma longa L. Previous studies identified different activities of curcumin, including anti-cancer, anti-inflammatory, antimicrobial, antioxidant, and antidiabetic activities. 1 Curcumin also supports its beneficial effects on various human diseases, including cancer, 2 diabetes, 3 neurological disorders, 4 and other inflammatory diseases. 5 However, low water solubility, quick metabolism, and poor bioavailability are significant limitations to the applications of curcumin in various medical and pharmaceutical subjects.
Integrating curcumin into nanocarriers using different methods is an appropriate and effective option to enhance the bioactivity of curcumin, increase its bioavailability and solubility, circulate in a short time, and be stored in the body, and at the same time overcome the physiological barriers of curcumin. 6 Since 2011, curcumin nanoparticles have been mentioned in more than 1500 publications in the NCBI PubMed database. 6 Researchers have developed various techniques for the production of nanocurcumin. The most popular techniques used in procedures for nanocurcumin include nanoprecipitation, single emulsion, trace emulsion, spray drying, wet milling, solvent evaporation, ultrasonication, coagulation technique, ion gelation, fractionation solid dispersion, thin film hydration, and emulsion polymerization. 7
Silver is one of the most preferred metallic elements for treating pathogenic problems due to its strong antibacterial activity and broad-spectrum antibacterial action. Recently, the application of nanoscience and nanotechnology has grown rapidly, leading to significant advances in the production of nanomaterials. As a result, nanoparticle productions have been gathered using a variety of approaches, such as physics, chemistry, and biology. Silver nanoparticles are widely used for the production of cosmetics and healthcare products, antibacterial textiles, wound dressings, antitumor drug carriers, etc., due to their excellent antibacterial properties. 7
The modern trend for synthesizing silver nanoparticles is to use green synthesis, in which bio-friendly components such as plants, bacteria, and fungi play the key role. Because plants, fungi, and bacteria contain numerous compounds working as reducing agents (for example, phenolic compounds, polyphenols, flavonoids, saponins, amino acids, vitamins, enzymes, and proteins) to synthesize silver nanoparticles.8,9
Gelatin is a protein generated through the hydrolysis of collagen. As a natural biofilm-forming agent, gelatin has properties such as high biocompatibility and water solubility, low immunogenicity, plasticity, adhesion, and adhesion promotion cells. It is widely used in the pharmaceutical and medical sectors. Gelatin could be produced in numerous forms, such as films and micro- or nanoparticles, depending on its application. 10
Polyvinylpyrrolidone (PVP) improves the bioavailability of insoluble drugs, and it has the ability to protect the active compounds against external conditions (pH, temperature, and oxygen). 11 PVP is used to stabilize the surface of the particles by controlling the growth and dispersion rate of the nanoparticles through interactions between the metal, carbonyl group, and pyrrolidine ring.12,13 Other polymers used in nanoparticle synthesis with similar functions as PVP are polyethylene glycol (PEG) and polyvinyl alcohol (PVA). PVA is an innocuous, dissolvable, biodegradable, and biocompatible material widely used in various applications such as membrane preparations, drug delivery, recycled polymers, and textiles. However, PVA has poor stability in water. PEG is also a polymer used in biomedical applications because of its non-toxicity, non-carcinogenic effects, and bioadhesion properties. 14 However, as time progresses, the tendency for active pharmaceutical ingredients (APIs) encapsulated within PEG to aggregate is greater than that of those encapsulated within PVP.15,16 In the synthesis of silver nanoparticles, PVP was used to prevent agglomeration, reduce the size of silver nanoparticles, and enhance their properties. 17
In this work, we synthesized the gelatin microgel-stabilized silver nanoparticles loaded with curcumin in the complex (GelCurAg), including gelatin, curcumin, and silver nanoparticles, which was stabilized with PVP to increase its solubility and dispersion in water. Its physical and chemical properties were investigated, and the antibacterial efficacy against Gram-negative and Gram-positive bacteria was examined. Curcumin and silver have limited antibacterial activities compared with GelCurAg. We also investigated the antioxidant and antiproliferation properties of the complex. Therefore, the combination of curcumin and nanosilver into a bioavailable combination enhanced the potential for intriguing pharmaceutical applications in the future.
