Abstract
Objective
To investigate the way in which prolylcarboxypeptidase (PRCP) protects against myocardial ischemia-reperfusion injury (MIRI) and the mechanisms that underlie it.
Method
A surgical ligation of the coronary artery was adopted to establish a myocardial ischemia-reperfusion model in male SD rats. Thirty-six rats were randomly divided into six groups: Normal group, Sham group, MIRI model group, empty vector (MIRI + EZ.null) group, PRCP overexpression (MIRI + PRCP) group, and nicorandil (MIRI + Nic) group, with 6 rats in each group. The rats received an injection of PRCP's adeno-associated virus 9 (AAV9) through the tail vein 3 weeks prior to the modeling.
Results
Compared with the Normal and Sham groups, the expression levels of NOD-like receptor thermal protein domain-associated protein 3 (NLRP3), Caspase-1, IL-18, IL-1β, and GSDMD in the MIRI group and MIRI + EZ-null group were significantly increased (
Conclusion
PRCP can lessen MIRI and protect cardiac function in rats by inhibiting NLRP3/Capase-1/IL-18/IL-1β signaling pathway-mediated cell pyroptosis.
Keywords
Introduction
One of the leading causes of cardiovascular disease-related deaths worldwide is coronary heart disease. 1 Effective medications for preventing myocardial ischemia-reperfusion injury (MIRI) in clinical practice are still lacking, despite advances in the interventional strategy to treating acute MI.2,3
It is unclear what role prolylcarboxypeptidase (PRCP) plays in MIRI. It has been demonstrated that PRCP is crucial to the management of MIRI. 4 PRCP is a lysosomal ProX carboxypeptidase capable of cleaving AngII to produce Ang1-7 at an optimal acidic pH.5–7 Endothelial cells can express PRCP, which can be produced in all of the arteries, veins, and capillaries of the human circulatory system. 8 It has been discovered that pathologic circumstances such as inflammation, hyperlipidemia, diabetes, obesity, and hypertension change PRCP expression.9,10 The involvement of PRCP in the pathophysiology of patients with experimental hypertension and cardiac transplantation has been identified as a key factor in renal and cardiac Ang1-7 generation. 11 Adam GN et al. showed that the unifying mechanism for hypertension and thrombosis in PRCPgt/gt mice is that PRCP deficiency is associated with increased ROS and cleaved caspase 3 in the endothelium. PRCP depletion is associated with transcriptional down-regulation of eNOS and uncoupling of endothelial cell nitric oxide production. PRCP increases cell growth and proliferation and neoangiogenesis. Absent PRCP led to increased neointima proliferation after balloon injury.12,13
Pyroptosis is a recently identified type of planned inflammatory necrosis of cells that is reliant on the GSDMD protein family's pore-forming activity. 14 It is typified by DNA fragmentation, chromatin condensation, and an apoptosis pathway that is dependent on caspase-1. Prior research has demonstrated its involvement in the pathophysiology of numerous cardiovascular disorders as well as its critical regulatory function in the onset and development of MIRI.15,16 Accordingly, comprehending the mechanism of pyroptosis could help narrow this signaling cascade and thus mitigate MIRI. 17 The NLRP3/Capase-1/IL-1β/IL-18 signaling pathway is a classic pathway of pyroptosis activation. 18 Pyroptosis is thought to be considered a significant component of inflammation in response to MIRI. The NOD-like receptor thermal protein domain-associated protein 3 (NLRP3) inflammasome, consisting of the NLRP3, ASC, and Caspase-1 proteins, may initiate pyroptosis which is a form of inflammatory programmed cell death. Activation of the NLRP3 inflammasome can exacerbate MIRI by boosting caspase-1-mediated thermal pyroptosis and the consequent release of pro-inflammatory cytokines IL-18 and IL-1β. 19 Thus, we postulated that PRCP could operate as a cardioprotective agent in MIRI by inhibiting the NLRP3/Capase-1/IL-18/IL-1β signaling cascade, which is consistent with anticipation of Adam.12,13
Materials and methods
Rats were euthanized with sodium pentobarbital (120 mg/kg), and hearts were excised. When the rat's heart stopped completely and the pupils were dilated, we determined that the rat was dead. All the studies followed the relevant Equator guidelines. 22
Indicators for detection
Echocardiography
Echocardiography (VEVO2100, VisualSonics) was used to detect cardiac function factors in rats in each group after the MIRI model was established. Following induction anesthesia with 5% isoflurane, the rats were placed supine on a heating plate at 37 °C, and their chests were cleansed before a tiny quantity of coupling agent was applied. Anesthesia was then maintained with 3% isoflurane. The detection frequency was set to 12.00 MHz, and the probe was held for 10 seconds. The left ventricular diastolic end-diastolic diameter (LVIDD), left ventricular systolic end-systolic diameter (LVIDS), ESV (contractile end-systolic left ventricular volume), EDV (diastole end-left ventricular volume), fractional shortening (FS), and ejection fraction (EF) were detected.
