Abstract
Head and neck cancer (HNC) is the sixth most common cancer in the world, and its incidence is increasing year by year. Due to the late-stage diagnosis and poor prognosis of HNC, as well as the limitations of traditional treatment methods, it is urgent to improve early detection rates and explore alternative treatment approaches. Graphene-based nanomaterials (GBNs) have been widely applied in biomedical fields due to their high surface area, excellent photothermal properties, and high loading capacity. This literature review introduces the functionalization and biocompatibility of GBNs, followed by a focus on their applications in the diagnosis and treatment of HNC. This includes their potential as bioimaging or biosensing platforms for diagnosis and monitoring, as well as their research progress in chemotherapy drug delivery, phototherapy, and gene transfection. The tremendous potential of GBNs as a platform for combination therapies is emphasized. Finally, in this literature review, we briefly discuss the toxicity and limitations of GBNs in the current research and provide an outlook on their future clinical applications in the diagnosis and treatment of HNC.
Introduction
Head and neck cancer (HNC), which includes cancers occurring in the oral cavity, pharynx, larynx, nose, salivary glands, and thyroid gland, is the sixth most common cancer globally, and 90% are head and neck squamous cell carcinomas (HNSCCs). 1 Due to the occult structure of head and neck tissues, as well as the lack of sensitivity and specificity in traditional diagnostic methods, two-thirds of patients are already in advanced stages when seeking medical treatment, and the 5-year survival rate after treatment is only 70–90%. 2 In addition to conventional imaging techniques and tissue biopsy, liquid biopsy, as a less invasive detection method, has also become an important new approach for cancer diagnosis that cannot be ignored. 3 For example, microRNAs, as a biomarker for HNC, have strong diagnostic and therapeutic capabilities. 4 Unfortunately, due to the heterogeneity of tumors, liquid biopsy-based biomarkers have not met the required sensitivity and specificity requirements in clinical trials, and have not yet been approved by the FDA for the detection of HNC. 5 Early monitoring methods for HNC still need to be further explored by researchers. Traditional treatment methods for HNC mainly include surgery, radiotherapy, chemotherapy, immunotherapy, and combination therapies. 6 Among them, surgical resection is the mainstream approach, but it is often accompanied by physiological function damage and various postoperative complications such as pharyngeal fistula, significantly reducing the patients’ quality of life. 1 Chemotherapy has also been used as a standard treatment option, but due to the non-targeting and short half-life of traditional chemotherapy drugs, they often exhibit serious toxic side effects and drug resistance, which greatly limits their long-term use. 7 In addition, the occurrence and development of HNC are related to immune escape and disruption of T cell signaling, and immunotherapy targeting this has become one of the treatment methods for HNC. 8 In clinical treatment, it is usually combined with other treatment methods such as radiotherapy, chemotherapy, and targeted therapy.9, 10 However, the off-target effects caused by complex genomics and genetic heterogeneity often lead to serious adverse reactions, coupled with the expensive cost of immunotherapy, which greatly limits its promotion.8, 10 Therefore, it is imperative to explore and develop new methods for the early diagnosis and treatment of HNC. With the continuous development of material science, nanomaterials have emerged as promising biomedical materials, mainly due to their ultra-small size (typically ranging from 1 to 100 nm), large surface area-to-volume ratio, and high tunability. 11 Among various nanomaterials, graphene-based nanomaterials (GBNs) stand out for their outstanding performance.
Graphene is a two-dimensional honeycomb lattice structure formed by a single layer of carbon atoms in a sp² hybridization pattern, with the p-orbitals of adjacent carbon atoms forming a π–π system. It is the world's thinnest new nanomaterial discovered so far, with a thickness of only 0.335 nm.12, 13 In 2004, Geim and Novoselov et al. 14 from the University of Manchester, UK, successfully isolated few-layer graphene (FLG) with a two-dimensional crystalline structure from graphite blocks using a mechanical exfoliation method, and this two-dimensional nanomaterial sparked a frenzy of excitement in the scientific community. As a two-dimensional crystal composed of a layer of carbon atoms, graphene has a unique atomic structure and mechanical properties compared with other polymer materials and is widely used in new energy batteries, 15 sensors, 16 displays, 17 coatings, 18 and composite materials 19 and other fields, which is well-deserved the king of materials. In 2008, Liu et al. 20 reported for the first time that graphene nanomaterials were used as carriers of poorly soluble and aromatic anticancer drug (SN38), demonstrating their enormous potential in the field of biomedicine and attracting widespread attention from scientists. GBNs, represented by graphene, graphene oxide (GO), reduced graphene oxide (rGO), and graphene quantum dots (GQDs), have become promising nano-platforms for various anticancer drug delivery and cancer treatment due to their good biocompatibility, large surface area, tunable functional groups, and excellent photothermal, electrical, and mechanical properties, 21 which endow them with easy modification, high drug-loading rate, 22 and strong photothermal conversion efficiency. 23 Furthermore, the novel imaging and sensing technology of graphene-based multifunctional nanocomposites can help to better understand the pathophysiology of cancer, 24 and it also exhibits unique performance in the diagnosis and combination therapy of cancer,25, 26 providing an exciting opportunity to improve the prognosis of HNC patients. 13 Therefore, GBNs bring new approaches to the diagnosis and treatment of HNC (Figure 1).

Application of graphene-based nanomaterials (GBNs) in the diagnosis and treatment of head and neck cancer (HNC).