Materials and methods
Materials
Curcumin, gelatin, and PVP were supplied by Biobasic, Canada (high purity). AgNO3, CH3OH, trichloroacetic acid (TCA), Sulforhodamine B (SRB), and DPPH (1,2- and 2,2-diphenyl-1-picrylhydrazyl) were obtained from China (99% purity). Tryptone and peptone were purchased from Bio Basic, Canada (high purity). Pathogens, including Escherichia coli (E. coli, ATCC 25922), Staphylococcus aureus (S. aureus, ATCC 25923), Pseudomonas aeruginosa (P. aeruginosa, ATCC 27853), and Bacillus cereus (B. cereus, ATCC).
NIH 3T3 cells were provided by the Cell Department, Biological Faculty, VNU University of Science, Vietnam National University Hanoi, and SK-MEL-28 cells were purchased from ATCC. All cell lines were grown in DMEM low glucose (1%) or RPMI supplemented with penicillin (400 U/mL), streptomycin (50 mg/mL), L-glutamine (300 mg/mL) (Pan-Biotech, USA), and 10% fetal bovine serum (Sigma, Deisenhofen, Germany, high purity) in a humidified environment of 5% CO2 at 37°C.
Preparation of the GelCurAg complex
Preparation of the GelCurAg complex followed the previous research with slight changes. 18
First, 2 mg of curcumin powder (Biobasic, Canada) was completely dissolved in 5 mL of methanol. Then, slowly add the curcumin solution into 20 mL of gelatin solution at different concentrations (0.05, 0.1, 0.2, and 0.5% w/v) using a magnetic stirrer (stirring speed: 1000 r/min/min) for 5 min. After that, slowly added 200 µL of 1% AgNO3 and 1 mL of PVP (0.4 mg/mL) into the solution, and the solution was stirred for 15 min at the stirring speed of 1000 r/min/min. The experimental solutions were irradiated with UV light for various times (10, 30, 60 minutes) to form a GelCurAg solution. The different controls were made the same way; however, they were not supplemented with AgNO3 or curcumin components.
Characterization of the GelCuAg complex
The GelCurAg spectra were recorded by using a UV-vis spectrophotometer (BioMate 3S, Thermo Scientific, USA) with wavelengths (ʎ) ranging from 300 nm to 600 nm for determining the presence of silver nanoparticles. Particle size and zeta potential were determined by a Zetasizer Nano instrument (Malvern Instrument Limited, UK). The morphology of GelCuAg was captured by a TEM image (JEM2100, Jeol, Japan). The FTIR spectrum determined the chemical structure of GelCuAg by using a Fourier transform infrared spectrophotometer (IRAffinity-1S, Shimadzu, Kyoto, Japan). The XDR pattern of nanoparticles was determined by an instrument (PANalytical Empyrean, Malvern Instrument Limited, UK).
DPPH antioxidant assay
The antioxidant activity of the sample was determined following the previous research. 19 The DPPH (1.1-diphenyl-2-picrylhydrazyl) assay was used for assessing the antioxidant properties of Gel0.1CurAg samples. The reaction solution was mixed with 0.5 mL of DPPH solution (100 ppm), 1 mL of methanol, and 0.5 mL of Gel0.1CurAg at varied final sample concentrations (48, 24, 12, 6, and 3 μg/mL). The positive control (PC) sample was made the same way, while the negative control (NC) used methanol instead of Gel0.1CurAg. The sample's absorbance at 517 nm was measured using a UV-vis spectrophotometer (BioMate 3S, Thermo Scientific, USA). The capacity of antioxidants in the sample was calculated by the equation in previous research. 19 The IC50 value was the estimated concentration of the sample to scavenge the 50% DPPH free radical.
