Abstract
Sepsis-induced cardiomyopathy (SIC), as a common complication in the intensive care unit, not only increases the complexity of patient care but also greatly enhances the risk of death. Currently, clinical management of SIC remains challenging, mainly due to the complexity of its pathogenesis and the lack of targeted therapies. Although the specific etiology of SIC is not yet fully understood, existing studies have revealed several vital pathological processes that are intertwined and contribute to the progression of the disease. This narrative review summarizes the existing pathogenesis of SIC, which involves multiple aspects including the inflammatory response, mitochondrial dysfunction, cell death mechanisms, immune regulation, and calcium homeostasis imbalance. Given the multifactorial pathogenesis of SIC, future studies need to explore the interactions between these mechanisms and how to intervene to develop more precise and effective therapeutic strategies to reduce mortality and improve prognosis in patients with SIC.
Keywords
Introduction
Sepsis, a severe condition originating from an imbalance in the body's response to fight infection, is often accompanied by conditions such as trauma, post-surgery, infectious diseases, or shock and becomes a non-negligible complication of these acute and critical illnesses. 1 According to the Global Burden of Disease Study 1990–2017 analyses, sepsis is still one of the main contributors to patient mortality worldwide. This condition is characterized by its ability to cause life-threatening multi-organ dysfunction, and statistics show that the overall mortality rate can reach approximately 35.5% within 90 days of onset. 2 It is noteworthy that before the COVID-19 outbreak, the number of new cases of sepsis worldwide was approximately 50 million per year. 3 Meanwhile, sepsis-induced cardiomyopathy (SIC), a severe complication of sepsis, particularly highlights the involvement of cardiac function. This condition leads to the inability of the heart to maintain blood perfusion to all organs of the body effectively, thus accelerating the deterioration of the disease, which is one of the critical factors in the mortality of sepsis. 4 In the United States, about 1.7 million sepsis patients require hospitalization each year, with the mortality rate in sepsis complicated by shock state hovering between 35% and 50%. It has been estimated that about 20% to 65% of sepsis patients develop varying degrees of abnormal myocardial function, especially those with concomitant SIC, where the risk of death is significantly elevated and may even be as high as 70% to 80%. 5 The understanding of SIC has been explored over a long period, and Figure 1 shows the more critical time points in the recognition of SIC.6–10 Early on, septic shock presents as a warm shock, i.e. hyperdynamic metabolism, increased cardiac output (CO), decreased peripheral vascular resistance, and tachycardia; after that, approximately 3/4 of patients present with hypokinetic manifestations, i.e. decreased left ventricular ejection fraction (LVEF) and ventricular dilatation. When sepsis induces cardiac dysfunction without structural changes in the myocardium, it is called SIC. Cardiac dysfunction is characterized by 1) a sudden drop in CO despite an increase in catecholamine levels, 2) a sudden drop in blood pressure after fluid resuscitation, 3) a sudden and reversible drop in LVEF, and 4) a sudden onset of bilateral ventricular dilatation. As a complex and severe complication of sepsis, SIC has been the focus of medical research and practice in terms of its clinical manifestations and diagnosis, therapeutic strategies, and underlying pathophysiological mechanisms. The clinical features of the disease are marked by a dramatic onset and impaired total cardiac function, as evidenced by a marked reduction in bilateral ventricular function, as well as a significant decrease in responsiveness to conventional therapies, such as fluid resuscitation and catecholamines, which have posed a considerable challenge to clinical treatment. 11 The study of SIC found that the “genomic storm” triggered by sepsis symbolizes a severe infectious reprogramming of the human genome, characterized by the massive synthesis of inflammatory mediators and vasoactive substances. This is characterized by the enormous synthesis of inflammatory mediators and vasoactive substances, including nitric oxide, interleukin-1β (IL-1β), IL-6, IL-8, IL-10, and tumor necrosis factor-α (TNF-α). 12 These substances also impair the responsiveness of myocardial fibers to calcium, impair responsiveness of myocardial fibers to calcium, interfere with mitochondrial function, and down-regulate β-receptors on cardiomyocyte membranes. Collectively, they may contribute to a long-term impairment of myocardial function.

