Abstract
Objective:
To evaluate and compare the impact of single- versus dual-antiplatelet (SAPT vs DAPT) therapy on coagulation profiles and postoperative outcomes in patients with peripheral arterial disease (PAD).
Methods:
Patients with PAD undergoing lower extremity revascularization from December 2020 to August 2023 were prospectively enrolled in this cohort study and followed for one year to record the incidence of thrombotic events (TEs). These events include arterial graft/stent occlusion or stenosis, as identified through Doppler ultrasonography or angiography. Patients were categorized by type of intervention (open vs. endovascular) and further stratified by thromboprophylaxis regimen (SAPT vs DAPT). Descriptive statistics were conducted to characterize each group. Differences in continuous variables were analyzed using Student's t-test, while categorical variables were evaluated using Fisher's exact test. Kaplan–Meier survival curves and cox proportional hazard assessed the thrombosis probability between groups.
Results:
A total of 157 patients were analyzed, of which 56 were open and 101 were endovascular. In the endovascular group, the majority of patients were on DAPT (61.4%), whereas in the open procedure, most individuals were on SAPT (78.6%). DAPT recipients exhibited a lower prothrombotic profile than SAPT recipients in either operative cohort (p < .05). However, the incidence of index TEs was similar between medication groups within each cohort over the one-year follow-up period. DAPT was associated with a decreased incidence of recurrent TEs in the open intervention group (p < .001), indicating its potential for reducing subsequent thrombotic complications following an index TE. Additionally, there was no significant difference in thrombosis probability between endovascular and open procedures (p = .73), nor between DAPT and SAPT groups across both procedure modalities (p = .98).
Conclusion:
While DAPT demonstrated lower prothrombotic thromboelastography with platelet mapping profiles compared to SAPT, this did not translate into differences in index TEs across surgical modalities.
Keywords
Introduction
PAD is symptomatic in approximately 50% of patients, and ranges in severity from claudication to critical limb-threatening ischemia (CLTI). 1 Medical management of symptomatic PAD includes exercise therapy, smoking cessation, and operative revascularization with antiplatelet medications and prophylactic cholesterol-lowering agents. These approaches aim to reduce cardiovascular mortality and morbidity, and improve PAD severity; however, when symptoms are lifestyle-limiting, surgical revascularization may be indicated.2,3
Current guidelines recommend long-term single-antiplatelet therapy (SAPT), with either low-dose aspirin or clopidogrel, for patients with PAD to decrease the risk of vascular disease progression and postoperative complications.4–6
For patients undergoing open surgical or endovascular revascularization for PAD, the use of dual antiplatelet therapy (DAPT) remains a topic of contention. DAPT with both aspirin and clopidogrel has been recommended for at least four weeks following percutaneous coronary artery stenting, which may be continued for a longer duration depending on patient co-morbidities and risk profiles. 7 However, the DAPT regimen postrevascularization for symptomatic PAD is poorly established and remains controversial. 8
Currently, there is a lack of studies that utilize viscoelastic assays to quantitatively evaluate the risk or benefit conferred by DAPT or SAPT postrevascularization for patients with symptomatic PAD. Established assays, such as thromboelastography with platelet mapping (TEG-PM), can provide objective measurements of the activity of one's platelets and response to antiplatelet therapies. Therefore, these assays can provide valuable insights illustrating the impact of SAPT or DAPT regimen on platelet activity, and, hence, shed light on the prophylactic utility of either regimen in this patient population. This prospective observational study aims to compare the coagulation profiles between patients on SAPT or DAPT following open surgical or endovascular intervention for lower extremity PAD to determine the impact of medication regimen on thrombotic potential. We hypothesize that patients placed on DAPT portray more favorable coagulation profiles and experience fewer thrombotic events (TEs) following operative treatment than their counterparts receiving SAPT prophylaxis.
Material and methods
Study population and study design
Patients over 18 years of age scheduled for lower extremity revascularization for PAD within the Massachusetts General Hospital vascular surgery department between December 2020 and August 2023 were enrolled in this prospective cohort study.