Cytotoxicity analysis
The cytotoxicity of Gel0.1CuAg on cells was measured using the SRB Assay in Cell Culture, as already reported. 20 SK-MEL-28 cells were seeded in a 96-well plate at a density of 4 × 103 cells per well and incubated at 37°C with 5% CO2 for 24 h. Following that, cell wells were added to the GelCuAg at varied concentrations of 3, 6, 12, 25, 50, and 100 µg/mL and continuously incubated for 48 h in an environment of 5% CO2 at 37°C. The cells were then fixed with 10% TCA on a well plate for 1 h before being stained with a 0.04% SRB solution for 15 min. Acetic acid (1%) was utilized to remove the remaining chemicals and solvents from the cell plates. Finally, using 10% Tris-HCl for dissolving the SRB crystals and calculating the absorbance of samples at 510 nm by an ELISA equipment (BioTech Power Wave XS, Winooski, VT, USA).
Antibacterial assay
The antibacterial activities of the samples were determined by the agar-well diffusion method, according to Mounyr Balouiri, 2018. 21 The Gel0.1CurAg at various silver concentrations of 0.23, 0.15, and 0.076 (µg/mL). Distilled water was used as a negative control (NC). Gel0.1Cur and curcumin (80 µg/mL) were used as various controls for comparison with samples containing silver nanoparticles. The positive control (PC) for E.coli and P.aeruginosa was ampicillin (5 µg/mL); the PC for B.cereus, and S.aureus was Korixone (2 µg/mL). The volume of the sample used for the antibacterial test was 30 μL. The zone of inhibition was measured by using a decimal ruler in millimeters.
Statistical analysis
In this study, the results were shown as mean ± standard error of the mean (the zone of inhibition of bacteria). Every experiment was repeated three times, and the data acquired were statistically assessed using suitable procedures.
Results
Characterization of GelCurAg solution
UV-vis spectroscopy, one of the most commonly used methods for determining the structural properties of AgNPs, was utilized to characterize them. The UV-Vis spectrum at wavelengths of 300–600 nm is often used to indicate the presence of silver nanoparticles, while the peak appeared at the 410–420 nm wavelength. In this study, the samples contained various concentrations of gelatin (0.05, 0.1, 0.2, and 0.5), and the one peak at 420 nm indicated the presence of silver nanoparticles in all samples. Independent experiments were conducted in the respective Figure 1. The nanosilver content is proportional to the absorbance value at 420 nm. The absorbance value of the