2,3,5-Triphenyltetrazolium chloride (TTC) staining
The size of the myocardial infarction area was determined using TTC staining. Take some myocardial tissue out of the freezer at −80 °C. Then, using a sharp knife, cut the tissue longitudinally into slices that are about 2 mm thick. Place the slices on an ice block and work your way up to the apex of the heart. Place the freshly cut tissue slices in a small glass culture dish containing 1%TTC staining solution (G-1017, Servicebio). Place the dish in a 37 °C water bath incubator in the dark for 15–30 minutes. Every 5 to 8 minutes, gently shake the dish and flip the slices over one or twice to make sure they have equal contact with the staining solution. Use a pipette to remove the TTC staining solution from the container and place it in another container in the refrigerator at 4 °C for future use. Then, rinse the tissue surface with tissue washing solution to observe the color change with the naked eye. Normal tissue appears pink, while necrotic tissue appears pale. Take a photo for record keeping. Finally, fix the tissue in 10% formaldehyde solution and store it in the dark. The images were captured, and the size of the infarct area of the heart was quantified using ImageJ software.
Hematoxylin–eosin (HE) staining
The Cardiac tissues were pre-fixed with 4% paraformaldehyde overnight, and the fixed rat heart was cut into 5 μm slices under paraffin-embedding. Subsequently, the slices were stained sequentially with hematoxylin for 3 minutes and then with eosin for 3 minutes. Finally, the sealed sections were observed using a light microscope (Nikon, Tokyo, Japan), which showed blue nuclei and red cytoplasm under the microscope. Picture acquisition was also performed.
TdT-Mediated dUTP nick end labeling (TUNEL)
TUNEL staining was used in order to assess cardiomyocyte apoptosis. In short, the cardiac tissue sections were fixed in 4% paraformaldehyde and then stained with TUNEL Assay Kit (G1507, Servicebio) and DAB (G1212, Servicebio)for cardiac sections according to the protocols provided by the manufacturer. ImageJ software was used to quantify the percentage of TUNEL-positive cells.
Western blotting
Total protein extraction was carried out with the utilization of RIPA buffer (P0013B, Beyotime Biotechnology), and the concentrations of protein were determined by means of the BCA Protein Assay Kit (P0010, Beyotime Biotechnology). After SDS–PAGE electrophoresis, the protein samples were transferred to PVDF membranes (Millipore). Remove the successfully transferred PVDF membrane and place it in 5% skimmed milk powder. Block it for 1 hour at room temperature on a shaker. Subsequently, refer to the instruction manual of the primary antibody and dilute the corresponding primary antibody against NLRP3 (DF7438, 1:500, Affinity), Casepase-1 (AF5418, 1:500, Affinity),1L-1β (AF5103, 1:500, Affinity), 1L-18 (DF6252, 1:500, Affinity), GSDMD (AF4012, 1:500, Affinity), GAPDH (T0004, 1:3000, Affinity) according to the appropriate ratio, and the membranes was incubated at 4 °C overnight with primary antibodies. The overnight primary antibody was washed thoroughly with TBST solution 3 times, each for 10 minutes, followed by incubation with the secondary antibody to goat anti-rabbit (1:2000, BA1056, BOSTER Biological Technology) in TBST solution. The PVDF membranes were then developed using ECL chemiluminescence for color visualization and quantified by ImageJ analysis software. GAPDH was used as an internal reference for detecting the expression level of proteins.
