Abstract
Acute hemorrhagic rectal ulcer (AHRU), though a relatively rare etiology of lower gastrointestinal bleeding, represents a critical clinical emergency characterized by sudden onset, painless, massive, and fresh rectal bleeding. While endoscopic hemostasis remains prominent therapy, its efficacy is limited in emergency settings by poor bowel preparation and obscured visualization. Rectal arterial embolization (RAE) offers a viable alternative, however, detailed descriptions of its application for AHRU in literature are lacking. Thus, we report a single-center experience of RAE in six patients with massive AHRU-related bleeding. Technical success was achieved in 83% (5/6) of cases, with clinical success was achieved in 100% cases (6/6). Rebleeding after initial RAE procedure occurred in 33% (2/6) of cases, one of which ultimately achieving hemostasis through transanal suturing after initial embolization failure. No major embolism-related complications were observed during follow-up. Moreover, complete embolization of rectal supplies demonstrated potential to reduce rebleeding risk, and empiric embolization may hold promise for managing refractory AHRU hemorrhage. These findings advocate for RAE as an effective alternative for AHRU hemorrhage, though further validation in larger cohorts is essential.
Keywords
Introduction
Acute hemorrhagic rectal ulcer (AHRU) is a relatively rare cause of lower gastrointestinal bleeding (LGIB), with a reported incidence of approximately 0.9–23%.1–6 It is characterized by sudden onset, painless, massive, and fresh rectal bleeding. Although the pathogenesis of AHRU remains unclear, its acute onset and potential to cause life-threatening hemorrhage necessitate urgent diagnosis and treatment.
Surgical intervention for LGIB is associated with significant invasiveness, substantial morbidity, and an estimated mortality risk of 15–30%.7,8 Consequently, minimally invasive techniques have become the preferred first-line therapeutic approach. The optimal hemostatic strategy for AHRU hemorrhage remains controversial. While endoscopic hemostasis is well-established as the most frequently utilized and effective modality in clinical practice, its efficacy in emergency settings is significantly limited by low bowel preparation rates and massive bleeding obscuring visual field clarity, with a high postprocedural rebleeding rate reported up to 48.3%.1,4–6,9–11 Transanal suturing has also been reported as a hemostatic method for AHRU hemorrhage, however, restricted visualization during the procedure often compromises adequate hemostasis of deep ulcers, with a post-procedural rebleeding rate of up to 42%.12,13
Transcatheter arterial embolization (TAE) has emerged as a minimally invasive, effective, and safe treatment for LGIB. In 1974, Bookstein et al. first reported the successful use of TAE in treating nine cases of gastrointestinal hemorrhage. 14 However, its application in gastrointestinal hemostasis was historically limited by a high risk of ischemia and infarction, ranging from 13% to 33%. 15 With the advent of coaxial microcatheter technology, the incidence of embolism-related complications has markedly decreased. Currently, TAE has been established as a first-line therapeutic strategy for LGIB. 16 In contrast, rectal arterial embolization (RAE) for AHRU hemorrhage has rarely been documented in the literature and has yet to achieve widespread clinical attention.17,18 Herein, we present our institutional experience with RAE for hemostasis in six cases of AHRU in detail.
In this study, the techniques outcomes and follow-up data of RAE for six patients with AHRU are reported. Technical success was achieved in 83% (5/6) of cases, with clinical success was achieved in 100% cases (6/6). Rebleeding after initial RAE procedure occurred in 33% (2/6) of cases, one of which ultimately achieving hemostasis through transanal suturing after initial embolization failure. No major embolism-related complications were observed during follow-up.
Digital subtraction angiography and embolization of gastrointestinal tract
Digital subtraction angiography was performed by three experienced interventional radiologists (with 32, 27, and 20 years of experience in interventional radiology, respectively). Under local anesthetic infiltration (1% lidocaine), percutaneous femoral access was established with a 5-French (5-Fr) introducer sheath (Radifocus® Introducer II, Terumo, Tokyo, Japan). Systematic angiographic evaluation included sequential catheterization of the celiac trunk, superior mesenteric artery, inferior mesenteric artery, and bilateral internal iliac arteries. Catheter selection was guided by vascular anatomy: an 80-cm 5-Fr Cobra angiographic catheter (Radifocus® Cobra, Terumo, Tokyo, Japan) or an 80-cm 5-Fr RH angiographic catheter (Radifocus® RH, Terumo, Tokyo, Japan) was employed. Selective angiography and embolization were achieved using a 135-cm 2.8-Fr microcatheter (Renegade® HI-FLO, Boston Scientific, Marlborough, MA, USA), which was advanced coaxially to the targeted arteries under fluoroscopic guidance. The employed embolic agents were determined by the operators, including 150–350 μm or 350–560 μm gelatin sponge particles (GS) (Embozene® Gelfoam, Alicon, Hangzhou, China), 350–560 μm polyvinyl alcohol particles (PVA) (Embozene® PVA, Alicon, Hangzhou, China) and 3 × 3.3-mm microcoil (MC) (VortX® Diamond, Boston Scientific, Marlborough, MA, USA). Postprocedural hemostasis at the femoral access site was achieved using a compression tourniquet (Work® ZXD, Shanyou, Hangzhou, China) followed by 6 h of bed rest with limb immobilization.
Definitions
AHRU is defined as sudden-onset, painless hematochezia with endoscopic confirmation of active hemorrhage or stigmata of recent bleeding from rectal ulcers, after exclusion of alternative etiologies including stercoral ulcers, inflammatory bowel disease, radiation proctitis, rectal mucosal prolapse syndrome, infectious colitis, colorectal malignancy, or postoperative anastomotic complications. Technical success is defined as complete angiographic occlusion of target vessels. Rebleeding is defined as recurrent rectal bleeding requiring further intervention following hemostatic therapy. Clinical success is defined as sustained bleeding cessation without requiring additional intervention. Embolism-related complications were classified based on the reporting standards of the Society of Interventional Radiology. 19 Minor complications result in no sequelae, while major complications (e.g. intestinal ischemia, necrosis, perforation, obstruction, or nontarget embolization) result in unplanned increase in the level of care, prolonged hospitalization, permanent adverse sequelae, or death.