The process of recognizing SIC. SIC: sepsis-induced cardiomyopathy.
Nonetheless, the imbalance of these mediators alone is insufficient to elucidate the entire pathogenesis of myocardial dysfunction fully. The pathogenesis of SIC is still unclear. Given the severe implications for patient outcomes, in-depth research into the mechanisms driving SIC is of paramount clinical importance. In addition, among the biomarker tests in sepsis, elevated markers of myocardial injury such as cardiac troponin I and N-terminal pro-B-type natriuretic peptide reflect the presence of myocardial damage and indicate the severity of the patient's condition and a trend toward a poorer prognosis. 1 Dynamic monitoring of these biomarkers has become an effective tool for assessing the degree of cardiac function impairment and predicting prognosis in sepsis patients. 2 As for diagnostic techniques, echocardiography, with its non-invasive, real-time monitoring, has become the method for assessing cardiac function in patients with SIC, especially for differentiating systolic from diastolic dysfunction.
Given the current lack of specific therapeutic options for SIC, clinical management is primarily guided by the principles of management of septic shock, with an emphasis on early recognition of sepsis, rapid control of the source of infection, and prevention of progression to SIC by modulating the imbalance in the immune response. However, for the diagnosis and management of SIC, there is an essential clinical value in early recognition and intervention, which may involve the use of continuous monitoring of CO. For example, by transthoracic echocardiography, pulmonary artery catheterization, or arterial waveform monitoring, cardiogenic shock is a severe condition that can occur even in the absence of acute myocardial infarction and is associated with significant morbidity and mortality. 3 Non-cardiac factors such as respiratory infections caused by sepsis account for approximately 20 percent of the etiology of cardiogenic shock and can also further exacerbate sepsis-induced organ dysfunction. 4 In addition, maintaining appropriate hemodynamic status, rationally applying vasoactive drugs, and comprehensive treatment strategies such as nutritional support and organ function maintenance are equally indispensable. 5 Using extracorporeal membrane oxygenation (ECMO) technology to treat SIC has reaped benefits in recent years. ECMO is an effective option for the treatment of patients with severe respiratory (venous-venous; VV) or refractory cardiogenic shock (venous-arterial; VA) when an experienced ECMO team performs this technique. 6
Although current research has identified that the pathogenesis of SIC may involve multiple factors, including excessive inflammatory response, mitochondrial dysfunction, dysregulated intracellular calcium homeostasis, aberrant cell death pathways, and immune dysregulation (Figure 2), the exact pathophysiological mechanisms remain largely elusive. This limited mechanistic understanding directly hinders the development of clinical treatment strategies, leaving current therapies predominantly at the stage of symptomatic support and lacking targeted interventions addressing core pathological processes. Unraveling the molecular regulatory networks underlying SIC holds significant scientific value and is key to overcoming therapeutic bottlenecks. By identifying critical targets driving disease progression, this research could catalyze the development of precision therapies, which is clinically urgent for reducing the high mortality rate associated with cardiovascular complications in sepsis patients. Thus, systematically decoding the multidimensional pathogenic mechanisms of SIC has emerged as a pressing dual challenge in critical care research and clinical translation.