Gel0.1CurAg solution was the highest, meaning the silver nanoparticle concentration in the samples was the greatest. Consequently, the concentration of gelatin was 0.1% used for further experiments. In contrast, in the control sample not exposed to UV, silver nanoparticles were not formed. Therefore, the UV-VIS spectrum, at wavelengths of 300–600 nm, did not show the peak appearing at 410–420 nm (Figure 1).

Independent experiments were conducted on the respective UV-vis absorption spectra of the GelCurAg (the gelatin microgel-stabilized silver nanoparticles loaded with curcumin) and the GelAg with various wavelengths (ʎ) in the range of 300-600 nm.
Independent experiments were conducted in the respective Figures 2 A, B, C; the FTIR spectra of the four test samples (Gel0.05CurAg, Gel0.1CurAg, Gel0.2CurAg, and Gel0.5CurAg) had the same peaks, and they were very different from the FTIR spectrum of curcumin, GelAg samples. This indicates that the curcumin was encapsulated inside a complex containing gelatin, silver nanoparticles, and PVP. The chemical functional groups OH, C = O, and C-O-C, corresponding to wavenumbers (cm−1) 3330.46, 1641.13–1644.02 cm−1, and 1013.41–1017.27 cm−1 were shown in the FTIR spectra of the 4 test samples (Gel0.05CurAg, Gel0.1CurAg, Gel0.2CurAg, and Gel0.5CurAg).22,23
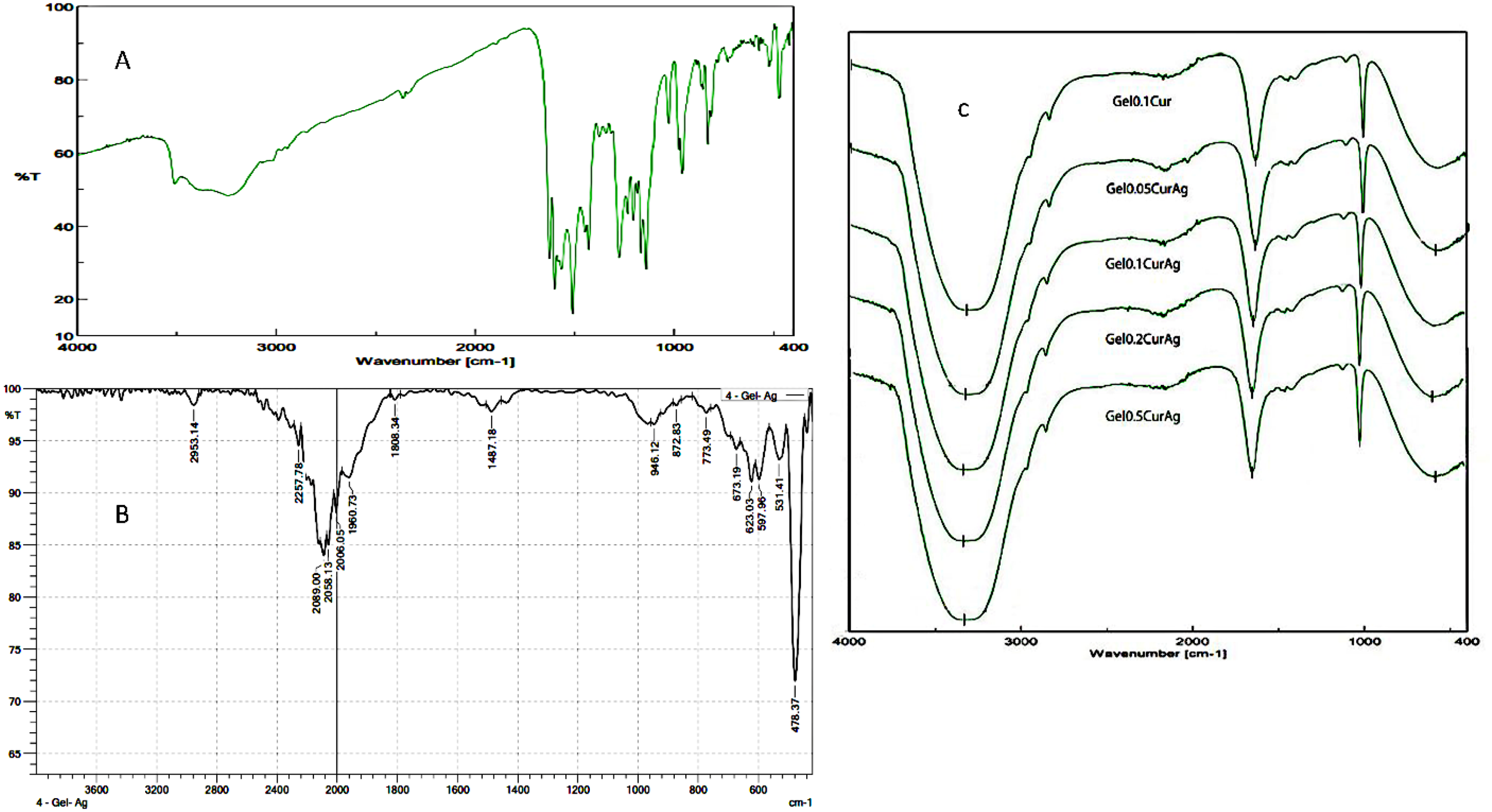
FTIR spectrum of curcumin (A), GelAg (B), and GelCurAg (C) (the gelatin microgel-stabilized silver nanoparticles loaded with curcumin) in the range of 0.05, 0.1, 0.2, and 0.5 (% w/v) gelatin.
Table 1 shows the size of the Gel.CurAg complex with the concentration of gelatin in the range of 0.05, 0.1, 0.2, and 0.5 (%) w/v, which was evaluated by a zetasizer instrument. The results shown in Table 1 indicated that the size and PDI (polydispersity index) of Gel0.1CurAg were the smallest. In addition, the particle size value and the PDI of the particles in the solution were important factors in determining the size uniformity of the nanoparticle. The polydispersity index is an important index describing the width and size range of the particle. In fact, the PDI value may range from 0 to 1. The smaller the PDI value, the more uniform the size of the nanoparticle, and vice versa. The smaller the size of particles, the better the dispersion of the particles in solution. Further, when using the gelatin ratio of 0.1% in complex Gel0.1.CurAg, the amount of silver nanoparticles was the highest (Figure 1). The Gel0.1Cu.Ag was chosen for further experiments.