Case reports
Case 1
A man in his early 50s with hypertension and diabetes was admitted to an external hospital for right-sided hemiplegia secondary to cerebral hemorrhage in May 2020. During hospitalization, he developed constipation requiring enema administration. On day 28, he exhibited massive hematochezia, prompting transanal suturing that achieved initial hemostasis and revealed multiple rectal ulcers. Recurrent massive hemorrhage occurred on postoperative day 6, necessitating transfer to our hospital in June 2020. On our hospital day 2, he presented with shock symptoms accompanied by fresh hematochezia and clots, requiring transfusion of 2 units of packed red blood cells (PRBCs). Emergency colonoscopy identified two unligated ulcers 8 cm proximal to the dentate line without active bleeding (Figure 1(A) and (B)), bleeding ceased spontaneously, however, visualization was obscured by intraluminal dark red blood and clots, deferring definitive evaluation until bowel preparation. Repeat colonoscopy on day 5 after bowel preparation revealed oozing from the unligated ulcers with no additional bleeding sources. Topical norepinephrine spray was applied endoscopically, yet recurrent hematochezia and shock on day 6 necessitated 3 units of PRBCs. Given two failed endoscopic attempts and persistent hemodynamic instability, emergency angiography was performed, revealing no active extravasation (Figure 1(C), (E), and (G)). Empiric embolization of all rectal arterial supplies was conducted using 350–560 μm GS until significant flow reduction (Figure 1(D), (F), and (H)), achieving immediate hemostasis. No rebleeding or major embolism-related complications occurred during 23 months of telephone follow-up.

Endoscopic and angiographic findings in Case 1. (A, B) Colonoscopy images show oozing from unligated ulcers (arrows) 8 cm proximal to the dentate line, with adjacent sutured ulcers (arrowheads) near the dentate line. (C) Selective microcatheter angiography of the superior rectal artery (SRA) via a 2.8-Fr microcatheter reveals no active contrast extravasation (arrows). (D) Postembolization angiography of the SRA via a 2.8-Fr microcatheter demonstrates marked flow reduction (arrows) following administration of 350–560 μm GS. (E) Selective angiography of the right inferior rectal artery (RIRA) (arrow) via a 2.8-Fr microcatheter shows no contrast extravasation. (F) Postembolization angiography of the RIRA via a 2.8-Fr microcatheter confirms flow stagnation (arrow) after 350–560 μm GS deployment. (G) Selective angiography of the left internal iliac artery (LIIA) via a 5-Fr Cobra catheter identifies no active contrast extravasation (arrow). (H) Postembolization angiography of the LIIA via a 5-Fr Cobra catheter demonstrates flow stagnation in the left inferior rectal artery (LIRA) (arrow) following selective administration of 350–560 μm GS via a 2.8-Fr microcatheter.
Case 2
A woman approaching 60s with hypertension and coronary atherosclerotic heart disease was admitted to our hospital in July 2020 for bilateral lower limb paraplegia secondary to a thoracic medullary tumor, which was detected 32 days prior to admission. On day 2, she developed massive hematochezia accompanied by shock symptoms, requiring 6 units of PRBCs. Emergency colonoscopy identified active bleeding near the dentate line, however, endoscopic hemostasis failed due to poor visualization (Figure 2(A)) and repeated scope slippage. Subsequent angiography revealed atherosclerotic occlusion of the right internal iliac artery (RIIA) (Figure 2(B)), with no active bleeding from the SRA (Figure 2(C)) or LIRA (Figure 2(D)). Given the bleeding origin in the rectum, embolization of the rectal arterial supplies was performed using 350–560 μm GS until significant flow reduction. Bleeding ceased spontaneously, and conservative management was initiated due to the patient's poor general condition. Recurrent massive hematochezia on postoperative day 3 prompted transanal suturing. Direct visualization revealed multiple ulcers adjacent to the dentate line, which were successfully ligated. Subsequent to discharge from our institution, surgical tumor resection was performed at an external hospital. No rebleeding or major embolism-related complications occurred during 23 months of telephone follow-up.