Quantitative PCR
Total RNA was obtained from SD rats’ cardiac tissue using Trizol reagent. RNA concentration and purity were determined using Maestro Nano Microvolume UV–Vis Spectrophotometer (Maestrogen, MN-913A). Reverse transcription reaction systems were prepared using HiScript III RT SuperMIX (Vazyme Biotech, R323-01) mixed with total RNA. In the PCR instrument, the reaction was terminated after 37 °C for 15 to 20 minutes, and 85 °C for 30 seconds. The quantitative real-time PCR (qPCR) reaction was performed using 4 μl of Template DNA with 10 μl 2× SYBR Green Master Mix (Bioer Technology, BSB25L1B), 0.4ul of ROX Reference Dye (optional), 4.8 fjkklμl of ddH2O, and a total of 20 μl of forward and reverse primers. 23 The reaction program was as follows: initial denaturation at 95 °C for 30 seconds, followed by 40 cycles at 60 °C for 30 seconds each and 15 seconds at 95 °C. After the completion of the reaction, the reaction was dissociated at 95 °C for 15 seconds at a slope of 1.6 °C/s, and then dissociated at 60 °C for 1 minute, with an increase in the temperature rate of 1.6 °C/s. The reaction was then warmed up to 95 °C for 15 seconds at a rate of 0.08 °C/s, and then amplified at 95 °C, with the following procedure. Finally, the temperature was warmed up to 15 seconds at 95 °C with a warming rate of 0.08 °C/s. Primer design was performed online through the National Center for Biotechnology Information(NCBI), primer-blast. The primer sequences designed are shown in Table 1. Amplification data were analyzed using the delta-delta Ct method. GADPH serves as an internal reference gene, reflecting relative changes in the RNA expression levels of genes.
Q-PCR primer sequences.

Statistical analysis
All the experiments conducted by the groups in this study were repeated at least 3 times. The data presented in the form of Mean ± SEM obtained from GraphPad Prism 8 (GraphPad, San Diego, CA, USA). Parametric tests were performed on the data. After tests of normality and Chi-square, we determined mean differences among groups using a one-way ANOVA with Tukey's correction for multiple comparisons.
The level of significance was set at 0.05.
Results
Effects of PRCP on MIRI and LV function
To evaluate the impact of PRCP against myocardial ischemia-reperfusion damage, we assessed the Infarct Size of SD rats. Compared with the MIRI group, PRCP, and nicorandil pretreatment effectively protected against the MIRI in the cardiac tissue (Figure 1B). To evaluate the impact of PRCP on the cardiac function of rats, we performed cardiac M-mode ultrasonography 24 hours after reperfusion (Figure 1A). We conducted a computational analysis of LV EF and fraction shortening (FS), both of which can correctly indicate the level of cardiac function (Table 2). Echocardiography showed a significant decrease in left ventricular fractional FS and EF after MIRI compared with the MIRI group. In contrast, PRCP overexpression improved FS and EF (Figure 1C, D).