This study was conducted in accordance with the principles of the Declaration of Helsinki (1975, as revised in 2013). The reporting of this study conforms to STROBE guidelines for observational studies. 9 The study protocol was approved by the Mass General Brigham institutional review board (Boston, MA, USAIRB #2022P001918) on 19 September 2022. Consecutive sampling was used to enrolled participants during the study period, provided they met inclusion criteria. Written or electronic consent was obtained from all study participants or the participant's legally authorized representative if the participant could not provide consent. All patient data was deidentified to ensure participant confidentiality and privacy.
Exclusion criteria included the inability to provide informed consent, the inability to undergo serial blood draws as outlined in the protocol, and pregnancy. Patients undergoing either open surgical (bypass, endarterectomy, or thrombectomy) or endovascular (angioplasty, stent, or deep venous arterialization) surgical revascularization were included, while those undergoing hybrid procedures were excluded from the analysis. A board-certified vascular surgeon decided which procedure modality and postoperative antiplatelet regimen was indicated based on their clinical assessments.
Participants were removed from the study if the index procedure did not result in successful revascularization due to either the inability to find a targetable lesion or the inability to access a lesion endovascularly.
TEG with platelet mapping assay
The TEG® 6 s Hemostasis Analyzer (Haemonetics Corporation, Boston, MA, USA) was used to generate coagulation profiles following blood sample collection. The TEG-Platelet Mapping 6 s System® utilized PlateletMapping® cartridges to measure the kinetic changes of a sample of heparinized whole blood as it clotted. This TEG-PM assay produced a qualitative assessment of platelet function. 10
The PlateletMapping® cartridge measured the maximal amplitude (MA) of blood clots under four different conditions using dried-in-place reagents. The first test utilized the Kaolin activation technique, which stimulated a strong thrombin response to ensure the complete breakdown of available fibrinogen and the full activation of all platelets. This method quantifies the sample's maximal clot strength (MAHKH). The second test inhibited thrombin and measured the blood clots Fibrin-only MA, depicting MA without platelet activation using reptilase, which directly converted fibrinogen to fibrin (referred to as MAActF). The final two assays also entirely inhibited thrombin and measured the blood clots MA when platelets were stimulated by the agonists adenosine diphosphate (MAADP) or arachidonic acid (MAAA). A decrease in MA in response to the agonist (ADP/AA) indicated the action of P2Y12 inhibitors or aspirin, respectively. Platelet reactivity can then be calculated as a percentage (%) using the formula:
Study procedures
Blood samples were collected into a 4.0 mL nongel sodium heparin Vacutainer for the TEG-PM analysis at baseline (prior to revascularization), and during the one-, three-, and six-month postoperative follow-up visits. For the purposes of this study, only data from the one-, three-, and six-month visits were analyzed. As per the manufacturer's instructions, the sample analysis was conducted after a 30-min incubation period, and within 2 hr of the blood draw.
Samples were assigned to the SAPT group if the participant reported using 81 mg aspirin or 75 mg clopidogrel and DAPT group if the participant received both medications. Patients were asked to report which antiplatelet agents they had taken within the last seven days at each blood draw visit.
Variables
Demographic information gathered encompassed age, sex, race, and ethnicity. For the purposes of this study, women and men were defined by their biological sex at birth rather than gender. Additionally, comorbid conditions such as hypertension, hyperlipidemia, diabetes, coronary artery disease, and renal status were documented. Medical personal history of deep vein thrombosis, pulmonary embolism, stroke, and MI were noted. Each participant's smoking history, body mass index (BMI), TEs, bleeding events, Rutherford score and runoffs were also recorded. Former smokers were defined as those who stopped smoking more than a year prior to their intervention. TEs were defined as arterial graft/stent occlusion or stenosis, as identified through follow-up Doppler ultrasonography (USG) or radiographic imaging. Occlusion/complete stenosis was defined as a complete narrowing of the lower extremity artery, seen as an absence of color within the graft/stent on Doppler USG. 11 Significant stenosis was defined as >70% narrowing, PSV >300 cm/s, and velocity ratio > 3.5, and graft flow velocity <45 cm/s. 11 The incidence of index TEs was tracked for both cohorts during the one-year follow-up duration of the study.
Bleeding events were defined as hemorrhages requiring intervention for control or blood transfusion. We did not differentiate between major and minor events due to the limited number of bleeding events.
