Abstract
Groundnut (Arachis hypogaea L.) is a vital leguminous oilseed crop, widely cultivated in tropical and subtropical regions due to its high nutritional and economic significance in food, feed, and oil purposes. It is a rich source of protein, oil, vitamins, minerals, and bioactive compounds with anti-inflammatory, anticancer, and anti-aging properties. Globally, groundnut production is approximately 54.2 million tonnes, with India contributing 10.1 million tonnes through rainfed cultivation. However, its productivity is constrained by drought, salinity, soil nutrient deficits, and disease infestations. Conventional farming depends on chemical inputs to enhance yield and productivity but negatively impacts soil health and fertility, reduces microbial diversity, and pollutes agroecosystems, creating an urgent need for sustainable alternatives. Microbe-based bioinoculants comprising nitrogen-fixers, phosphorus solubilizers, potassium solubilizers, sulphur oxidizers, other plant growth-promoting rhizobacteria (PGPR), mycorrhizal fungi, and cyanobacteria offer an alternative approach to enhance the growth and yield of groundnut through various direct and indirect mechanisms, including augmenting nutrient absorption, improving quality parameters, suppressing plant pathogens, stimulating plant defence, and increasing resilience to abiotic stresses. This narrative review examines the diversity, benefits, and growth-promoting mechanisms of rhizospheric, phyllospheric, and endophytic microorganisms associated with groundnut. Additionally, molecular docking of groundnut root exudate metabolites, produced upon microbial inoculation, with stress-responsive proteins highlights the significance of microbial inoculants in mitigating drought and salinity stresses. This review synthesizes recent advances in microbial inoculant applications, highlighting their potential to revolutionize sustainable groundnut cultivation. Therefore, microbial inoculants provide a promising solution ensuring sustainability and assurance of food security amid global difficulties.
This is a visual representation of the abstract.
Introduction
Groundnut (Arachis hypogaea L.), or peanut, is a vital leguminous and oilseed crop of the Fabaceae family, widely grown in tropical and subtropical regions for its nutritional and economic significance. It is rich in protein (25 g), oil (47–53%), and vitamins such as B1 (0.64 mg), B3 (12 mg), B5 (1.76 mg), B6 (0.34 mg), B9 (240 μg), E (8.33 mg) and minerals such as magnesium (168 mg), calcium (92 mg), phosphorus (76 mg), iron (4.58 mg) and zinc (3.27 mg) approximately per 100 g of seed. It also contains bioactive compounds including flavonoids, resveratrol, and phytosterols possessing antioxidant, anti-inflammatory, anticancer, and anti-aging properties.1,2 Beyond human consumption, it serves as a key source of edible oil, the food industry (peanut butter, confectionery, snacks, protein supplements), livestock fodder, and soil fertility enhancement through nitrogen fixation, reducing dependence on synthetic fertilizers. 3
Global groundnut production reached approximately 54.2 million tonnes across 30.5 million hectares, with China leading at 18.3 million tonnes, followed by India at 10.1 million tonnes exporting 680.70 thousand metric tonnes in 2023–2024 (860.68 million USD).4,5 In India, groundnut cultivation is predominantly rainfed, with the Kharif season (June to October) accounting for the largest cultivated area (about 4000 hectares) and the highest production (around 8000 tonnes), compared to the Rabi and summer seasons. Figure 1 illustrates the recent trends in groundnut cultivation, production, and derived products from global and Indian perspectives. Since Kharif cultivation relies heavily on monsoon rainfall, any variation in precipitation patterns can lead to drought, adversely impacting germination, biomass accumulation, and pod development subjecting to 85% yield loss in groundnut estimated at 50 million United States Dollars (USD) annually, particularly in arid and semi-arid regions. 6 Soil salinity and nutrient deficiency further exacerbate these issues by causing osmotic stress, nutrient imbalances, and ion toxicity, ultimately leading to poor plant growth and yield losses of 53.6% and 40%, respectively.7,8 Moreover, inadequate soil fertility, recurrent pest occurrences, and opportunistic pathogen breakouts could profoundly affect the crucial growth stages of groundnut, such as flowering, peg formation, and pod development, impeding nutrient absorption and diminishing overall output. 9 Hence, there is an urgent need to develop an alternative sustainable approach to strengthen soil health and overall groundnut productivity.

Trends in groundnut cultivation, production, and derived products: global and Indian perspectives. Panel (A) illustrates the seasonal distribution of groundnut cultivation in India, showcasing variations in area, production, and productivity across the Kharif, Rabi, and summer seasons. Panel (B) presents a global comparison of groundnut production, highlighting China and India as the leading producers. Data for both panels were obtained from FAOSTAT, 2024 (https://www.fao.org/faostat/en/#data/QCL). Panel (C) provides a state-wise analysis of groundnut production in India during the Kharif season over two consecutive years (source: Angrau groundnut outlook report – June to May 2023-24 – search). Panel (D) depicts trends in groundnut-derived products, including oilseed, protein meal, and oil, indicating fluctuations in production over recent years (source: https://www.fas.usda.gov/data/oilseeds-world-markets-and-trade-02112025).
In response to increasing food demand, modern agriculture has relied heavily on chemical fertilizers to enhance crop yields. Chemical inputs dominate because of their cost, widespread accessibility, and immediate effect in supporting crop productivity. For groundnut, the recommended NPK fertilizer application is 25:75:25 kg/ha, which yields an oil yield of 49.94% and a dry pod yield of about 2605.78 kg/ha. 10 However, excessive use of chemical fertilizers has led to environmental degradation, including soil nutrient depletion, contamination of soil and water resources, and loss of soil microbial diversity. 11 Continuous fertilization alters soil pH, disrupts microbial communities, and reduces the bioavailability of essential nutrients, ultimately decreasing crop productivity. 12 Moreover, unsustainable land use practices contribute to land degradation, climate change, and global warming, adversely affecting crop growth, yield, and overall agricultural sustainability. 13
To mitigate these issues, sustainable agricultural practices incorporating organic amendments and microbial-based bio-inoculants have emerged as viable alternatives. These microbial inoculants enhance soil health, improve nutrient mobilization and solubilization, and promote plant growth while reducing reliance on chemical inputs.14,15 Compared to chemical fertilizers, microbial inoculants nurture soil health by improving its structure and aggregation, nutrient cycling regulating pH, supporting soil microbial communities, enriching fertility by organic matter decomposition, and aiding in heavy metal degradation.16,17 The use of microbial inoculants in agriculture began during the 19th and 20th centuries with the discovery of Rhizobium in leguminous crops which was later purified and patented first by Nobbe and Hiltner in 1896 which laid the foundation for the commercialisation of microbial inoculants and their application in crop cultivation. 18 Nitrogen fixers, phosphorus solubilizers, potassium mobilizers, and plant growth enhancers such as plant growth-promoting rhizobacteria (PGPR), mycorrhizal fungi, and cyanobacteria improve groundnut productivity. Beneficial strains of microorganisms such as Rhizobium phaseoli S18, Rhizobium pusense S6R2, Pantoea dispersa YBB19B, Enterobacter cloacae S23, and Bacillus tequilensis NBB13 enhance nutrient acquisition, stimulate phytohormone synthesis, improve stress tolerance, and act as biocontrol agents against pathogens by producing antibiotics, lytic enzymes, hydrogen cyanide (HCN), bacteriocins, ACC (1-aminocyclopropane-1-carboxylate) deaminase, and siderophores.19–21 They also compete with pathogens for resources and induce systemic resistance in plants, thereby mitigating infections and promoting overall plant health. 22 Microbial inoculants are applied through seed treatment, seedling root immersion, soil application, or foliar spray to ensure efficient colonization and optimal plant growth. These methods enhance nutrient uptake, improve soil fertility, and contribute to long-term agricultural sustainability. 23
Despite extensive research on microbial inoculants in agriculture, there is still a lack of comprehensive reviews detailing their effectiveness in groundnut cultivation. This review explores the microbial diversity, benefits, and mechanisms by which functional microbial groups such as rhizosphere bacteria, phyllosphere bacteria, endophytic bacteria, mycorrhizal fungi, and cyanobacteria contribute to groundnut productivity. Furthermore, it investigates how microbial inoculants influence root exudate metabolites, which interact with stress-responsive proteins under saline and drought conditions. Understanding these microbe-plant interactions through molecular docking and functional analysis provides insights into improving groundnut production under challenging agro-climatic conditions. This narrative review offers a holistic perspective on the potential of microbial inoculants in addressing key challenges in groundnut cultivation, reinforcing their role in enhancing plant growth, resilience, and overall agricultural sustainability.
This review is based on an extensive literature search (2000–2024) across databases including Google Scholar, PubMed, ScienceDirect, SpringerLink, and CAB Abstracts, following the SANRA guidelines for narrative reviews. 24 Key search terms such as ‘microbial inoculants’, ‘groundnut-associated microorganisms’, ‘plant growth promotion’, ‘soil health improvement’, and ‘stress alleviation’ were utilized. Nearly 50 peer-reviewed articles, book chapters, and conference materials were analysed to examine the role of beneficial microbes in sustainable groundnut production.
