Abstract
The incidence of cancer is increasing and evolving as a major source of mortality. Nanotechnology has garnered considerable scientific interest in recent decades and can offer a promising solution to the challenges encountered with traditional chemotherapy. Nanoparticle utilization holds promise in combating cancer and other diseases, offering exciting prospects for drug delivery systems and medicinal applications. Metallic nanoparticles exhibit remarkable physical and chemical properties, such as their minute size, chemical composition, structure, and extensive surface area, rendering them versatile and cost-effective. Research has demonstrated their significant and beneficial impact on cancer treatment, characterized by enhanced targeting abilities, gene activity suppression, and improved drug delivery efficiency. By incorporating targeting ligands, functionalized metal nanoparticles ensure precise energy deposition within tumors, thereby augmenting treatment accuracy. Moreover, beyond their therapeutic efficacy, metal nanoparticles serve as valuable tools for cancer cell visualization, contributing to diagnostic techniques. Utilizing metal nanoparticles in therapeutic systems allows for simultaneous cancer diagnosis and treatment, while also facilitating controlled drug release, thus revolutionizing cancer care. This narrative review investigates the advancements of metal nanoparticles in cancer treatment, types and mechanisms in targeting cancer cells, application in clinical scenarios, and potential toxicity in medicine.
Introduction
The prevalence of multifactorial diseases like cancer is escalating, emerging as a major global health crisis and primary cause of mortality. In 2022, there were an estimated 20 million new cancer cases and 9.7 million deaths. The estimated number of people who were alive within 5 years following a cancer diagnosis was 53.5 million. About one in five people develop cancer in their lifetime, approximately one in nine men and one in 12 women die from the disease. 1 Projections suggest a staggering 75% annual rise in new cases over the next two decades.
In the evolution of solid cancer, there are four steps: noninvasive tumor, local invasive cancer without metastasis, local invasive cancer with lymph node metastasis, and eventually systemic disease. For the first three phases, local treatment, including lymph node dissection, may cure the disease. The choice of local treatment depends on the tumor characteristics, but surgery remains important in many of these cancers. Gastric cancer is one of the typical tumors which remain locally invasive, with or without nodal metastasis, but without systemic metastasis for a rather long period. 2 Chemotherapy remains a cornerstone in cancer treatment, encompassing various drugs such as alkylating agents, DNA-damaging agents, anti-metabolites, plant alkaloids, terpenoids, and biological agents. However, contemporary chemotherapy is plagued by drawbacks, notably its lack of target specificity, which can compromise clinical outcomes.3,4 Chemotherapy has significant drawbacks, including adverse effects such as nausea, hair loss, and organ toxicity, variable efficacy across different cancers and stages, development of drug resistance, damage to healthy tissues, psychological distress for patients, and high costs with accessibility issues. 5
Another strategy involves upregulating target enzymes, such as mutant topoisomerase II, which reduces drug activation and accelerates drug degradation, leading to drug inactivation through conjugation with increased glutathione levels. Chemotherapeutic agents play critical roles in DNA repair, cell cycle regulation, and p53 activity, yet their efficacy can be compromised by significant adverse effects. The effectiveness of many anticancer drugs is hindered by issues like poor solubility and limited absorption.6,7 Techniques such as surfactants, polymorphism, salt formation, co-crystal formation, and co-solvent use are employed to improve the solubility of poorly soluble anticancer drugs in water. However, achieving dissolution of these drugs often requires high concentrations of surfactants and co-solvents, which can lead to undesirable side effects. Their low solubility and poor penetration severely limit the clinical efficacy of modern chemotherapy against solid tumors.8,9
Solid tumors impose physical barriers that impede the therapeutic efficacy of drugs. Key factors governing drug penetration include the preservation of intercellular connections among malignant cells, despite their dedifferentiation, in both primary tumors and metastatic lesions. 10 Furthermore, nests of malignant epithelial tumor cells are shielded from tumor vasculature by layers of extracellular matrix (ECM) proteins, including fibronectin, collagen, laminin, and elastin. 11
Nanotechnology's distinctive physical and functional attributes have garnered considerable scientific interest in recent decades. Nanotechnology develops nanoparticles (NPs) or nanomaterials, which can be naturally or synthetically derived and range from 1 to 100 nm in size. 12 It has had a profound impact on medical equipment, such as imaging probes, drug delivery systems, and diagnostic biosensors in pharmaceutical practice. The use of nanotechnology in cancer treatment offers notable advantages over traditional applications.13,14
An NP is an extremely small unit whose size is expressed in nanometers (nm; 1 nm = 10−9 m). Both naturally occurring and artificially produced NPs are products of human activity. Manufactured NPs possess distinct material properties due to their submicroscopic size, and they have potential uses in healthcare, engineering, environmental remediation, and catalysis, among other fields. 15 The 2008 International Organization for Standardization (ISO) standard defines an NP as a distinct nano-object with all three Cartesian dimensions less than 100 nm. Similar specifications for one-dimensional nano-objects, such as nanofibers and nanotubes, and two-dimensional nano-objects, such as nanodiscs and nanoplates, were found in the ISO standard. 16 But in 2011, the European Union Commission adopted a more detailed but broad definition: “Any substance, whether natural, accidental, or man-made, that has particles in one or more of its exterior dimensions falling between 1 and 100 nm in size for at least 50% of the particles in the number size distribution, whether in an unbound form, as an aggregate, or as an agglomeration.” 17
