Abstract
Congenital extrahepatic portosystemic shunt (CEPS) is a rare vascular malformation, classified into type I (complete absence of the portal vein) and type II (presence of a hypoplastic portal vein). This anomaly may lead to severe complications, including liver tumors, hepatopulmonary syndrome (HPS), and pulmonary hypertension. We report the case of a girl in her early teens with type II CEPS and a prior history of hepatocellular carcinoma (HCC), who subsequently developed HPS. Due to the complexity of the vascular anatomy, conventional femoral vein catheterization was unsuccessful. A trans-splenic approach, performed under ultrasound guidance, enabled successful embolization of the shunt using an 18 mm vascular plug. The patient's symptoms resolved completely within 3 months postprocedure, with normalization of oxygen saturation and resolution of orthodeoxia. This case underscores the efficacy of trans-splenic access in complex CEPS cases and highlights the importance of portal pressure monitoring prior to shunt closure to minimize the risk of complications such as superior mesenteric vein thrombosis.
Keywords
Introduction
Congenital extrahepatic portosystemic shunt (CEPS) was first described by Abernethy in 1793. 1 Since then, various pathological presentations and multiple surgical and interventional radiological treatment strategies have been reported.2–5 CEPS can manifest as either end-to-side (type I) or side-to-side (type II) shunts, both of which can be life-threatening.3,6–8 The management of CEPS depends on the type of vascular malformation, and the presence or absence of intrahepatic portal branches significantly influences the treatment strategy.2,5,9 Type I CEPS is more commonly observed in women and children and is frequently associated with a broader range of congenital anomalies and clinical complications compared to type II shunts. Due to the absence of a functional portal vein and the complex vascular anatomy, liver transplantation has traditionally been considered the definitive treatment for type I cases.3,6,10 In contrast, type II CEPS can be managed using either surgical or endovascular approaches. 3 Precise mapping of the vascular anatomy is essential before selecting an appropriate treatment modality.4,9 While computed tomography (CT) and magnetic resonance imaging (MRI) are widely used for this purpose, they may not always be sufficient for diagnosing type I CEPS, making balloon occlusion angiography an indispensable diagnostic tool. 9 Due to phenotypic plasticity, even patients with poorly developed intrahepatic portal venules on imaging may benefit from a staged endovascular approach. 4 Closure of the portosystemic shunt is essential to prevent CEPS-related complications, including hepatic tumors, hepatic encephalopathy, hepatopulmonary syndrome (HPS), and pulmonary hypertension (PHT). 7 Type II CEPS can be effectively treated by occluding the shunt using a vascular plug or by graft stenting of the inferior vena cava. These procedures are typically performed via femoral or jugular vein access.3,4,8 However, catheterization can be technically challenging, particularly when advancing against the direction of vascular flow. In such scenarios, alternative access routes, such as transhepatic or trans-splenic approaches, have been successfully employed—especially in pediatric patients undergoing interventional procedures.2,3,8
Herein, we present the case of a girl in her early teens with type II CEPS who developed HPS and was successfully treated using a catheter-based approach.
Case presentation
A girl in her early teens was referred to the Department of Pediatric Pulmonology at Ankara University Faculty of Medicine, Ankara, Turkey, with exertional dyspnea, cyanosis, and digital clubbing. She was admitted for further evaluation and treatment in October 2020.
Her medical history revealed a diagnosis of hepatocellular carcinoma (HCC), for which she had undergone a right hepatectomy at the age of 5. Although HCC is rare in pediatric patients with congenital portosystemic shunts (CPSS), McLin et al. 11 highlighted that both benign and malignant liver nodules may develop, particularly in cases of extrahepatic CPSS. Although type II CEPS had been identified on CT (Figure 1), shunt closure had not been performed during follow up.