Microorganisms associated with groundnut
The microbial diversity of groundnuts is crucial as it enhances their growth and development under diverse stress circumstances and increases soil quality. 25 These encompass multiple bacterial and fungal taxa recognized for their roles in nitrogen fixation, phosphate solubilization, disease suppression, and growth enhancement.19,26 This diversity includes both culturable and unculturable bacteria that exert different influences on groundnut development. Understanding groundnut-associated microbial diversity is essential for identifying nitrogen fixers, phosphate solubilizers, plant growth-promoting endophytes, rhizobacteria, and stress-resilient microbes. These microbes enhance soil health, induce systemic resistance, and reduce reliance on synthetic fertilizers, promoting sustainable agriculture.27,28 Figure 2 represents the role of beneficial microorganisms in promoting groundnut growth and sustainability.

Role of beneficial microorganisms in groundnut growth and sustainability
Plants interact with diverse microbial communities that inhabit different ecological niches, including the rhizosphere (root-associated microbes), phyllosphere (leaf-associated microbes), and endophytes (microbes residing within plant tissues). Among these, culturable microorganisms that can be isolated and developed using specific media constitute a minor yet significant fraction of microbial diversity. 29 Despite their limited representation, they play a crucial role in elucidating direct plant-microbe interactions. Several nitrogen-fixing bacteria, such as Rhizobium and Bradyrhizobium, have been isolated from root nodules (Table 1). Similarly, methylotrophic bacteria, including Pseudomonas and Bacillus, have been identified in the phyllosphere, stems, and root zones, exhibiting plant growth-promoting attributes that enhance groundnut growth under field conditions.29,30
List of rhizobia nodulating groundnut.

Microbial diversity includes a vast array of uncultivable microorganisms, such as bacteria, archaea, and fungi, that cannot be cultured under laboratory conditions. 31 Consequently, various methodologies such as metagenomics, next-generation sequencing (NGS) (including Illumina and PacBio), 16S rRNA and ITS sequencing, shotgun metagenomics, metatranscriptomics, and metaproteomics were employed to elucidate the presence of both culturable and unculturable microorganisms associated with groundnut rhizosphere soil, as well as to identify the functional genes within the microbial communities and their roles in nutrient cycling, stress resistance, and disease suppression.32,33
Rhizospheric bacteria
The rhizosphere is the area surrounding plant roots where exudates are released by the plants, affecting microorganism interactions and enhancing plant development and yield. Rhizosphere bacteria promote plant growth in many ways, hence diminishing the need for chemical fertilizers. 42 The rhizosphere of groundnut contains a diverse array of microbes, including bacteria, fungi, actinobacteria, and other microbial entities. The structure and behaviour of microbial communities in the groundnut rhizosphere, together with their beneficial features, must be confirmed by pot cultures and field trials to create prospective bioinoculants. The rhizosphere of groundnut was predominantly composed of the phyla Actinomycetota (formerly Actinobacteria), Pseudomonadata (formerly Proteobacteria), Saccharibacteria, Chloroflexota (formerly Chloroflexi), Acidobacteriota (formerly Acidobacteria), and Cyanobacteriota (formerly Cyanobacteria). In drought-impacted soils, the populations of Actinomycetota and Acidobacteriota increased throughout the seedling and pod development phases, whereas Cyanobacteriota and Gemmatimonadota (formerly Gemmatimonadetes) proliferated during the flowering phase. 43 The mature groundnut rhizosphere displayed an increased abundance of Gammaproteobacterota, Bacillota, and the Bacteroidota (formerly Cytophaga-Flavobacteria-Bacterioides) group, but the Rhizobiaceae family of Pseudomonadota was absent. 44 The research assessed the microbial community of the groundnut rhizosphere and geocarposphere (soil region surrounding developing pod) by 16S rRNA amplicon sequencing, revealing a higher abundance of Acidobacteriota that facilitated seed germination. In contrast, Pseudomonadota, Actinomycetota, and the species of Dyella and Burkholderia exhibited greater abundance in the rhizosphere. 45 In the seed germination phase, Pseudomonas indica predominated, but Bacillus was more prevalent during the vegetative stage in the groundnut rhizosphere. 31 Rhizobium giardinii NET30 and Rhizobium tropici NCHA22 were identified as fast proliferators of rhizobia. 34 Enterobacter arachidis Ah-143 isolated from the groundnut rhizosphere, contains the nifH gene that facilitates atmospheric nitrogen fixation. 46 Co-inoculation of rhizosphere isolates Serratia marcescens EG 10 and Trichoderma harzianum with Bradyrhizobium sp. USDA 3456, led to enhanced vegetative growth, nodule numbers, and pod yield through plant growth-promoting activities such as indole acetic acid production, siderophore synthesis, and phosphorus solubilization. 47
Endophytic bacteria
Endophytes are microorganisms that live either inside the plant tissues either intercellularly or extracellularly, without harming the host plants. 48 The bacteria linked to groundnut found in root nodules are essential for plant growth, nutrient uptake, stress mitigation, and disease suppression. These microorganisms are specialized for particular plant tissues, forming mutualistic partnerships that confer essential advantages to the host plant, which are vital for sustainable agricultural methods. 49 Groundnut root nodules contain a diverse array of endophytic microorganisms, including rhizobial endophytic (RE) bacteria and non-rhizobial endophytic (NRE) bacteria, also known as passenger endophytes (PE), which inadvertently enter the nodule and are regarded as potential biofertilizers and biocontrol agents. Root nodules are specialized structures resulting from the symbiotic relationship between bacteria and the host plant, mostly housing nitrogen-fixing bacteria, particularly Rhizobium and Bradyrhizobium. 25 They transform atmospheric nitrogen (N₂) into ammonia (NH₃), which is accessible for plant absorption to produce amino acids and proteins.
Research on groundnut nodule endophytes indicated that these microorganisms were classified into the phyla Alphaproteobacterota, Betaproteobacterota, Gammaproteobacterota, and Bacillota. The groundnut encompasses various non-rhizobial endophytic genera, including Agrobacterium, Pantoea, Bacillus, Herbaspirillum, Burkholderia, Mistsuaria, Methylobacterium, Pseudomonas, Enterobacter, and Klebsiella. These endophytes exhibit plant growth-promoting characteristics that augment crop development and represent potential bioinoculants for sustainable groundnut agriculture.19,30 Among the 98 bacterial isolates, the majority belongs to the phyla Pseudomonadota comprising Alphaproteobacterota (3), Betaproteobacterota (3), and Gammaproteobacterota (45) as well as Bacillota (44) and Bacteroidota (3). Among these, 58 endophytic bacteria were associated with the bunching type of groundnut (VRI 2), while 40 were linked to the semi-spreading phenotype (VRI 7) (Figure 3(A)). 19 The metagenomic investigation indicated that groundnut nodules harbour actinobacterium Micromonospora, which offers antibacterial characteristics and synthesizes secondary compounds that bolster the groundnut's defence mechanisms. 50 This indicates that root nodules function as microhabitats for diverse bacteria that enhance plant development, soil health, and nutrient availability.