According to that definition, even if an object's other dimensions fall outside of the range of 1–100 nm, it just requires one of its characteristic dimensions to fall within that range to be classified as an NP. (As atomic bond lengths are obtained at 0.1 nm, a lower limit of 1 nm is utilized.) 18
NPs exhibit remarkable physical and chemical properties, such as their tiny size, chemical composition, structure, and extensive surface area to volume ratio, rendering them versatile and cost-effective for various applications and enabling greater exposure of active molecules per dose administered. 19 Additionally, NPs can enhance absorption, increase blood circulation of the encapsulated compounds, and improve the delivery of active ingredients to targeted sites. 19 NP utilization holds promise in combating cancer and other diseases, offering exciting prospects for drug delivery systems (nanocarriers), diagnostics (nanoimaging), and medicinal NP applications. 5 Metal NPs serve as multipurpose agents among all the NPs. Several types of metallic NPs such as gold, silver, iron, iron oxide, zinc, titanium dioxide, cerium oxide, nickel, copper, silicon, magnesium, barium, calcium, and bismuth-based metal NPs have been investigated as a potential in cancer treatment. The leading role of metal NPs against cancer is increasing day by day.20–22 The aim of this narrative review is to highlight researchers about the potential of metal NPs in cancer treatment, the advantages of metallic NPs, and toxicity concerns of the metal NPs in clinical application.
Cancer comprises a diverse spectrum of diseases characterized by the uncontrollable proliferation of abnormal cells within various parts of the body. Each type of cancer presents unique characteristics and challenges. For instance, breast cancer commonly originates in the milk ducts or lobules of the breast tissue and can be effectively managed with early detection through regular screenings and self-examinations.23–26 Lung cancer, often linked to smoking and environmental pollutants, begins in the lungs’ epithelial cells and spreads rapidly, manifesting symptoms like persistent coughing and chest pain. 27 Prevention strategies such as smoking cessation and reducing exposure to toxins play crucial roles in mitigating its impact. 28 Colorectal cancer, which affects the colon or rectum, frequently develops from precancerous polyps and underscores the importance of routine screenings like colonoscopies. Lifestyle modifications such as maintaining a balanced diet and engaging in regular exercise also significantly contribute to reducing the risk of colorectal cancer.29–31 Leukemia, a blood and bone marrow malignancy, disrupts normal immune function with rapid growth of abnormal white blood cells. Treatment options vary from chemotherapy to stem cell transplantation, depending on the type and stage of leukemia. 32 Prostate cancer arises in the prostate gland and is prevalent among males, often without early symptoms, necessitating regular PSA screenings. 33 Lymphoma affects the lymphatic system, including lymph nodes, with Hodgkin's and non-Hodgkin's types prominent.34,35 Melanoma originates from melanocytes and is a significant skin cancer. Pancreatic, ovarian, and bladder cancers pose challenges in early detection and treatment due to late diagnoses and specific symptoms. 36 Kidney cancer, primarily renal cell carcinoma, develops in the kidneys, while thyroid cancer originates in the thyroid gland, potentially causing hormone imbalances. Cervical cancer, linked to HPV, affects the cervix, emphasizing the importance of early detection and treatment for improved outcomes in these cancers. 37
Inorganic and metallic nanoparticle
Metal NPs are multifunctional agents that provide several opportunities in various biological applications, such as thermal ablation, radiation augmentation, and diagnostic tests. It is caused by the metal NPs’ strong surface electromagnetic field, a variety of optical properties, simple surface chemistry, surface functionalization, and simplicity of manufacture. 38 Due to their unique properties, NP-based systems enable theragnostic, continuous evaluation and therapy, and real-time chemotherapeutic monitoring, making them a safer and more effective alternative to conventional system treatments. 39
To target specific cells, these NPs may be given either alone or in combination with antibodies, peptides, or DNA/RNA. Metal NPs may have their surfaces modified with biocompatible polymers (such polyethylene glycol) to enhance gene transport and silencing, as well as prolong the pharmacological effect.40,41 Gold, silver, or a mix of the two metal NPs’ optical characteristics may be fine-tuned to the desired wavelengths by adjusting their size, composition, and form; for example, noble metals with a core/shell or alloy structure can range from 1 to 100 nm in size. As a result, they are suitable for photothermal applications and imaging under native tissue. 42
Furthermore, noble metal NPs effectively transform light or radio frequencies into heat, allowing thermal ablation of specific cancer cells. A short overview of metal NPs in cancer therapy is given in the section below, which helps readers better comprehend these particles and their potential benefits for specific clinical outcomes. 43
Mechanisms of metallic NPs in targeting cancer cells
Metal NPs are brand-new anticancer medicines that are completely selective for cancer cells. The potential anticancer activity of NPs is influenced by their physicochemical characteristics, which may be attributed to either their inherent or extrinsic aspects. One of its inherent, or internal, anticancer properties is its antioxidant activity, which slows the growth of tumors. 44 Because of the inherent anticancer properties of metal NPs, researchers have been very interested in using them to combat the initiation, growth, and progression of tumors. One of the extrinsic aspects is the utilization of outside stimuli, such as in hyperthermia, where metal NPs act as co-adjuvants and are triggered by outside radiations like IR or X-rays to form free radicals that destroy cancer cells. The intensification of ionizing radiation's harmful action is another aspect 45 (Figure 1).