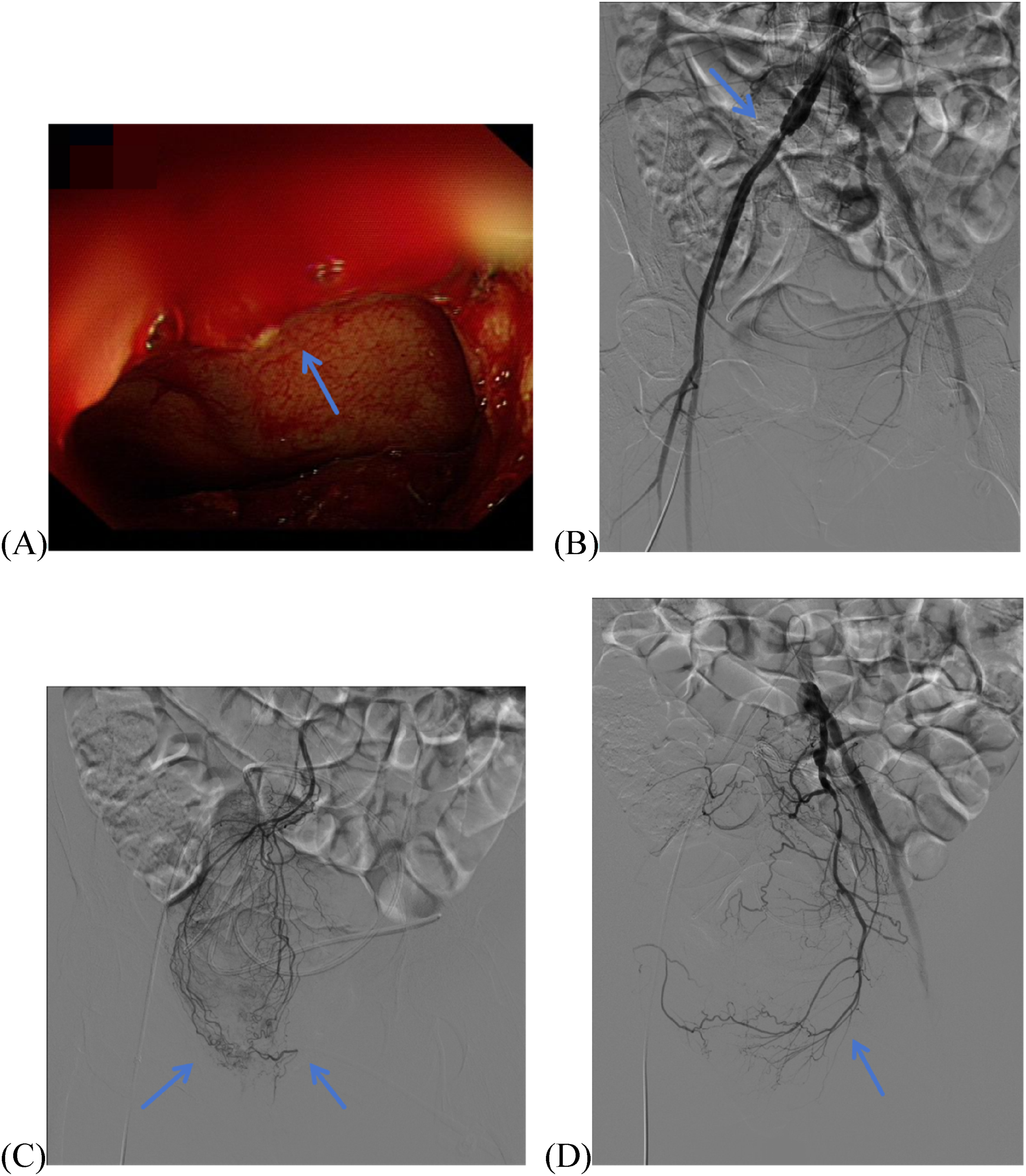
Endoscopic and angiographic findings in Case 2. (A) Colonoscopy demonstrates active hemorrhage (arrow) adjacent to the dentate line. (B) Selective angiography of the right common iliac artery via a 5-Fr Cobra catheter reveals atherosclerotic occlusion of the RIIA (arrow). (C) Selective microcatheter angiography of the SRA identifies no contrast extravasation (arrows). (D) Selective angiography of the LIIA via a 5-Fr Cobra catheter identifies no active contrast extravasation (arrow).
Case 3
A woman in her early 30s was admitted to our hospital in September 2019 for paraplegia secondary to a cervical medullary tumor, which was detected 22 days prior to admission. During hospitalization, she was unable to defecate spontaneously, necessitating enema therapy. On day 9, she developed massive hematochezia with clots accompanied by shock symptoms, requiring transfusion of 6 units of PRBCs, bleeding ceased spontaneously, and colonoscopy was deferred for bowel preparation. On day 11, recurrent hematochezia with fresh blood and fecal matter precipitated hemorrhagic shock, requiring 5 additional PRBCs units. Given inadequate bowel preparation and hemodynamic instability, emergency angiography was performed, revealing contrast extravasation from the left terminal branch of superior rectal artery (LT-SRA) (Figure 3(A)). Catheterization of the bleeding artery was achieved using a 2.8-Fr microcatheter, followed by embolization with 150–350 μm GS and a 3 × 3.3 mm MC (Figure 3(B)). All other rectal arteries were embolized with 350–560 μm GS until significant flow reduction. Immediate hemostasis was achieved, and the patient was discharged on postoperative day 7. Three days following discharge, colonoscopy performed at an external hospital revealed a solitary rectal ulcer. Subsequently, the patient underwent surgical resection of the tumor at the same institution. No rebleeding or major embolism-related complications occurred during 32 months of telephone follow-up.

Angiographic findings in Case 3. (A) Selective angiography of the inferior mesenteric artery (IMA) via a 5-Fr RH catheter demonstrates active contrast extravasation (arrow) from the LT-SRA. (B) Postembolization angiography following selective microcatheterization of the LT-SRA and subsequent occlusion with 150–350 μm GS and a 3 × 3.3 mm MC confirms complete resolution of contrast extravasation (arrow).
Case 4
A man in his mid-40s with hypertension presented with spontaneous intracerebral hemorrhage and right-sided hemiplegia was admitted to our hospital for surgical management in November 2020. On postoperative day 8, he developed hemorrhagic shock secondary to massive hematochezia with clots and fecal matter, necessitating 4 units of PRBCs. Given inadequate bowel preparation and hemodynamic instability, emergency angiography was performed, revealing contrast extravasation from the LT-SRA (Figure 4(A)), which was embolized with 350–560 μm PVA via a 2.8-Fr microcatheter, achieving immediate hemostasis (Figure 4(B)). Recurrent hematochezia 18 hours later required 4 additional PRBCs units and repeat angiography, demonstrating active bleeding from the LIRA (Figure 4(C)), successfully embolized with 350–560 μm GS and four 3 × 3.3 mm MCs (Figure 4(D)). A third hemorrhage on postoperative day 30 prompted angiography revealing contrast extravasation from the RIRA (Figure 4(E)), managed with 350–560 μm PVA and two 3 × 3.3 mm MCs (Figure 4(F)). Bedside colonoscopy on postoperative day 12 post-bowel preparation identified multiple rectal ulcers. No rebleeding or major complications occurred during 19 months of telephone follow-up.