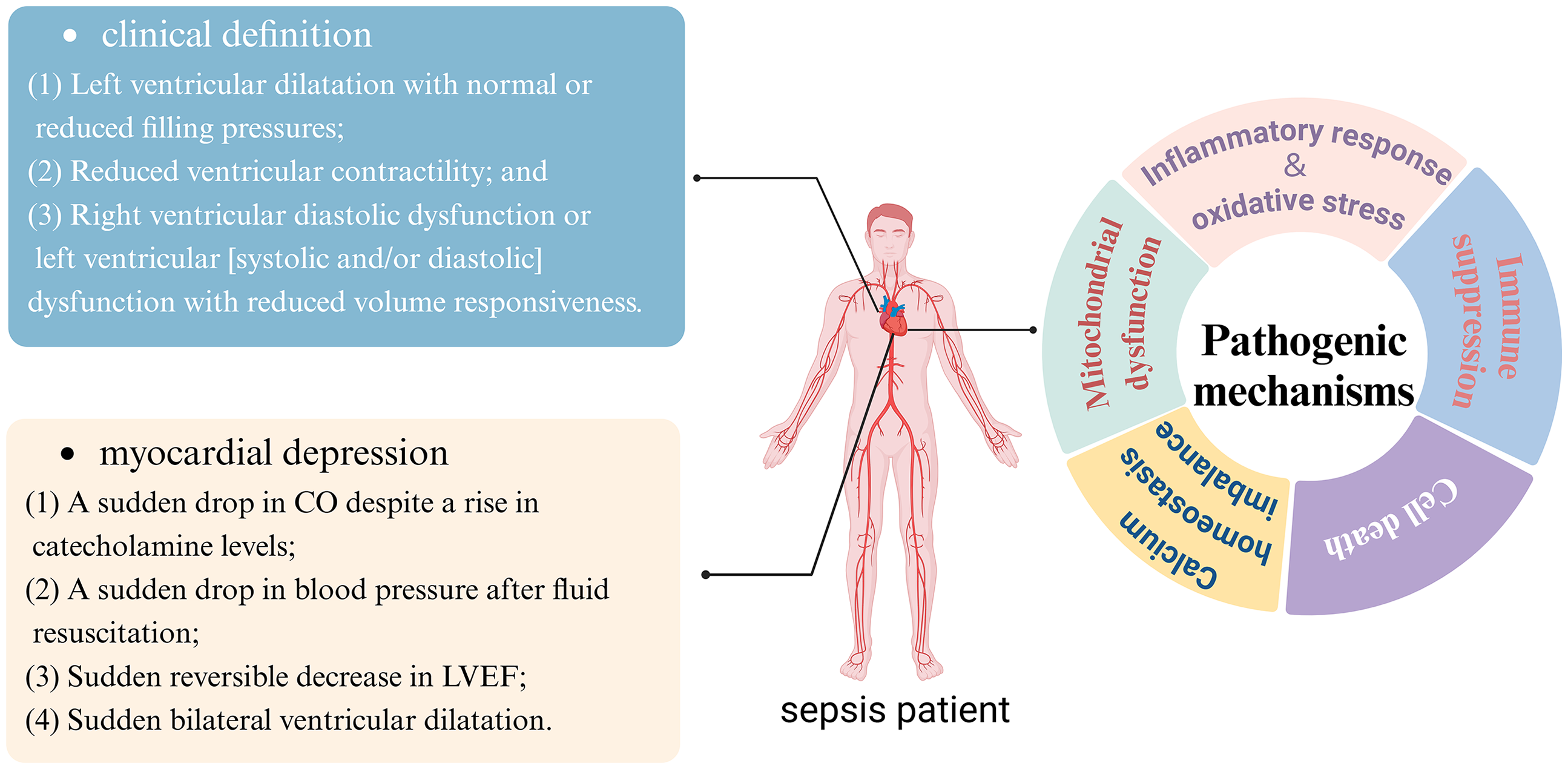
Clinical definition, cardiac dysfunction, and pathogenic mechanisms of SIC (created by Biorender). SIC: sepsis-induced cardiomyopathy.