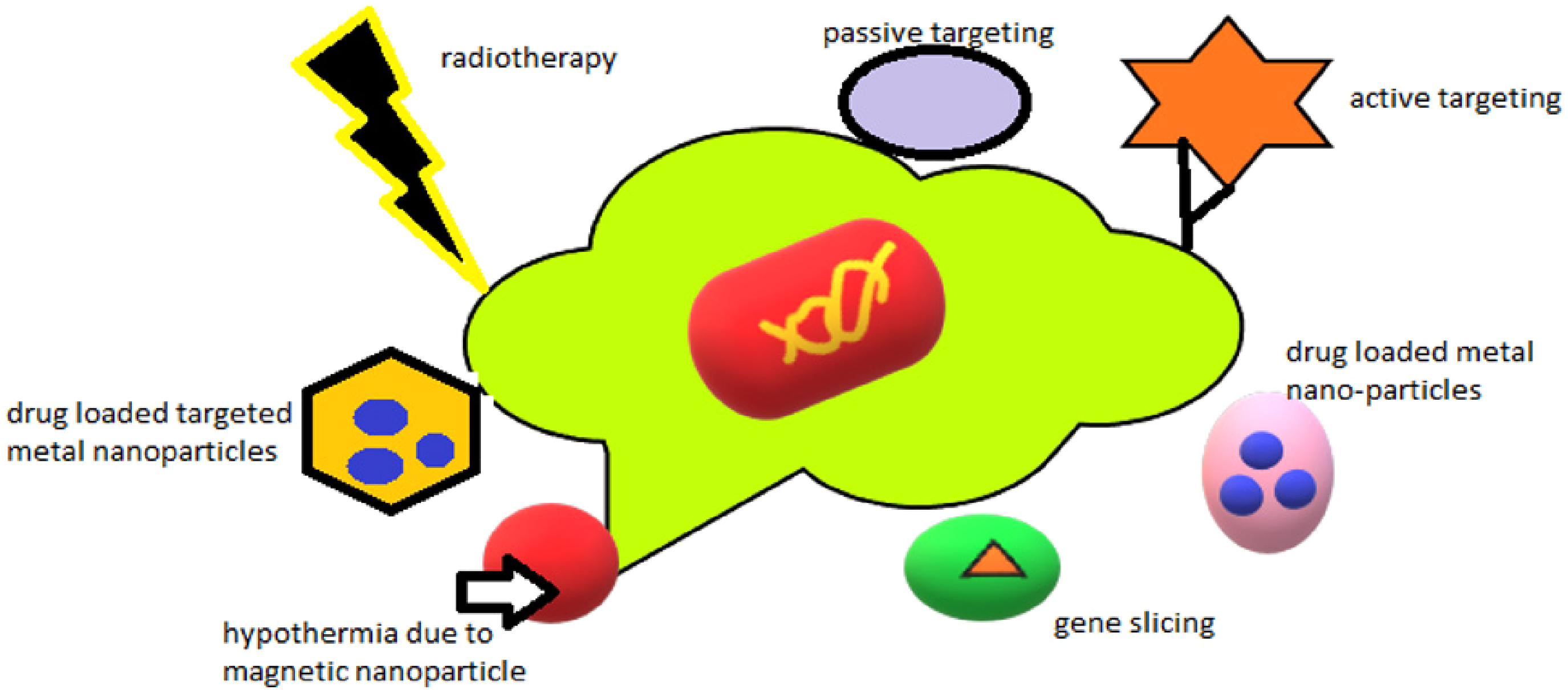
Mechanism involved in cancer targeting.