Independent experiments showed the size of the GelCurAg complex at different gelatin concentrations in the range of 0.05, 0.1, 0.2, and 0.5% by using the zetasizer instrument.

We investigated the characterization of Gel0.1CurAg by using TEM images, zeta potential, and XRD patterns.
The Gel0.1CurAg complex's XRD pattern showed diffraction peaks at 2θ = 38.1°, 44.3°, and 64.4°. These peaks were related to the (111), (200), and (220) silver planes (JCPDS card No. 03-0921) (Figure 3A). TEM pictures at a magnification of 50,000X revealed the shape of the Gel0.1CurAg complex. According to the results in Figure 3B, the Gel0.1CurAg complex was spherical in shape and is approximately 50 nm in size. The size of the nanoparticles determined by TEM images is much smaller than that shown in Table 1. This difference may be due to the accuracy of the equipment and the dispersion of the nanoparticles in the solution. In addition, Zetasizer Nano (Malvern Instruments, Worcestershire, UK) determined the zeta potential of the Gel0.1CurAg complex to be around 11.2 (Figure 3C).
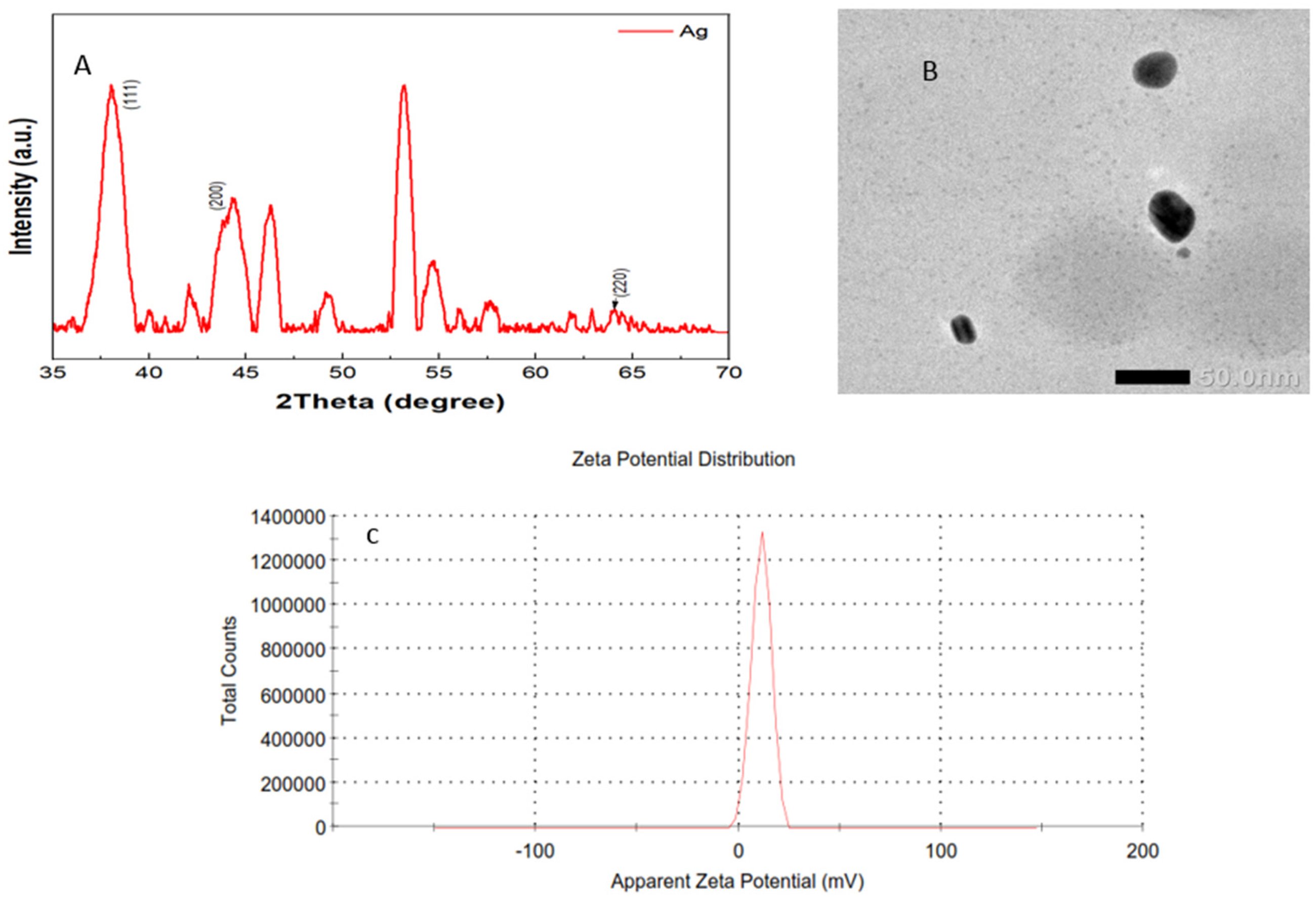
Characterize the XRD pattern (A), TEM image at a magnification of 50,000X (B), and the zeta potential (C) of the Gel0.1CurAg complex (the gelatin microgel-stabilized silver nanoparticles loaded with curcumin).
We continuously investigated the UV spectra of Gel0.1Ag, Gel0.1Cur, and Gel0.1CurAg in the range of 300–600 nm wavelength.
Figure 4 showed that when AgNPs and curcumin are combined, the absorption peak of Gel0.1CurAg is shifted slightly to the right (420 nm) due to the presence of silver nanoparticles. In the Gel0.1CurAg solution, the UV-vis spectrum was also a wide peak, and the absorbance is higher compared to Gel0.1Cur. The UV-Vis spectrum of the Gel0.1CurAg was investigated for the time exposed to UV under 10, 30, and 60 min (Figure 4). The result indicated that the peak at 420 nm was highest at 10 min. It may be a consequence of the decomposition of gelatin under long-term UV irradiation. When