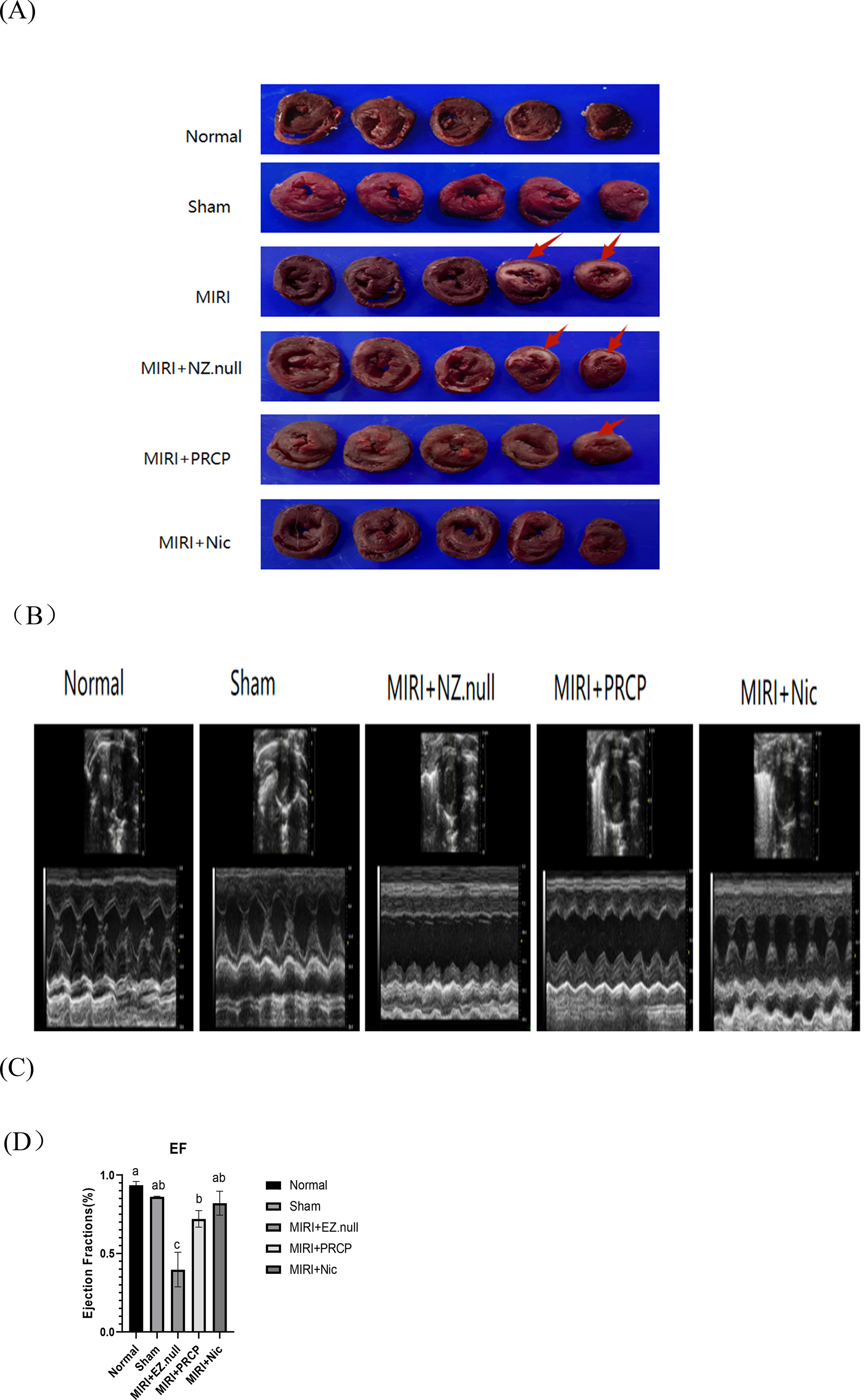
Effects of PRCP on MIRI and LV function. (A) Echocardiography assessing EF and FS in rats after MIRI.PRCP reduces infarct size and enhances myocardial contractile function. (B) The infarct size in MIRI rats was evaluated using the 2,3,5-triphenyltetrazolium chloride (TTC) staining method. After TTC staining, normal tissues remain unstained, while infarcted tissues appear pale white. The area of infarction is marked by the red arrow in the figure. (C) Histogram of the EF results in (A). (D) Histogram of the FS results in (A). Data are presented in the form of Mean ± SEM. Different letters denote significant differences.
Cardiac ultrasound results.

LVIDD, left ventricular diastolic end-diastolic diameter; LVIDS, left ventricular systolic end-systolic diameter; ESV, contractile end-systolic left ventricular volume; EDV, diastole end-left ventricular volume; FS, fractional shortening; EF, ejection fraction. Different letters denote significant differences.
Effect of PRCP on MIRI-induced mitophagy pyroptosis
Comparison of the degree of cardiomyocyte apoptosis in each group was interest. Cardiomyocytes are specialized heart cells that are fully differentiated and cannot be renewed or regenerated. Recent studies have shown that MIRI can trigger cardiomyocyte apoptosis or necroptotic apoptosis, and the apoptosis resulting from cardiac injury is irreversible.