Active or passive tumor targeting
One key feature of metal NPs is their ability to target particular tumors with different therapeutic agents while avoiding biological barriers. This reduces the toxicity and side effects of chemotherapeutics. 46 Consequently, metal NPs provide a platform that supports the synergy of anticancer drugs’ antitumor action. Metal NPs attack tumor cells passively and actively. The increased permeability and retention (EPR) effect occurs when NPs of a given size gather more in cancerous tissue than in normal tissue. Tumor tissues are more macromolecule-permeable than healthy tissues due to their weak lymph node apertures, and poor endothelium. 47 This makes it easier for the NPs to penetrate into and destroy malignant cells. Due to their distinct shapes and increased vascular permeability, metal NPs provide a chance to specifically target cancer cells. 48
Functionalized NPs are used to deliver chemotherapeutic medicines, which not only increases the medication's therapeutic efficacy and furthermore aids in the prevention of medication resistance. 49 To target “medulloblastoma cells” (Daoy-2 and Daoy cell lines with overexpressed HER-2 and moderate HER-2 levels, respectively) and high-grade “glioma cells” (A431 and U373 and U87 with overexpressed interleukin-13 receptor alpha 2 levels, respectively), immunoshells, also called antibody-tagged gold silica nanoshells, were developed. “Medulloblastoma cells” (Daoy-2) exhibited high tumor development and a substantial HER2 binding affinity, in contrast to “Daoy,” which lacks “HER-2.” 50 The situation was analogous with “U373 and U87,” where significant targeting was detected but A431 was not. Customized microbes, according to these results, allow for more targeted delivery without harming healthy cells. 48
Gene silencing
The process of selectively blocking the synthesis of a particular translational product using an antisense molecule is known as gene silencing. Small interfering RNA and antisense DNA both significantly and favorably contribute to the downregulation of specifically expressed genes inside tumor cells. 51 How much siRNA suppresses tumor genes after transfection, how much siRNA is delivered, and how long the silencing response lasts are all factors in transfection success. Metal NPs are ideal carriers for RNA and DNA, which have negative charges, due to their very positive surface charges. Furthermore, metal NPs, such as gold NPs (AuNPs), target messenger RNA (mRNA) and interact with cell biomolecules to increase cytotoxicity. 50
This effectively addresses concerns regarding therapeutic RNA delivery by increasing the half-life of siRNA and lowering the dose needed, protecting it from RNase action with lipid coating, providing chemical stability through poly (β-aminoester) coating, and preventing common detachment from the vectors. For effective transfection (MagRET) gene silencing, magnemite NPs (Y-Fe2O3) are used. 52 It is made by applying lanthanide Ce(3/4+) cation to the surface of a magnetite core. Additionally, the [CeL(n)](3/4+) cation/complex facilitates the coordinative chemistry that binds the magnemite core to polyethylamine (PEI). 53
PEI oxidation reduced the toxicity in vivo. It was discovered that the success rate of research using long non-coding RNA, microRNAs, and mRNAs rose by 80%–100% in a range of cells. For the purpose of siRNA transfection, “CHO-K1 and HeLa cell” lines were subjected to testing with core-shell “nanoparticles of superparamagnetic iron oxide” that had been treated with polycationic polymers, namely branching polyethyleneimine or polyhexamethylene biguanide. 54 The results demonstrated that the modified branching polyethyleneimine outperformed the unmodified counterpart. Superparamagnetic iron oxide nanoparticles (SPIONs) are enhanced in their efficiency by magnetofection, which involves the application of an external magnetic field. These NPs were determined to be less harmful than polyhexamethylene biguanide and unmodified polycationic branching PEI. 50
Hyperthermia and radiotherapy
High-intensity radiation is used in radiation treatment to either stop the growth of malignant cells or eradicate them. The ions’ tremendous kinetic energy from repeated collisions leaves a significant energy accumulate in the cells that they traverse. Tumor cells’ RNA production and DNA replication are halted by the supplied energy, which eventually results in cell death. Ionizing radiation therapy, or radiotherapy, works better as an adjuvant to treatment when administered in conjunction with hyperthermia. 55 This produces a symbiotic result. In order to get the intended effects from radiation treatment in the event of hyperthermia, a time-dependent interaction is crucial. Though it may seem perfect to treat tumor cells simultaneously with radiation and heat, the opposite is thought to be more beneficial. The hyperthermic reaction to the tumor location and increased radiation targeting are two benefits of metal NPs. When combined with radiation and metal treatment for cancer, response rates rise by 16%–26%. 56
A potentially effective drug for combination treatment is AuNPs. After subjecting AuNPs to moderate hyperthermia (∼41 °C) for 20 min and administering a radiation dosage (10 Gy using 125 kV X-rays), the impact of the AuNPs-mediated hyperthermia and ionizing radiations was seen. When comparing radiation treatment alone to combination therapy, it was shown that the tumor volume doubling time (the amount of time needed for the tumor to double in volume, regardless of starting tumor size) was longer with radiation therapy alone. Radiation treatment is more successful when AuNPs are used because of their radio sensitization effect. Treatment failure was mostly caused by the ability of breast cancer xenografts exposed to radiation treatment alone to develop stem cells into the remaining tumor. Combining therapies addresses the problem of intrinsic radio-resistance in cancer stem cells within the remaining tumor, enhancing the overall treatment of cancer.57–60