Statistical analysis
The data were stratified by procedure type: open surgical intervention and endovascular intervention. Within each procedure type, further stratification was performed based on the postoperative medication regimen, categorizing subjects into SAPT and DAPT. This stratification enabled the evaluation of potential differences between these medication regimens within each intervention type.
Continuous variables were compared using an unpaired Student's t-test to compare means between the antiplatelet therapy group, while categorical variables were analyzed with Fischer's exact test to assess differences between these groups. Fischer's exact test was used due to the small sample size between the groups.
A bar chart analysis was performed to visually illustrate the distribution of TEG-PM values between the cohorts. Kaplan–Meier survival curves were generated to assess thrombosis probability in relation to procedure modality as well as antiplatelet regimen. A Cox proportional hazards regression model was performed to evaluate the association between different antiplatelet regimens and the risk of TEs. To account for potential confounders, a multivariable regression analysis was conducted, adjusting for variables including sex, age, BMI, and smoking status. This analysis aimed to isolate the effect of antiplatelet therapy on thrombotic risk, while controlling for the impact of these key demographic and clinical factors. Additionally, to minimize selection bias, patient characteristics were thoroughly assessed to ensure comparability across treatment groups.
Thrombosis probability was defined as the time from the date of the procedure to the date of the event. For those who did not have a TE, the date of the last follow-up was used for this time to event analysis. Short-term adverse events were reported only if they were deemed relevant and had the potential to impact the TEG results.
A p-value of <.05 was considered statistically significant. Unpaired student's t-test and Fischer's exact test were performed using GraphPad. Kaplan–Meier curves were generated using R version 4.3.0 (2023-04-21) (R Foundation for Statistical Computing, Vienna, Austria) and R studio version 2023.09.0 + 463.
Results
Study population
Of the 303 patients enrolled during the study period, 146 patients were excluded for reasons such as hybrid procedure election (N = 47), screen failure (N = 4), and loss to postoperative follow-up (N = 95) (Figure 1). Of the 157 patients analyzed, 64% underwent endovascular intervention and 36% underwent open surgical procedures (Figure 1). In the open surgical group, patients were more likely to receive a SAPT regimen compared to DAPT (n = 44, 78.6% vs n = 12, 21.4%; p < .0001). Conversely, in the endovascular group, patients were more likely to have a DAPT regimen than SAPT (n = 62, 61.38% vs n = 39, 38.61%; p < .002). In both open and endovascular groups, the number of patients receiving SAPT and DAPT were comparable. Similarly, there were no significant differences in demographics comorbidities, Rutherford score and runoffs between the procedure cohorts (Table 1). Patients with a Rutherford classification of 0, 1, or 2 are usually managed nonoperatively, however 17 study participants in this category underwent surgical interventions. These 17 patients had previously undergone lower extremity interventions and were identified through noninvasive vascular laboratory surveillance to have hemodynamically significant stenosis at the site of prior treatment. Although they had not developed severe symptoms from the restenosis, they were taken to the operating room for revascularization to mitigate the risk of graft failure.

Study flow chart.
Demographics and comorbidities of patients undergoing open and endovascular interventions (N = 157).

Abbreviations: GFR: glomerular filtration rate (mL/min); DVT: deep vein thrombosis; PE: pulmonary embolism; MI: myocardial infarction; BMI: body mass index; SAPT: single antiplatelet therapy; DAPT: dual antiplatelet therapy.
Note: We stratified according to antiplatelet regimen for each intervention to compare them. We used Fisher exact test to compare categorical variables and Student's t for continuous variables. Patients in the open surgical group were more likely to receive SAPT, while those in the endovascular group were more likely to receive DAPT. Across both groups, patients on SAPT and DAPT showed comparable demographics and comorbidities, with no significant differences.
Blood was collected at one-month (1 M), three-month (3 M), and six-month (6 M) visits. Therefore it was expected that 303 endovascular cohort and 168 open surgical cohort samples would be available for analysis. The cause for the discrepancy between the number of samples available and expected was multifactorial. Firstly, some patients were lost to follow-up or denied blood draws. Secondly, patients had to be on a SAPT or DAPT regimen for at least seven days to be classified as either category. Additionally, in cases where a patient's medication regimen at a single collection point differed from the majority of their samples, the deviant sample was excluded from analysis. This approach guaranteed that each patient and their corresponding samples were consistently classified under a single medication regimen. In the endovascular cohort, 24% of 1 M samples, 55% of 3 M samples, and 46% of 6 M samples were missing or excluded. In the open cohort, 20% of 1 M samples, 71% of 3 M samples and 53% of 6 M samples were missing or excluded.