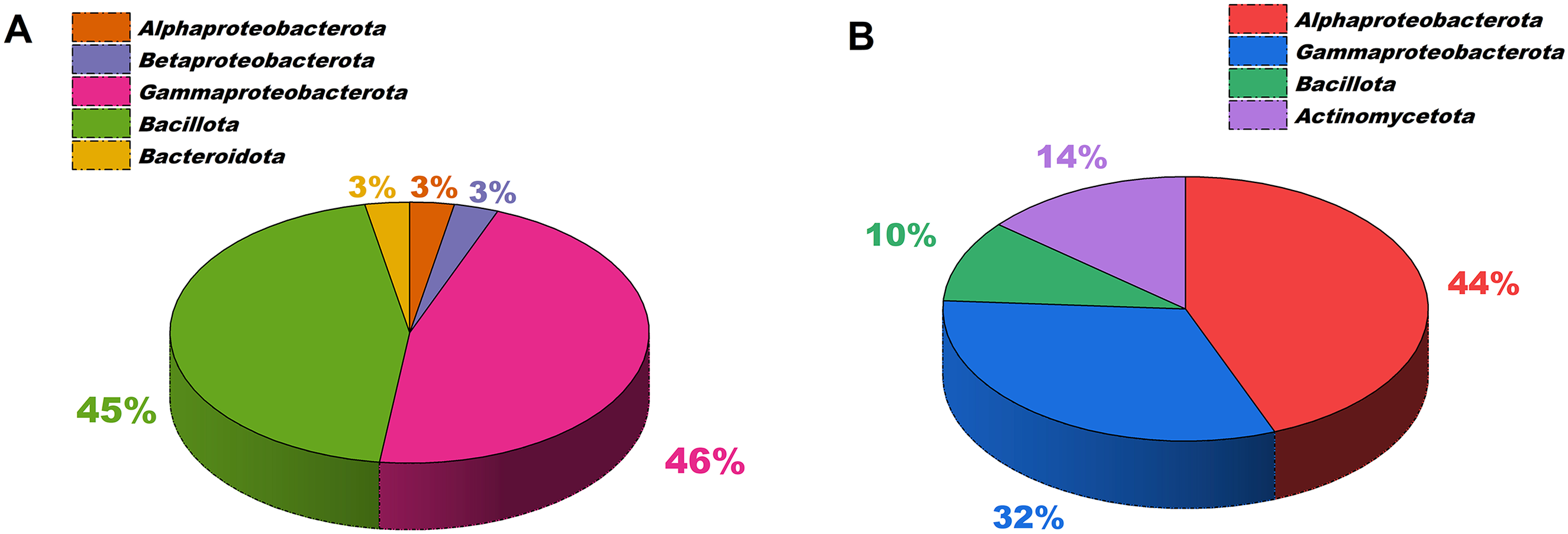
The co-inoculation of RE with PE in groundnut resulted in an increased yield. Enhanced nodule quantities were noted in plants administered with Rhizobium phaseoli S18 and Pantoea dispersa YBB19B, as well as Rhizobium mayense S19 in conjunction with Kosakonia oryzae ESB1. 19 The application of the groundnut endophytic bacterium Bacillus firmus J22N improved kernel and biomass yield. It enhanced plant development and bolstered the plant's resilience to salt stress by generating antioxidants and osmoprotectants. 27 The simultaneous inoculation of Rhizobium pusense S6R2, Rhizobium phaseoli S18, Pantoea dispersa YBB19B, Bacillus altitudinus TBB5A, and Rhizobium pusense S6R2 with Bacillus tequilensis NBB13 led to increased production of IAA and ACC deaminase, enhanced phosphorus solubilization, and improved nodule formation under saline conditions. 21 The co-inoculation of Bradyrhizobium with rapidly proliferating Klebsiella, isolated from groundnut nodules, colonized the existing nodules and improved crop development. 51 The seed treatment with non-rhizobial endophytic bacteria such as Klebsiella pneumoniae AG4, Pseudomonas aeruginosa ABG5, Chryseobacterium indologenes AM2, and Enterobacter cloacae ACP3, resulted in increased nitrogen fixation, phosphorus solubilization, siderophore synthesis, and ACC deaminase activity. 52 Bacterial isolates such as Bacillus sp. GL2 and Paenibacillus glycanilyticus YMR3, inhabit the root tips of groundnut seedlings endophytically, enhancing groundnut biomass yield, nodule quantity, and jasmonic acid levels, thereby bolstering plant defence against pathogens. 53
Phyllospheric bacteria
The leaf surface, known as the phyllosphere, hosts a unique microbial community. These encompass microorganisms that enhance plant health, growth, and stress resilience, including bacteria and fungi. Methylobacterium, Pseudomonas, Bacillus, Streptomyces, Aspergillus, and Trichoderma are microorganisms present in the phyllosphere. 54 Eighty bacterial isolates of methylotrophs were obtained from fifteen groundnut genotypes. Among them, 29 were identified as pink-pigmented methylotrophs (PPFM), whereas 51 were classified as non-pink-pigmented methylotrophs. These methylotrophs represent phyla including Alphaproteobacterota (35), Gammaproteobacterota (26), Bacillota (8), and Actinomycetota (11) (Figure 3(B)). They safeguard plants from detrimental bacteria and fungi by synthesizing antibiotics. 29 These bacteria establish a competitive milieu for infections and impede their reproduction by colonizing the phyllosphere. 55 They enhance the plant's capacity to endure stress from salinity, drought, ultraviolet radiation, and nutrient deficiencies. Furthermore, several microorganisms produce antioxidants that protect plant cells from oxidative damage, including superoxide dismutase, peroxidase, and catalase. 27 Some phyllosphere microorganisms are known to synthesize plant growth-promoting substances like auxins, gibberellins, and cytokinins, which contribute to the enhancement of shoot and root development, as well as overall plant vigour. 56
The in vitro seed germination study, performed by inoculating methylotrophic bacteria isolated from the phyllosphere of groundnut genotypes, demonstrated that these isolates had various growth-promoting characteristics. Seed treatment with Methylobacterium populi TMV7-4, Methylobacterium thiocyanatum VRI7-A4, Methylobacterium radiotolerans VRI8-A4, Pseudomonas psychrotolerans ICGV-5, and Pseudarthrobacter phenanthrenivorans TMV7-A3 resulted in a greater quantity of root hairs, enhanced radicle length, increased shoot length, extended root length, and elevated dry weight. 29 Treating groundnut with Bacillus firmis GRS123, isolated from the rhizosphere, and Bacillus megaterium GPS 55 and Pseudomonas aeruginosa GPS21, both isolated from the phylloplane, resulted in a 26% increase in plant biomass. 57 The assessment of methylotrophs isolated from the groundnut phylloplane indicated that Rhizobium pusense TMV2-6 exhibited superior ACC deaminase activity, Pseudomonas psychrotolerans K-TMV7-6 demonstrated enhanced acetylene reduction assay activity, Pantoea stewartii subsp. indologenes N-CO6-5 generated elevated levels of IAA, and Pantoea dispersa ICGV-2 produced increased exopolysaccharide and solubilized phosphorus. 58 A significant improvement in seed germination was observed in a pot culture experiment with the phyllospheric Methylorubrum aminovorans PPFM-Tm13 encapsulated in polyvinyl alcohol nanofiber. 59
The neighbor-joining phylogenetic tree, constructed with MEGA7 software using 16S rRNA gene sequences of plant growth-promoting bacteria isolated from groundnut, classified these bacteria into the phyla Pseudomonadota, Bacillota (Gram-positive, Low G + C), Actinomycetota (Gram-positive, High G + C), and Bacteroidota including Flavobacterium. 60 Bacteria are classified within the Pseudomonadota phylum, including Bradyrhizobium, Rhizobium, and Methylobacterium, which are positioned distinctly in the phylogenetic tree. Bradyrhizobium established a distinct cluster independent from Methylobacterium. Rhizobium established a distinct clade apart from the cluster comprising Bradyrhizobium and Methylobacterium. Betaproteobacterota and Gammaproteobacterota constituted a distinct cluster apart from Alphaproteobacterota. Actinomycetota constituted a distinct group apart from the Bacillota. Additionally, Bacteroidota established a distinct and profound phylogenetic lineage from the other bacterial phyla (Figure 4).
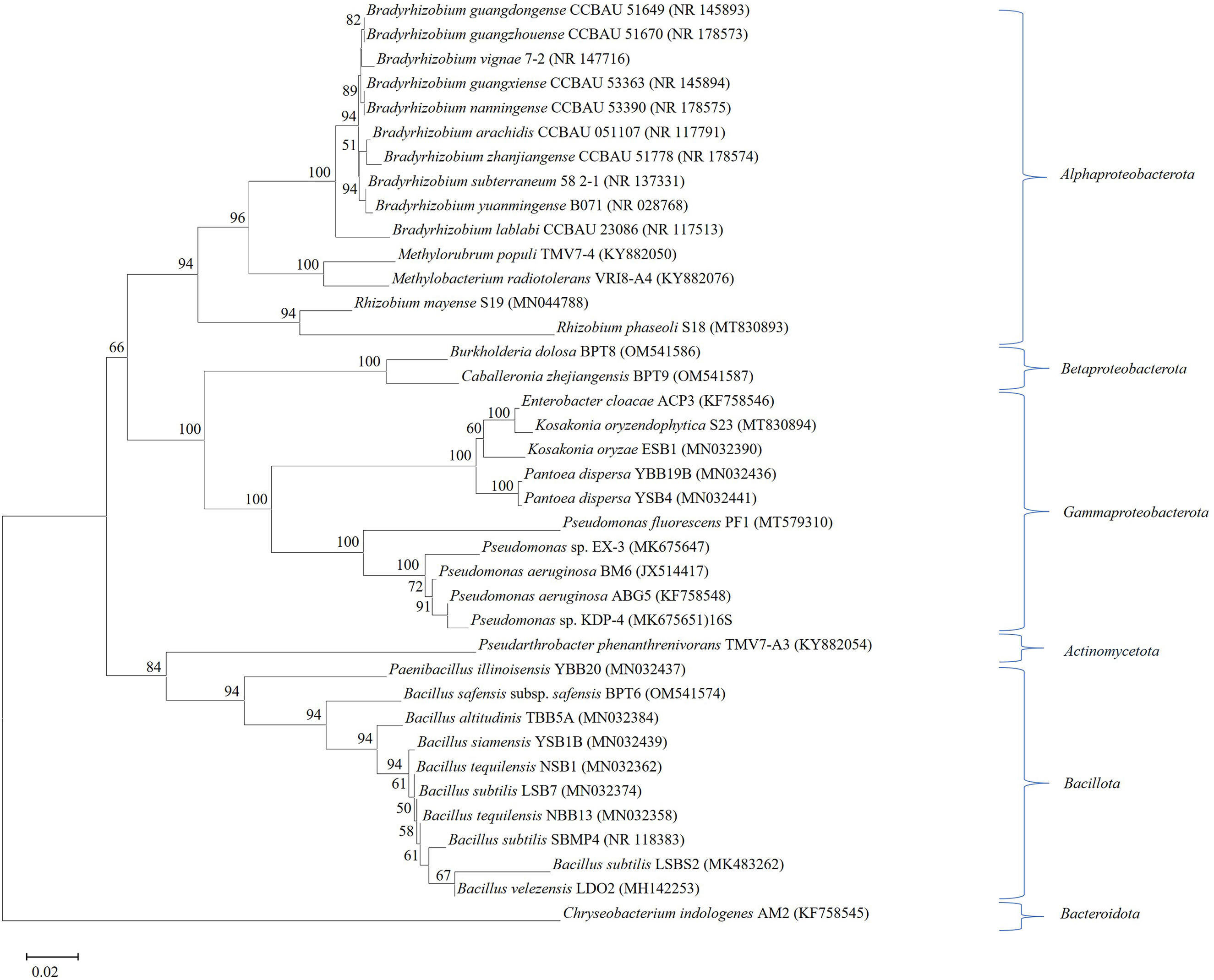
Phylogenetic relationships of plant growth-promoting Bacteria isolated from groundnut. Neighbour-joining phylogenetic tree based on 16S rRNA gene sequences of potential plant growth-promoting bacteria isolated from groundnut. Numbers at the nodes indicate bootstrap values, expressed as percentages of 1000 replications; only values >50% are shown. Bar, 0.02 changes per nucleotide position (Tamura et al. 60 ).