Hyperthermia and chemotherapy
It is common practice to combine chemotherapy with hyperthermia therapy with metal NPs to maximize the effectiveness of both treatments. Among examples of concurrent treatment with a much greater targeted success rate, hyperthermic intraoperative peritoneal chemotherapy stands out. 61 The NPs of “iron” (MF66) and “Nucant multivalent pseudopeptide” (N6L; MF66-N6L), “doxorubicin” (DOX; MF66-DOX), or a combination of the two (MF66-N6LDOX) were shown to be beneficial in treating breast cancer athymic nude mice models. The findings demonstrated that under alternating magnetic fields, there is an exceptional heating potential of around 500 W/g. Iron NPs functionalized with “N6L and MF66-N6LDOX” are more selective and effective in destroying cancer than ligand-free MF66-DOX. Tumor cells were shown to slowly absorb the medication, leading to significant slowing of tumor development (40% reduction from initial tumor volume) and, in some instances, total elimination of the tumor.62,63
Types and application of metallic nanoparticles
The metal NPs can be divided into noble and non-noble metals-based NPs. Nanoparticles (NPs) for cancer treatment can be based on noble metals like gold, silver, platinum, and palladium, as well as non-noble metals. 64
Magnetic nanoparticles (iron/nickel) and zinc oxide nanoparticles
Iron nanoparticles
The therapy and diagnostics of cancer have made substantial use of iron NPs because of their plasmonic and magnetic capabilities. When non-toxic wavelength radiations such as oscillating magnetic fields or near infrared radiation (NIR) are present, iron oxide NPs undergo a transformation into reactive oxygen species (ROS) stimuli that can kill tumor cells. Because iron oxide NPs are covalently coupled to tissue-specific molecular determinants, they provide site-specific targeting, which is their primary benefit. 44 Due to their ability to be magnetic, iron oxide NPs lessen the harmful side effects of cancer treatment by converting radiant energy from light into heat or ROS when a localized external magnetic field is applied. 65
Because iron oxide NPs are magneto sensitive, they are used in conjunction with chemotherapy drugs like doxorubicin to improve the therapeutic result of conventional doxorubicin treatment. The anticancer effect is enhanced by doxorubicin's paramagnetic characteristics and the potential for electron transitions into nano-complexes with more free radicals because of an external magnetic field. It is shown that PEG-coated iron oxide nanocubes cause the tumor's ECM to break down when subjected to magnetic stimulation. 66 Consequently, during the three-cycle heating process, NPs are redistributed, preventing the growth of the tumor. In situations of hyperthermia, heat treatment over 40 °C is often utilized to kill cancer cells. Iron oxide NPs, which may collect energy from outside sources like near-infrared radiation, can reach the tumor site and transform heat into energy. 67 When cerium oxide, titanium, magnesium, and iron oxide nanoparticles are exposed to radiofrequency oscillating magnetic fields, they experience a reorientation of their magnetization, which produces heat. Recent research by Sadhukha and colleagues examines the efficacy of tumor-targeted SPIO NPs for lung cancer magnetic hyperthermia. 68 Furthermore, iron NPs boost the lethal impact by inducing ROS formation. Using monosaccharide-coated iron oxide NPs, Wydra and colleagues found that they increased apoptotic cell death and the production of ROS inside cells. Only during therapy may this be tolerable, but following therapy, patients may have significant health risks due to the obstruction of the body's ability to eliminate NPs. 69 Therefore, to stop the development of ferromagnetic NPs, iron oxide NPs in the range of 30–40 nm are chosen. One may choose a larger size, but only up to a point known as the superparamagnetic limit, beyond which iron oxide NPs begin to exhibit a propensity to transform into ferromagnetic NPs. For biomedical applications, it is essential to prevent unwanted particle aggregation that might affect the magnetic behavior of NPs. 70 Sometimes it is better if they are smaller so they can serve as a heat source for a smaller tumor region. The removal of the magnetic field causes SPIONs to lose their magnetism. By acting as radiosensitive agents and catalyzing the creation of ROS, iron oxide NPs, when administered as citrate and malate-coated SPIONs with an X-ray source, aid in the death of tumor cells. 71
Gold nanoparticles
AuNPs are showing promise as an adjuvant in cancer treatment. Gene silencing, medication delivery, radiation, and hyperthermia are some of the applications for AuNPs (Figure 2). AuNPs’ surface may be functionalized with a variety of biomolecules to enable site-specific targeting. AuNPs coated with PEG are utilized to slow down the body's rapid removal of AuNPs and facilitate the direct coupling of other molecules of interest. By circumventing the first metabolic pathway and facilitating passage of the blood–brain barrier, it enhances the effectiveness of treatment. 72 Because of their tiny size, NPs are able to evade the reticuloendothelial barrier, which results in improved EPR of the medication for an extended period of time. A photothermally regulated drug delivery system was shown using the hydrogel matrix used to create the gold sulfide nano shells. After NIR is absorbed, a burst mechanism causes the drug to escape from the hydrogel. When combined with ionizing radiation, mice treated with AuNPs coated with PEG, which are used to treat breast cancer and adenocarcinoma, have a better survival rate. 73

Function and properties of gold nanoparticles.