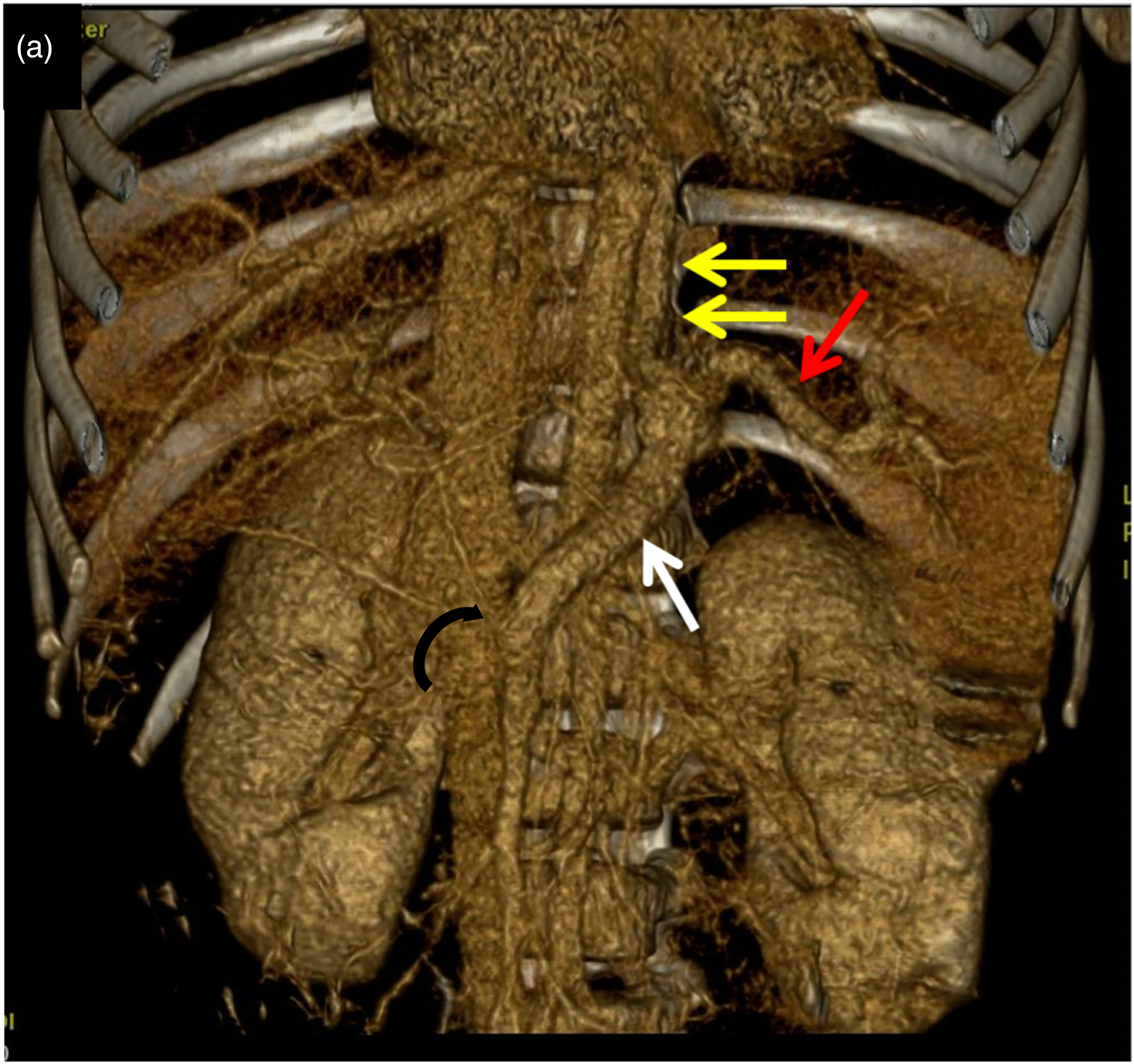
Preoperative 3D CT showing hypoplastic portal vein and azygos vein formation. 3D: three-dimensional; CT: computed tomography.
Upon admission, her peripheral oxyhemoglobin saturation (SpO2) on room air was 89% in the sitting position and 91% in the recumbent position. Arterial blood gas analysis revealed hypoxemia, an increased alveolar-arterial gradient, and normal carbon dioxide levels, accompanied by orthodeoxia. She did not exhibit polycythemia, liver dysfunction, or elevated alpha-fetoprotein levels. There was no clinical or laboratory evidence of ascites, anemia, or edema.
Chest X-ray and electrocardiogram findings were unremarkable. Echocardiographic evaluation revealed no signs of pulmonary hypertension (PHT). Contrast echocardiography with agitated saline demonstrated delayed appearance of contrast after three cardiac cycles. Thoracic CT angiography did not identify any arteriovenous communication. MRI revealed multinodular lesions in the left lobe of the residual liver.
Based on these findings, the patient was diagnosed with type II CEPS accompanied by hepatopulmonary syndrome (HPS), multinodular hepatic lesions, and HCC. Previous Doppler ultrasound imaging had been unsuccessful in visualizing the intrahepatic portal veins (Table 1).
Time line and course of the disease from first diagnosis to initial treatment.

CEPS: Congenital extrahepatic portosystemic shunt.
Figure 1: Preoperative Virtual Reality Reconstructed 3D images image (a) shows hypoplastic portal vein connected to SMV via thin venous branch (curved arrow). Portal venous system after joining splenic (red arrow) and mesenteric veins (white arrows) continues as azygos vein (double arrows).