In scientific studies, researchers have used various animal and cellular models to delve deeper into the pathological mechanisms of SIC. The cecum ligation perforation model and the lipopolysaccharide (LPS) intraperitoneal injection model are classical approaches to simulate human sepsis and myocardial injury. In contrast, the LPS-induced cellular model provides a platform to study the pathogenesis of SIC at the molecular level. 14 These models offer essential clues to understanding the complex pathophysiological processes of SIC and lay the foundation for developing new therapies. This review is guided by the Scale for the Assessment of Narrative Review Articles. 7
Pathogenesis of sepsis-induced cardiomyopathy
Inflammatory response and oxidative stress
Studies on the pathological mechanisms of SIC have gradually revealed the central roles of inflammatory response and oxidative stress. PAMPs refer to pathogen-associated molecular patterns (PAMPs), a class of molecular patterns specific to pathogens, such as bacterial LPSs and viral double-stranded RNAs, which are recognized by the pattern recognition receptors (PRRs) of the immune system. DAMPs are damage-associated molecular patterns (DAMPs), a class of molecular patterns released upon cellular damage or death, such as DNA in the nucleus, adenosine triphosphate (ATP) in the cytoplasm, etc. 8 They can be recognized by the PRRs of the immune system, ATP in the cytoplasm, etc. Key mediators in these immune responses are PRRs, including toll-like receptors (TLRs), NOD-like receptors (NLRs), and RIG-I-like receptors (RLRs). TLRs on immune cell surfaces detect PAMPs like bacterial LPSs and viral RNA. Their activation produces pro-inflammatory cytokine, contributing to myocardial inflammation and dysfunction during sepsis. 9 Intracellular NLRs recognize PAMPs and DAMPs released from injured cells. Activation can form inflammasomes, leading to interleukin release, which exacerbates inflammation and myocardial injury in SIC. 10 Cytosolic RLRs, such as RIG-I, identify viral RNA, triggering the production of type I interferons and pro-inflammatory cytokines that can injure endothelial cells and contribute to cardiac complications in sepsis. 11 The activation of these PRRs results in a cascade of pro-inflammatory mediators that worsen myocardial dysfunction during sepsis. Understanding the roles of TLRs, NLRs, and RLRs is vital for developing targeted therapies to reduce myocardial injury in septic patients. SIC will bind PAMPs and DAMPs to PRRs, triggering the corresponding signaling pathways, such as the nuclear factor-κB signaling pathway and the mitogen-activated protein kinase signaling pathway. The activation of these signaling pathways leads to the production of large amounts of pro-inflammatory cytokines, such as TNF-α and IL-1β. The release of these cytokines causes a disturbance in the immune response, leading to myocardial dysfunction. 12 Busch et al. demonstrated that in SIC, the inflammatory center of inflammatory vesicles, NLRP3, is an essential involved protein that causes increased expression of the downstream inflammatory factor IL-1β, which leads to cardiomyocyte atrophy, impairs myocardial systolic and diastolic function, and causes severe myocardial injury. 13
The release of large amounts of myocardial inhibitory factors triggers a storm of inflammatory factors in the septic state, which is an essential cause of cardiac dysfunction and high patient mortality. Inflammatory factors, especially TNF-α and IL-1β, play a leading role in the pathogenesis of SIC. 14 It has been suggested that overexpression of inducible nitric oxide synthase (iNOS) in shock patients is associated with vasodilation and hypotension, that the myocardium is similarly capable of producing iNOS, and that its overexpression generates an increased cycle of NO in inflammatory and other cells, which is closely associated with septic cardiac dysfunction. 15 Inflammatory cytokines such as TNF-α and IL-1β do not only trigger immune responses but can also increase reactive oxygen species (ROS) production, and this exacerbates oxidative stress. 16 Canakinumab, a monoclonal antibody targeting IL-1β has been investigated in several clinical trials. In a study published in Circulation in 2018, canakinumab was evaluated for its effects on cardiovascular events in patients with a history of myocardial infarction and elevated high-sensitivity C-reactive protein. The results indicated that canakinumab reduced the incidence of recurrent cardiovascular events, suggesting a role for IL-1β inhibition in cardiovascular disease management. 17 Meanwhile, oxidative stress is another important factor contributing to septic cardiac dysfunction, and the use of free radical scavengers has been shown to improve cardiac function in septic mouse models. 18 In the septic state, the body is attacked by pathogens (e.g. bacteria), and the immune system is activated, releasing large amounts of pro-inflammatory cytokines and activating immune cells. 19 Excessive release of these inflammatory mediators and activation of immune cells leads to increased intracellular ROS levels, creating oxidative stress. ROS production usually originates from multiple channels, including the mitochondrial respiratory chain, NADPH oxidase, protein kinase, etc. Increased ROS may lead to various cellular injuries, including lipid peroxidation, protein oxidation, and DNA damage, which can lead to cardiomyocyte dysfunction and death. 20
Although altered levels of circulating immune cells and inflammatory factors are known to be tightly linked to SIC, the specific molecular link interaction mechanisms need to be explored and elucidated in greater depth.