TEG-PM parameters
Within the open intervention group, those that received SAPT had significantly greater MAAA (37.4 ± 16.6 vs 19.7 ± 9.0, p < .001), ADP % aggregation (76.0 ± 29.7 vs 52.9 ± 33.4, p < .05) and, AA % aggregation (53.0 ± 35.1 vs 11.5 ± 15.2, p < .001) values than those receiving DAPT on average (Figure 2). In the cohort of patients that underwent endovascular interventions, blood samples from patients receiving SAPT demonstrated significantly greater MAAA (40.5 ± 19.5 vs 30.5 ± 18.5, p < .01) and AA % aggregation (59.0 ± 40.3 vs 27.8 ± 31.9, p < .001) values than those who received DAPT (Figure 2).
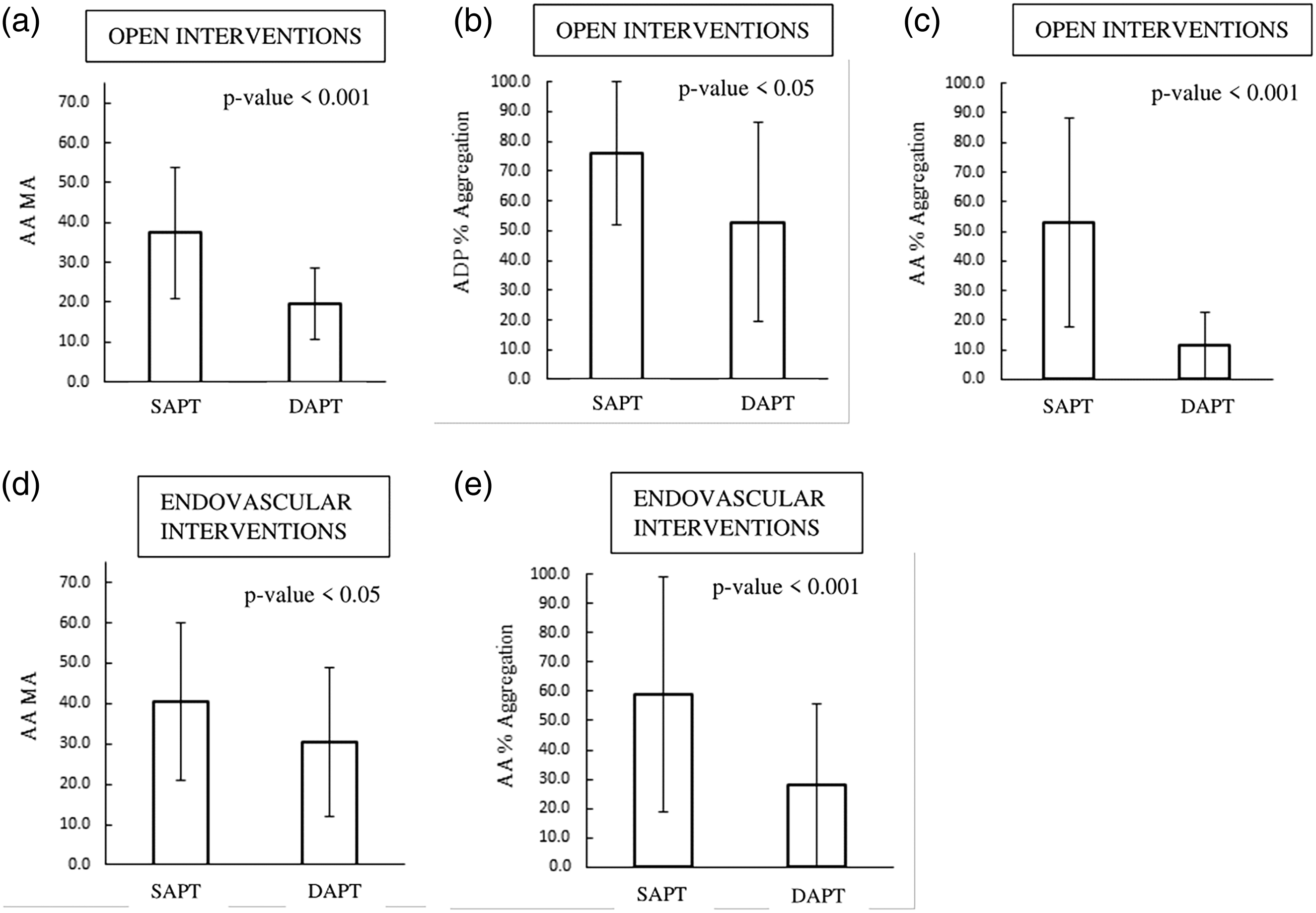
Comparison of TEG-PM parameters between SAPT and DAPT users within the open intervention cohort.
