Abstract
Pulmonary artery sarcoma (PAS) is a rare but aggressive malignancy often misdiagnosed as pulmonary embolism (PE) due to similar clinical presentations. This case report presents an early 40s female with progressive dyspnea, hemoptysis, high pulmonary arterial pressures (112 mmHg), and right heart strain unresponsive to anticoagulation therapy. Imaging findings, including cardiac magnetic resonance and positron emission tomography-computed tomography, ultimately revealed PAS. This case highlights the importance of considering rare etiologies, such as PAS, in patients with PE-like symptoms refractory to standard treatment. Early detection using advanced imaging techniques can improve diagnostic accuracy and outcomes in this rare malignancy.
Keywords
Introduction
Pulmonary artery sarcoma (PAS) is a rare primary vascular tumor, with an incidence of 0.001–0.003%. 1 It most commonly affects the pulmonary artery, and its prognosis is typically poor. The clinical presentation of PAS overlaps with pulmonary embolism (PE), often leading to misdiagnosis and prolonged inappropriate treatment, such as anticoagulation therapy. PAS arises from primitive mesenchymal stem cells, which possess the potential to differentiate into various cell types, contributing to its diagnostic complexity. The non-specific nature of PAS symptoms and its radiologic resemblance to PE make it particularly challenging to diagnose. This report aims to provide insight into the clinical and imaging features that may aid in distinguishing PAS from PE, emphasizing the importance of early diagnosis in improving patient outcomes.
Case presentation
An early 40s woman with no significant medical history presented with a 2-month history of progressive dyspnea. Initially, her dyspnea was exertional, occurring after heavy physical activity and accompanied by chest pain that resolved with rest. Her condition gradually worsened, with dyspnea triggered by emotional stress and associated with cyanosis. After an episode of hemoptysis following an emotional conflict, she sought medical care. She also experienced weight loss of 4 kg within these 2 months. Two days later (October, 2018), the patient was referred to Guangdong Provincial People's Hospital (Guangzhou, China) for further evaluation.
On examination, the patient was febrile (temperature: 38.5 °C), tachycardic (111 bpm), and tachypneic (31 breaths/min) with a blood pressure of 117/71 mmHg. Jugular venous distension was noted, and cardiac auscultation revealed a loud second heart sound (P2). The lungs were clear, and no peripheral edema was present. She had no history of coronary artery disease, hypertension, diabetes, rheumatologic conditions, smoking, alcohol use, or a family history of cardiovascular or malignancy-related diseases.
Electrocardiography showed sinus tachycardia and an S1Q3T3 pattern and T-wave inversions in leads V1-V4, indicating possible right heart strain (Figure 1). A chest X-ray revealed an enlarged cardiac silhouette and pulmonary hilar prominence (Figure 2(a)). Transthoracic echocardiography demonstrated right atrial and ventricular enlargement, moderate tricuspid regurgitation, increased right ventricular wall thickness (6.1 mm), and severe pulmonary hypertension (estimated pressure: 80 mmHg) (Table 1). Laboratory tests showed elevated NT-proBNP (2977 pg/mL), near-normal D-dimer levels, and mildly elevated inflammatory markers (ESR, CRP) and neuron-specific enolase (25.5 ng/mL; reference: 0–16.3 ng/mL). Arterial blood gas analysis indicated hypoxemia with a pO₂ of 57 mmHg. Abdominal ultrasound and lower limb venous Doppler studies were unremarkable.