the gelatin molecules break down into small fragments, several of the AgNPs were not encapsulated for agglomeration into larger particles. As a result, the AgNPs concentration decreased, and the SPR band intensity decreased. Curcumin is extremely light-sensitive and breaks down rapidly in the presence of light. Therefore, when irradiated under a UV lamp for a long time, the absorbance decreases with each wavelength.

Independent experiments showed the UV spectrum of Gel0.1Ag, Gel0.1Cur, and Gel0.1CurAg complex (the gelatin microgel-stabilized silver nanoparticles loaded with curcumin) ranging from 300 to 600 nm wavelength (A) and the UV-vis spectrum of Gel0.1CuAg in different time-exposed UV light (B).
Antibacterial activity
The results of the antibacterial activity of the Gel0.1Cur and Gel0.1CurAg solutions at various silver concentrations are shown in Table 2. Overall, Table 2 indicated that the antibacterial ability of GelCurAg solution depends on the presence of silver nanoparticles because GelCur didn’t inhibit the growth of bacteria at the same concentration of curcumin. Likewise, at a silver concentration of 0.076 μg/mL, the Gel0.1CurAg solution also did not show clear antibacterial activity. When increasing the amount of silver in the GelCurAg solution, it may be seen that the antibacterial ability increases through the bacterial inhibition zone. The antibacterial inhibition zones ranged from 9 mm to 16 mm at concentrations of silver in the range of 0.15 μg/mL to 0.23 μg/mL. This property is clearly shown in E. coli and P. aeruginosa bacteria; both have resistance zones from 1.1 cm to 1.5 cm. The Gel0.1Cur solution has antibacterial activity against the E. coli strain and P. aeruginosa, while the Gel0.1CurAg is against 4 strains of bacteria, including E. coli, P. aeruginosa, S. aureus, and B. cereus.
Inhibition zones of samples against bacteria consist of E. coli, P. aeruginosa, S. aureus, and B. cereus.

“−” weak or no antibacterial activities; PC: positive control; NC: negative control.
