Abstract
Objective
To study the impact of kinetic glomerular filtration rate (kGFR) on clinical decision making and its implications on drug dosing compared to that of estimated GFR (eGFR) using chronic kidney disease epidemiology collaboration (CKD-EPI) equation in critically ill patients with acute kidney injury (AKI) admitted in a tertiary level intensive care unit (ICU).
Methods
Cross-sectional, prospective, observational study design. All patients admitted to Medical ICU, Fortis Hospital, Bangalore with AKI defined as per AKI network (AKIN) criteria. Patients were recruited after approval from the scientific and institutional ethics committee, with written informed consent. Serum creatinine values at admission and further values were noted. GFR was calculated using both formulas (CKD-EPI and kGFR) and documented at all intervals of creatinine sampling. Drugs requiring renal dose modification along with the dosing were documented. Sample size was calculated after a pilot study and a total of 107 patients were analyzed.
Results
Incidence of AKI was 12.84%. The mean (±SD) eGFR was 37.25 (±29.4) and kGFR was 42.5 (±33.2), (p-value .003). 70 (65.42%) patients required drug dose change when kGFR was used. Dosing changes from Day 1 to Day 5 are 53/104 (50.9%), 39/81 (48.1%), 12/26 (46.1%), 2/9 (28.5%), 1/2 (50%). Predominant dose changes were for antimicrobials: vancomycin (35.7%), acyclovir (23.1%), and meropenem (23%).
Discussion
Drug dosing using different methods of GFR calculation showed a difference in the dosing in 65.42% of patients with AKI. Accounting for change in creatinine over time using kinetic GFR may lead to better drug dosing in critically ill patients with AKI.
Conclusion
Our study shows that calculating GFR using kGFR formula instead of CKD-EPI may change drug dosages among patients with AKI admitted in ICU. By replacing conventional GFR estimation formulas with kGFR we may reduce the drug dosing inaccuracies that are currently prevalent in this cohort of patients.
Introduction
Acute kidney injury (AKI) is a common clinical condition found in critically ill patients associated with high mortality, morbidity and increased length of hospital stay. 1 Accurate assessment of renal function is important in diagnosis and is instrumental in proper dosing of drugs eliminated by the kidney as well as timely application of renal protective therapeutic strategies. Assessment of glomerular filtration rate (GFR) using creatinine in the critically ill has been shown to be widely inaccurate. 2 Creatinine values are influenced by muscle mass, age, gender, race, and disease states. In the critically ill patients are often catabolic, immobilized and are on drugs that could influence tubular secretion.
Renal dose adjustment of drugs eliminated by the kidneys is based on GFR values. Inaccurate assessment of GFR can cause under dosing, overdosing causing toxicity and increased patient morbidity and mortality. The gold standard of accurate GFR assessment using radionucleotide markers is largely limited to research studies and is not feasible in routine clinical practice. Estimated GFR using various formulas are therefore used for clinical decisions. These estimation formulas use age, gender, race, and body surface area along with serum creatinine to overcome the limitations of serum creatinine alone for GFR assessment.
The chronic kidney disease epidemiology collaboration (CKD-EPI) equation developed in 2009 has been used to estimate GFR, diagnose AKI, and guide drug dosing. 3 Despite the known inaccuracy of the formula in AKI settings, it continues to be used to guide drug dosing in the critically ill. This formula has been validated only in chronic kidney disease states where creatinine values are in a steady state. Using these formulae in critically ill patients with AKI, in whom creatinine levels change over hours, can lead to highly inaccurate measurements of GFR which can have an impact on many clinical decisions, notably drug dosing.
There is a need to streamline interpretation of creatinine kinetics and accurately estimate renal function in the acute setting. Chen proposed a simple mathematical model to estimate GFR using initial creatinine content, volume of distribution, creatinine production rate, and quantitative difference between consecutive plasma creatinine over a given time period. 4 This method has been added to the Intensive Care Medicine agenda in 2017 as a method of assessing AKI that requires more research. 5 Accounting for rate of change of creatinine can make estimation formulas more accurate than the other formulas where the level of creatinine is assumed to be constant. Pelletier et al. in 2019 published a study showing a good correlation between kinetic GFR with Chen's method and measured GFR using cystatin C in AKI. 6
There is no published data to date studying the drug dosage based on kinetic GFR in critically ill patients with AKI. The current data published shows kinetic GFR to predict more accurate GFR when compared to other estimation formulas like CKD-EPI formula, MDRD formula and the Cockcroft-Gault formula. 7 We sought to see the impact of using kinetic GFR (kGFR) for drug dosing when compared to the CKD-EPI formula (eGFR). We also evaluated different drugs that required a change in dosing as well as whether patient variables and comorbidities influenced the calculation of the GFR using the two formulae. We believe our study further enhances our understanding of AKI dynamics and GFR changes in critically ill patients.
Aim: To study drug dosing decisions using kinetic GFR (kGFR) versus estimated CKD-EPI GFR (eGFR)
Objectives:
Primary objective: To assess differences in drug dosing based on kGFR versus eGFR
Secondary objectives:
To assess the correlation between kGFR and eGFR To compare kGFR versus eGFR in ICU patients categorized as per demographic parameters and disease spectrum To assess outcomes based on kGFR
Materials and methods
The study protocol was accepted by the institution ethics committee (IEC) of Fortis Hospital Bannerghatta road (IEC number: IEC/018/2020 dated June 06, 2020) Bangalore Karnataka, India prior to enrollment. This was a cross sectional, prospective observational study done in medical ICU in Fortis Hospital from the period of June 2020 to May 2021. The procedures followed were in accordance with the Helsinki Declaration of 1975 as revised in 2013. Consecutive sampling methods were followed. All patients >18 years who are admitted in Medical ICU and diagnosed with AKI as per AKI network (AKIN) criteria, defined as a 50% to 300% increase in serum creatinine from baseline were included in the study. 1 Urine output calculation for classifying patients into different stages of AKI was not done as this would require a period of 24 h and patients might have received diuretics or other drugs that could confound this result. A written informed consent was taken from the patient's legally acceptable representative before enrollment in the study. Patients were either severely ill or were deeply sedated on mechanical ventilation and lacked the decision-making capacity to provide informed consent. Patients who were pregnant, had end stage renal disease or patients who underwent hemodialysis after one value of creatinine were excluded from the study. Patients were followed up for their entire stay in ICU. The reporting of this study conforms to STROBE guideline. 8
The following data were collected: demographic characteristics, admitting diagnosis, past medical history, baseline creatinine if available, fluid balance, and the need for dialysis. Serum creatinine values at admission and further consecutive values were also noted during the patients’ stay. All patient details were deidentified during collection of the data. GFR was calculated using both CKD-EPI formula as well as kinetic GFR formula using a computer-based calculator and was documented at all intervals of creatinine sampling. Kinetic GFR was calculated only after two creatinine samples were available. Drugs requiring renal dose modification along with the dosing as per GFR calculated using both formulae were documented once second creatinine value was collected. Drug dose was decided based on Renal Handbook edition 4. 9
CKD-EPI formula used
eGFR = 141 * min (Scr/κ,1)α * max (Scr/κ, 1)−1.209 * 0.993Age * 1.018 [if female] * 1.159 [if black]
Scr is serum creatinine (mg/dL), κ is 0.7 for females and 0.9 for males, α is −0.329 for females and −0.411 for males, min indicates the minimum of Scr/κ or 1, and max indicates the maximum of Scr/κ or 1.
Kinetic GFR formula used:
kGFR = [Steady-State Plasma Creatinine * Creatinine Clearance/Mean Plasma Creatinine] *(1 − [(24*Delta Plasma Creatinine)/(Delta Time (h)* Max Delta Plasma creatinine/day)]).
Sample size estimation was done using Open Epi software version 2.3.1. According to our pilot study of 10 patients, the proportion of patients requiring drug dosing change based on kGFR were 50% and considering 80% power of the study, 95% confidence interval and relative error of 20%. The sample size was estimated to be 99. 835 patients were screened out of which 107 patients were included. There was no missing data.
Statistical analysis
Categorical variables were expressed as frequency and proportions. Drug dosing changes were expressed as a proportion of the total number of patients. To compare values of GFR and to see the change in different disease conditions and demographics, Paired t test was used.
Results
The overall incidence of AKI in our study was 12.84%. Table 1 shows the clinical demographic variables of our study. Table 2 shows the renal characteristics of study cohort at the time of admission. The mean ± SD creatinine was 2.7 ± 1.8 mg/dL. Most AKI subjects in our study had creatinine less than 2 mg/dL (62.6%). The fluid balance was positive in 88.8% of our study patients at the time of admission.
Clinicodemographic variables of the subjects.