Endophytic fungi
Endophytic fungi inhabit the internal tissues of plants and derive advantages through direct and indirect methods, such as nutrient acquisition, synthesis of phytohormones, antibiotics, and siderophores, as well as the induction of resistance to diverse biotic and abiotic challenges. 28 Inoculation of endophytic fungus by immersion of groundnut seedlings in a Phomopsis liquidambaris B3 fungal suspension enhanced the chemotactic interaction of rhizobia with groundnut by altering the phenolic acids and flavonoids in root exudates. This interaction resulted in an increased number of nodules, enhanced nitrogen fixation efficiency, improved physiological parameters, and increased kernel yield in a monocropping system. 61 A pot culture investigation on the co-inoculation of the endophytic fungal biocontrol agent Trichoderma harzianum and the nitrogen-fixing Bradyrhizobium sp. enhanced groundnut seed germination and increased biomass yield. 62
Mycorrhizal fungi
Mycorrhizal fungi serve as a significant bioinoculant for enhancing soil structure by producing extraradicular hyphae that facilitate macroaggregate formation. Arbuscular mycorrhizae, such as Glomus mosseae, synthesize a specific glycoprotein called glomalin, which functions as a binding agent, aggregating soil particles.63,64 Groundnut plants infected with Glomus mosseae exhibited improved growth due to higher nutrient absorption from the soil and increased production. 65 Arbuscular mycorrhizal fungi diminished the reliance on phosphorus fertilizers and enhanced phosphorus uptake by plants, resulting in higher nodulation and nitrogen fixation. 66 The utilization of commercial Rhizobium fertilizer in conjunction with AM fungus taxa such as Glomus, Gigaspora, and Acaulospora facilitated a tripartite symbiotic relationship with groundnut, enhancing plant development and yield. 67 The integration of Ca2+ with arbuscular mycorrhizae modifies the groundnut rhizosphere microbiota and enhances soil enzyme activity in conditions of salt stress. The populations of the phyla Pseudomonadota and Bacillota, as well as the Sphingomonas sp., increased following the application of mycorrhizal fungi. 68
Cyanobacteria for amelioration
Cyanobacteria, such as Anabaena oryzae and Nostoc muscorum, enhance nutrient uptake in groundnuts through nitrogen fixation, phosphate solubilization, potassium mobilization, calcium uptake, and IAA production. They fix atmospheric nitrogen via heterocysts, where nitrogenase converts N₂ into NH₃, assimilated through the glutamine synthetase-glutamate oxaloacetate transaminase (GS-GOGAT) pathway. Phosphate solubilization occurs through the secretion of organic acids (gluconic, citric, and oxalic) and phosphatases, releasing bioavailable phosphorus for root absorption. Potassium mobilization involves acid-mediated weathering of K-bearing minerals, making K+ ions available for uptake via root K transporters. Calcium solubilization is facilitated by organic acid excretion and biofilm-induced pH modifications, enhancing Ca2+ absorption. 69 Additionally, cyanobacteria produce IAA, promoting root elongation and lateral root formation, and increasing nutrient uptake efficiency. These mechanisms collectively improve soil fertility, plant growth, and stress resilience, reducing dependence on chemical fertilizers and supporting sustainable groundnut cultivation. 70
Mechanisms of growth promotion of groundnut by plant growth-promoting bacteria
The diverse microbial communities associated with groundnut, spanning rhizosphere, endophyte, and phyllosphere niches, play a pivotal role in improving soil health, nutrient availability, and plant resilience. These microorganisms contribute not only by fixing nitrogen, solubilizing phosphorus, and mobilizing essential minerals but also by modulating plant hormone levels, producing bioactive compounds, and enhancing systemic resistance against biotic and abiotic stresses. Understanding the specific mechanisms by which plant growth-promoting microorganisms (PGPM) exert their beneficial effects is crucial for harnessing their full potential and helps optimize microbial inoculants in sustainable groundnut cultivation. The interactions between groundnut and beneficial microorganisms are dynamic, involving multiple direct and indirect pathways that collectively support plant growth, enhance stress tolerance, and suppress pathogens. 71 The growth-promoting characteristics of prospective microorganisms linked to groundnut are listed in Table 2.
Plant growth promoting traits of the potential of microorganisms associated with groundnut.

Direct mechanisms
Nitrogen fixation
Biological nitrogen fixation is an energy-dependent process that consumes 16 ATP molecules and 8 hydrogen molecules, converting di-nitrogen into two ammonia molecules. In a symbiotic relationship, plants provide 12 g of carbon source (glucose) to bacteria for each gram of nitrogen fixed. 81 The inoculation of Rhizobium in groundnuts promotes plant development, augments production, and sustainably boosts soil health. 72 Rhizobium fixes 112–152 kg of nitrogen per acre annually in groundnut. 82 Bradyrhizobium sp. was identified as the predominant nitrogen-fixing bacterium in groundnut, promoting growth and yield. 26 The application of Bradyrhizobium and Trichoderma harzianum, each at 2 g/kg of seeds, in conjunction with gypsum and the prescribed fertilizer dosage, resulted in increased production in groundnut. 83 Biochar enhances soil fertility, sequesters significant quantities of carbon, and contributes to climate change mitigation. A study on utilizing rice husk char as a carrier material for the soil application of Rhizobium demonstrated enhancements in soil nitrification, improved nodule formation and yield, and carbon sequestration, consequently mitigating greenhouse gas emissions. 84 The application of a consortium of bacterial biofertilizers, including Azospirillum, Azotobacter, Rhizobium, and phosphobacteria, in conjunction with AM fungus, enhanced the biochemical parameters such as chlorophyll and protein content in groundnut kernels. 85
Nodule formation by Rhizobium sp. in groundnut
Most studies in groundnuts demonstrated that the root nodules are majorly formed by the slow-growing Bradyrhizobum sp. and scarcely by the fast-growing Rhizobium sp. The rhizobial isolates, Rhizobium huautlense BN 20, Rhizobium pusense BN 23, and Rhizobium alkalisoli BN 50 were found to nodulate groundnut and promote plant growth under stressed conditions. 86 Nitrogen fixation by Rhizobium encompasses multiple steps, beginning with plant signalling and culminating in the formation of nitrogen-fixing nodules (Figure 5). The roots of legumes exude flavonoids into the soil, which attract Rhizobium for infection. This bacterium adheres to the root hair surface through the mediation of a plant protein known as lectin. Following infection, the bacterial transcriptional regulator protein (NodD) was activated, which stimulates the transcription of nodulation genes, resulting in the production of Nod factors (signalling molecules known as lipo-chito oligosaccharides [LCOs]). Nod factors are responsible for the production of nodules in leguminous plants. Upon Nod factors initiation, the root hairs undergo deformation and curling, creating a structure that encapsulates Rhizobium. At this stage, infection threads are established, enabling the bacterium to infiltrate the cortical region, where Rhizobium cells are liberated from the infection threads into individual root cells. They transform into specialized forms known as bacterioids, which can convert atmospheric nitrogen into ammonia. These structures are encased by plant membranes that create an optimal environment for Rhizobium development. These bacterioids develop and form root nodules. 87

Mechanism of Rhizobium nodulation in groundnut through root hair entry
Nodulation by Bradyrhizobium sp. in groundnut
Bradyrhizobium sp. is chemotactically drawn to groundnut root hairs by flavonoids (daidzein, genistein, and chrysin) exuded by the plants. The typical mechanism for rhizobial entry into groundnut involves intercellular penetration through cracks in the root and cortex, leading to the formation of an infection thread. 88 Bradyrhizobium infiltrates the root via the intermediate lamella between neighbouring axillary hair cells and infects substantial basal cells linked to certain axillary root hairs, resulting in the formation of determinate nodules. 89 The development of nodules in legumes by nitrogen-fixing Bradyrhizobium entails several stages. The bacterium located at the lateral roots adjacent to the axillary root hairs infects the roots of legumes. Upon entering the roots, the bacterium disseminates between the plant cells and infiltrates the interior tissues of the roots. In the root cortical region, they initiate proliferation and develop nodule primordia (precursors of root nodules). As bacterial cells grow, plant cells experience morphological alterations and develop immature nodules that subsequently mature into fully developed bacteroids, which are structures that fix atmospheric nitrogen (Figure 6). 90

Mechanism of Bradyrhizobium nodulation in groundnut through crack entry. The process of root nodule formation in groundnut through the crack entry mechanism facilitated by Bradyrhizobium. The process begins when Bradyrhizobium enters near the base of axillary root hairs at the lateral root junction. The bacteria then spread intercellularly through loosened cell walls, creating a pathway for colonization. Once inside, the bacterial cells enter plant cells and undergo division within the nodule primordia, leading to the initiation of nodule formation. As the process progresses, young nodules develop and mature into fully functional nodules containing differentiated bacteroids (Sharma et al. 90 ). These bacteroids play a crucial role in biological nitrogen fixation, enhancing the plant's ability to acquire atmospheric nitrogen and promoting overall growth and productivity.