Initial catheterization of the portosystemic shunt was attempted via the femoral vein under fluoroscopic guidance. However, due to the complex vascular anatomy, femoral access was unsuccessful in advancing the catheter through the shunt. Therefore, diagnostic angiography via trans-splenic access was performed to delineate the anatomical configuration of the shunt and to assess its suitability for endovascular closure. The patient's left hypochondriac region was sterilized using chlorhexidine solution. Under ultrasound guidance (Sono Site, USA), the most inferior peripheral splenic venule was punctured using a 21 G, 7 cm micropuncture needle (Merit Medical, Utah, USA). Splenoportography via a 4 Fr sheath revealed a 10 mm diameter shunt between the splenic and hemiazygos-azygos veins. Portal pressure and right atrial pressure were recorded as 16 and 12 mmHg, respectively. Following pressure measurements, heparin (100 IU/kg) was administered, and a 0.035'' exchange Glidewire® (Terumo, Tokyo, Japan) was advanced into the inferior vena cava through the shunt via the splenic sheath. The wire was captured and externalized via the right femoral vein using a 15 mm Amplatzer Goose Neck Snare device (Medtronic). The femoral sheath was replaced with a 7 Fr, 90 cm sheath (Arrow, Teleflex®, Morrisville, NC), and over the through-and-through wire, a long sheath was advanced within the shunt near the splenic vein junction. A vascular plug (Cera-Vascular Plug, Lifetech Scientific, Shenzhen, China) was deployed within the shunt without disconnecting the delivery system. Subsequent splenoportography through the splenic sheath showed redirection of flow to the portal system and visualization of hypoplastic intrahepatic portal branches after plug occlusion (Figure 2). Portal pressure measured 23 mmHg 20 minutes following test occlusion, after which the plug was released (Figure 3). The splenic puncture tract was embolized using a Gelfoam torpedo. Low molecular weight heparin (0.2 mg/kg) was administered for 10 days. No antibiotic prophylaxis was used during or after the angiographic procedure.

Schematic and angiographic images of our patient with CEPS. CEPS: Congenital extrahepatic portosystemic shunt.
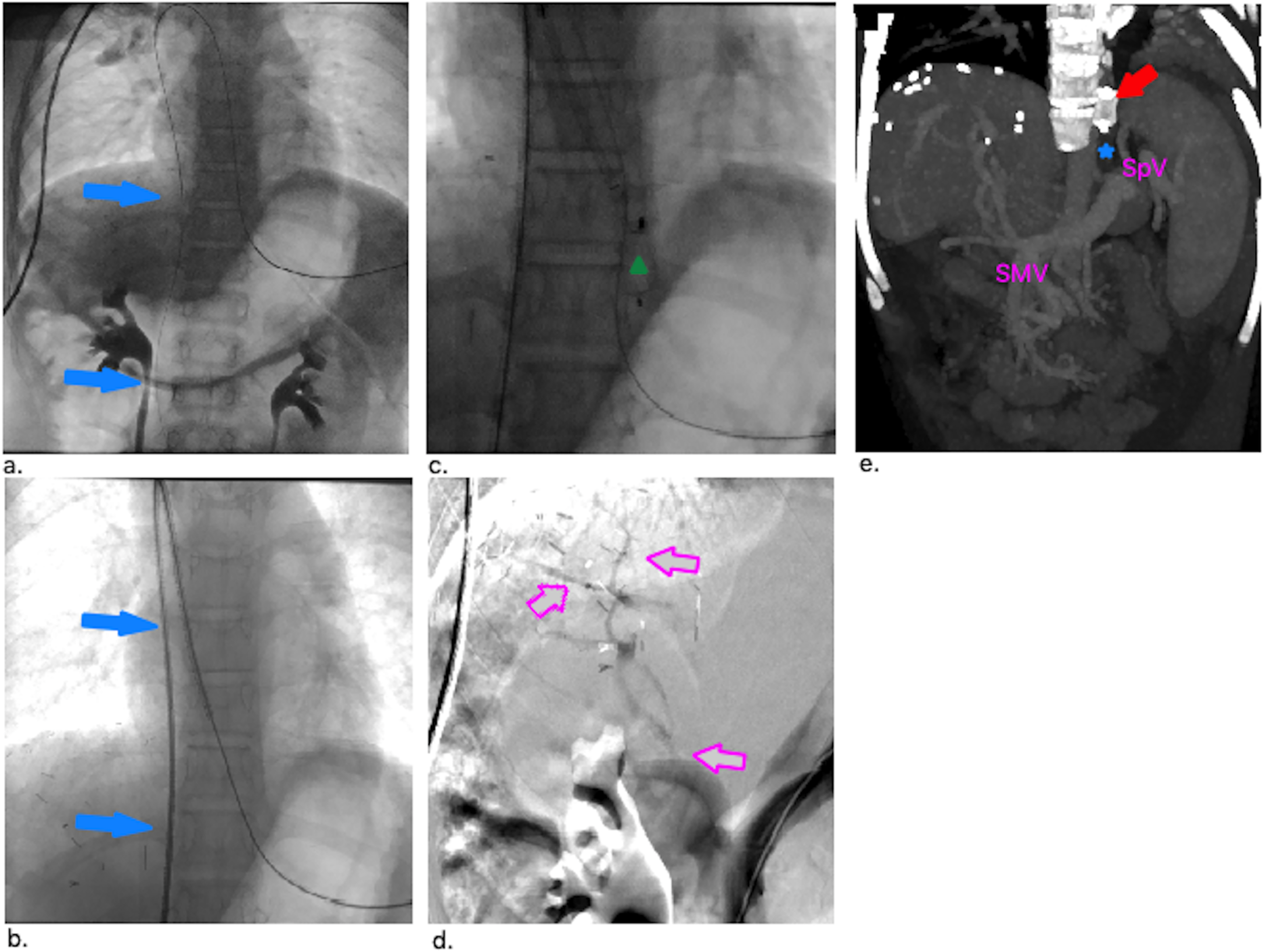
Angiographic images showing shunt wiring, vascular plug deployment, and postocclusion changes.
Figure 2: Schematic illustration, and pre- and postprocedural images of a female patient in her early teens with type II CEPS presenting with HPS. (a) Schematic drawing of the malformation. (b) Merged angiographic images of the shunt obtained by antegrade and retrograde catheters. CEPS: Congenital extrahepatic portosystemic shunt. HPS: Hepatopulmonary syndrome.
Figure 3: (a, b) Through-through wiring of the shunt and placement of the long sheath near splenic vein junction (blue arrows). (c) Stored image of vascular plug after successful deployment shown with green triangle. (d) Demonstration of the hypoplastic intrahepatic portal branching after plug occlusion (pink arrows). (e) Vascular plug (red arrow) and thrombosed shunt (blue star) shown at CT images. SpV: splenic vein, SMV: superior mesenteric vein.