24
Compared with the Normal group and the Sham group, the establishment of the MIRI model can upregulate the degree of cardiomyocyte apoptosis in rats (

Effect of PRCP on MIRI-induced mitophagy pyroptosis. (A) Apoptosis of tissue cells was observed through TUNEL staining. The MIRI group displayed a much higher level of cardiomyocyte pyroptosis (by approximately 10%) compared with the Normal group and the Sham group. And after PRCP or nicorandil pretreatment, the number of TUNEL-positive cells was notably decreased. Scale bar = 50 μm.
Effects of PRCP on MIRI-induced myocardial tissue injury
The normal group and the sham group showed well-ordered myocardial tissue without cell necrosis or edema in the interstitium. No obvious inflammatory cell infiltration was observed. The myocardial tissue in the MIRI and MIRI + NZ.null groups was disordered and edematous, with large numbers of vacuolated deformities and a paler myocardial color. Moderate inflammatory cell infiltration was observed. The myocardial tissue in the MIRI + PRCP and MIRI + Nic groups was relatively intact, with only a small amount of inflammatory cell infiltration (Figure 3A). Overexpression of PRCP and nicorandil can both inhibit the degree of myocardial injury in MIRI rats.

Effects of PRCP on MIRI-induced myocardial tissue injury. (A) Representative HE staining. In the figure, blue arrows mark eosinophils, red arrows mark neutrophils, green arrows mark lymphocytes, black arrows mark macrophages, and purple arrows mark plasma cells. Scale bar = 50 μm.
Effects of PRCP on NLRP3 signaling pathway
To explore the connection between pyroptosis and MIRI, the expression of GSDMD, we measured the expression of GSDMD, a crucial executor of pyroptosis. In contrast to the Normal group and the Sham group, Western blot analysis revealed that (Figure 4A) inflammatory markers (NLRP3, GSDMD, caspase-1, IL-18, and IL-1β) were significantly upregulated in the MIRI group. Overexpression of PRCP by adeno-associated virus transfection and nicorandil intervention can all result in a downregulation of NLRP3, Caspase-1, IL-18, IL-1β, and GSDMD expression in rat cardiac tissue (

Effects of PRCP on NLRP3 signaling pathway. (A) Western blot was used to detect NLRP 3, GSDMD, Caspase-1, IL-18, IL-1β. (B) Relative expression of NLRP3, GSDMD, Caspase-1, IL-18, IL-1β mRNA was detected by q-PCR. There were more than three of these duplicates. Mean ± SEM were used to express the data. Different letters denote significant differences.
Discussion
Both the kallikrein–kinin system (KKS) and the renin–angiotensin system (RAS) are crucial in regulating the physiology of kidney, heart, and blood vessel. The engagement of these two Interlaced pathways is of essential significance in various frequent pathology conditions, such as inflammation, heart failure, renal disease, and diabetes. Whereas both KKS and RAS are highly controlled systems that identify distinct receptors presented on endothelial cells, they are able to precisely modulate each other's effects through the sharing of multifunctional enzymes (PRCP, angiotensin-converting enzyme). PRCP has surfaced as a protease that offers cardioprotective benefits, highlighting its significance for cardiovascular health. It has been reported that the downregulation of PRCP, together with the enhancement of RAS activity, may cause further worsening of left ventricular (LV) systolic dysfunction in spontaneously hypertensive rats (SHRs). 25 Recent studies provide compelling evidence that PRCP can protect the heart during myocardial ischemia-reperfusion. 4 But some of the potential mechanisms remain unclear and have not yet been fully explained.