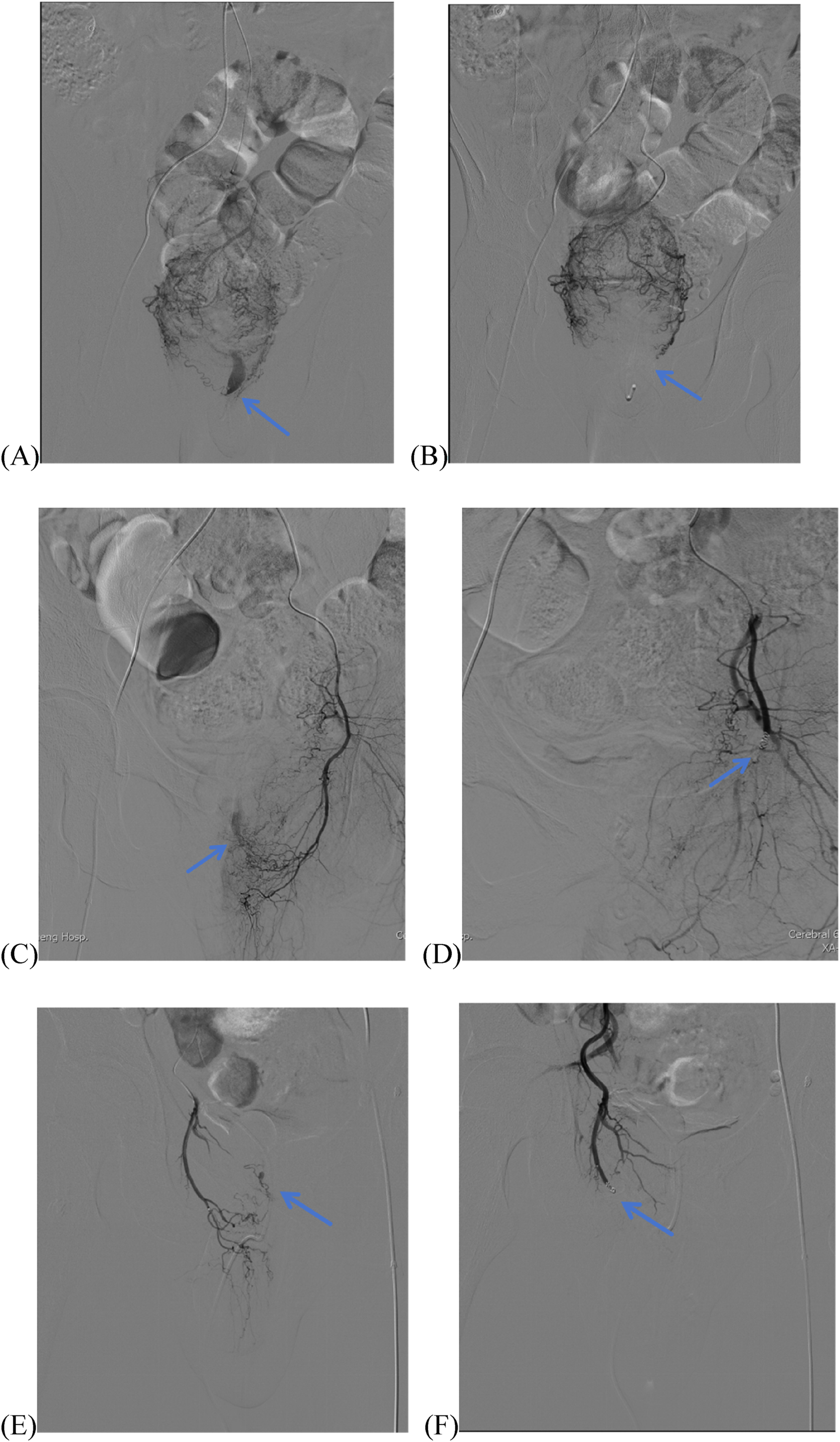
Angiographic and endoscopic findings in Case 4. (A) Initial angiography session: selective microcatheter angiography of the SRA demonstrates active contrast extravasation (arrow) from the LT-SRA. (B) Postembolization angiography confirms complete resolution of extravasation (arrow) following occlusion of the LT-SRA with 350–560 μm PVA via a 2.8-Fr microcatheter. (C) Second angiography session: selective angiography of the LIIA via a 5-Fr Cobra catheter reveals active contrast extravasation (arrow) from the LIRA. (D) Postembolization angiography of the LIIA demonstrates resolution of extravasation (arrow) after occlusion of the LIRA with 350–560 μm GS and four 3 × 3.3 mm MCs via a 2.8-Fr microcatheter. (E) Third angiography session: Selective microcatheter angiography of the RIRA via a 5-Fr Cobra catheter identifies active contrast extravasation (arrow). (F) Postembolization angiography of the RIRA confirms resolution of extravasation (arrow) following occlusion with 350–560 μm PVA and two 3 × 3.3 mm MCs via a 2.8-Fr microcatheter.
Case 5
A woman in her mid-40s with hypertension and diabetes was admitted to our hospital in May 2021 for spontaneous intracerebral hemorrhage requiring emergent external ventricular drainage, followed by intracranial hematoma evacuation on the following day. Postoperatively, she developed massive hematochezia with clots and fecal matter on day 10, accompanied by hemorrhagic shock, necessitating 3 units of PRBCs. Due to inadequate bowel preparation and hemodynamic instability, emergency angiography was performed, revealing contrast extravasation from the right terminal branch of the SRA (RT-SRA) (Figure 5(A)). Catheterization using a 2.8-Fr microcatheter achieved targeted embolization with 350–560 μm GS, with postembolization angiography confirming cessation of extravasation (Figure 5(B)). Both inferior rectal arteries were embolized with 350–560 μm GS until significant flow reduction. However, repeat angiography demonstrated new contrast extravasation from the LT-SRA at the anastomotic junction between the LT-SRA and RT-SRA (Figure 5(C)), which was successfully occluded with 350–560 μm GS (Figure 5(D)). Hemostasis was achieved, and the patient was transferred to an external hospital on postoperative day 4. Colonoscopy eight days post-discharge after bowel preparation revealed a solitary rectal ulcer. No rebleeding or major embolism-related complications occurred during 12 months of telephone follow-up.