In this narrative literature review, we briefly summarize the functionalization methods of GBNs to enhance their biocompatibility. Additionally, we focus on the research progress of GBNs in the diagnosis and treatment of HNC, including chemotherapy drug and gene delivery, phototherapy (including photodynamic therapy and photothermal therapy), and combination therapy, emphasizing the GBNs as good conductors of heat and electricity to become popular materials for cancer imaging and detection. Lastly, we discuss the toxic effects and limitations exhibited by GBNs in animal experiments. This narrative review aims to stimulate broader interest in GBN-related applications and provide new insights for future diagnosis and treatment of HNC. This review is guided by the Scale for the Assessment of narrative review articles (SANRA). 27
Functionalization modification and biocompatibility of GBNs
As with any new material, understanding the biocompatibility of GBNs in vitro and vivo is an essential part of their journey toward clinical applications. 28 Although graphene has been widely applied in numerous fields due to its unique properties, its strong hydrophobicity severely affects its dispersibility in water, which limits the application of pure graphene in biomedical fields. 13 GO is the oxidized form of G, containing abundant oxygen-containing groups such as hydroxyl, carboxyl, and carbonyl. Compared to pure graphene, it has superior dispersibility in water. The presence of carboxyl and hydroxyl groups also makes it easier to modify and functionalize, making it an excellent material in the field of nanomedicine and having broad development prospects. 29 So researchers have explored appropriate surface functionalization or modification of GBNs to improve their water solubility, biocompatibility, and stability, thus enhancing their biomedical value. 29
Covalent and non-covalent functionalization modification are the two main strategies for modifying graphene and its derivatives. 30 Covalent modification typically refers to grafting the oxygen-containing functional groups of GBNs with functional macromolecules with high hydrophilicity through covalent bonding to enhance their biocompatibility. 12 The graphene polymer hybrid nanomaterials obtained by Roberto Muñoz et al. through two-stage covalent functionalization have been shown to improve the hydrophobicity of graphene while enhancing biocompatibility and have the potential to become a platform for resistive humidity sensors. 31 Another research team conducted sequential covalent modification of GO with dual functional groups and found that it has better stability compared to non-covalent modification, making it more suitable for coupling biomolecules (such as drugs, proteins, and peptides) for biomedical applications. 32 Non-covalent modification, on the other hand, involves the physical adsorption of polymers or biomolecules (such as various peptides, enzymes, and DNA) onto GBNs using non-covalent forces (such as π–π stacking, hydrogen bonding, van der Waals forces, and electrostatic interactions) in order to improve the stability and safety of GBNs while reducing their cytotoxicity.12, 33 Commonly used polymers for surface functionalization include polyethylene glycol (PEG), polyethyleneimine (PEI), chitosan (CS), and hyaluronic acid. Among them, PEG, as a hydrophilic and biocompatible polymer, is the most extensively studied for modifying GBNs.29, 34 In 2008, Liu et al. synthesized and modified nanographene oxide (NGO) using PEG and loaded SN38 through π–π stacking, resulting in the formation of NGO-PEG-SN38 composite. This composite not only efficiently kills tumor cells but also possesses satisfactory biosafety and water solubility, which exemplifies the significant advantages of PEG-functionalized GBNs. 20 PEI is another polymer widely used for surface modification of GBNs. Feng et al. 35 functionalized GO using PEI through non-covalent electrostatic interactions. The resulting GO-PEI composite exhibited improved physiological stability compared to GO, reduced cytotoxicity, and enhanced gene transfection efficiency. According to reports, GBNs can be non-covalently functionalized with compounds with antibacterial activity to improve their antibacterial performance. 36 When amino-functionalized, it can reduce the toxicity of the material itself and serve as a highly selective and sensitive fluorescent probe for detection.37, 38 In addition, the nano materials generated by non-covalent functionalization of GBNs with metal nanoparticles and certain polymers can be developed into various biosensors and biosensors.39, 40 Therefore, researchers can choose the most suitable surface functionalization strategy for GBNs based on their specific goals and requirements, aiming to improve biocompatibility and enhance the cell-materials interactions, making them an excellent nanomaterial for applications in HNC.
The application of GBNs in the diagnosis of HNC
Currently, the detection methods for HNC mainly rely on endoscopy, imaging techniques, and tissue biopsy; however, the majority of patients are diagnosed at advanced stages and the late survival rate is only 20–60%. 5 Early detection of cancer can significantly improve the cure rate. Therefore, it is necessary to develop and innovate early detection methods for cancer. 41 The advancement in nanotechnology has opened up new possibilities in this regard. In recent years, the unique physicochemical properties, large specific surface area, and good biocompatibility of GBNs have made them a powerful nanomaterial for applications in cancer diagnosis, including biosensing and bioimaging. 42
Bioimaging
Imaging is the first step in cancer diagnosis and treatment. GBNs have been reported to be used in a variety of bioimaging systems due to their excellent properties.43–45 Among them, GQDs have received great attention in the field of biomedical imaging due to their wide absorption spectrum in the visible and near-infrared range, certain intrinsic enzymatic activity, and unique structure.46–48 Table 1 lists several studies of GBNs applied to bioimaging of HNC.
Application of GBNs in bioimaging.

Note: FA, folate acid; FR, folate receptor; PAI, Photoacoustic imaging; GE11, an EGFR antagonist peptide; FLI, fluorescence imaging; AF750-6Ahx-Sta-BBN, a GRPR-specific Alexa Fluor 750 (AF750)-labeled bombesin (BBN) antagonist.
Ding et al. 49 designed an exosome-mimetic nanoenzyme vesicle using the intrinsic peroxidase-like activity and surface grafting of GQDzyme and modified it with folate (FA)-conjugated natural erythrocyte membrane. They demonstrated its potential as a non-toxic, safe, and efficient tumor-targeted H2O2-responsive photoacoustic imaging (PAI) contrast agent. It not only successfully targeted the folate receptor (FR) overexpressed in nasopharyngeal carcinoma CNE-2 cells but also observed an increasing photoacoustic signal with prolonged time in vivo experiments on tumor-bearing mice, with a significantly increased half-life (>24 h). Another study constructed a GE11-modified GQDs nanoprobes targeting the highly expressed EGFR on CNE-2 cells, and its uptake in tumor cells and the delivery and release process of doxorubicin (DOX) were sensitively detected by a fluorescence resonance energy transfer (FRET) system. One hour after intravenous injection, a distinct fluorescence signal could be observed in the tumor. 50 This indicates that imaging-guided targeted drug delivery is a feasible strategy to reduce false-positive signals of tumor imaging and minimize the toxic side effects of chemotherapeutic drugs in normal cells, achieving the integration of diagnosis and treatment. 52 In addition, Li et al. 51 synthesized a nanoprobe by conjugating a gastrin-releasing peptide receptor (GRPR)-specific bombesin (BBN) antagonist with nanographene oxide (NGO) via hydrogen bond and π–π bonds. Immunofluorescence analysis demonstrated that it exhibited bright and stable fluorescence when internalized into oral squamous cell carcinoma HSC-3 cells. In conclusion, targeted nanoparticles hold great potential for enrichment and precise imaging in tumor tissues, making them promising fluorescent probes for labeling cancer cells.