Mitochondrial dysfunction
The normal functioning of the heart as a highly energy-demanding organ dramatically depends on the production of ATP by mitochondria within cardiomyocytes. Notably, mitochondrial dysfunction is a central element in the pathogenesis of SIC. In recent years, the relationship between SIC and mitochondrial dysfunction has received much attention. 21 The primary manifestation of mitochondrial dysfunction in SIC is a disturbance in mitochondrial energy metabolism, resulting in decreased ATP production and increased ROS production. 22 These changes lead to diminished cardiac contractility and altered cardiac function in patients with sepsis. Inflammatory factors such as TNF-α and IL-6 play an essential role in this process, and they exacerbate the condition by exacerbating mitochondrial damage and promoting apoptosis in cardiomyocytes. It has been shown that 1.5 h after exposure to TNF-α in an ex vivo model, there was an increase in caspase 8 activity accompanied by a decrease in mitochondrial membrane potential and a decrease in neurotoxicity in cells pretreated with TNF-R1 antibody, suggesting that TNF-α exerts its neurotoxicity through TNF-R1 and causes acute mitochondrial damage. 23 Studies have shown that mitochondrial biogenesis is often impaired in SIC, which hinders the heart's recovery from septic injury. Interventions that use specific drugs or therapeutic strategies to enhance mitochondrial biogenesis and function have shown promise in experimental models. Research has also focused on mitochondrial dynamic homeostasis, including the balance between fusion and division. Dysregulation of this process leads to mitochondrial fragmentation and dysfunction, further exacerbating cardiac dysfunction in sepsis. 24 The potential therapeutic benefits of targeting mitochondrial dysfunction in SIC have also been explored, such as mitochondria-targeted antioxidants and drugs stabilizing the mitochondrial membrane, which have shown potential in preclinical studies.
Cardiomyocyte mitochondria in patients with SIC often show abnormal morphological and functional changes, including excessive mitochondrial fragmentation, increased oxidative stress, decreased mitochondrial membrane potential, and impaired mitochondrial membrane integrity, contributing to disease progression. 25 Stem cell exosome therapy may provide a new perspective for treating SIC by alleviating calcium overload in mitochondria 26 . Abnormal myocardial mitochondrial function is characterized explicitly in SIC by decreased transmembrane potential, altered membrane permeability, disorganized cristae arrangement, and swelling. 26 The mechanisms behind this series of changes are complex, covering a variety of pathological processes such as inflammatory factor release, NO production, oxidative stress, calcium overload, membrane pore opening, energy coupling imbalance, autophagy, and apoptosis. In particular, NO produced by mitochondria through the NOS pathway is an essential signaling molecule with critical roles in vasodilatation, inhibition of the mitochondrial respiratory chain, regulation of pro-inflammatory cytokines, and modulation of platelet function; however, excess NO can lead to mitochondrial dysfunction and interfere with energy metabolism in cardiomyocytes. This may be related to the activation of regulatory factors such as PGC-1α, NRFs, and ERRs during mitochondrial biogenesis. 27 In addition, research published in 2021 showed that administration of MitoQ in a mouse model of sepsis reduced mitochondrial superoxide production and protected mitochondrial function, thereby preventing diaphragmatic muscle weakness. Thus, MitoQ may be a promising therapeutic strategy for treating sepsis. 28 In summary, the complexity of mitochondrial dysfunction in SIC requires us to understand its mechanisms from multiple perspectives and explore targeted therapeutic strategies. The mode of mitochondrial damage is shown in Figure 3.