Clinical outcomes: TEs
In the overall cohort, bypass stenosis or occlusion was the most common thrombosis event reported (n = 18). Thrombosis events were more frequent in the endovascular group compared to the open surgical group (13 vs five events). Subjects on DAPT experienced more bypass-related events than those on SAPT. However, when comparing antiplatelet therapies and their associated events, no statistically significant differences were observed within each procedure type. Similarly, there was no significant difference in bleeding events between groups (open SAPT: 2 vs DAPT: 2, p = .19; endovascular SAPT: 0 vs DAPT: 1, p = 1).
Kaplan–Meier curves
In the open procedure cohort, the Kaplan–Meier curve demonstrated a sharp drop off in thrombosis event-free survival, with approximately 50% of patients experiencing a thrombosis event within 8.7 months. In contrast, the endovascular group showed a more gradual decline in thrombosis event-free survival. At 10.47-month mark, the thrombosis probability is around 50% for the endovascular cohort. There was no statistically significant difference in thrombosis probability between the endovascular and open procedure groups (p = .73). This suggests that the type of procedure (endovascular vs open) does not significantly influence thrombosis outcomes in this cohort (Figure 3). Similarly, there was no statistically significant difference in thrombosis probability between the two treatment groups (DAPT and SAPT) in open and endovascular procedures (p = .98) (Figure 3).

Thrombosis probability comparing endovascular and open procedures. (a) Thrombosis probability over time is compared between two groups of patients who underwent endovascular procedures: those receiving dual antiplatelet therapy (DAPT) and those receiving monoantiplatelet therapy (SAPT). Both groups showed similar probabilities of thrombosis. (b) Similarly, for patients who underwent open procedures, the thrombosis probability over time was comparable between both groups (SAPT and DAPT). (c) The endovascular (Endo) group is represented by the yellow line, while the open surgical group is shown in red. The survival curves for both groups exhibit a similar trajectory, indicating comparable thrombosis probabilities across the follow-up period.
In the open procedure group, subjects receiving SAPT showed a 25% lower thrombosis probability compared to those on DAPT (HR: 0.75, p = .67); however, this reduction continued not statistically significant after adjusting for age, sex, BMI, and smoking status. Similarly, in the endovascular group, there was no effect in the hazard in thrombosis probability between SAPT and DAPT (HR: 1.04, p = .92) following the same adjustments. These findings suggest that the choice between SAPT and DAPT does not significantly impact thrombosis risk in either group under the adjusted conditions (Table 2).
Comparison of events between patients on single antiplatelet therapy (SAPT) and dual antiplatelet therapy (DAPT) in endovascular and open procedures.

Abbreviations: N = total number of events in each group.
Note: In the overall cohort, bypass stenosis or occlusion was common (n = 18), with more thrombosis events in the endovascular group (13 vs 5) and higher bypass-related events in DAPT compared to SAPT. However, no statistically significant differences were observed in antiplatelet therapies or bleeding events across procedure types.