In this study, we examined the impact of PRCP overexpression preconditioning on MIRI-modeled rats, and the results showed that it improved a number of harmful changes, including vascular morphological abnormalities, and reduced reflow area and infarct size and decreased cell pyroptosis. Proinflammatory cytokines such as 1L-18, and 1L-1β increase in the early stage after infarction. Excessive inflammation, whether it occurs early or persists in a chronic and unrestrained manner, can lead to significant and irreversible impacts on the structure and function of the myocardium. Thus, Promptly addressing the inflammatory response is crucial to preventing any negative outcomes. 26 As an ATP-sensitive K + (KATP) channel opener (KCO), nicorandil exerts cardioprotective effects in the face of MIRI. It reduces the risk of major adverse cardiovascular events and the no-reflux phenomenon, lowers infarct size, and lessens the occurrence of ventricular arrhythmias in sufferers with AMI. Therefore, we chose nicorandil as the positive control drug. 27 The results indicated that nicorandil significantly decreased the size of the infarcted myocardial area and reversed the increases in EF and FS caused by MIRI, and PRCP showed similar efficacy to nicorandil.
The focus on pyroptosis has become evident as a potential therapeutic approach for treating various cardiovascular diseases, such as atherosclerosis and myocardial infarction. The expression of basic inflammatory vesicle components under normal physiological conditions alone is insufficient to trigger pyroptosis. Consequently, we can propose that altering the expression of inflammasome components could serve as a potential therapeutic approach. The use of trimetazidine reduces the myocardial infarction caused by MIRI and H/R models by blocking the TLR4/MyD88/NF-κB/NLRP3 pathway, which can mediate pyroptosis through GSDMD. 28 Zhang et al. 29 demonstrated that metformin treatment inhibited NLRP3 inflammasome activation by preventing cell death via the AMPK pathway in a Langendorff rat heart model with MIRI and NRCMs with H/R. In addition, metformin treatment reduced apoptosis, decreased the size of myocardial infarction, and inhibited cellular fibrosis. Cinnamic acid possesses antioxidant and anti-inflammatory properties, which is a compound extracted from Cinnamomi ramulus. It protects cardiomyocytes from MIRI by inhibiting the NLRP3/Caspase-1/GSDMD signaling pathway and attenuating oxidative stress. 30 As our positive control, nicorandil was demonstrated by Chen et al. to reduce focal death in rats with myocardial infarction by inhibiting the TLR4 / MyD88 / NF-κB/NLRP3 signaling pathway. 31 Our study also confirmed that nicorandil can reduce scorch death in rats with MIRI, which is consistent with previous studies, and overexpression of PRCP can achieve the same effect.
This work has a few limitations. The main limitation of this study is the poor quality of immunohistochemical staining. Factors such as the heating time for antigen retrieval, the dilution ratio of the antibody, the incubation time of the antibody, and the DAB staining duration all impact stain quality. At the same time, using positive and negative control slides can help prevent false-positive results. In subsequent studies, we will further optimize experimental conditions to enhance the quality of the results. Furthermore, the possibility of atypical pyroptosis signaling pathways or other forms of cell death should not be ruled out, which warrants additional investigation.
Conclusion
In this study, SD rats underwent MIRI model establishment, which resulted in obvious myocardial infarction and tissue damage, as well as cardiac dysfunction. At the same time, the expression of cell pyroptosis and related pathway proteins increased in terms of both protein and mRNA levels. We can reverse these unfavorable outcomes by overexpressing PRCP in rats. We can conclude that overexpression of PRCP may alleviate cell pyroptosis by inhibiting the NLRP3/Caspase-1/IL-18/IL-1β pathway, thereby improving heart function and exerting cardioprotective effects in SD rats with MIRI.
Footnotes
Acknowledgments
The authors would like to express our gratitude to all those who participated in this study.
Authors’ contribution
Luxiao He performed writing review and editing, writing the original draft, visualization, validation, methodology, investigation, software, formal analysis, and data curation.
Qiuping Lu and Ye Luo performed writing original draft, formal analysis, and data curation.
Youfa Qin did writing the original draft, resources, supervision, project administration, funding acquisition, and conceptualization.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of competing interest
The authors state that they are not aware of any competing financial interests or personal relationships that might seemingly have affected the work described in this paper.
Funding
The authors disclose that they received the following grants for research, writing and/or publication of this article: Major Research Project Incubation Fund of Dongguan Songshan Lake Central Hospital (SZR005), Dongguan Science and Technology Bureau, China (No. 202071715024129, No. 20231800940392), and Scientific Research Project of Guangdong Provincial Bureau of Traditional Chinese Medicine (20201368).