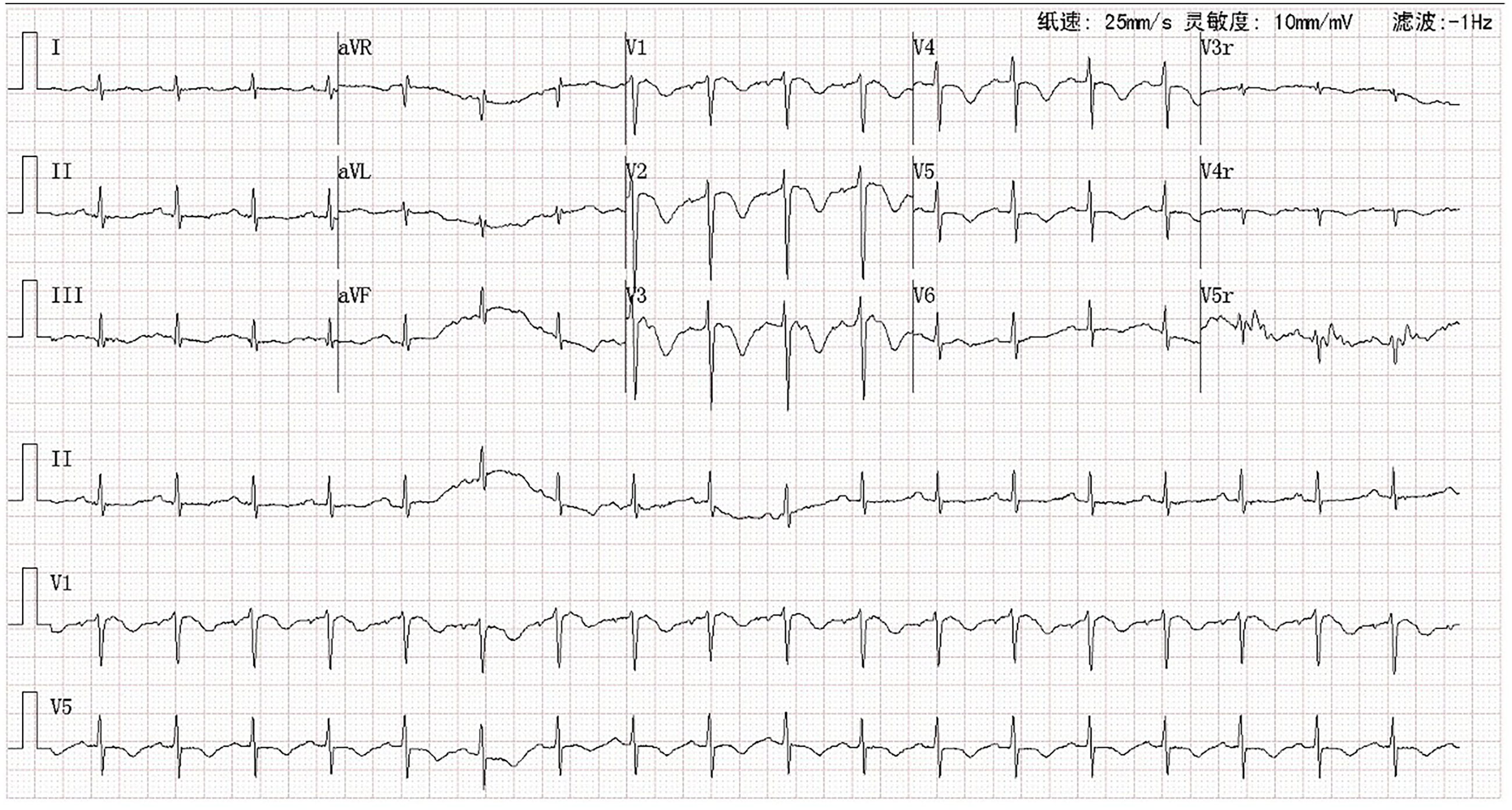
Electrocardiogram showing possible right ventricular strain.