Discussion
Our study of 157 PAD patients (64% endovascular, 36% open surgical) revealed significant differences in platelet function between SAPT and DAPT groups. In the open surgical cohort, SAPT patients showed significantly higher MA AA (37.4 ± 16.6 vs 19.7 ± 9.0, p < .001), ADP % aggregation (76.0 ± 29.7 vs 52.9 ± 33.4, p < .05), and AA % aggregation (53.0 ± 35.1 vs 11.5 ± 15.2, p < .001) compared to DAPT patients. Similarly, in the endovascular group, SAPT patients demonstrated higher MA AA (40.5 ± 19.5 vs 30.5 ± 18.5, p < .01) and AA % aggregation (59.0 ± 40.3 vs 27.8 ± 31.9, p < .001). While initial TEs were comparable between SAPT and DAPT groups in both interventions, recurrent TEs were significantly higher in SAPT patients undergoing open surgery (15.9% vs 0%, p < .001).
TEG® is a well-established viscoelastic technique for assessing hemostasis. It is widely used in operating rooms and surgical intensive care units, particularly in transplant, cardiac, and trauma surgery. The application of TEG to identify prothrombotic conditions in peripheral vascular disease, including detection of venous thromboembolic events and assessment of clinical outcomes after carotid artery stenting, represents an emerging and promising area of research. 12 Majumdar et al. have identified a statistically significant increase in MAAA for SAPT samples, alongside increased platelet aggregation and decreased platelet inhibition, all of which suggest a more prothrombotic state in patients on SAPT compared to DAPT, regardless of the type of intervention received. 13 In this study, using TEG-PM to analyze the blood samples of patients undergoing operative lower limb revascularization for symptomatic PAD, it is consistently evident that patients receiving DAPT postoperative prophylaxis convey less coagulable profiles than those receiving SAPT. This phenomenon was observed in patients who underwent either open or endovascular intervention of symptomatic PAD. Our results complement and build on the findings of Majumdar et al., which used TEG-PM to investigate the impact of SAPT and DAPT on platelet reactivity in patients with PAD but did not extend their investigation to evaluate the clinical outcomes of these cohorts following revascularization.
More importantly, the key question is whether the significant effect of DAPT on TEG-PM coagulation profiles translates into meaningful clinical outcomes. In patients who underwent endovascular revascularization, the incidence of index TEs in those receiving SAPT and DAPT was similar (20.4% vs 25.0%, respectively; p = .7) despite the significantly more favorable coagulation parameters seen in those receiving DAPT. Similarly, among the open intervention cohort, patients receiving SAPT experienced a similar incidence of index TEs as those receiving DAPT (25.6% vs 25.8%, respectively) despite those receiving DAPT having more favorable coagulation parameters on TEG-PM. It is, however, noteworthy that DAPT decreased the occurrence of recurrent TEs in both the open and endovascular revascularization groups compared to SAPT use, albeit only significantly in the open intervention group. Therefore, patients receiving DAPT were as likely to experience a TE as those receiving SAPT in either operative cohort; however, patients were less likely to experience recurrent TE when treated with DAPT.
In our study, when comparing medication regimens, irrespective of the procedure type, we also found that the SAPT group showed greater platelet activity indicative of a prothrombotic state. These results suggest that a DAPT regimen would confer a lower risk of postrevascularization TE. The multispecialty guidelines also favor DAPT as it was associated with a lower risk of limb-related events in symptomatic patients postrevascularization. 14 The SVS guidelines even recommend that DAPT may be prolonged for six months in patients undergoing repeated intervention, while its use for greater than six months after endovascular revascularization was associated with less frequent cardiovascular and MALEs than those who used DAPT for less than six months or those on SAPT.4,15,16 DAPT use after lower extremity revascularization has also been shown to reduce all-cause mortality and major amputation significantly. 8 The CHARISMA trial also showed that DAPT regimens were associated with a significant benefit when it comes to MACCE in patients at high risk of ischemia (history of symptomatic PAD, stroke, or MI), although DAPT did not significantly differ in terms of benefits in their overall study population, which is similarly reflected in our overall patient sample. 16 They also reported an increased risk of moderate bleeding with this drug regimen. It was suggested that this could be reduced by lowering the dose of aspirin since the ATC meta-analysis reported a 22% reduction in MACEs with 75 to 150 mg of aspirin per day. This lower dose of aspirin was found to be as effective as the higher dosage but with a lower risk of bleeding. 17
Our findings also complement the recent European Society for Vascular Surgery 2023 Guidelines 4 on antithrombotic therapy. While the guidelines suggest that DAPT may be considered for up to six months postendovascular intervention in patients without high bleeding risk (Class IIb, Level C), our study provides novel mechanistic insights through TEG-PM analysis supporting this recommendation. We demonstrated that DAPT recipients exhibited significantly improved coagulation parameters compared to SAPT users, though this did not translate into reduced index TEs. Notably, our finding of reduced recurrent TEs with DAPT in the open surgery group (0% vs 15.9%, p < .001) suggests potential benefits in specific high-risk populations, aligning with the guidelines’ emphasis on individualized antithrombotic approaches.