The bioimaging capability of GBNs provides a better option for the in vivo detection of HNC. However, there is still limited literature on this topic, and more studies are needed in the future to confirm the application potential of GBNs.
Biosensing
Cancer biomarkers are cancer-related biomolecules (such as proteins, nucleic acids, and enzymes) expressed in human cells, tissues, or body fluids. They play a crucial role in cancer screening, early detection, and guiding treatment. 53 Biosensors, with their lower limits of detection (LOD), achieve higher sensitivity in detecting biomarkers and have the potential to overcome the limitations of current detection methods. Therefore, they are used for early diagnosis of cancer. 54 Due to their large photothermal conversion area, excellent electrochemical performance, and low toxicity, GBNs have been developed and designed as cancer biosensors for detecting important biomolecules. 55 Especially GO, based on its large volume-to-surface ratio, efficient fluorescence quenching properties, and good specificity and stability, has been developed into biosensors for various applications,56, 57 including the detection of protein, 58 glutathione, 59 cancer markers, 60 and genes. 61 Table 2 summarizes the applications of GBNs in the field of biosensors for HNC in recent years.
Application of GBNs in biosensing.

Note: OC, oral cancer; FAM, a fluorescent dye; HCR, hybridization chain reaction; NH2-IL-rGO-ssDNA, amine-ionic liquid functionalized reduced graphene oxide; MEL, melamine; BSA, bovine serum albumin.
In 2011, Wang et al. 68 developed a GO-based fluorescent dye-conjugated oligomer probe, which achieved high sensitivity and selectivity in the detection of Escherichia coli (E. coli). Subsequently, a large number of GO-based fluorescent biosensors have emerged and been applied in various medical fields. MicroRNA-205 (miR-205) is an important biomarker for evaluating radiation resistance in nasopharyngeal carcinoma (NPC), and the development of new methods for detecting miR-205 is of significant importance for individualized therapy of NPC. 69 GO exhibits efficient fluorescence quenching properties, and the fluorescence signal of the dye-labeled single-stranded DNA (ssDNA) probe which is bound to GO through π–π stacking can be rapidly quenched. The fluorescence signal accumulated by the hybridization of the target with the probe enables the achievement of the detection purpose. 70 Yang et al. 63 adsorbed FAM-modified oligonucleotides as recognition elements on the surface of GO, forming a highly sensitive fluorescence-sensing platform for detecting miR-205 with a detection limit of 1.18 nM. The team also discovered that the binding of only 0.3 U duplex-specific nuclease (DSN) to the GO sensor could achieve incredible fluorescent signal cascade amplification. 64 In addition, a novel fluorescence biosensor based on GO and hybridization chain reaction (HCR) designed by Qin et al. also achieved fluorescence signal amplification in a cost-effective manner and demonstrated the superior ability of miR-205 for imaging and localization in living cells. 65 The development of these GO-based biosensors aimed at capturing the biomarker miR-205 provides a promising platform for early detection and clinical diagnosis of NPC. However, time-consuming fluorescent labeling and complex chemical modifications still pose some limitations to GO-based biosensors.71, 72 In addition, the biosafety issues of GO also limit its clinical applications, posing challenges to its industrialization. 73
Graphene-based electrodes offer advantages over other carbon-based nanomaterials in terms of electrocatalytic activity and electrical conductivity, thus providing graphene with great opportunities in the field of electrochemistry. 74 Its role in the diagnosis of HNC has been demonstrated in research. Previously, researchers developed a gold nanomaterials coupled titanium dioxide (TiO2) specific electrode sensor for detecting HNC biomarker squamous cell carcinoma antigen (SCC-Ag), demonstrating its high sensitivity and selectivity. 75 Similarly, Verma et al. 62 utilized the synergistic properties of gold nanoparticles (AuNPs) and rGO to fabricate an electrochemical immunosensor using AuNPs-rGO as a nanocomposite transducer for non-invasive detection of oral cancer biomarker IL-8. This biosensor has been proven to be a rapid (9 minutes), portable, cost-effective detection method with a wide linear range. In recent years, an increasing number of head and neck squamous cell carcinomas (HNSCC) have been found to be associated with human papilloma virus (HPV) infection. 76 Researchers have explored the detection of PYHIN gene family members as DNA sensors in HPV-positive HNSCCs and found that the expression of interferon-inducible protein 16 (IFI16) and melanoma 2 protein (AIM2) may be associated with HPV infection, but the complexity of genes and interactions between proteins may become confounding factors that affect experimental results.77, 78 HPV16 is the most common high-risk HPV subtype associated with HNC. 76 Farzin et al. 66 designed an electrochemical gene sensor for the detection of HPV16 DNA in HPV16-positive HNC patients. This sensor utilized a composite material of multi-walled carbon nanotubes (MWCNT) and amine-ionic liquid functionalized reduced graphene oxide (NH2-IL-rGO) to provide a high specific surface area nanoplatform for hybridization between the ssDNA probes and the target HPV16 DNA strands. The sensor demonstrated ultra-high sensitivity with a minimum detection limit of 1.3 nM. In addition, Liquid biopsy based on saliva biomarker detection has been proposed as a non-invasive diagnostic method for the detection of HNC, especially oral cancer. 79 Edoardo Farnesi et al. characterized interleukin-8 (IL-8) and lysozyme (LYZ) in saliva using a biosensor based on surface-enhanced Raman scattering (SERS) and molecular dynamics (MD) and explained their relationship with oropharyngeal squamous cell carcinoma (OSCC). 80 Relying on the convenience of saliva liquid biopsy, a portable and easy-to-use electronic biosensor based on rGO has been developed for the rapid, efficient, and repeatable quantitative detection of two biomarkers (CEA and CYFRA 21-1) in oral OSCC. 67 Due to the multifactorial nature of cancer, simultaneous detection of multiple biomarkers is often necessary. 54 The low concentration of biomarkers in saliva makes the development of biosensors based on liquid biopsies challenging; therefore, biosensors with high sensitivity and low detection limits will be the direction of researchers’ future efforts. 79
The rational design and optimization of these GBN-based sensors have undoubtedly opened up new frontiers in the field of biomarker research and explored their potential in cancer diagnosis. In this context, it is expected that we can develop cancer biomarker monitoring devices with higher specificity, higher sensitivity, affordability, and portability, thereby improving the early detection rate and prognosis of HNC.