Phosphate solubilization
The utilization of phosphate solubilizing bacteria as biofertilizers transforms insoluble phosphorus into a soluble form via mechanisms such as soil pH reduction through organic acid production and H+ release, cation chelation, and organic phosphorus mineralization, thereby enhancing phosphorus uptake by plants. 91 Inoculating groundnut with Bacillus thuringiensis B1 in acidic soils enhanced phosphate availability and protein yield. 92 Providencia rettgeri TPM23 is a prospective plant growth-promoting bacterium isolated from encrusted environmental surface soils with significant phosphate solubilization capability, which mitigates the impact of salt stress in groundnut. 93 Acinetobacter pittii YMX11, Pseudomonas indica TPMX14, Brevibacillus shisleri TPMX3, Chryseobacterium lathyri YMX8, Paenibacillus illinoisensis TMX9, Gordonia terrae TPMX16, and Ensifer sesbaniae TPMX5 isolated from groundnut rhizosphere are recognized for their salt-tolerant phosphate solubilizing capabilities, making them potential bioinoculants for groundnut to promote plant growth under saline conditions. 94
Serratia sp. S119, isolated from groundnut nodules, solubilizes inorganic phosphates via gluconic acid synthesis, also synthesizes the phytohormone IAA, and inhibits the growth of the pathogen Sclerotinia sclerotiorum, responsible for sclerotinia rot or stem rot. 95 The phosphate-solubilizing rhizospheric bacteria Bacillus subtilis, isolated from groundnut, may serve as a promising bioinoculant. 96 The assessment of the prolonged impacts of inoculating the phosphate-solubilizing bacterium Burkholderia cepacia ISOP5 and the photosynthetic purple non-sulphur bacterium Rhodopseudomonas palustris ISP-1 as biofertilizers for groundnut demonstrated an enhancement in seed protein content and an increased relative abundance of genes associated with nitrogen metabolism. 97
Potassium solubilization
Potassium solubilizing bacteria typically employ mechanisms such as pH reduction, chelation, and acidolysis. 98 Potassium-solubilizing Micrococcus varians and Corynebacterium kutscheri, when applied to groundnuts, were observed to enhance the quantity of pods and seeds per plant. 99 The pot culture experiment on groundnut utilizing Bacillus sp. PG10, isolated from the rhizosphere of pearl millet, demonstrated superior potassium solubilization capacity, increased seed germination, and enhanced morphological characteristics. 100
Sulphur oxidation
Sulphur oxidizers convert reduced forms of sulphur such as sulphides, thiosulphates, and elemental sulphur into sulphate making it available to plants. Pot culture and field trials including the simultaneous administration of Thiobacillus sp. LCH (an autotrophic sulphur-oxidizing bacterium) and Rhizobium TNAU14 in the groundnut variety ALR 2 increased both nodule quantity and nodule dry weight. 74 Co-inoculation of Rhizobium with sulphur-oxidizing bacteria, namely Thiobacillus strains, in field settings characterized by sulphur-deficient soils resulted in enhanced nodulation, higher plant biomass, elevated accessible sulphur in the soil, an 18% augmentation in pod yield, and improved oil content. 74 Groundnut seeds inoculated with sulphur-oxidizing bacteria and Rhizobium, along with gypsum application to the soil, augmented the available sulphur and improved yield. 101
Phytohormone production
Microorganisms that promote plant growth synthesize phytohormones like auxin (indole-3-acetic acid), cytokinin, gibberellins, ethylene suppressor (1-aminocyclopropane-1-carboxylate deaminase), and abscisic acid. 102 Pot and field trials including the inoculation of pseudomonads from the groundnut rhizosphere have demonstrated the production of IAA and siderophores, the solubilization of inorganic phosphate, inhibition of Aspergillus niger, Aspergillus flavus, and Sclerotium rolfsii, as well as the enhancement of plant growth and yield. 103
Siderophore production
Siderophores are low molecular weight (200–2000 Da), high-affinity iron-chelating compounds synthesized in iron-deficient environments. They solubilize Fe3+ for absorption, replace detrimental pesticides, detoxify heavy metal pollutants, and serve as potential biocontrol agents and biosensors. Siderophore-producing bacteria encompass Pseudomonas fluorescens (ferribactin), Trichoderma sp. (coprogen), Fusarium sp. (fusigen), Klebsiella pneumoniae (enterochelin), Streptomyces pilosus (Desferrioxamine B), Bacillus subtilis (bacillibactin), and Rhizobium meliloti (rhizobactin).104,105 The field inoculation of the catecholate-type siderophore bacillibactin and Bacillus subtilis LSBS2 enhanced iron chelation, oil content, total biomass, and improved plant immunity. 106 The chelation of iron by the siderophores inhibited the growth of plant pathogens indirectly by making iron unavailable to them. 107 A study on the dihydroxymate type of siderophore produced by the Rhizobium isolates namely RM-1, RM-2, and RM-3 from groundnut rhizosphere exhibited antifungal activity against the collar rot pathogen Sclerotium rolfsii. 108
Indirect mechanisms
Microbial inoculants in disease suppression in groundnut
Microbial inoculants act as biocontrol agents for plant pathogens, enhancing crop production and protection, ensuring sustainability, and food safety, and reducing the need for toxic chemicals. 109 The talc-based formulation of the biofertilizer Azospirillum brasilense Az 204 and the biocontrol agent Pseudomonas fluorescens strain PF1, applied to soil in groundnut cultivation, improved root and shoot growth. 78 Pseudomonas aeruginosa AMAAS 57, which produces 2,4-diacetylphloroglucinol (2,4-DAPG), was isolated from the groundnut rhizosphere and demonstrated efficacy in suppressing soil pathogens, specifically Sclerotium rolfsii and Aspergillus niger, responsible for stem rot and collar rot, respectively. 110 A pot culture experiment showed that the simultaneous inoculation of AM fungus (Glomus fasciculatum) and the biocontrol agent (Trichoderma viride) significantly reduced Sclerotium rolfsii in groundnut and enhanced plant growth. 111
Induction of systemic resistance
Plant growth-promoting microorganisms (Rhizobium sp., Glomus sp., Trichoderma sp., Pseudomonas sp., and Bacillus sp.) indirectly induced systemic resistance in plants. They activate the plant's defensive mechanisms against diverse diseases upon exposure to specific triggers. Induced systemic resistance (ISR) is facilitated by the jasmonic acid or ethylene signalling pathways, resulting in enhanced plant health and productivity. The ISR response in plants entails the activation of certain defence genes, and the synthesis of pathogenesis-related proteins (PR proteins), phytoalexins, phenolics, and exopolysaccharides that directly impede pathogens. 112 Microbial elicitors such as lipopolysaccharides, terpenoids, and flagellin stimulate defensive mechanisms against infections. 113 Pseudomonas aeruginosa RP2, when colonizing groundnut, demonstrated an induced defence response by inhibiting the stem rot pathogen, Sclerotium rolfsii, via the root exudate metabolites benzoic acid and salicylic acid. 114 A study on groundnut treatment using Methylobacterium sp. PPFM-Ah with challenge inoculation of pathogenic Aspergillus niger or Sclerotium rolfsii demonstrated enhanced activity of peroxidase (PO), phenylalanine ammonia-lyase (PAL), and β-1,3-glucanase, which elicited systemic resistance against rot pathogens. 55 The seed treatment with Methylobacterium radiotolerans VRI8 A4 in groundnut elicited induced systemic resistance (ISR) against seed-borne fungal infections. 115
Antibiosis
Microbial inoculants produce some bioactive compounds which inhibit the plant pathogens, supporting plant health. Bacillus velezensis LDO2, an endophytic bacterium from groundnut, synthesizes a range of antifungal and antibacterial compounds, including bacilysin, butirosin, bacillaene, difficidin, fengycin, macrolactin, and surfactin, which inhibit plant diseases and enhance plant growth. 116 In vitro, research on Pseudomonas aeruginosa P4, which colonizes the groundnut rhizosphere, demonstrated the production of the antibiotic pyocyanin, which exhibits antibiosis against Fusarium oxysporum. 117
Hydrolytic enzymes
Hydrolytic enzymes such as chitinase, glucanase, protease, and cellulase produced by microbial inoculants can degrade the cell walls of plant pathogens. The enzyme α-1,4-N-acetylglucosaminidase derived from Bacillus subtilis AF1 isolated from the pigeon pea rhizosphere region inhibited the germination of urediniospores of the rust pathogen Puccinia arachidis. 118 Pseudomonas aeruginosa strain Kasamber 11, Bacillus subtilis SBMP4, and Pseudomonas aeruginosa A1K319 isolated from iron-deficient rhizospheric soil exhibited elevated production of hydrolytic enzymes cellulase, pectinase, and chitinase. 119
Beyond enhancing nutrient availability and plant growth, microbial inoculants play a crucial role in improving crop quality. Their ability to influence key physiological and biochemical processes translates into better yield, higher nutrient content, and improved produce quality. In groundnut, the application of beneficial microbes such as Rhizobium, phosphate-solubilizing bacteria, and arbuscular mycorrhizal fungi has been shown to enhance kernel yield, oil content, and protein levels. Let's explore how these microbial inoculants contribute to both the productivity and quality of groundnut.