While previous studies have examined clinical outcomes of SAPT versus DAPT in PAD patients, our study uniquely combines TEG-PM analysis with clinical outcomes to provide objective measurements of antiplatelet therapy effects. This prospective observational study offers an objective comparison between SAPT and DAPT regimens. These findings lay the foundation for further investigation with larger sample sizes to enhance the understanding and management of postoperative thrombosis events and their recurrence.
This study has its own set of limitations. First, the number of patients recruited and TEG-PM samples may not be sufficient to power this study adequately. This may have underestimated the difference in the incidence of TEs between SAPT and DAPT users in either operative group. Second, we included patients undergoing any lower extremity revascularization, disregarding lesion location and characteristics. Therefore, a randomized method with consistent selection criteria is required to influence practice. Additionally, many factors may contribute to thrombotic risk, such as inflammation, perioperative blood loss, and transfusions. This study did not specifically consider these factors when investigating the variability in platelet response between medication groups. We also did not specifically examine the effects of clopidogrel and aspirin resistance in this study, which may affect our comparisons. Further research is necessary to fully elucidate the optimal antiplatelet regimen for PAD patients to improve clinical outcomes and minimize adverse events. Lastly, this was an exploratory, observational study and as such there was no power study performed. However, for our future studies, we will use the data and patient size from this study to calculate power.
Conclusion
While DAPT may positively influence coagulation parameters, indicating a less prothrombotic state compared to SAPT, this did not translate into a clinically significant reduction in the incidence of initial TEs postrevascularization for patients with PAD. These findings suggest that while DAPT may offer some advantages over SAPT in managing patient's postrevascularization, the choice of therapy should be tailored to the patient's specific clinical context, considering the complex interplay of coagulation dynamics and thrombotic risks.
Supplemental Material
sj-docx-1-sci-10.1177_00368504251324332 - Supplemental material for Single versus dual antiplatelet therapy impact on coagulation/thrombosis post PAD revascularization
Supplemental material, sj-docx-1-sci-10.1177_00368504251324332 for Single versus dual antiplatelet therapy impact on coagulation/thrombosis post PAD revascularization by Adriana A Rodriguez Alvarez, Shiv S Patel, Isabella F Cieri, Samir Ghandour, Mounika Boya, Sasha P Suarez, Aniket Agrawal, Ivy Lee, Lois Owolabi, Mohit Manchella and Anahita Dua in Science Progress
Footnotes
Acknowledgment
We extend our sincere gratitude to Will Teeple for his invaluable support and contributions to the laboratory.
Author contributions
Adriana A. Rodriguez Alvarez: design of the work, analysis, interpretation, and drafting; Shiv S. Patel: conception, design of the work, analysis, interpretation, and drafting; Isabella F. Cieri: data collection and drafting; Samir Ghandour: data collection and drafting; Mounika Boya: data collection, and editing; Sasha P. Suarez: data collection and editing; Aniket Agrawal: data collection and editing; Ivy Lee: data collection and editing; Lois Owolabi: editing and data collection; Mohit Manchella: editing and data collection; Anahita Dua: final approval, supervision, editing, critical revision, and funding.
Consent to participate
Written or electronic consent was obtained from all study participants or the participant's legally authorized representative if the participant could not provide consent.
Data availability
Data will only be shared following publication, upon receiving a written request and approval from the corresponding author.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Institutes of Health (NIH) R21 Federal Grant (Grant No. R21AG07731).
Ethical considerations
The study protocol was approved by the Mass General Brigham institutional review board (IRB #2022P001918).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