Splenic contusion and postprocedural hypersplenism were identified on CT imaging performed after the embolization. However, bleeding from the splenic puncture site was effectively controlled without the need for additional interventions and gradually resolved during the hospital stay with close clinical and laboratory monitoring and supportive care. Hypersplenism resolved by the fourth day following the procedure. The patient was discharged 10 days postprocedure with a room air SpO2 of 95% at rest and a normal orthodeoxia test. At the 3-month follow up, she remained asymptomatic and normoxemic.
This case report was conducted in accordance with the principles of the Declaration of Helsinki. All patient details have been anonymized to ensure confidentiality. Informed written consent for treatment was obtained from the patient's legal guardians. The reporting of this case adheres to the CARE guidelines. 12
Discussion
There are several mechanisms involved in the development of HPS in the context of CEPS. Elevated levels of endothelin-1 (ET-1) contribute to HPS by persistently stimulating nitric oxide synthase.13,14 Additionally, translocation of intestinal bacteria may activate alveolar macrophages, leading to upregulation of inducible nitric oxide synthase (iNOS) expression. Increased ET-1 levels within the shunt circulation promote pulmonary vasodilation and angiogenesis. These vascular changes contribute to ventilation-perfusion mismatch, impaired oxygen diffusion, the formation of arteriovenous shunts, and ultimately the clinical manifestation of hepatopulmonary syndrome.13,14
Hepatic neoplasms, including regenerative nodular hyperplasia, hepatic adenoma, and hepatocellular carcinoma (HCC), have been reported in patients with CEPS. These lesions are thought to arise due to impaired portal venous inflow, resulting in compensatory augmentation of hepatic arterial perfusion and abnormal hepatocellular proliferation. Over time, these tumors may demonstrate progressive growth; however, they may stabilize or even regress following shunt closure during long-term follow up.7,11,15
In type II CEPS, early shunt closure is recommended to prevent and/or resolve complications and clinical manifestations. Although the present patient underwent right hepatectomy 7 years prior, it appears that shunt ligation was not planned or performed at that time. A study by Baiges et al. emphasized the importance of shunt closure not only as a treatment strategy for existing complications, but also as a prophylactic approach in asymptomatic patients to prevent severe outcomes such as hepatopulmonary syndrome, pulmonary arterial hypertension, and hepatocellular carcinoma. This preventative strategy may positively influence disease progression, particularly in patients at high risk for these life-threatening complications. 16
New-onset multinodular hepatic lesions and HPS were detected during follow up in this case. Therefore, long-term surveillance is essential for the timely identification of potential complications in CEPS patients, especially in type II cases without prior surgical or interventional shunt correction.