Combined effect of microbial inoculants and other treatments on groundnut growth, yield, and quality parameters
Microbe-based bioinoculants play a crucial role in the growth and development of groundnut from seed germination to the pod development stage. It significantly enhances the quality indices of groundnut, including pod yield, protein, and oil content. The groundnut production is augmented by 65% with the synergistic application of Rhizobium and phosphorus fertilizer. 120 A study examining the impact of quality parameters of groundnut, utilizing zinc solubilizing bacteria and liquid formulations of recommended NPK fertilizer doses, yielded increased kernel yield, haulm yield, and an enhancement of 6.7 kg of oil per hectare, along with an approximate increase of 4.4 kg in protein content per hectare. 121 A pot culture experiment utilizing the synergistic application of biofertilizers, comprising phosphate solubilizing bacteria and arbuscular mycorrhizal fungus, alongside homo-brassinolide, which modulates phytohormones, yielded a 30% rise in kernel production and a 45% enhancement in oil content. 122 Inoculation with Rhizobium resulted in a maximum oil content of around 46% in the kernels. 123 Field experiments demonstrated a groundnut kernel yield of 2173 kg per hectare, accompanied by a shelling percentage of 79%, achieved through the application of a prescribed dosage of phosphatic fertilizer, phosphate solubilizing bacteria, arbuscular mycorrhizal fungi, and farmyard manure. 124 The co-inoculation of PGPR Stenotrophomonas maltophilia BJ01 facilitated groundnut growth by increasing the synthesis of auxin, phenolic compounds, flavonoids, antioxidant levels, and osmoprotectants in nitrogen-deficient conditions. A field experiment conducted with nitrogen treatment in conjunction with Rhizobium and phosphobacteria demonstrated enhanced growth and yield characteristics, achieving a kernel yield of 2656 kg/ha. 125 The application of Rhizobium and Pseudomonas striata for seed treatment in groundnut enhanced the yields of dry pods and haulm. Furthermore, it led to an augmentation of microbial populations and organic carbon levels in the post-harvest soil. 126 The application of a liquid formulation of zinc-solubilizing bacteria combined with NPK in groundnut enhanced crop physiology, yielding a kernel output of 2114 kg/ha. 121 The administration of a 100% recommended dose of phosphorus (RDP) combined with Farm Yard Manure, phosphate solubilizing bacteria, and arbuscular mycorrhizae resulted in the greatest shelling percentage of 79.23% and a test weight of 42.22 g in groundnut, surpassing the application of 100% RDP alone. 124 The utilization of indigenous strains of sulphur-oxidizing bacteria (Pseudomonas aeruginosa AAU PF 3 and Bacillus tropicus AAU SOB 1) and a zinc-solubilizing bacterium (Beijerinckia fluminensis AAU ZSB F2) in groundnut, combined with a prescribed dosage of fertilizers, gypsum, and zinc sulphate, enhanced the oil content by 53% and protein content by 31.82% in the kernel. 127 The application of liquid bioinoculants including N-fixing Bradyrhizobium, and the zinc solubilizing bacterium, Pseudomonas striata, resulted in the maximum dry pod yield of 29.10 quintals per hectare. 128 The application of Aspergillus terreus, a zinc-solubilizing fungus, at a dosage of 2.5 litres per hectare alongside 100% recommended dose of fertilizers (RDF) yielded superior pod (14.96 g) and haulm (20.18 g) production per plant. 129 The combined application of phosphorus (60 kg P₂O₅/ha) and Rhizobium inoculant (6 g /kg of seed) resulted in an average yield of 2812.5 kg/ha with a benefit-cost ratio of 3. 130 The P fertilizer input was reduced by 15 kg from the recommended RDF (75 kg) with the use of microbial inoculants.
In addition to enhancing the yield and quality of groundnut, microbial inoculants contribute to overall plant health by improving soil fertility and microbial diversity. Their beneficial effects extend beyond nutrient enrichment, as they also help plants withstand various environmental stresses. This resilience is particularly crucial in the face of abiotic stresses, which can severely limit groundnut productivity.
Microbial inoculants in the alleviation of abiotic stresses in groundnut
Among the various abiotic stresses, soil salinity and drought pose significant challenges to groundnut cultivation, affecting plant metabolism, nutrient uptake, and overall yield.
Salt stress
Soil salinity, among other abiotic stimuli, adversely impacts plant growth and development while contributing to land degradation 131 . Research conducted to investigate the impact of salt stress on groundnuts demonstrated that soil salinity modifies the rhizosphere bacteria. The genera Sphingomonas and Microcoleus predominated in salt-affected soils, exhibiting elevated metabolic activities that aid plants in enduring salt stress via several processes including signalling. 132 The phyla Cyanobacteriota and Pseudomonadota exhibited an increase, while Acidobacteriota demonstrated a decline following salt treatment. 76 Research on bacterial genera inhabiting the spermosphere of groundnut identified Bacillus, Massilia, Pseudarthrobacter, and Sphingomonas. The genus Bacillus exhibited increased phosphatase activity, which facilitated phosphorus absorption and improved germination under saline stress. 133 Pot culture experiments on groundnut using PGPR strains Pseudomonas aeruginosa AMAAS57 and Pseudomonas aeruginosa BM6 resulted in enhanced production of phenols and amino acids, which help mitigate free radicals and act as osmolytes under salt stress, respectively. 134 Gas chromatography-mass spectrometry (GC-MS) metabolomics and metagenomic research indicated that intercropping groundnut with sorghum enhanced salt stress resistance through the regulation of root metabolites and the presence of beneficial microbes. 135 A study on inoculation of halotolerant rhizobacterial strains (Klebsiella sp MBE02, Pseudomonas sp. MBE05, Agrobacterium tumifaciens MBEO1, and Ochrobactrum anthropic MBE03) isolated from halophyte Arthrocnemum indicum in groundnut exhibited N fixation, P solubilization, ACC deaminase production reducing ethylene levels by breaking down ACC (precursor of ethylene) into α-ketobutyrate and ammonia, ion homeostasis, and oxidative stress mitigation under salt stress. 136 The inoculation of arbuscular mycorrhizal fungi increased the abundance of the bacterial phyla Pseudomonadota and Bacillota, as well as the fungal taxa Ascomycota and Haematonectria. This led to enhanced groundnut yield in saline-alkali soils. 137 Plant-promoting fungi (PGPF), specifically Trichoderma asperellum and Trichoderma virens, enhanced plant development during the vegetative phases of groundnut under salinity stress. 138 The treatment of endophytes (Bacillus firmus J22N; Bacillus tequilensis SEN15N; and Bacillus sp. REN51N) in groundnut led to increased pod yield under salt stress by producing ROS scavenging enzymes, ACC deaminase, and accumulating proline. 27 The simultaneous inoculation of Rhizobium pusense S6R2, Rhizobium phaseoli S18, Pantoea dispersa YBB19B, Bacillus altitudinus TBB5A, Rhizobium pusense S6R2 and Bacillus tequilensis NBB13 resulted in enhanced IAA and ACC deaminase production, increased phosphorus solubilization, and improved nodule formation under saline stress conditions. 21 Enterobacter cloacae S23, an endophytic passenger in groundnut, acquired elevated levels of proline during saline stress. 139
Drought stress
The populations of Actinomycetota, Acidobacteriota, Cyanobacteriota, and Gemmatimonadota proliferated in the groundnut rhizosphere under drought stress, enhancing the drought tolerance of groundnut. 43 The inoculation of the endophytic fungus Phomopsis liquidambaris B3 enhanced the colonization of root-associated AM fungi, particularly Gigaspora margarita, and conferred drought resistance. 140 Drought-resistant Pseudomonas isolated from the groundnut rhizosphere produces ACC deaminase and exopolysaccharides, reducing ethylene generation, improving water retention, and promoting root growth under drought stress. 141 Pseudomonas strain PCKR-2, isolated from the groundnut rhizosphere, had the highest phosphorus solubilization index and improved plant development under drought conditions. 142 The consortium of Agrobacterium deltaense AMT1, Caballeronia zhejiangensis BPT9, Burkholderia dolosa BPT8, and Bacillus safensis BPT6 improved plant growth and augmented kernel and oil yield under drought conditions. 143 The endophytic drought-tolerant bacterium Bacillus amiloliquefaciens TG4 (Pj2), isolated from the roots of Prosopis juliflora, demonstrated many plant growth-promoting properties in groundnut when inoculated under drought stress conditions. 77 The endophytic bacteria Rhizobium pusense S6R2, Enterobacter cloacae S23, Rhizobium phaseoli S10R2, Bacillus altitudinus TBB5A, and Rhizobium mayense S11R2 serve as effective bioinoculants for groundnut to mitigate drought stress which accumulates a significant amount of osmolytes such as proline, trehalose, and glycine betaine causing osmolyte adjustment, scavenge free radicals, maintain membrane integrity. 144
The role of microbial inoculants in alleviating abiotic stresses in groundnuts extends beyond direct plant growth promotion and stress mitigation. These beneficial microbes influence key physiological and biochemical pathways, leading to osmolyte accumulation, oxidative stress reduction, improved nutrient uptake, and enhanced microbial interactions in the rhizosphere. A crucial aspect of this interaction is the modulation of root exudate composition. Microbial inoculation influences metabolite profiles in root exudates, which in turn shape plant–microbe interactions under stress conditions. Understanding these changes in metabolite profiles provides insights into how microbial inoculants indirectly contribute to stress tolerance, thereby establishing a mechanistic link between abiotic stress alleviation and root exudate modifications.