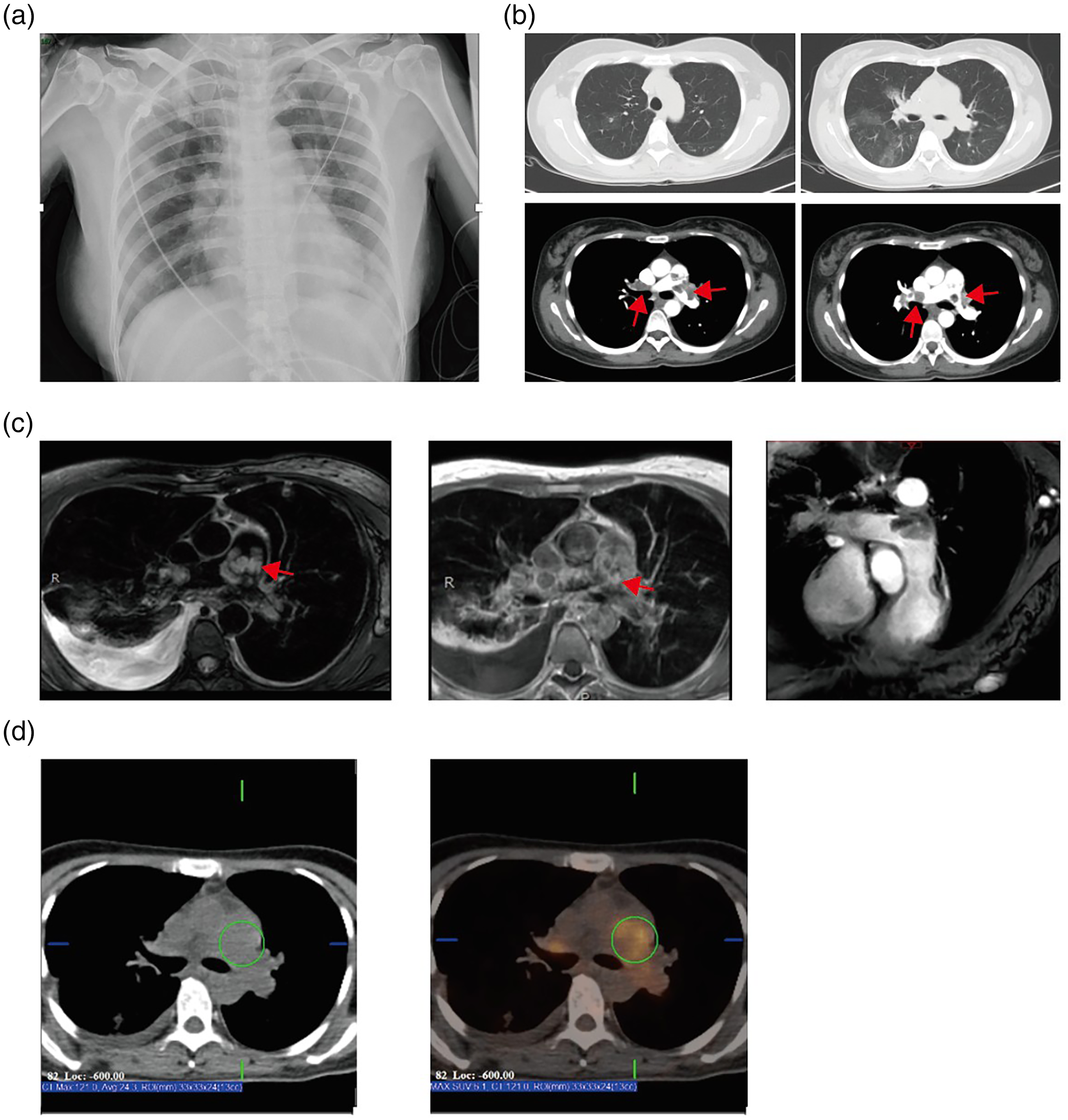
Imaging findings of the patient. (a) Chest radiograph; (b) pulmonary artery CT: lung window showing patchy opacities, mediastinal window showing multiple filling defects in the main and bilateral pulmonary arteries (arrows indicate filling defects); (c) MRI revealing a cancerous lesion in the pulmonary arteries (arrow indicates high-signal focus within the pulmonary artery); (d) PET-CT showing filling defects in the pulmonary arteries with increased glucose uptake. PET-CT: positron emission tomography-computed tomography.
Echocardiographic findings of the patient.

CT pulmonary angiography identified filling defects in the main and bilateral pulmonary arteries (Figure 2(b)), with atypical features, including patchy ground-glass opacities suggestive of pulmonary infarction. Despite a high Wells score (5.5), the near-normal D-dimer level and atypical imaging raised suspicion for non-thrombotic causes. Nevertheless, thrombolysis with urokinase (1,200,000 U) was administered at a local hospital due to suspected pulmonary embolism (PE), high Wells score, and clinical presentation of progressive dyspnea. The patient's symptoms worsened, leading to transfer for advanced management.