Mitochondrial damage in SIC (created by Biorender). SIC: sepsis-induced cardiomyopathy.
Cell death
In recent years, mechanisms of cell death have gained prominence in scientific research and are particularly important in SIC. Therapeutic strategies that modulate apoptotic pathways are expected to improve the cardiac prognosis of septic patients. For example, the inhibition of apoptotic signaling pathways or enhancement of the expression of anti-apoptotic proteins can reduce apoptosis in cardiomyocytes, thereby protecting myocardial function. 29 MCL-1 and BCL-XL anti-apoptotic proteins play essential roles in cytoprotective mechanisms, and it has been found that MCL-1 and BCL-XL protect against carfilzomib-mediated cytotoxicity in multiple myeloma in the stroma. 30 It provides new ideas and possibilities for the future treatment of SIC. Some studies have shown that sepsis-induced cardiac dysfunction can be effectively ameliorated by inhibiting the apoptotic process. 31 For example, in a mouse sepsis model constructed using LPS, controlling ROS generation, limiting cardiomyocyte apoptosis, and decreasing inflammatory response were essential for alleviating sepsis-induced myocardial injury. In addition, the transmembrane protein TMEM43 has been shown to intervene in the imbalance of iron metabolism in cardiomyocytes and inhibit the iron death pathway, thereby counteracting sepsis-associated cardiac damage. 32 In a recent research development, Liu and his team's study in 2024 revealed that low-dose application of the oncology therapeutic drug olaparib positively modulated mitochondrial dynamics in SIC, significantly elevated the rate of mitochondrial autophagy and effectively attenuated the phenomenon of myocardial iron death, demonstrating the potential of pharmacological interventions in the preservation of cardiac muscle function. 33 Another concurrent study highlighted that controlling ROS-related ferritin accumulation in SIC protects the myocardium and maintains its function, underscoring the importance of iron death regulation. Regarding the mechanism of cell death in SIC, GSDMD proteins and the toxic fragments generated by their cleavage play a crucial role in mediating cell membrane perforation, triggering cell lysis, and promoting the release of the pro-inflammatory factors IL-18 and IL-1β, which exacerbate inflammatory responses and tissue damage. 34 A schematic diagram of the mode of septic myocardial cell death is shown in Figure 4.

Immune regulation and cell death in sepsis-induced cardiomyopathy.
In addition, the scientific community has gradually recognized the negative role of neutrophils in septic myocardial injury, especially the excessive formation of the neutrophil extracellular trapping network, which may be one of the mechanisms of direct damage to cardiomyocytes. The current diversified studies on cell death mechanisms have enriched our understanding of the regulatory network of cell death and opened up new perspectives for in-depth analyses of the pathological mechanisms of SIC, indicating that future therapeutic strategies in this field will be more precise and efficient.