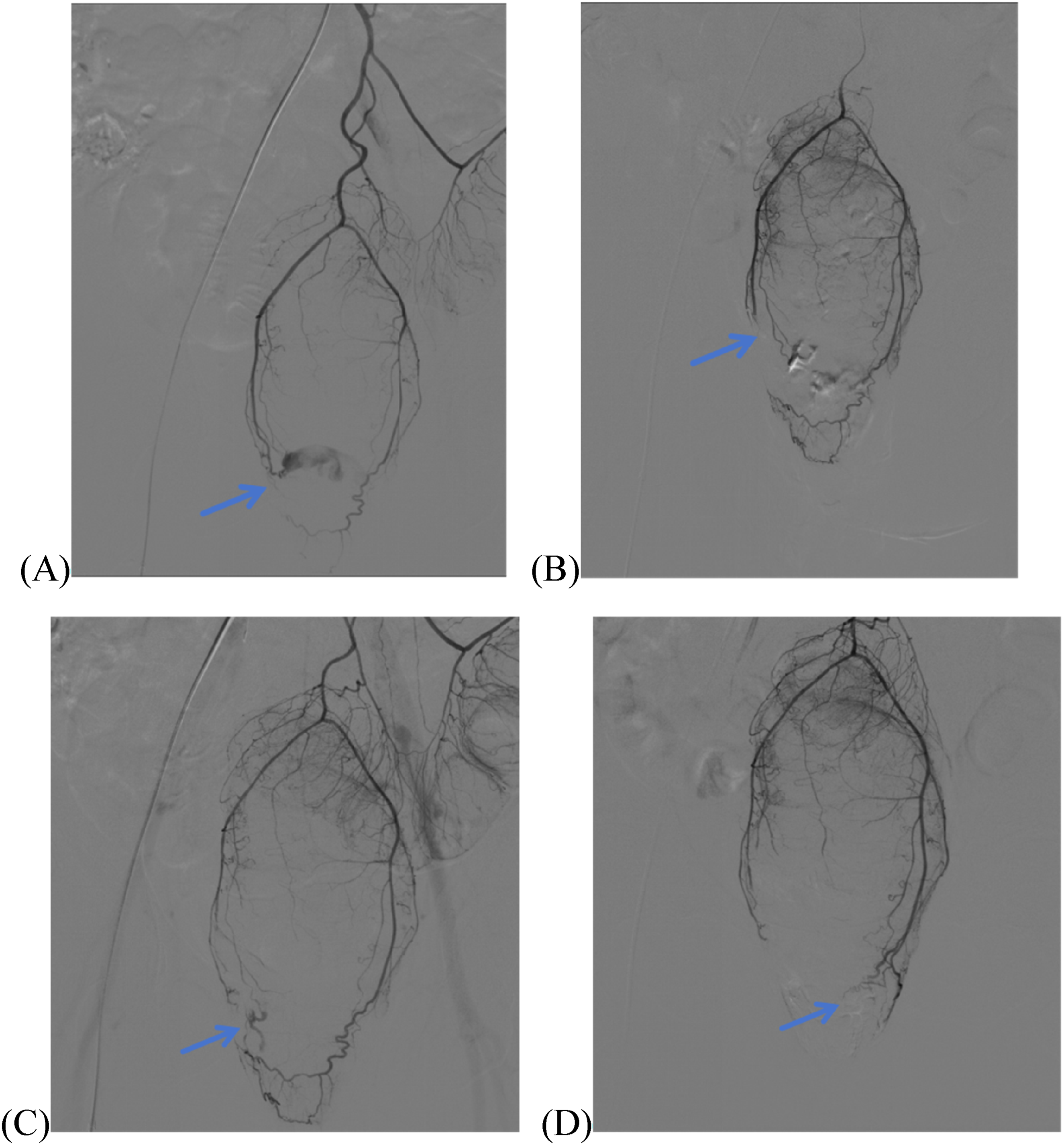
Angiographic findings in Case 5. (A) Selective angiography of the IMA via a 5-Fr Cobra catheter demonstrates active contrast extravasation (arrow) from the RT-SRA. (B) Postembolization angiography of SRA via a 2.8-Fr microcatheter after selective microcatheterization and targeted occlusion of the RT-SRA with 350–560 μm GS confirms complete cessation of extravasation (arrow). (C) Following embolization of both inferior rectal arteries, selective angiography of the IMA via a 5-Fr Cobra catheter revealed new contrast extravasation (arrow) from the LT-SRA at the anastomotic junction between the LT-SRA and RT-SRA. (D)Postembolization angiography of SRA via a 2.8-Fr microcatheter after selective microcatheterization and targeted occlusion of the LT-SRA with 350–560 μm GS confirms complete cessation of extravasation (arrow).
Case 6
A woman in her mid-60s with hypertension, diabetes, and chronic heart failure was admitted to our hospital in August 2020 for spontaneous intracerebral hemorrhage with left-sided hemiplegia, requiring emergency intracranial hematoma evacuation. Postoperatively, she developed massive hematochezia with clots and fecal matter on day 8, accompanied by hemorrhagic shock, necessitating 2 units of PRBCs. Due to patient's poor condition and hemodynamic instability, emergency angiography was performed, revealing active contrast extravasation from the LT-SRA (Figure 6(A)). Catheterization using a 2.8-Fr microcatheter achieved targeted embolization of the bleeding artery with 350–560 μm PVA and two 3 × 3.3 mm MCs until complete cessation of extravasation (Figure 6(B)). Embolization of nonbleeding rectal arteries was performed with 350–560 μm GS until angiographically confirmed flow reduction. Hemostasis was achieved, and colonoscopy on postoperative day 8 revealed multiple rectal ulcers with minor oozing observed at one ulcer site requiring no intervention (Figure 6C). No rebleeding or major embolism-related complications occurred during 22 months of telephone follow-up.