The application of GBNs in the treatment of HNC
Due to their unique photothermal properties, high biocompatibility, and affordable cost, GBNs are considered to have great potential to overcome the limitations of traditional cancer treatment methods, so the development of novel cancer therapies based on GBNs has gained widespread research attention 81 . Here, we review various strategies involving GBNs in the treatment of HNC in recent years.
Chemotherapy
In recent years, chemotherapy has been used as a standard treatment option with good anticancer efficacy. However, traditional chemotherapy drugs have shortcomings such as non-targeting, instability, and short half-life, which often lead to drug-related toxic side effects and drug resistance, greatly limiting their effectiveness in the treatment of HNC. 7 Therefore, it is imperative to explore and develop a targeted drug delivery system that can accurately target tumors. Thanks to the exceptional drug-loading capacity of graphene nanomaterials, reaching up to 200%, they have been designed as efficient delivery and sustained-release platforms for various anticancer drugs 82 ; moreover, different functionalization strategies have endowed them with controllable targeted delivery capabilities while enhancing the stability and biosafety of the system. 83 Figure 2 illustrates the mechanism of GBNs mediated targeted delivery of chemotherapy drugs to HNC cells. Table 3 summarizes the applications of GBNs in HNC chemotherapy in recent years.

Mechanism of GBNs mediated targeted delivery of chemotherapy drugs to HNC cells.
Applications of GBNs in chemotherapy.
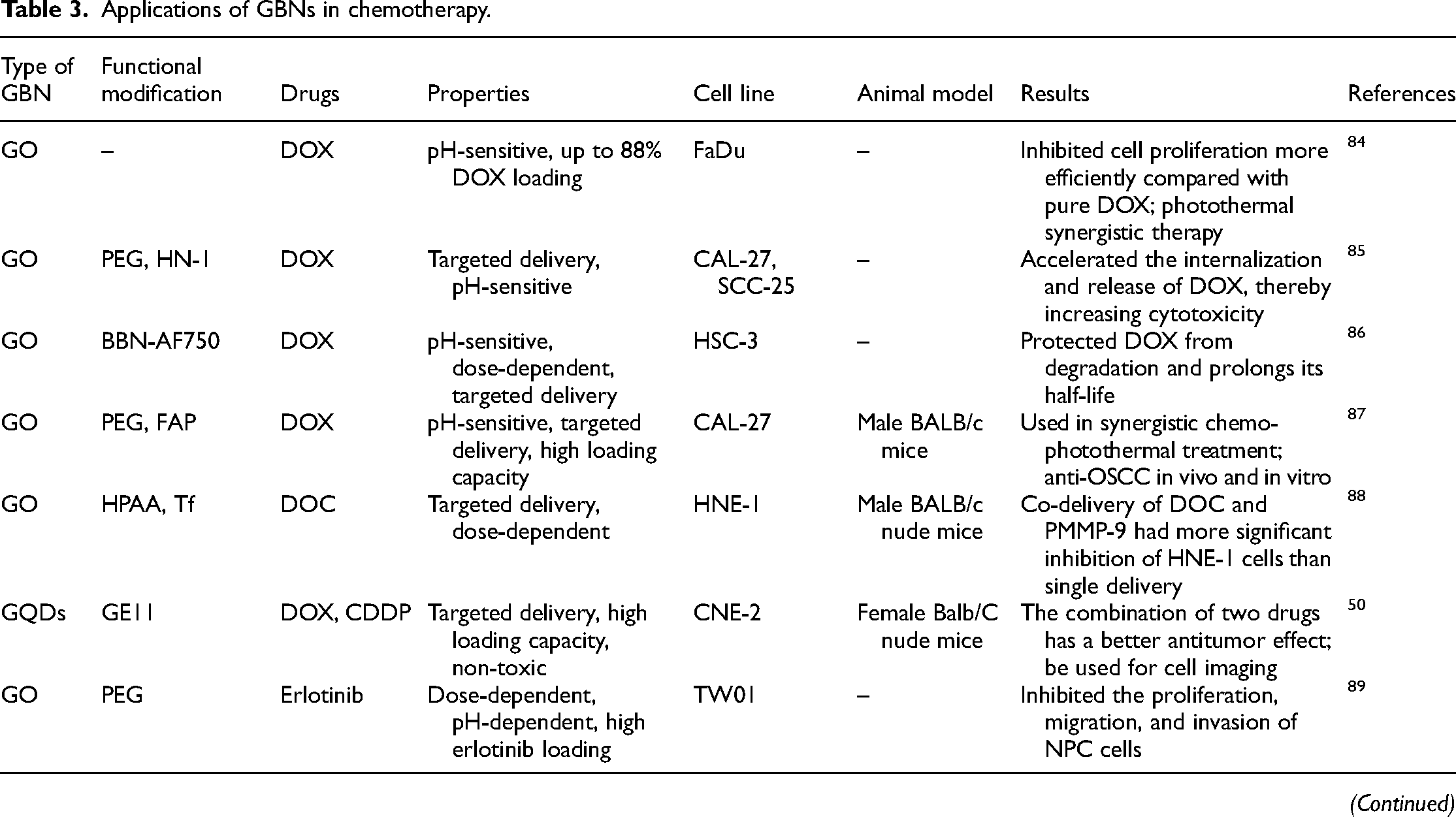
Note: HN-1, a 12-amino acid peptide TSPLNIHNGQKL; BBN-AF750, bombesin antagonist peptides conjugated to Alexa Fluor 750; PMMP-9, matrix metalloproteinase-9 shRNA plasmid; GE11, an EGFR antagonist peptide; MPC, 2-(methacryloyloxy)ethyl phosphorylcholine.