Immune regulation
The dynamic homeostasis of the immune system plays a central regulatory role in the development of SIC. A groundbreaking study by Zhang et al. revealed the critical role of immune cells, particularly cardiac-resident macrophages expressing high levels of triggering receptor expressed on myeloid cells 2 (TREM2), in maintaining mitochondrial homeostasis in cardiomyocytes and counteracting septic cardiac dysfunction. 35 Conversely, sepsis-induced immune dyshomeostasis manifests as an initial cytokine storm followed by protracted immunosuppression. Overproduction of anti-inflammatory mediators (e.g. IL-4, IL-10, IL-37) dampens pathogen clearance. It promotes “immunoparalysis"—a state marked by impaired leukocyte function and secondary infections that exacerbate cardiac injury. This process not only inhibits the regular release and signaling of pro-inflammatory factors but also promotes a state of widespread immunosuppression, providing a breeding ground for pathogens and further worsening the condition. The activation of the TLR has a vital role in immune mechanisms, and it plays an essential role in myocardial suppression caused by various diseases. TLR is expressed in the hearts of patients with sepsis, and it has been confirmed that it plays a vital role in myocardial suppression caused by sepsis. It has been reported in the literature that the knockdown of TLR3 can inhibit the apoptotic signaling pathway mediated by Fas/FasL in cardiomyocytes, and the activation of the upstream signaling molecule of TLR4, CD14 leads to the increase of the expression of inflammatory mediators. 36 Intriguingly, TLR2 exhibits cardioprotective effects, underscoring the functional duality of TLR signaling SIC.
Thus, immunomodulatory mechanisms in SIC highlight their critical position in controlling disease progression and outcome. This highlights the therapeutic potential of attenuating extreme immunosuppressive states by modulating the immune response and the vital importance of maintaining immune homeostasis in managing sepsis and its complications. Therefore, future therapeutic strategies should focus on the precise identification and modulation of immune imbalances, intervening early in the course of the disease to reverse immunosuppression and thus mitigate the dangers of sepsis and its cardiac complications (Figure 4).
Dysregulation of Ca2+ homeostasis
Another important myocardial injury mechanism in sepsis is impaired calcium ion regulation. Calcium ions (Ca2+), a key mediator of myocardial contraction and diastole, are mainly stored in the endoplasmic reticulum of cardiomyocytes. There are two calcium channel subtypes, L-type, and T-type, on cardiomyocyte membranes, with L-type calcium channels dominating, significantly enriched in atrial and ventricular myocyte membranes, particularly in the T-tubule region of the sarcoplasmic reticulum (SR). In sepsis, releasing inflammatory factors and oxidative stress may trigger endoplasmic reticulum stress in cardiomyocytes, which may disrupt Ca2+ homeostasis and lead to mitochondrial calcium overload and impaired energy metabolism. Ca2+, essential mediators of myocardial excitation-contraction coupling, are tightly regulated by three key molecular systems in cardiomyocytes: the ryanodine receptor (RyR) responsible for SR Ca2+ release during systole, the sarcoplasmic reticulum Ca2+-ATPase (SERCA) that reabsorbs Ca2+ into the SR during diastole, and the Na+/Ca2+ exchanger (NCX) extruding cytosolic Ca2+ via electrochemical gradients. In sepsis, systemic inflammation, and oxidative stress disrupt this regulatory triad, leading to RyR hyperphosphorylation (enhancing SR Ca2+ leakage), SERCA downregulation (impairing Ca2+ reuptake), and NCX dysfunction (reducing cytosolic Ca2+ extrusion), collectively causing cytosolic Ca2+ overload and mitochondrial Ca2+ accumulation. 37 The ensuing mitochondrial Ca2+ overload uncouples oxidative phosphorylation, exacerbates ROS production, and triggers lipid peroxidation. At the same time, cytosolic Ca2+ elevation activates calpain-dependent cleavage of pro-apoptotic proteins and calcium/calmodulin-dependent kinase II (CaMKII), promoting mitochondrial permeability transition pore opening and cytochrome c release, thereby accelerating cardiomyocyte apoptosis. Furthermore, mitochondrial ROS generated from Ca2+ overload activates NLRP3 inflammasomes, amplifying IL-1β and IL-18 production, which in turn disrupts Ca2+ handling, forming a vicious cycle between calcium dysregulation and inflammation. Notably, controversies exist regarding the interdependence of these pathways: while Shrestha et al. observed LPS-induced sinus node dysfunction via impaired M2 muscarinic acetylcholine receptor-gated inwardly rectifying potassium channel (M2R-GIRK) signaling (reducing membrane Ca2+ aggregation) independent of systemic inflammation, 38 Ni et al. demonstrated that pyruvate kinase M2 (PKM2) preserved Ca2+ homeostasis while suppressing inflammatory markers (e.g. TNF-α, IL-6), suggesting Ca2+-inflammation crosstalk. 