With advances in technology, interventional radiological approaches to CEPS treatment have significantly improved. Portal pressure screening and measurement of the portal-to-systemic pressure gradient are critical during intervention. It is recommended to maintain portal pressure below 18 to 25 mmHg and a portosystemic gradient below 10 mmHg to prevent complications such as mesenteric vein thrombosis.3,9 Femoral, jugular, and transhepatic routes are most commonly employed for portosystemic shunt embolization. 3 The through-and-through wiring technique offers advantages for catheterization and may help avoid wire kinking or device delivery difficulties. Trans-splenic access has proven to be a safe option for various interventional procedures, particularly in CEPS occlusion. 17 Although not previously reported in the literature, we hypothesize that the use of large-caliber splenic sheaths (≥7 Fr) may increase the risk of hemorrhagic complications compared to smaller sheaths (≤5 Fr) 17 . For this reason, femoral access was preferred for plug deployment. Gelfoam torpedo was chosen for splenic tract embolization, as it has shown comparable efficacy to other embolization techniques. 17 A vascular plug was used for shunt occlusion; however, other successful treatment methods have also been described in the literature. 3
In this case, no complications related to endovascular embolization with the vascular plug were observed. The patient was discharged 10 days postprocedure with a room air SpO2 of 95% and a normal orthodeoxia test. At the 3-month follow up, she was normoxemic and remained asymptomatic at her most recent evaluation.
Conclusion
CEPS should be managed using either surgical or interventional radiological procedures to prevent and/or resolve associated complications. Prior to occluding the portosystemic shunt, it is essential to perform portal pressure measurements to minimize the risk of shunt occlusion-related complications, such as superior mesenteric vein thrombosis.
Learning points
CEPS has to be treated with either surgical or interventional radiological techniques to prevent and/or resolve related complications. Trans-splenic access could be used safely in pediatric population. Measuring the portal pressure is mandatory before terminating the portosystemic shunt to prevent shunt occlusion-related complications, such as superior mesenteric vein thrombosis.
Footnotes
Acknowledgements
The authors would like to thank all medical staff involved in the diagnosis, treatment, and follow up of the patient presented in this case report. We also extend our gratitude to the pediatric and radiology teams at Ankara University Faculty of Medicine for their valuable support during the clinical management and data collection processes.
Ethical considerations and patient consent
This study has been conducted in accordance with the ethical standards outlined in the 1964 Declaration of Helsinki and its later amendments. Written informed consent was obtained from the patient and/or their legal guardian prior to inclusion in this study. Institutional Ethics Board approval was not required for this case report.
Author contributions/CRediT
ECÇmade significant contributions to the concept, study design, data acquisition, and analysis and/or interpretation of the data. Drafted the initial version of the manuscript, revised it critically for important intellectual content, approved the final version for publication, and agreed to be accountable for all aspects of the work to ensure its accuracy and integrity. EUB contributed to the analysis and/or interpretation of the data, drafted significant portions of the manuscript, revised it critically for important intellectual content, approved the final version for publication, and agreed to be accountable for all aspects of the work to ensure its accuracy and integrity. SKS contributed to the analysis and/or interpretation of the data, participated in manuscript drafting and revisions for intellectual content, approved the final version for publication, and agreed to be accountable for all aspects of the work to ensure its accuracy and integrity. FZ contributed to project development, provided critical insights into the manuscript during revisions, reviewed the article for important intellectual content, approved the final version for publication, and agreed to be accountable for all aspects of the work to ensure its accuracy and integrity. SF contributed to the analysis and/or interpretation of the data, provided guidance and supervision throughout the project, revised the manuscript critically for intellectual content, approved the final version for publication, and agreed to be accountable for all aspects of the work to ensure its accuracy and integrity. NÇ provided critical oversight and supervision of the study, contributed to the manuscript's critical revision for intellectual content, approved the final version for publication, and agreed to be accountable for all aspects of the work to ensure its accuracy and integrity.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data supporting the findings of this case report are included within the article. Due to patient confidentiality, raw data are not publicly available but can be provided by the corresponding author upon reasonable request.