Antioxidant activity of the samples
The DPPH method is commonly used to determine the antioxidant activity of compounds and extracts to evaluate their scavenging capacity for evaluating their capacity to scavenge free radicals. Table 3 shows that the IC50 value of Gel0.1CurAg was 4.87 µg/mL. The PC was use of ascorbic acid with an IC50 value of 4.9 µg/mL.
The antioxidant activities of Gel0.1CurAg solution.

Antiproliferative activity of the Gel0.1CurAg solution
The cytotoxicity activities of the Gel0.1CurAg solution were investigated on SK-MEL-28 cells and NIH 3T3 cells. The IC50 value on SK-MEL-28 cells and NIH 3T3 cells was IC50 = 3.98 µg/mL and 5.62, respectively. However, the toxicity difference of Gel0.1CurAg between SK-MEL-28 cells and NIH 3T3 cells was not significant.
Discussion
Poor solubility is one of the most challenging problems in the pharmaceutical industry. Numerous APIs have low bioavailability because of their insoluble properties. For many decades, nanotechnology was applied to create nanoparticle size and increase the solubility, dispersion, and bioavailability of APIs. The size of APIs is one of the most important features that must be determined before their application as medicines. Larger particles have a low total surface charge, resulting in less contact surface between the particle and solvent, poorer dispersion, and less solubility. Therefore, numerous techniques used in the fabrication and production of nanoparticles are applied in the biomedicine and pharmaceutical industries to reduce the size of material particles. 24 Techniques to change the size of particles are varied and abundant, depending on the characteristics of APIs. It may use physical or chemical impact or a combination of these methods to control the size of particles within certain limits.
Gelatin is a protein derived from the hydrolysis of collagen. It is a natural biofilm-forming agent with characteristics such as high biocompatibility and water solubility, low immunogenicity, flexibility, adhesion, and cell adhesion. Therefore, it is widely used as an excipient component for manufacturing drugs. Depending on its application, gelatin can be prepared in many forms, including films and microparticles or nanoparticles. 25
A variety of polymers have also been investigated as stabilizers for the synthesis of nanoparticles, for example, PVP. PVP has excellent characteristics such as biodegradable, water-soluble properties, superior binding properties, and a stabilizing effect for suspensions and emulsions.26,27 Hence, PVP was used in manufacturing drugs to improve the bioavailability of insoluble drugs, stabilization, and binding the different compounds in one complex. 27 Besides, several biopharmacological activities of silver nanoparticles, such as toxic effects, anti-cancer, and antimicrobial activities, have been reported in previous research.28,29
Many factors affect the stability of nanosilver in solution, including pH, metal ion concentration, and abundance of biomolecules. Destabilization of nanosilver solution leads to loss of biopharmaceutical activities. Therefore, using a stabilizing agent such as PVP in the synthesis of silver nanoparticles was one way to get desired colloidal stabilizers. 17 In the synthesis of nanoparticles, the size and the shape of AgNPs were significantly affected by the concentration of the polymers. In previous research, chemical, 30 physical, 31 and green methods 9 were used to synthesize silver nanoparticles from AgNO₃ solution. Recently, green methods have been widely used for the synthesis of silver nanoparticles in various medical applications. Silver nanoparticles were synthesized by using different plant extracts containing proteins, lipids, carbohydrates, carotenoids, vitamins, polyphenols, flavonoids, alkaloids, and saponins, serving as reducing agents to transform Ag + into Ag0 (silver nanoparticles).32–34 Silver nanoparticles are widely used in biomedical applications because of their low toxicity for human cells and stability over a wide temperature range. Green synthesis of silver nanoparticles is a method that has been developed recently to replace physical and chemical synthesis, which has many disadvantages, such as using toxic chemicals and consuming a lot of energy. 32 In addition, in this study, gelatin and UV radiation were used to create silver nanoparticles and attach them to the desired structure complex containing both component silver nanoparticles and curcumin.