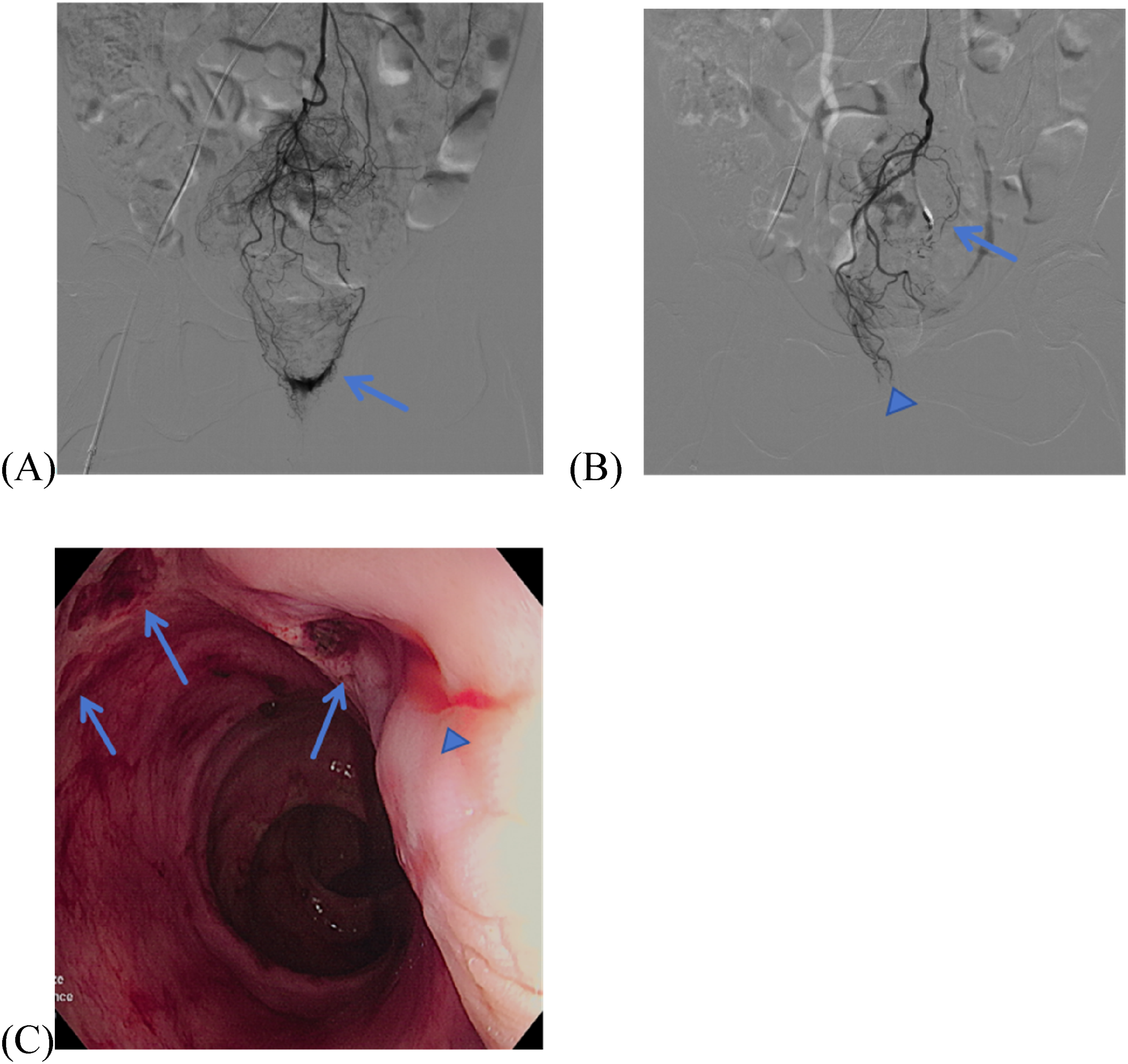
Angiographic and endoscopic findings in Case 6. (A) Selective angiography of the SRA via a 2.8-Fr microcatheter demonstrates active contrast extravasation (arrow) from the LT-SRA. (B) Postembolization angiography demonstrates complete resolution of contrast extravasation (arrow) following occlusion of the LT-SRA with 350–560 μm PVA and two 3 × 3.3 mm MCs via a 2.8-Fr microcatheter. Concurrently, embolization of the RT-SRA with 350–560 μm GS resulted in significant flow reduction (arrowhead). (C) Postoperative colonoscopy on day 8 reveals multiple rectal ulcers (arrows), with minor oozing observed at one ulcer site (arrowhead) requiring no intervention.
The reporting of this study conforms to CARE guidelines. 20 We have obtained all patients’ consent to treatment and deidentified all patient details to ensure anonymity.
The basic information, diagnosis, and treatment of the patients are shown in Table 1.
Basic information, diagnosis and treatment of patients.
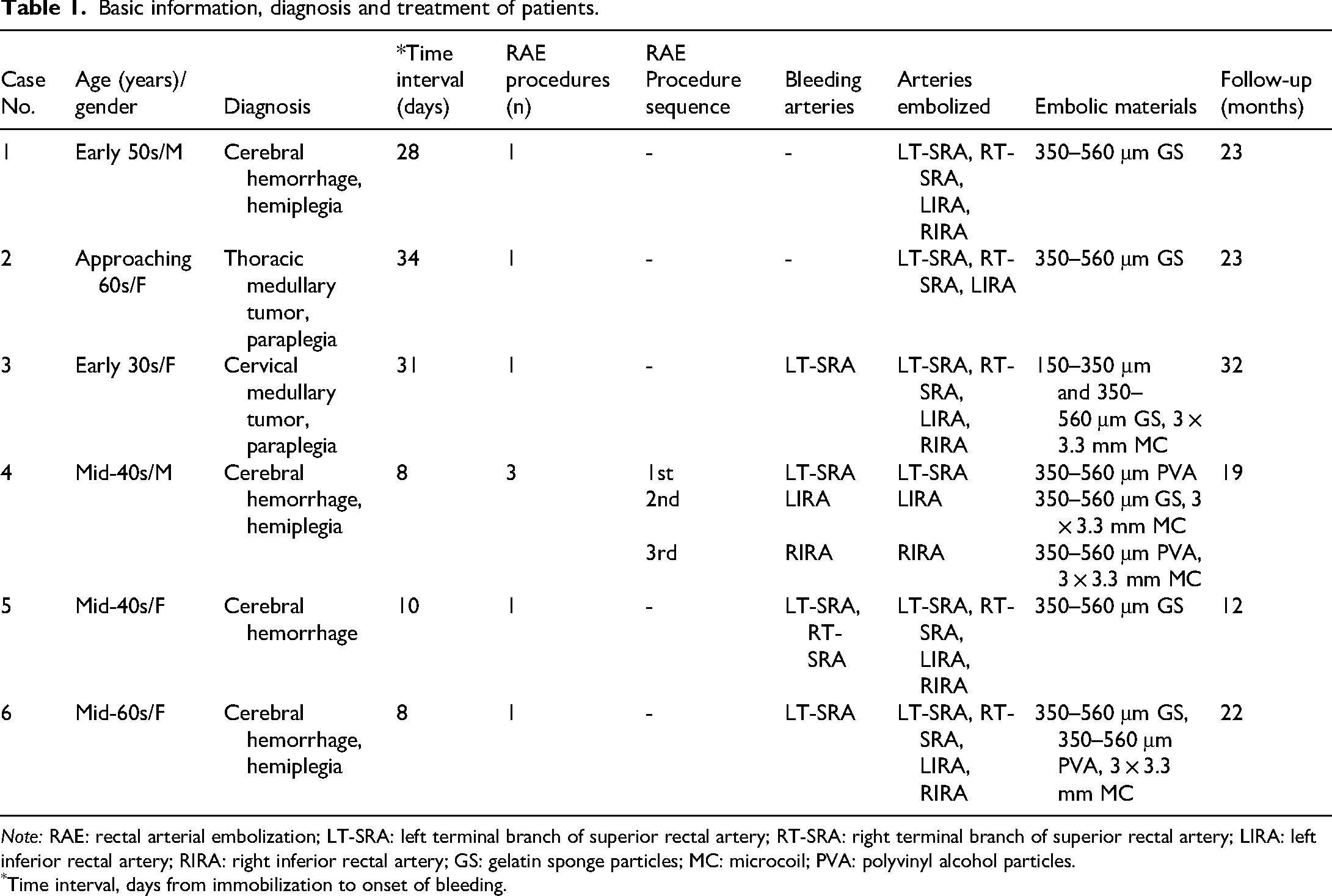
Note: RAE: rectal arterial embolization; LT-SRA: left terminal branch of superior rectal artery; RT-SRA: right terminal branch of superior rectal artery; LIRA: left inferior rectal artery; RIRA: right inferior rectal artery; GS: gelatin sponge particles; MC: microcoil; PVA: polyvinyl alcohol particles.