Effect of microbial inoculants in root exudate metabolites
Root exudates serve as a primary interface between plants and soil microbiota, influencing microbial colonization, nutrient cycling, and stress responses. The composition of root exudates is dynamic and significantly modulated by microbial inoculants, particularly under abiotic stress conditions. Beneficial microbes alter the secretion of organic acids, amino acids, phenolics, and secondary metabolites, thereby enhancing nutrient availability and stress resilience in groundnuts. Understanding the interplay between microbial inoculants and root exudate metabolites provides insights into optimizing plant–microbe interactions for sustainable stress management in groundnut cultivation. Microbial inoculants influence root exudate metabolites, enhancing stress resilience in groundnuts through microbial interactions and biochemical defence mechanisms.
Previous research indicated that the co-inoculation of rhizobial endophytes (RE) Rhizobium phaseoli S18, Rhizobium pusense S6R2, Rhizobium pusense S6R2 with passenger endophytes (PE) Pantoea dispersa YBB19B, Bacillus tequilensis NBB13, Enterobacter cloacae S23 and Pseudomonas aeruginosa RP2 in groundnut resulted in modifications to root exudate metabolites under saline and drought-stressed conditions.19–21 These metabolites exhibit antibacterial, antiviral, antioxidant, and insect-repellent activities, enhancing plant defence and abiotic stress tolerance (Table 3, Figure 7). After endophytic bacterial colonization, groundnut plants produce a variety of signalling molecules like 2,4-di-tert-butylphenol, 1-nonadecene, and nonadecanol which help to attract beneficial microorganisms. Metabolites such as 1-tridecene, hexadecenoic acid, 1-hexadecanol, cyclotetracosane, and pentatriacontane possess antimicrobial properties and act as natural defence mechanisms against soil-borne pathogens. Root exudate metabolites such as quassin, and epoxylanostan are produced in response to drought and saline stress to assist plants in coping with the stress by modifying microbial community and promoting beneficial interactions but their precise molecular functions require further exploration.

Heat map represents the variation in the root exudate metabolites of groundnut under different soil conditions due to the inoculation of rhizobial and passenger endophytes. The heat map was constructed using the retention time of root exudate metabolites from groundnut under different conditions, such as (1) uninoculated control, (2) RE and PE inoculated, (3) Pseudomonas aeruginosa RP2 inoculated, (4) saline-stressed condition (uninoculated control), (5) saline-stressed condition (inoculated with RE and PE), (6) moisture-stressed condition (uninoculated control), and (7) moisture-stressed condition (inoculated with RE and PE) was analysed by gas chromatography–mass spectrometry (GC-MS).19–21
Functional properties of root exudates under different inoculation and stress conditions in groundnut.

Molecular docking of root exudate metabolites produced from rhizobial and passenger endophytes with stress-responsive protein under saline and drought stress conditions
Molecular docking serves as a powerful tool to explore ligand–protein interactions, providing insights into how root exudate metabolites influence stress-responsive pathways at the molecular level. By docking metabolites from microbial-inoculated groundnut root exudates with key stress-related proteins such as DREB, Syntaxin, VIT1, and LEA, we can predict their binding affinities and potential roles in enhancing plant tolerance to salinity and drought stress. These computational analyses help bridge the gap between biochemical metabolite profiling and functional stress adaptation, offering promising leads for enhancing abiotic stress resilience in groundnuts.
Recent studies indicate that molecular docking is an essential method for elucidating interactions between ligands and receptors. 145 Molecular docking approaches have been utilized to predict probable interactions between small compounds and diverse plant target proteins. 146 This study utilized the compounds from root exudates produced by the co-inoculation of rhizobial and passenger endophytes under saline and drought stress conditions and docked them with four previously identified stress proteins.19–21
Dehydration-responsive element-binding (DREB) proteins are crucial for regulating plant responses to abiotic stressors, including salt. DREB proteins play a crucial role in identifying and attaching to particular DNA sequences in the promoter regions of stress-responsive genes, known as DRE/CRT motifs (dehydration-responsive element/C-repeat). 147 Moreover, the Syntaxin protein significantly influences plant development and stress responses in Arabidopsis by modulating cell wall formation, pH levels, and reactive oxygen species (ROS) levels. 148 The VIT1 protein is essential for iron homeostasis in plants during drought conditions. It contributes to drought resistance by positively interacting with tetracyclic diterpenoids, which mitigate oxidative stress during water scarcity. 149 The LEA protein is essential for plant drought survival by stabilizing cellular structure, regulating ion and osmotic equilibrium, and mitigating oxidative damage by lowering reactive oxygen species in plant cells. 150 All proteins were modelled utilizing SWISS-MODEL in the current investigation (Figure 8(A) and (B)). Flexible loops were adjusted in restricted regions of the protein structure. The Computed Atlas of Surface Topography (CASTp) was utilized to identify the active binding locations of the protein (http://sts.bioe.uic.edu/castp/index.html). 151 For ligand production, the metabolites from root exudates were chosen, and their structures were obtained from PubChem in a structured data file. 152 The AutoDock Vina module in PyRx 0.8 and BIOVIA Discovery Studio Visualizer (https://www.3ds.com/products-services/biovia/) were employed for molecular docking to evaluate binding energies and ligand-receptor interactions. 153
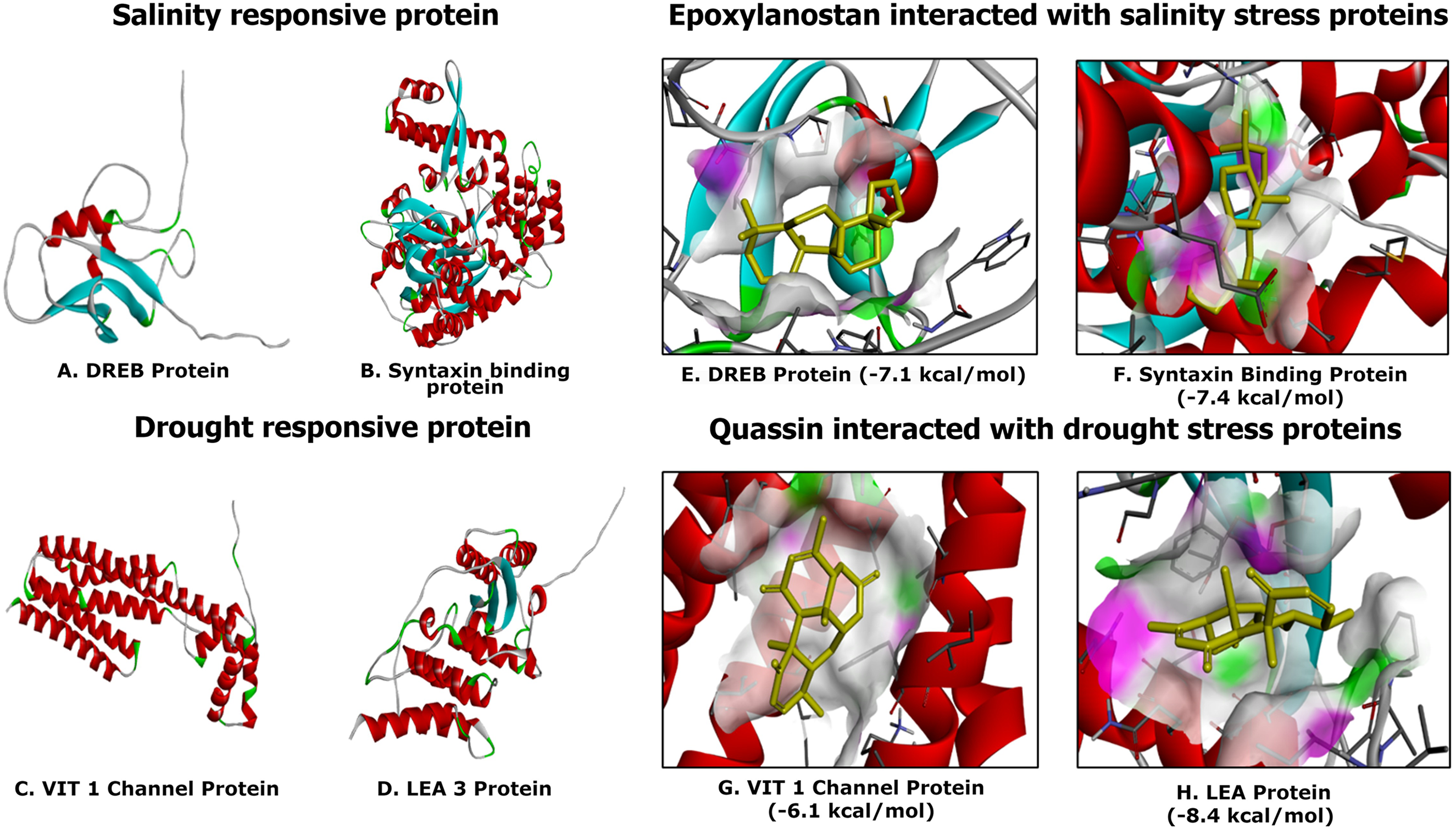
Molecular docking analysis of stress-responsive proteins. Images A and B represent salinity-responsive proteins, where A (DREB protein) and B (syntaxin
Among the identified compounds evaluated against the salinity and drought-responsive protein (Figure 8(C) and (D)), Epoxylanostan interacts with the DREB (dehydration-responsive element-binding) protein with a binding affinity of −7.1 kcal/mol, forming hydrogen bonds with TRP 63. As DREB proteins regulate stress-responsive gene expression, this interaction suggests a possible role of Epoxylanostan in stress mitigation. Similarly, syntaxin-binding protein, which is involved in vesicular trafficking and cellular stress adaptation, binds Epoxylanostan with a higher affinity of −7.4 kcal/mol, forming hydrogen bonds with ASN 173 and engaging in Van der Waals interactions with THR 87, GLN 163, ILE 175, and VAL 174. Conversely, acetic acid displayed the lowest binding affinity, with −2.9 kcal/mol in the DREB protein and −3.5 kcal/mol in the syntaxin-binding protein. The binding affinity values, expressed in kcal/mol, indicate the strength of interaction, with more negative values signifying stronger binding.