39 Recent advances highlight therapeutic potential through targeting Ca2+ regulators— transmembrane BAX inhibitor motif-containing protein 6 (TMBIM6) was shown to maintain mitochondrial Ca2+ homeostasis by inhibiting voltage-dependent anion channel 1 (VDAC1) oligomerization, thereby improving sepsis-induced mitochondrial fission and cardiac dysfunction while experimental interventions such as RyR stabilizers (e.g. dantrolene) and SERCA2a gene therapy demonstrate efficacy in restoring Ca2+ handling in preclinical models. 40 These findings underscore calcium overload as a central hub linking mitochondrial dysfunction, apoptosis, and inflammation in septic cardiomyopathy, though precise molecular cascades warrant further elucidation.
Outlook
While mechanistic insights into SIC pathogenesis have been illuminating, they must now drive therapeutic innovation. However, the clinical potential of promising therapeutics remains unproven in trials targeting SIC, limited by three translational barriers: (i) etiological heterogeneity—the diversity of sepsis triggers requires precise target selection; (ii) temporal urgency—rapidly progressing cardiac insufficiency narrows the window for intervention, hence the need for early biomarkers; (iii) interconnectivity of pathways—the single therapies targeting a single mechanism may not be able to counteract redundant pathological networks, requiring combinatorial strategies. Emerging technologies promise to overcome these barriers.
Conclusion
This review discusses the translation of SIC from mechanistic research to clinical treatment. By deeply dissecting the pathogenesis of SIC, researchers have identified multiple potential therapeutic targets. However, the clinical application of these therapies still faces three significant challenges: etiological heterogeneity, time urgency, and pathway interconnectivity. Emerging technologies offer the possibility to overcome these obstacles. By tightly integrating drug development with the molecular mechanisms of SIC, breakthroughs in diagnostic prediction, therapeutic interception, and personalized treatment options are expected to transform this fatal syndrome into a manageable complication.
Supplemental Material
sj-pdf-1-sci-10.1177_00368504251329190 - Supplemental material for Exploring the pathogenesis of sepsis-induced cardiomyopathy: Multilayered mechanisms and clinical responses
Supplemental material, sj-pdf-1-sci-10.1177_00368504251329190 for Exploring the pathogenesis of sepsis-induced cardiomyopathy: Multilayered mechanisms and clinical responses by Jinfang Xue, Ning Zhou, Quan Li, Ruijie Wang, Yan Li, Huadong Zhu and Chuanzhu Lv in Science Progress
Footnotes
Acknowledgments
We are grateful to Miss Ren Yuan for her help in writing and revising this article.
Author contributions statement
JFX and ZN were responsible for the design of the study, data organization image production, and drafted the manuscript. CZL and HDZ were in charge of supervising the experiments and revising the manuscript draft. QL, RJW, and YL participated in the creation of forms and modification of pictures. All authors contributed to writing the manuscript, approved the final version, and had final responsibility for the decision to submit it for publication. The corresponding author confirms that all listed authors meet authorship criteria and that no others meeting the criteria have been omitted.
Availability of data and materials
All data generated or analyzed during this study are included in this published article [and its supplementary information files].
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National High Level Hospital Clinical Research Funding;2022-PUMCH-D-005; CAMS Innovation Fund for Medical Sciences; 2019-I2M-5-023. Hainan Provincial Key Research and Development Project; ZDYF2020112.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