Due to their structural characteristics, additional AuNPs are being researched for gene silencing treatment. Due to their propensity to accumulate at the tumor site 4–5 h after treatment, AuNPs provide a chance for sustained laser hyperthermia therapy. 74
Titanium dioxide nanoparticles
Titanium dioxide works by photodynamic treatment (PDT), which produces ROS that cause cell death by activating hydrophobic compounds using visible or ultraviolet electromagnetic radiation. 75 Nevertheless, the radio thermal efficacy has been compromised due to its poor solubility. Initially, PDT made use of inorganic and polymeric NPs as passive carriers to make hydrophobic titanium dioxide more soluble. Instead of using conventional PS molecules, titanium dioxide might be used directly as a photosensitizing agent. 76
Titanium dioxide NPs are thought to have great promise for cancer treatment owing to their increased photocatalytic activity and improved UV absorption efficiency. 77 Furthermore, a significant factor in determining anticancer efficacy is the UV light range needed for cancer treatment. Surface functionalization is employed using titanium dioxide NPs to get around this restriction. It has been noted that the surface chemistry, cell type, and particle cores of NPs all affect the survivability of cells. 78
In particular, the –COOH group is less hazardous than the –NH2 and –OH groups. The findings demonstrate that functionalized titanium dioxide NPs with surface engineering may specifically target cancer cells. When titanium dioxide NPs were tested on malignant cell lines of T-24, HeLa, and U937 cells, they were discovered to be integrated into the cytoplasm and cell membrane. 79 Less penetration into tumor cells and a lack of selectivity for malignant cells are the main drawbacks of UV and visible light. The NIR range, which is typically in the 700–1000 nm range, may be employed to promote greater tissue penetration for titanium dioxide treatment. 80 Titanium dioxide NPs are stable under light radiation, have a longer half-life, and stay in the body for longer. In the presence of NIR, recently crystallized shells made of NaYF4:Y63+, Tm3+NaGdF4:4b3þ cores, and titanium dioxide as a coating material on the core's surface were created and shown to be effective. When used as a tumor model in female Balb/c mice, upconversion nanoparticles (UCNPs)@titanium dioxide-based UV light-mediated photodynamic therapy (PDT) had less potent tumor inhibitions than UCNPs@titanium dioxide irradiated by IR light. 81
Zinc oxide nanoparticles
Nanoparticles of zinc oxide (ZnO NPs) insert themselves into cells and induce DNA damage, much like genotoxic medicines. In terms of mitotic and interphase (apoptotic) mortality, ZnO NPs had the greatest impact on T98G cancer cells, a moderate impact on KB cells, and little to no effect on HEK cells, which are normal cells. This feature has led to ZnO NPs being intensively studied in cancer research as of late. 82 Dose-dependent response is seen in ZnO NPs. Melanoma cell death rate and ROS generation are modulated by dose-dependent ZnO NPs. Cell viability, apoptosis rate, and mRNA level of apoptosis genes were studied in HepG2 (liver cancer) and MCF-7 (breast cancer) cancer cells in relation to dosage variation. At a very low concentration of 25 µg/ml of ZnO NPs in HepG2 cells, the cell viability as assessed by 2,5-diphenyl-2H-tetrazolium bromide (MTT) assay test was less than 10%, and there was a dose-dependent decreasing response. At higher concentrations of ZnO NPs, apoptosis will be accelerated. We used quantitative real-time polymerase chain reaction (PCR) to measure the “mRNA levels of apoptotic markers” (P53, Bax-2, bcl-2, and Caspase-3). In comparison to untreated cells, treated cells showed a 1.9-fold increase in the mRNA level of the tumor suppression gene p53, a 2.5-fold drop in the mRNA level of the ant apoptotic gene “bcl-2,” and a 2.7-fold decrease in the mRNA level of the pro-apoptotic gene Bax. 83
In treated cells, caspase-3 mRNA expression was 1.8 times greater than in untreated cells. Levels of mRNA expression regulate cell death. A quantitative real-time PCR analysis found that ZnO NPs downregulated bcl-2 mRNA levels and upregulated p53 and Bax mRNA levels. The caspase activation process is facilitated by opening the mitochondrial membrane and allowing soluble proteins to be released into the cytosol from the intermembrane gap. 84 The electrostatic property of the ZnO NPs determines the surface charges that may be present under various acidic and basic circumstances. There are a number of therapeutic drugs that may be conjugated with them. In addition to their usage as photodynamic agents, their high ROS production triggers apoptotic cell death. 85 Metalloproteins have anticancer action, since their therapy was shown to reduce cell viability. Asparaginase-loaded ZnO NPs have excellent selectivity, efficacy, and stability as an anticancer drug. Chemotherapeutic medicines, such as paclitaxel and daunorubicin, are administered in conjunction with ZnO NPs to reduce their toxicity. 86
Cerium oxide nanoparticles
Cerium oxide nanoparticles, often known as CNPs, are a novel and potentially useful weapon in the battle against malignancies with applications ranging from radiation therapy to the transport of drugs. NPs made of cerium oxide have the remarkable ability to kill tumor cells while shielding healthy tissues in the area from harmful radiation and oxidative stress. 87 CNPs are responsible for an increase in both the oxidative stress and the apoptotic cell death that occurs in malignancies. Because the Warburg effect causes the extracellular environment of a tumor to decrease from 7.1 (in normal cells) to 6.7 (a little acidic), it is considered that malignant cells are acidic and that their cytoplasm is somewhat more “alkaline (pH > 7.4)” than the cytosol of regular cancer cells. 88
The specificity of CNPs toward cancer cells has been demonstrated by their ability to inhibit catalase and superoxide dismutase (SOD) activities in acidic environments. This is because ionizing radiation produces superoxide, which is stable at low pH and increases the radiation's toxicity due to the buildup of H2O2. 89