Time interval, days from immobilization to onset of bleeding.
Discussion
AHRU predominantly occurs in bedridden hospitalized patients with severe comorbidities, as reported in the majority of literature.1,5,10,12,21 Although the etiology of AHRU remains unclear, a decreased blood flow in the rectum secondary to arteriosclerosis or prolonged immobilization is hypothesized to be the primary pathophysiological mechanism.5,12,21,22 In our study, all six patients were bedridden due to neurological disorders and developed massive AHRU hemorrhage during hospitalization, with a median interval of 19 days (range: 8–34 days) from immobilization to bleeding onset, consistent with previous study. Notably, Case 2 had a documented history of arteriosclerosis. Additionally, cases 1 and 3 exhibited a history of constipation, and emerging evidence suggests that defecatory dysfunction may contribute to AHRU pathogenesis.17,18 Although rare, AHRU remains clinically significant due to its propensity for life-threatening hemorrhage necessitating aggressive intervention. Therefore, heightened clinical suspicion for AHRU should be maintained in patients with neurological diseases and prolonged immobilization.
Colonoscopy hemostasis is currently the common therapeutic approach for AHRU hemorrhage, as colonoscopy enables both diagnosis and treatment. The reported initial hemostasis success rate ranges from 88% to 100%.1,4–6,9–11 However, colonoscopic management faces significant challenges, particularly in cases of massive hemorrhage where adequate bowel preparation is unattainable. Achieving effective hemostasis may be compromised by poor visual field due to active bleeding and fecal residue in the rectum.23,24 Muramatsu et al. conducted a multicenter retrospective study involving 149 AHRU patients, the largest to date, and reported that pretreatment laxatives were administered in 57 patients (38%), with full rectal visualization achieved in only 71 patients (48%). The authors emphasized that even after attempted cleansing, persistent stool or active bleeding often obscured endoscopic visualization during procedures. 1 In a study by Jung et al., colonoscopy and sigmoidoscopy hemostasis succeeded in 22 patients (88%), while three patients (12%) with failed initial hemostasis required surgery due to uncontrolled bleeding. 4 In our series, Case 1 experienced rebleeding following endoscopic intervention. In Case 2, endoscopic intervention failed due to massive hemorrhage obscuring the visual field and ulcer proximity to the dentate line, which precluded effective therapeutic maneuvers. For cases 3–6, colonoscopic therapy was contraindicated owing to persistent massive bleeding and significant fecal retention within the bowel, as confirmed by clinical evaluation. Transanal suturing has been explored as an alternative.3,13 Hung et al. retrospectively analyzed 26 patients undergoing transanal suturing for AHRU hemorrhage, reporting immediate bleeding control in all cases. However, insufficient hemostasis of deep ulcers led to rebleeding in two patients, precluding repeat suturing due to massive bleeding. 12 This finding highlights the limitation of transanal suturing for deep ulcers. Similarly, in our study, transanal suturing failed to achieve adequate hemostasis for deep ulcerations in Case 1, whereas successful hemostasis was attained in Case 2 through suturing of dentate line-proximal ulcers.
The advent of coaxial microcatheter systems, advancements in embolic agent biocompatibility, and refinements in embolization techniques have significantly reduced the risk of embolism-related complications. TAE is now widely accepted as a safe and effective intervention for managing LGIB.16,25,26 To our knowledge, RAE for AHRU hemorrhage has been scarcely documented in the literature. For instance, Costouros et al. reported successful hemorrhage control via RAE in three patients with benign rectal ulcers, while Hendrickson et al. achieved hemostasis in two out of four similar cases.17,18 In Muramatsu et al.'s study, 84 (56%) underwent endoscopic therapy, with one refractory case ultimately managed by RAE. 1 Matsumoto et al. described two patients in whom endoscopic hemostasis failed due to obscured visualization from active hemorrhage, necessitating RAE. 5 Notably, these prior studies lack detailed descriptions of RAE procedural protocols or embolic agents. In contrast, our study provides a detailed analysis of RAE application in six cases of AHRU. Technical success was achieved in 83% (5/6) of procedures. Rebleeding following initial RAE occurred in 33% (2/6) of cases. Clinical success was achieved in all cases (6/6), with Case 2 ultimately achieving hemostasis through transanal suturing after initial embolization failure. All RAE procedures were well-tolerated. During a median follow-up of 22.5 months (range: 12–32 months), no major embolism-related adverse events were observed.