The tumor microenvironment typically presents an acidic pH (pH 6.5–6.8), and pH-sensitive drug delivery systems constructed based on the pH difference between tumor tissue and normal cells can significantly improve the bioavailability of anticancer agents while reducing toxic side effects on normal cells. 93 The toxic side effects of DOX and the possible multi-drug resistance of HNC subtypes have limited the therapeutic efficacy of DOX in HNC. 94 Singh et al. 84 designed a pH-responsive nanoconjugate by coupling DOX to GO nanosheets, which exhibited higher anti-proliferative ability against FaDu hypopharyngeal cancer cells and achieved a drug release rate of up to 88% under acidic conditions. Li et al. 85 explored the controlled release of DOX in oral squamous cell carcinoma cells (CAL-27 and SCC-25 cell lines) using a GO nanosystem, and DOX also achieved a release rate of 70% under acidic conditions and rapidly internalized into tumor cells. All these findings demonstrate the potential of pH-sensitive GO-DOX in the treatment of HNC. Additionally, the introduction of the targeting functional peptide HN-1 (TSPLNIHNGQKL) enabled DOX to precisely target tumor cells. Surface modification with targeting ligands has been shown to enhance the ability of nanosystems to deliver drugs accurately to tumor cells. The research team also reported a dual-sensitive GRPR-targeted GO delivery system loaded with the pro-apoptotic peptide BBN-AF750 and the anticancer drug DOX. In the multifunctional synergistic treatment of HNSCC, DOX was slowly released in HSC-3 cells, achieving precise tumor targeting and visualized therapy. 86 Recently, this team synthesized a novel nanoparticle delivery system (NPF@DOX) by loading fibroblast activation protein (FAP) targeting peptide onto PEG-functionalized graphene oxide. This system not only achieved a high drug-loading rate and reduced the off-target effects of DOX but also demonstrated synergistic enhanced antitumor effects through combined photothermal therapy both in vitro and in vivo. 87 Targeting molecule transferrin (Tf) has also been demonstrated to enhance tumor targeting sensitivity. Liu et al. 88 modified GO with redox hyperbranched poly(amido)amine (HPAA) and coupled it with Tf to achieve chemotherapeutic sensitization. Compared to free docetaxel (DOC), this delivery system, Tf-HPAA-GO, exhibited higher targeting capacity for DOC and increased cytotoxicity against nasopharyngeal carcinoma cells. Furthermore, graphene quantum dots modified with EGFR antagonistic peptide Ge11 (GQDs@Ge11) could simultaneously deliver cisplatin (CDDP) and DOX to nasopharyngeal carcinoma CNE-2 cells, demonstrating stronger anticancer activity than using single drugs. 50 The dual drug delivery system based on PEGylated nano graphene oxide (pGO) could also attenuate the toxicity of CDDP and DOX on normal cells. 95 Thus, these findings highlight the vast potential of GBNs in the field of cancer treatment, particularly in synergistically controlling the targeted delivery of multiple chemotherapy drugs.
PEG, as a non-toxic and non-immunogenic polymer material, is commonly used in drug delivery and drug modification to improve pharmacodynamic properties and drug distribution in vivo and to enhance drug solubility. 96 Lan et al. 89 loaded erlotinib onto PEG-coated graphene oxide (GO-PEG) and found that this drug carrier significantly reduced the IC50 of erlotinib in TW01 cells. It achieved a significant inhibitory effect on nasopharyngeal carcinoma cells with a lower dose of erlotinib and exhibited pH and dose-dependent responses. Cisplatin, commonly used in chemotherapy for HNC, often causes renal function damage and gastrointestinal side effects, limiting its long-term use. 97 Therefore, carboplatin with high biosafety is usually used instead of cisplatin in clinical practice.98, 99 However, due to its low price and wide antitumor range, cisplatin still plays an important role in the field of antitumor. Platinum-based nanomedicine and its targeted modification undoubtedly point out a bright path for future anticancer therapy. 100 Researchers have utilized PEGylated GQDs to load cisplatin for the treatment of OSCC, demonstrating that PEG-functionalized nanomaterials improve the intratumoral accumulation of cisplatin, reduce toxic side effects, and successfully counteract chemotherapy resistance under hypoxic conditions, thereby achieving stronger cisplatin-mediated in vivo and in vitro killing of oral squamous cell carcinoma cells. 90 Multifunctional PEGylated GQDs have improved the size and morphology of nanosystems and exhibit good photostability, making them a promising candidate for multimodal anticancer therapy. 46 By introducing hydrophilic polyethylene glycol methacrylate (PEGMA) onto the surface of situ graphene nanocarriers modified with iron oxide nanoparticles (IONPs), it was possible to improve the solubility of DOX and increase the targeting sensitivity to thyroid cancer cells, which resulted in the inhibition of proliferation and induction of apoptosis of CAL62 cells. 91 PEGylated drugs have made significant progress, providing patients with more and better therapeutic options for cancer treatment. Besides, chitosan, which is biodegradable and biocompatible, has also been used for functionalized modification of GBNs. By using CS-functionalized encapsulated graphitic carbon nanocages (GCNCs), Li et al. 92 successfully increased the drug-loading capacity while improving the sustained-release effect of 5-fluorouracil (5-FU) in the treatment of NPC.