Under drought stress, VIT1 Channel Protein (Vacuolar Iron Transporter 1), crucial for ion homeostasis, interacts with Quassin (−6.1 kcal/mol), forming hydrogen bonds with TYR 254 and GLN 250, suggesting a role in maintaining ionic balance under saline conditions. LEA (Late Embryogenesis Abundant) protein, which plays a protective role against dehydration-induced damage, binds strongly with Quassin (−8.4 kcal/mol). The interaction involves hydrogen bonding with ASN 173 and Van der Waals interactions with ILE 175, GLY 105, GLY 85, and THR 87. This suggests that quassin may help stabilize cellular proteins and maintain structural integrity under water-deficient conditions. Conversely, the ligand acetic acid displays the lowest binding affinities of −2.8 kcal/mol in VIT 1 channel protein and −3.1 kcal/mol in LEA 3 protein (Table 4). The inoculation of Bacillus pseudomycoides augmented the expression of the Dreb1 gene using volatile chemicals. Molecular docking revealed two compounds, 2,6-ditert-butylcyclohexa-2,5-diene-1,4-dione, and 3,5-ditert-butylphenol, with binding energies of −6.2 and −6.5 kcal/mol, respectively, suggesting their potential to inhibit MYB (myeloblastosis protein) protein production under drought stress conditions. 154 Similarly, essential oils, particularly taxol, augment drought resilience in Eucalyptus grandis by influencing the VIT1 protein. Molecular docking and dynamics simulations demonstrate that the Taxol–6IU4 complex exhibits considerable binding affinity and stability, implying that this interaction may enhance drought resistance in plants. 149 The incorporation of quercetin dihydrate into the active region of syntaxin proteins may impair their functionality, therefore affecting downstream processes in Chinese flowering cabbage to mitigate salinity stress effects. 155 Nonetheless, the molecular mechanisms underlying drought tolerance in mung beans, emphasizing molecular docking and molecular dynamics simulations to investigate interactions between ABA agonists and a modelled drought-related protein, reveal prospective avenues for improving drought resilience. Epoxylanostan and quassin, exhibiting significant binding affinity for drought and salinity proteins, indicate potential functions in stress tolerance, establishing them as attractive candidates for further research on abiotic stress.
Binding energies and interacting residues of root exudate metabolites with salinity and drought-responsive protein.

Limitations of microbial inoculants in groundnut cultivation
Though microbial inoculants benefit groundnut production in numerous aspects, they temporarily alter the indigenous beneficial soil microbial communities resulting in a shift in microbial diversity and function. Direct effects include competitive, inhibitory (antagonism), or synergistic microbial interaction while the indirect effects include modifications in root exudate patterns and metabolites influencing soil microbial communities. 156 It may disrupt the essential biological processes occurring in soil such as nutrient cycling, organic matter decomposition, plant-microbe interaction, natural symbiosis, biocontrol, and biodegradation of contaminants. Therefore, it is necessary to ensure the safety of indigenous species. A better understanding of strains, their adaptability, interaction studies with other microbes, and field evaluation and site-specific testing of microbe-based inoculants under varied soil types and environmental conditions are needed to ensure the consistent performance of microbial inoculants. 157 Assessment of soil microbiome to select compatible microbial strains with native microbiota, adopting co-inoculation or consortia or synthetic microbial communities (SynCom) promote synergistic benefit, employing molecular methods including metagenomics and qPCR helps in monitoring inoculation impacts on soil microbiota, addition of organic amendments and sustainable agricultural practices could potentially improve the benefits of microbial inoculants. SCAR (sequence characterized amplified region) markers specifically identify the introduced strains from the native microbiota. Knocking out of specific genes from the microorganism could be effectively used to study their role in plant–microbe interaction. Developing new formulation techniques such as polymer-based encapsulation and freeze-dried powder to enhance the shelf life and stability of microbial inoculants. Ensuring regulatory framework prevents the contamination of the environment.
Conclusion
Microbial inoculants play a key role in sustainable groundnut production by enhancing plant growth, facilitating nutrient uptake, improving soil fertility and microbial diversity, augmenting the plant's resilience to environmental stresses, and reducing the dependence on chemical inputs. This review highlighted the significance of groundnut-associated microorganisms, their mechanisms of plant growth promotion, their functional role in drought and salinity stress alleviation, and their effects on root exudate metabolites in stress signalling. Molecular docking studies further revealed the interactions between these metabolites and key stress-responsive proteins (DREB, Syntaxin, VIT1, LEA), demonstrating their role in plant stress adaptation. This review emphasizes the need to integrate microbial inoculants with organic amendments for long-term soil fertility and productivity. Policy support, research investments, and large-scale field trials are crucial for the widespread adoption and practical implementation of bioinoculants in sustainable groundnut cultivation.
Future thrust
The isolation and characterization of native microbial strains are crucial for enhancing sustainable groundnut production. The application of culture-independent methods to investigate soil microbial communities and reveal the functional and taxonomic diversity of indigenous microorganisms in response to bio-inoculant introduction remains to be clarified. The inquiry is necessary to determine the precise mechanisms by which the inoculant confers benefits. Many beneficial microbes are utilized as inoculants and are anticipated to possess multiple modes of action, which can be leveraged in various manners contingent upon the conditions at the inoculation site.
To optimize the use of microbial inoculants, a more systematic approach is required in strain selection, considering environmental parameters, microbial persistence, and interactions within microbial communities. Researchers should mainly focus on strain selection, its mechanisms, and multisite trials to assess their efficacy across diverse agro-climatic conditions, extending the technology from lab to land. Synthetic microbial communities offer a promising strategy to study and harness complex microbial interactions for improved plant resilience and productivity. Moving beyond single-strain inoculants, formulating SynCom-based inoculants can enhance microbial cooperation, nutrient acquisition, and stress adaptation in groundnut cultivation.
The adoption, integration, and optimization of microbial inoculants, along with organic amendments ensure long-term soil fertility, lower input costs, and benefit farmers through higher yield. Through provision subsidies, the establishment of regulatory frameworks for quality control, allocation of funds for research grants, and extension services to enhance farmers’ awareness could promote sustainable soil health. By integrating microbial inoculants with modern biotechnological approaches, we can advance groundnut production systems toward enhanced yield, resilience, and environmental sustainability.
Footnotes
Acknowledgements
The authors extend their gratitude to Periyakaruppan Jegan and Manimaran Gajendiran for their remarkable support with bioinformatics tools, which significantly contributed to the data analysis process.
Authors contribution
KKS contributed to writing the original draft, visualization, formal analysis, data curation, and conceptualization. RA contributed to reviewing and editing, supervision, resources, formal analysis, data curation, conceptualization, and validation. EAY contributed to writing, formal analysis, data curation, and conceptualization. DB, ST, and SV contributed to the formal analysis and review of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