To summarize, colonoscopy and transanal suturing offer diagnostic utility through direct visualization, however, the identification and treatment of the bleeding site in the rectum could be difficult in cases of massive, life-threatening AHRU hemorrhage due to impaired visualization and dentate line-proximal ulcers. In such critical scenarios, RAE presents distinct advantages: (1) it eliminates time-consuming bowel preparation requirements, (2) provides etiology-independent therapeutic intervention through targeted arterial occlusion rather than lesion-specific management, and (3) combines minimally invasive access with rapid procedural completion. In our case series of six patients with AHRU managed by RAE, RAE demonstrated clinical efficacy in achieving hemostasis for individuals with hemorrhagic shock. Therefore, RAE represents a viable alternative hemostatic approach when endoscopic therapy fails or is technically unfeasible.
Post-treatment rebleeding remains a significant challenge in the management of AHRU hemorrhage, with reported rates ranging from 20% to 48.3% after initial therapy.4,5,9,10,12,27,28 In Muramatsu et al.'s study, 35 patients (23%) experienced rebleeding following successful primary endoscopic hemostasis, with 13 (37%) of the rebleeding cohort attributed to different bleeding sites, meanwhile, the authors frequently observed extensive pulsating vessels on the ulcer base. 1 In Jung et al.'s cohort of 25 patients, 22 patients (84%) were identified multiple rectal ulcers. 4 Our study similarly demonstrated multiple ulcers and intermittent hemorrhage in cases 1, 2, 4 and 6, with cases 4 and 5 exhibiting multiple arterial bleeding. These characteristics collectively contribute to therapeutic challenges and high rebleeding rates. Anatomically, the rectum receives vascular supplies from the superior rectal artery (SRA), the middle rectal artery (which often has anatomical variants) and the inferior rectal artery, interconnected by an extensive anastomotic network.29,30 This anatomic redundancy was exemplified in Case 5, where hemorrhage occurred at the anastomotic junction between the left and right terminal branches of the SRA, explaining the high vascular involvement and rebleeding rates in AHRU. Sustained hemostasis was achieved in cases 1, 3, 5, and 6 through complete embolization of rectal feeding arteries, whereas postprocedure rebleeding occurred in cases 2 and 4 owing to incomplete embolization of rectal feeding arteries. The absence of major embolism-related complications during follow-up may be attributable to the selective use of GS, a temporary embolic agent, for angiographically negative rectal arteries. Based on these findings, we propose complete embolization of all rectal arteries as a strategy to mitigate rebleeding risk in AHRU. However, given the hypothesized role of rectal hypoperfusion in AHRU pathogenesis and our limited cohort of six patients, the long-term consequences of complete arterial occlusion warrant further investigation.
Angiographically negative results are frequently observed in acute gastrointestinal bleeding, particularly in LGIB. 31 In Case 1, intermittent hemorrhage posed a significant challenge for RAE. Despite the absence of active contrast extravasation on angiography, empiric embolization of all rectal arteries was performed based on prior endoscopic confirmation of the rectal bleeding origin, achieving clinical success by reducing vascularity in the affected bowel segments. However, most published studies on empiric embolization focus on upper gastrointestinal bleeding, due to the higher incidence of adverse events associated with LGIB interventions.32,33 Few reports describe empiric embolization for LGIB, for instance, Rachapalli et al. reported a case of rectal hemorrhage managed with empiric embolization of the SRA, achieving clinical success. 34 Given the aforementioned anatomic characteristics of rectal feeding arteries, which parallels that of the upper gastrointestinal tract, empiric embolization may hold promise for managing refractory AHRU hemorrhage, though further investigation is required to validate its safety and efficacy in larger cohorts.
TAE for gastrointestinal arterial hemorrhage employs various embolic agents, including temporary embolic agents such as GS and permanent embolic agents like PVA, MC, and N-butyl-2-cyanoacrylate (NBCA). Embolic agents selection is generally influenced by operator experience, catheter size, and vascular anatomy. 26 However, no evidence-based consensus regarding optimal embolic material selection. In our institution, NBCA was not routinely utilized due to limited availability. Emerging evidence suggests that combined embolic agents may achieve superior outcomes compared to the use of single material. 29 In our series, cases 3, 4, and 6 demonstrated sustained hemostasis of bleeding collateral arteries following dual-agent embolization (MC + PVA or MC + GS). Interestingly, Case 5 achieved successful bleeding control with GS alone. Given that GS are used as temporary embolic agents and are relatively safe, we selectively employed GS for nonbleeding collateral arteries to minimize permanent ischemic sequelae.
Conclusions
Overall, AHRU represents a life-threatening complication in bedridden patients with neurological disorders. Our study suggests RAE as a safe, minimally invasive, and effective alternative for AHRU hemorrhage, circumventing endoscopic limitations in active massive bleeding scenarios. Moreover, complete embolization of rectal supplies demonstrated potential to reduce rebleeding risk, and empiric embolization may hold promise for managing refractory AHRU hemorrhage, yet large-scale studies are warranted to evaluate long-term ischemic risks linked to rectal hypoperfusion. Standardized protocols and multidisciplinary collaboration are imperative to optimize embolic strategies and integrate RAE into clinical practice.
Footnotes
Author contributions
Conceptualization: Y.G. and W.D.; methodology: Y.G., H.W., and W.D.; formal analysis: W.D.; investigation: W.T., H.J., and L.H.; resources: Y.G., H.Liu., H.Liao., and X.M.; data curation: Y.G., H.W., and W.D.; writing—original draft preparation: Y.G. and W.D.; writing—review and editing: Y.G., H.W., X.M., and W.D.; supervision: Y.G. and W.D.; project administration: Y.G., H.J., and W.D.; funding acquisition: Y.G., H.J., and W.D. All authors have read and agreed to the published version of the manuscript. Y.G., H.W., W.T., H.J., L.H., H.L., H.L., and X.M. share first authorship.
Data availabilitys tatement
All data generated or analyzed during this study are included in this published article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Ethics Committee of the Naval Medical University, Shanghai, China (Approval No: 2023SL033/2023, Date of Approval: 25 April 2023).
Funding
This study was supported by Shanghai Municipal Health Commission (code: 20224Y0100).
Informed consent statement
We have obtained all patients’ consent to treatment and deidentified all patient details to ensure anonymity.