In summary, scientists have loaded cancer chemotherapeutic drugs onto GBNs and successfully constructed a new drug delivery system for the treatment of HNC by functionalizing various polymers and introducing targeting ligands. These multifunctional systems exhibit excellent biocompatibility and realize efficient targeted delivery of chemotherapeutic drugs, which perfectly overcome the limitations of traditional chemotherapeutic regimens. Unfortunately, graphene-based nanodelivery platforms loaded with commonly used chemotherapy drugs for head and neck cancer (such as paclitaxel, docetaxel, methotrexate, etc.) are rarely studied in head and neck cancer, while there are more studies in lung cancer and breast cancer.101, 102 This potential of GBNs provides us with new ideas for future research into new treatment strategies for head and neck cancer.
Phototherapy
Traditional cancer treatment modalities, including surgical resection, radiotherapy, and chemotherapy, have limitations such as multiple contraindications and severe adverse reactions, which fail to significantly improve the quality of life for patients. 103 Phototherapy, including photothermal therapy and photodynamic therapy, has demonstrated significant superiority as a non-invasive, low-toxicity, and short-cycle tumor treatment method. 104 GBNs with excellent optical properties have been explored for phototherapy in HNC, and their mechanism is shown in Figure 3. Table 4 summarizes the applications of GBNs in phototherapy in recent years.

The mechanism of GBNs in phototherapy for HNC.
Application of GBNs in phototherapy.

Note: Fe3O4@Au/rGO NSs, Fe3O4@Au/reduced graphene oxide nanostructures; RT, radiotherapy; NGODs-AA, ascorbic acid-assisted nitrogen-doped graphene oxide dots; CIS, cisplatin; PGO, plasmonic graphene oxide.
Photothermal therapy (PTT)
PTT is a cancer treatment technique that utilizes the conversion of absorbed near-infrared (NIR) light energy into thermal energy in the presence of a photosensitizer to locally heat cancer tissues, thereby enhancing tumor-killing efficiency. 110 Due to the attractive features of high absorption capacity in the NIR region, good biocompatibility, and ease of functionalization, GBNs can be used as photosensitizers or carriers for photosensitizers in cancer PTT, 12 especially rGO, which has several times higher NIR light absorption capacity than GO due to its smaller size of 20 nm. 111 Hu et al. 112 applied dopamine-functionalized rGO loaded with the photosensitizer indocyanine green for the PTT of breast cancer, demonstrating that this composite material exhibited stronger photothermal effects compared to pure rGO or dopamine-rGO. Li et al. 92 demonstrated that both GCNCs and GCNCs/CS, with good biocompatibility, can serve as excellent photosensitizers for PTT of NPC. Under 808 nm laser irradiation, the nasopharyngeal carcinoma cells of the tumor-bearing mice could be rapidly eliminated, and it was found for the first time that increasing the initial temperature can achieve more significant photothermal conversion effects. Nanoparticles containing noble metals, such as AuNPs, have been widely used for the treatment of HNC due to their multifunctionality and efficient photothermal conversion efficiency. 113 Ardakani et al. 105 coated Fe3O4@Au nanoparticles on rGO nanosheets, resulting in Fe3O4@Au/rGO composites with enhanced biocompatibility and photothermal effects, achieving a high photothermal conversion efficiency of up to 61% and effectively killing hypopharyngeal cancer KB cells. It is evident that GBNs have a very promising application prospect in the field of PTT in HNC.
Photodynamic therapy (PDT)
PDT, as a promising tumor ablation treatment method, has been clinically used to treat various cancers, such as skin cancer, 114 esophageal cancer, 115 breast cancer, 116 and bladder cancer. 117 It relies on the cytotoxic effect induced by reactive oxygen species (ROS) generated by the photosensitizer under light irradiation to kill cancer cells. 110 The environment of tumor cells is often hypoxic, and ROS, as a tumor suppressor, creates a toxic environment for them. 118 GBNs have been demonstrated to overcome the drawbacks of conventional photosensitizers in terms of poor water solubility and stability, making them useful candidates as photosensitizers for PDT. 119 In an experimental study, nitrogen-doped graphene oxide dots (NGODs) were utilized as photosensitizer, and the composite material NGOD-AA, constructed with the assistance of a hole scavenger ascorbic acid (AA), exhibited remarkable cytotoxicity and photodynamic effects against head and neck cancer cells (HONE-1), almost all of them were killed within 10 minutes of treatment, while normal cells remained unaffected. The high concentration of H2O2 generated by NGODs under white light irradiation has been proven to be the cause of inducing apoptosis and necrosis in cancer cells. 106 GQDs have been proposed as a promising photosensitizer due to their unique optical and catalytic properties. 120 Zhang et al. designed PEG-conjugated GQDs, which showed excellent photoluminescent characteristics and high efficiency in generating singlet oxygen (1O2) under 560 nm laser irradiation, demonstrating the tremendous potential for PDT in OSCC both in vivo and in vitro. 107 The small-sized GQD-PEG has also been proven to facilitate the penetration of the photosensitizer zinc phthalocyanine (ZnPc) and its accumulation in the nucleus of tumor cells, thus enhancing ROS-induced cytotoxicity. 108 PDT has shown remarkable success in enhancing cancer-specific cell killing and reducing toxic side effects, making it a prominent cancer therapeutic approach.