The SOD enzyme increases DNA damage by preventing the G2/M transition, making it more radiosensitive. In addition to their extrabiological mechanism of DNA damage, CNPs function as radio sensitizers because they may inhibit hTERTHPNE in normal cells while facilitating apoptotic cell death in malignant cells (L3.6pl). Hence, they have been shown to be safe for human use in business and medicine. 90
The cytotoxic effects of redox-active CNPs are limited to tumor cells exclusively. CNPs possess anti-invasive, pro-oxidant, and proapoptotic activities in squamous cell carcinoma of the skin and melanoma. These actions are achieved by increasing the number of ROS in malignant cells, which ultimately results in the apoptosis of only these cells. 44 Because they do not harm skin stromal cells, CNPs are preferable to traditional chemotherapy. Additionally, CNPs are used for the administration of doxorubicin and other chemotherapeutic treatments, with the goals of reducing doxorubicin toxicity, improving the specificity of the drugs, and stabilizing the drug molecules. In conclusion, CNPs provide a fresh perspective on cancer treatment when added to traditional chemotherapy. 91
Silver nanoparticles
Research has shown that silver nanoparticles (AgNPs) may raise levels of oxidative stress, alter the fluidity of cell membranes, and induce cell death in tumors. 92 The radiosensitizing agent is AgNPs. To make cells more susceptible to radiation, radiosensitizers work by releasing Ag+ cations, which trap electrons and raise cellular oxidative stress. Also, they lower the quantity of ATP within the cells and boost the formation of ROS. After gliomas were treated with AgNPs and then radiation, researchers saw an antiproliferative and proapoptotic impact. 93
“Glioma cells” (rat C6 glioma cells, human U251 and SHG-44 glioma cells) exhibit an increase in radiation-induced necrosis when exposed to AgNPs ranging in size from 20 to 100 nm, according to in vitro investigations. 94 Accumulating larger AgNPs results in diminished performance. Tested against U251 cells, 20 nm AgNPs show superior radiation sensitivity compared to 50 and 100 nm AgNPs. This was seen in SHG-44 and C6 cells as well. In the in vivo experiments, 6 mV X-rays are given either alone or in combination with AgNPs to rats that have C6 glioma cells. Combination treatment increased therapeutic effectiveness and tumor killing power without evident systemic harm, according to the findings of this investigation. 95
Silicon nanoparticles
The use of silica nanoparticles (SNPs) improves the drug delivery of anticancer medications; the use of lower doses of pharmaceuticals is possible due to the NPs’ low toxicity and tumor accumulating impact, as shown in the studies. SNPs may rapidly assemble because of the hydrogen bond interaction between silanol groups. Improving bio-distribution and cellular uptake requires overcoming this restriction. 96 The surface of the NPs is coated with trihydroxysilylpropyl carboxylate groups, which significantly affects the surface charge of SNPs reducing aggregation and improving stability in aqueous solutions through electrostatic repulsion. An ester link and a hydroxyl group covalently bind the medicine to the SNPs, strengthening a lactose circle and preventing the drug's broad discharge. 97
Mice with pancreatic cancer were treated with chemotherapy medicines such gemcitabine and paclitaxel, and it was shown that silica is an effective transporter for these drugs. Mesoporous silica, which has pores of varying sizes and shapes, may entrap the medication, preventing it from degrading and preventing its premature release before it reaches its target. 98 The key concern is that SNPs may be harmful. Thus, the acute toxicity of SNPs is investigated, and the results demonstrate that the mice do not experience any harm. Following this, other writers detailed the process of creating camptothecin-delivering surface-modified SNPs, including their design, production, and biological assessment. 99
Copper nanoparticles
Copper oxide NPs (CONPs) are produced by synthesizing various plant extracts, such as Acalypha indica and Ficus religiosa. This approach is known as green synthesis of NPs, and it has been shown to be dependable, easy, non-toxic, and environmentally beneficial. These NPs have been shown to have a cytotoxic impact on human lung cancer cells, leading to apoptosis and an increase in ROS production. 100 Mice with melanoma and lung cancers that have spread may be treated in vitro with CONPs utilizing B16-F10 cells. These cells have also shown the cytotoxic and anticancer effects of CONPs. They discovered that as the specificity rose, the survival rate of the tumor-bearing mice improved. After 7 days, a mouse given a dose of 2 mg/kg of CONPs had its main organs removed and examined to ascertain the pace at which the organs were being eliminated from the body. The results demonstrated that CONPs were extracted from the organ promptly and securely. The in vitro study's author also observed that CONPs might target HeLa cells’ mitochondria, release cytochrome C, and activate caspases 3 and 9. 101
Potential toxicity of using nanoparticles and metallic NPs in medicine
NPs pose significant health risks to humans due to their potential toxicity, influenced by factors such as quantity, route of administration, time of exposure, shape, surface structure, charge, chemical composition, and solubility. 20 The physicochemical properties of NPs differ from those of larger particles, and their small size allows them to easily enter the human body, crossing membranes to access cells, tissues, and organs that larger particles cannot reach. NPs exhibit higher chemical reactivity, leading to increased production of ROS, which can cause inflammation, oxidative stress, and damage to membranes, proteins, and DNA. 102