Curcumin is a polyphenol derived from the roots of Curcuma longa L. that has been shown to have powerful antioxidant, anti-inflammatory, anti-cancer, and antibacterial properties. 1 However, curcumin has a limited bioavailability due to its low solubility in water and solvents. In addition, curcumin's bioavailability is easy to reduce by the impacts of pH, temperature, and light. Silver nanoparticles have excellent antibacterial properties, too. Curcumin is the most typical polyphenol and has a capacity to synthesize silver nanoparticles. In this work, we produced the GelCurAg complex at the nanoscale (50 nm) to improve curcumin solubility and dispersion in water, as well as the complex's antibacterial activity when compared to just the curcumin components. Further, curcumin encapsulated in the GelCurAg complex coated with PVP showed that it increases the stability and solubility of the complex in water. Hence, the result in Table 2 indicated that the GelCurAg complex has excellent antibacterial activities, while the single curcumin or silver had weak or no antibacterial activities at the same test concentrations.
The antibacterial activity of curcumin and silver has been noted in many previous studies30,31,35 against many bacteria strains, including Staphylococcus aureus, Escherichia coli, Enterococcus faecalis, Pseudomonas aeruginosa, Klebsiella pneumonia, Bacillus subtilis, Enterobacter aerogenes, and Proteus mirabilis. Because curcumin is poorly soluble in water and is limited by its bioavailability, the antibacterial activity of natural curcumin is weak in biomedical applications. Silver is in metallic form or silver salt, making it difficult to apply in the biomedical field, too. Therefore, the combination of curcumin and silver in a nanocomplex has enhanced the ability to synergistically act against bacterial strains.
Although curcumin and silver have antimicrobial properties in many previous studies, in this study, curcumin and silver were created in a bioavailable combination to increase their antimicrobial activities. Several previous studies have combined curcumin and silver nanoparticles for enhancing its bioavailability, pharmaceutical properties, and specificity compared to each separate component.36,37 This tendency has been an interesting research subject in recent years.
In addition, Gel0.1CuAg has antiproliferation properties on SK-MEL-28 with an IC50 value of 3.98 µg/mL and the antioxidant property of this complex was a good result. However, the antiproliferative activity of the GelCurAg complex on the SK-MEL-28 cell line is not really much different from that on the NIH 3T3 cell line. We will have further experimental cytotoxicity conducted on the GelCurAg complex on the different cancer cell lines in the future to evaluate better its antiproliferative properties. Developing a nanocarrier system to enhance the pharmaceutical properties of insoluble APIs is a valuable research goal. In this study, we succeeded in developing a GelCurAg complex on a nanoscale based on gelatin and PVP. The complex's enhanced bioavailability and antibacterial properties indicate its potential biopharmaceutical application not only for curcumin and silver nanoparticles; it also applies to other insoluble APIs in the same way. It suggests the development of an effective drug nanocarrier for biopharmaceutical applications. However, this study lacked sufficient data on complex stability and API release, which will be addressed in future experiments.
Conclusions
We successfully created the GelCurAg complex, including gelatin, curcumin, and silver nanoparticle biofilms with PVP in the average size of 50 nm. The GelCurAg complex increased the solubility and dispersion in water when combined with only a curcumin solution. It has antioxidant activity with an IC50 value of 4.87 μg/mL and antiproliferation on SK-MEL-28 with an IC50 value of 3.98 µg/mL. GelCurAg solution has superior antibacterial properties against 04 bacterial strains, including E. coli, P. aeruginosa, S. aureus, and B. cereus. The antibacterial activities of these solutions are better compared to the single curcumin component. The excellent biopharmaceutical activities of the GelCurAg complex expand the significant biopharmaceutical application directions of curcumin and silver nanoparticles. Although this study is not entirely about a nanocarrier for biomedicinal applications, we continue to improve our research to develop a drug delivery complex in nanocarriers in the future.
Footnotes
Acknowledgement
The authors thank MS.c. Bui Thi Van Khanh for technical assistance in cell viability.
Author's contributions
Conceptualization, financial support and supervision done by LNNT; formal analysis done by SCD and DHQ; investigation done by NTLN, SCD, NNH, DHQ, BTVH, VDM, and VTT; methodology done by LNNT, BTVH, and SCD; software done by NTLN, NNH, SCD, and VDM; validation done by NTLN; writing—original draft done by LNNT; review and editing done by LNNT, BTVH, SCD, and DHQ. All authors have read and agreed to in its published version.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The datasets used and analyzed during the present study are available from the corresponding author on reasonable request.