Combination therapy
PTT and PDT have emerged as novel tumor treatment modalities with advantages such as non-invasiveness, high targeting specificity, and minimal adverse reactions. However, their standalone use faces limitations in achieving complete eradication of the entire tumor tissue and concerns regarding the biosafety of photothermal materials. 104 To overcome the limitations of monotherapy, a series of phototherapy-based multimodal combination therapies have been extensively investigated. GBNs, which are easily functionalized, serve as ideal candidates as both photosensitizers and drug carriers, allowing for the combination of chemotherapy and PTT to achieve a synergistic effect of “1 + 1 > 2”. Studies have demonstrated that PEG-functionalized GO nanosheets loaded with DOX have achieved synergistic anticancer effects of PTT and chemotherapy, which are more effective than standalone chemotherapy or PTT. 121 The PEI-functionalized graphene oxide hydrogel loaded with DOX can overcome the limitations of chemotherapy drugs through the enhanced permeability and retention (EPR) effect. This combination of chemotherapy and PTT reduces breast cancer cells to 33%. 122 In the study by Li et al. 87 , GO-PEG functionalized with DOX and FAP-targeting peptide through π-π bonds and hydrogen bonds was used as a photosensitizer. In vitro and in vivo experiments demonstrated that the efficacy of DOX combined with PTT in treating OSCC was greater than using either treatment alone. This system precisely targeted CAL-27 cells, thereby minimizing toxic side effects. They also found that NIR radiation triggered the pH-dependent release of DOX within tumor cells. In another study, GCNCs/CS loaded with 5-FU were used for chemo-photothermal synergistic therapy of NPC, and the results showed that no more tumors were detected in CNE tumor-bearing mice after 6 days of 808 nm laser irradiation. The combined effects of 5-FU and phototoxicity suppressed tumor cell growth. 92 Recently, researchers have used plasma graphene oxide (PGO) as a photothermal agent mixed with cisplatin to construct a 3D-printed implant, which effectively combined chemotherapy and photothermal therapy to reverse the chemotherapy resistance of cisplatin in oral cancer treatment, significantly reducing the survival rate of tumor cells while also observing an increase in the expression of pro-apoptotic genes. 109 It can be seen that the tumor synergy therapy participated by GBNs can increase the lethality of chemotherapy to tumors on the basis of PTT and maximize the antitumor efficacy, which provides a new and effective strategy to enhance tumor eradication for the treatment of HNC.
Tumor immunotherapy is able to activate the defense system to attack and eliminate tumors, and the combination of phototherapy and immunotherapy has opened up a new era in the treatment of HNC. 123 Zhang et al. 107 used GQD-PEG as a photosensitizer. Under the 560 nm laser, they observed not only a stronger ablation effect on OSCC than the control group but also a significant infiltration of CD8+ T-lymphocytes as well as an increase in induced recruitment of inflammatory cytokines (tumor necrosis factor-α and interferon-γ). This promotes the microenvironment towards a favorable direction for immunotherapy. Recently, the team combined PD-L1 monoclonal antibody (aPD-L1) with the photosensitizer ZnPc for photo-immunotherapy of OSCC. The ultra-small-sized (5 nm) GQD-PEG nanoparticles targeted to the nucleus can activate the innate immune response mediated by typeⅠinterferon, increase infiltration of cytotoxic T-lymphocytes, and alter the tumor microenvironment, achieving the conversion of “immune-cold” tumors to “immune-hot” tumors. 108 Furthermore, GO-PEG, as a photothermal material, can also achieve antitumor effects by inhibiting the polarization of tumor-associated macrophages M2. 124 Therefore, phototherapy's ability to alter the “soil” for tumor cell growth and metastasis provides a new strategy for the immunotherapy against tumors. In addition, the hybrid material Fe3O4@Au/rGO synthesized by Ardakani et al. demonstrated significant and efficient destruction of oral squamous cell carcinoma KB cells (88.1%), proving that the combination of radiotherapy and PTT is also a good candidate for antitumor treatment. 105
Due to the complexity of the tumor microenvironment, cancer multi-mode collaborative therapy has become a trend. 125 In this context, GNBs, which have the advantages of low cost, good biosafety, and high photothermal conversion rate, stand out as a promising material for the combined application of PDT, PTT, chemotherapy, radiotherapy, and other methods against HNC. 126
Gene therapy
Gene therapy refers to the method of introducing nucleic acids into target cells through certain approaches to correct or compensate for genetic defects in order to treat diseases. 127 Previously, the development of gene therapy for cancer has been limited by the lack of ideal vectors. 128 With the advancement of nanotechnology, GBNs have been discovered as candidate vectors for gene transfection due to their low cost, high biosafety, and multifunctionality. 29 For example, GO-PEI complexes, obtained by functionalizing negatively charged GO with positively charged PEI, have shown high gene transfection efficiency and low cytotoxicity as gene delivery vectors in HeLa cells. 35 MicroRNAs are important biomarkers for many cancers and have been used as therapeutic targets for gene therapy in OSCC. 129 Ou et al. used GO-PEI complexes to deliver miR-214 inhibitors targeting PTEN and P53 in oral squamous cell carcinoma cells. The results showed that, compared to liposomes and naked inhibitors, GO-PEI-miR-214 inhibitor complexes had a higher transfection rate (approximately 50%) and significantly inhibited the invasion and expression of miR-214, resulting in a 46% reduction in tumor volume growth in xenograft mice. In addition, the targeted effect of this complex significantly reduced side effects. 130 Due to its favorable physicochemical properties, GO, as a non-toxic gene delivery carrier, can be further combined with chemotherapy to enhance the anticancer effect. Liu et al. 88 reported the successful application of synergistic chemotherapy and gene therapy in the treatment of NPC. GO complexes conjugated with redox HPAA and targeting molecule Tf were used to co-deliver DOC and matrix metalloproteinase-9 shRNA plasmid (PMMP-9) to tumor cells, exhibiting a significantly higher inhibition rate (more than 64%) in HNE-1 cells compared to the individual use of DOC and PMMP-9. The co-delivery system of drugs and genes not only improves the sensitivity of chemotherapy drugs but also avoids drug resistance and adverse reactions, demonstrating great potential in tumor combination therapy.