Nanomaterials can induce toxicity in various human systems, including the pulmonary, reticuloendothelial, neurological, and cardiovascular systems, and may also have endocrine-disrupting or immunological effects. They primarily enter the body through inhalation, ingestion, and skin contact. Ingestion may occur accidentally or intentionally, and after ingestion, some NPs may be absorbed by the body, moving into vital organs and tissues, causing side effects. Topical use of nanomaterials can also be harmful, as certain nanomaterials can penetrate deeper layers of skin within 24 h of exposure. Neonatal toxicity is another concern, as exposure to NPs can severely harm pregnant women, leading to oxidative stress, inflammation, placental damage, delayed neonatal development, fetal abnormalities, neurotoxicity, and reproductive dysfunction in infants. 103
There are currently no established protocols for assessing the potential toxicity of NPs or for restricting exposure to the vast quantities of these newly generated substances. The tiny size and huge surface area of nanomaterials enhance their reactivity with biological targets, which means that hazardous effects are still a possibility, despite the distinct advantages. However, there are several safety and ecological concerns with nanotechnology-based consumer and commercial goods. 104 There are still a lot of unanswered questions about the long-term viability and safety of consumer and commercial products made with nanotechnology. Genotoxicity, organelle failure, and cellular inflammation are all regulated by oxidative stress, which also regulates NP toxicity. The activation of oxidative enzymatic pathways leads to the generation of free radicals based on oxygen or nitrogen, which in turn causes oxidative stress. 105
Protein, DNA, and lipid damage, mitochondrial and endoplasmic reticulum malfunction, and eventually apoptosis or ferroptosis result from the intracellular free radical scavenging mechanism's failure to defend against prolonged oxidative stress. Metal NPs will not enter clinical practice until every step of the process, from production to therapy, is carefully controlled for risks and toxicity. 106 A study found that in both freshwater and saltwater habitats, in addition to wastewater, silver binds strongly to organic and inorganic sulfur. Due to the lower solubility of silver sulfide, sulfidation significantly reduces the toxicity of AgNPs, potentially limiting their short-term environmental impact. The use of green synthesis, which involves microbes, plants, and algae, to produce NPs has been proposed as a way to lessen the environmental impact and high concentration of harmful compounds involved in traditional chemical and physical procedures. 107
Compared to physical and chemical methods, the green way to synthesis appears to be safer and better for the environment. Copper NPs were synthesized by mixing an acetate solution with an extract of the leaves of the Eclipta prostrata plant, without the need for surfactants or external energy sources. 108
Copper NPs derived from the extract of E. prostrata leaves were tested for their cytotoxic and antioxidant qualities. Environmental impact was reduced by the biosynthesized copper NPs, and E. prostrata leaf extract was found to be an effective copper ion reduction. 109 The produced copper NPs were cytotoxic to HepG2 cells, as shown by in vitro anticancer tests. To further reduce their toxicity, metal NPs may be coated with various polymers. The delivery of cisplatin prodrug to the bone was accomplished by combining alendronate bone-seeking moiety with PEG-coated NPs manufactured from a Zn2+ coordination polymer. 110
A great deal of knowledge on the influence of NP structure on biological characteristics has been gained from several studies using various metal NPs. According to the methodology, human fibroblast malignant melanoma (Ht144) cells are used to determine the reactive capacity of zinc oxide (ZnO) and its composites; Gil and colleagues carried out a thorough experimental examination. According to experimental findings, adding silver to ZnO NPs greatly enhances their capacity for photo-oxidation. Due to their lack of specificity, anti-cancer medications almost always generate substantial systemic toxic effects, which in turn increase side effects and multiple drug resistance. 111
Conclusion
Research in cancer chemotherapy remains a formidable challenge for achieving improved clinical outcomes. Targeted drug delivery offers promise by enhancing efficacy and reducing side effects. Nanotechnology has revolutionized biomedical research, with metal NPs emerging as potential therapies for cancer. Metal NPs possess unique physical and chemical properties that make them attractive for research, including cost-effectiveness, ease of synthesis, and tunable size and shape. Green synthesis has eliminated concerns about toxic chemicals. Noble metal NPs exhibit distinctive plasmonic properties useful for monitoring therapeutic carriers in the body, aiding diagnosis and treatment monitoring. Non-noble metal NPs offer hyperthermia and magnetic properties at lower costs. Despite promising results in preclinical and clinical trials, challenges such as toxicity and regulatory requirements remain.27,112,113 Addressing these challenges, including establishing new clinical guidelines and optimizing administration methods, could unleash the full potential of metal NPs in cancer therapy and drug delivery against cancer.
Footnotes
Acknowledgments
Only the listed authors contributed to this work and no other individuals or collaborators were involved in this work.
Authors’ contributions
Ali Al-Samydai designed the study, involved in planning, and supervised the work. Maha N. Abu Hajleh and Farah Al-Sahlawi contributed to the design, data collection, and implementation of the study along with results analysis and to the writing of the manuscript. Hamdi Nsairat drafted the final copy of the manuscript and designed the figures. Arwa Al Khatib developed the theoretical framework and data interpretation. Moath Alqaraleh and Alia K. Ibrahim conceived the study and were in charge of overall direction, editing, and planning.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Research ethics and patient consent
This study did not involve any experiment on animal or human subjects.