At our facility, with the patient's consent, anticoagulation therapy with enoxaparin (4000 U, q12h) was initiated, later transitioned to rivaroxaban (20 mg daily). Riociguat (0.5 mg BID) and bosentan (62.5 mg BID) were added, but her pulmonary arterial pressures continued to rise (from 80 mmHg to 112 mmHg), and clinical improvement was absent. Given the lack of response, chronic thromboembolic pulmonary hypertension 2 or PAS 3 was considered (Figure 3).

Echocardiography showing a hypoechoic mass (size: 29 × 21 mm) at the bifurcation of pulmonary trunk, extending into left and right pulmonary arteries (arrow indicates intraluminal thrombus).
Further imaging supported the suspicion of PAS. Cardiac magnetic resonance imaging revealed contrast enhancement of the pulmonary artery filling defects, indicating a mass rather than thrombus (Figure 2(c)). Positron emission tomography-computed tomography showed increased glucose uptake within the filling defects, suggestive of a malignant process 4 (Figure 2(d)).
The patient underwent surgical resection of the pulmonary artery mass. Intraoperative findings revealed diffuse, white-yellowish, cauliflower-like tumor tissue filling the main and bilateral pulmonary arteries 5 (Figure 4(a)). Histopathological examination with hematoxylin and eosin (H&E) staining confirmed a spindle cell sarcoma with moderate nuclear atypia and myxoid changes (Figure 4(b)). Immunohistochemistry was positive for desmin and Ki-67 (80%+), and molecular pathology identified MDM2 gene amplification via fluorescence in situ hybridization (Figure 4(c)–(e)). The pathological report confirmed the diagnosis of endarterial sarcoma. 6

Pathological findings of the patient. (a) Gross pathology of the pulmonary arterial tumor; (b) H&E staining of the pulmonary arterial tumor (arrow indicates vascular wall components, with atypical tumor cells on the right); (c) IHC: Desmin (+); (d) Ki67 (80%+); (e) molecular pathology: MDM2 fluorescence in situ hybridization (FISH) (+) (arrow indicates positive signal). H7E: hematoxylin and eosin; IHC: immunohistochemistry.
Postoperatively, the patient required extracorporeal membrane oxygenation due to hemodynamic instability from severe pulmonary hypertension. Despite aggressive support, she developed recurrent airway bleeding and multi-organ dysfunction syndrome. After discussions with her family, life-sustaining treatment was withdrawn, and the patient passed away shortly thereafter.
The reporting of this study conforms to CARE guidelines. 7
Discussion
PAS is a rare malignancy, presenting similarly to PE, such as dyspnea, chest pain, and right heart strain. 8 Differentiating PAS from PE is challenging due to overlapping clinical features and imaging findings. In this case, the patient was initially misdiagnosed with PE, as imaging revealed filling defects in the pulmonary arteries. However, the near-normal D-dimer and lack of response to anticoagulation therapy raised suspicion of PAS. 9 PAS typically presents with a mass in the pulmonary artery, which can be misinterpreted as PE on imaging. Unlike PE, PAS often involves the arterial wall and can infiltrate surrounding tissue, though it typically lacks the mass effect seen in lung cancer. Histopathological confirmation is essential; molecular pathology testing of the MDM2 gene suggests that targeted therapy against this gene may benefit patients with such mutations. 10 Surgical resection remains the most effective treatment, though prognosis is often poor due to late presentation.
Conclusion
This case highlights the importance of considering PAS in patients with PE-like symptoms refractory to anticoagulation. PAS may be mistaken for PE, leading to delays in diagnosis and suboptimal treatment. Early diagnosis through advanced imaging and histopathological evaluation is crucial, as surgical resection is the only intervention shown to improve survival. Clinicians should maintain a high index of suspicion for PAS, especially in cases of suspected PE with atypical or non-responsive symptoms.
Footnotes
Acknowledgements
The authors wish to gratefully acknowledge the patient and family.
Ethical considerations
Approval by the Institutional Review Board is not required for individual case report.
Informed consent
Written informed consent was obtained from the patient's family for their anonymized information to be published in this article.
Author contributions/CRediT
YXY did resources, visualization, and writing—original draft and editing. LJF did conceptualization, resources, supervision, methodology, project administration, and editing. CJH did conceptualization, supervision, funding acquisition, writing-original draft, and writing—review and editing.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Beijing Great Physician Commonweal Foundation (Grant No.CSCF2022B01).
Conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Data available on request from the authors.