Toxicity and limitations of GBNs
Currently, the research and development of GBNs in the biomedical field are rapidly proliferating, and the associated biosafety is a major concern in the scientific community. The toxic effects of GBNs on organisms have been widely reported. Previous studies have shown that oxidative stress caused by excessive production of ROS is one of the important mechanisms underlying GBN-induced cellular toxicity. 131 Wu et al. 37 found that high levels of oxidative stress from GQDs can lead to mitochondrial dysfunction and subsequently induce ferroptosis in microglial cells. Meanwhile, the sharp edges of GQDs allow them to penetrate cell membranes leading to cell membrane damage, which has raised concerns about their potential neurotoxicity. 132 Furthermore, Zhang et al. 133 demonstrated that GO or rGO can also induce cardiotoxicity by altering lipid peroxidation markers and myocardial enzyme activity. The animal toxicity of GBNs is an important indicator for assessing their biocompatibility. It has been found that GO can accumulate in mammalian alveoli and remain there for a prolonged period, leading to dose-dependent toxic effects such as pulmonary edema, pulmonary fibrosis, and granuloma formation, thereby affecting lung function. Additionally, GO can also accumulate in the liver and spleen, impacting organ function.73, 134 When studying the therapeutic efficacy of GQDs nanomaterials against OSCC, Wei, Zhang, and their respective teams applied PEG to functionalize GQDs and found that GQD-PEG reduced the accumulation of the material in the liver, lungs, and spleen.90, 112 Similarly, when using CS-wrapped GCNCs for the treatment of NPC, no significant changes in liver and kidney function were observed in tumor-bearing mice. 92 Therefore, this shows that surface modification by various polymers such as PEG and CS can significantly reduce the in vivo toxicity of GBNs and improve their biosafety. Another study has proved that the size of GO may directly impact its cellular toxicity, with smaller-sized GO being a safer material for mitigating pulmonary toxicity. 135
The larger specific surface area and abundant functional groups of GBNs give them strong protein adsorption capabilities. The formation of a “protein corona” by adsorbed proteins by GBNs poses a significant challenge to drug delivery systems as it may lead to functional loss of GBNs.13, 136 This material-protein interaction increases the size of the GBNs, which can cause capillary blockage and thrombus formation when administered intravenously. 137 In an in vivo experiment of injecting graphene and graphene oxide nanosheets intraperitoneally into rats, Soledad Aguado Henne et al. found that prothrombin and thrombin time significantly increased with dose, resulting in prolonged clotting time. 138 In addition to causing thrombosis and coagulation, Tatsiana Kulahava et al. confirmed in their study that GQDs can interact with the red blood cell components of human blood after intravascular injection, inducing red blood cell lysis and hemolysis under visible light. 139 Previously, Ding et al. 140 had demonstrated in a study that GO-PEI could inhibit T lymphocyte immune responses by inducing plasma membrane damage, showing serious blood toxicity. Therefore, the blood toxicity of GBNs cannot be ignored. Strict evaluation of GBNs should be conducted before their clinical application. Furthermore, when used in cancer immunotherapy, GBNs may have certain counterproductive effects. The excessive secretion of immune-related pro-inflammatory cytokines may increase the risk of downregulation of the body's own defense mechanisms.107, 141 Graphene oxide nanoparticles are also reported to be able to induce the polarization of macrophages into the M1 phenotype, thus promoting inflammation in the body. 142 Current research indicates that the toxicity of GBNs primarily depends on their physicochemical properties, accumulation in tissues and organs, and interactions with cells. Researchers have successfully improved the biocompatibility and water solubility of the material through various surface modification methods such as covalent modification or non-covalent modification, which have overcome the limitations of GBNs to a large extent. 12 In addition, a study by Sayan Mullick Chowdhury et al. suggests that functionalizing graphene nanoribbons with lower and suitable concentrations of PEG-DSPE (PEGyl-phospholipids) may reduce toxicity to blood components. 143
Although the use of GBNs in the medical field is increasing, their clinical application is hindered by the limited toxicity assessment only at the animal level. 33 Therefore, it is necessary to conduct long-term, thorough, and comprehensive toxicity assessments of GBNs at the human level to ensure their safe use in the human body. In addition, there are still many challenges in the green preparation and processing technology of GBNs, and it is hoped that cost-effective and sustainable GBNs will be manufactured in the near future. 144 At the same time, there are still few studies on the use of commonly used chemotherapy drugs for head and neck cancer, such as paclitaxel and methotrexate, in GBNs, which also makes our study limited to a certain extent, and provides ideas and directions for future research.
Conclusions
In the past few decades, GBNs have become one of the most promising materials for research in the biomedical field due to their unparalleled structure and properties. This article provides an overview of the potential of GBNs in the diagnosis and treatment of HNC. The high near-infrared absorbance and unique physical properties of surface-functionalized GBNs make them suitable for biomedical imaging, while various types of biosensing platforms for detecting head and neck cancer biomarkers have also been developed. Furthermore, their ability to serve as novel nanocarriers for anticancer drugs and gene transfection can be attributed to their excellent chemical stability, high targeting specificity, and good biocompatibility. We also emphasize the outstanding contributions of GBNs as photosensitizers or photosensitizer carriers in phototherapy (PTT and PDT) for HNC, as well as their potential for combined cancer treatment with other therapeutic modalities. It should be noted that the safe, reliable, highly efficient, and low-toxicity multifunctional GBNs offer possibilities for the integration of diagnosis and treatment in HNC.
The development of nanomaterials with minimal toxicity and improved biodistribution for the diagnosis and treatment of cancer is of utmost importance in the future of biomedical research. The toxicity and long-term biosafety assessments of GBNs require collaborative efforts from researchers with different disciplinary backgrounds, who can work together and integrate various factors to modify and improve GBNs, thereby facilitating their clinical translation. We believe that in the near future, GBNs will undoubtedly make substantial progress in the diagnosis and treatment of HNC.
Footnotes
Abbreviations
Authors’ contribution
Jiali He contributed to literature search, visualization, and writing–original draft preparation. Chen Lin and Yanghao Hu contributed to investigation, supervision, and revised the manuscript. Hongxia Deng and Shanshan Gu contributed to funding acquisition and project administration. Zhisen Shen contributed to idea for the review, conceptualization, writing–reviewing and editing, project administration, and funding acquisition. All authors contributed to conceptualizing and writing the manuscript. All authors read and approved the final manuscript. All authors contributed to the article and approved the submitted version.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ningbo Clinical Research Center for Otolaryngology Head and Neck Disease (grant number 2022L005) and Ningbo Top Medical and Health Research Program (grant number 2023030514).
Ethics statement
This article is a review article, and it does not involve related animal and patient studies.