Renal characteristics at the time of admission.

CKD: chronic kidney disease; EPI: epidemiology; eGFR: estimated glomerular filtration rate; kGFR: kinetic glomerular filtration rate; AKI: acute kidney injury.
GFR was calculated using both CKD-EPI formula as well as kinetic GFR formula and was documented at all intervals of creatinine sampling. The mean CKD-EPI-eGFR was 37.25 ± 29.4 and mean kGFR was 42.5 ± 33.2.
Among 107 patients who were included in our study with AKI, 104 patients (97.19%) had drugs that required renal dose modification. Both eGFR and kGFR were calculated and needed for dosing change compared based on values. Out of 104 patients on drugs requiring dosing adjustment, 34 (31.77%) patients had same dose requirement, whereas 70 (65.42%) patients had a different dose for at least one drug when kGFR formula was applied. Table 3 shows the frequency distribution of drug dosing change in our study.
Frequency table for drug dose change when using kGFR.

eGFR: estimated glomerular filtration rate; kGFR: kinetic glomerular filtration rate.
Most patients who required drug dosing changes required the changes in the first five days. Figure 1 shows frequency of drug dosing change day-wise when two GFRs were compared. As days in ICU progressed number of patients in the cohort reduced but percentage requiring change persisted to be high reflecting significant changes in drug dosing if kGFR formula is used.

kGFR: kinetic glomerular filtration rate.
A total of 11 drugs that require renal dose modification were prescribed in our study. Majority of the drugs were antimicrobials. Among all the drugs, vancomycin was the drug that required maximum drug dose change (35.7%) followed by Acyclovir (23.1%) and Meropenem(23%). Figure 2 shows percentage of change required for various drugs.
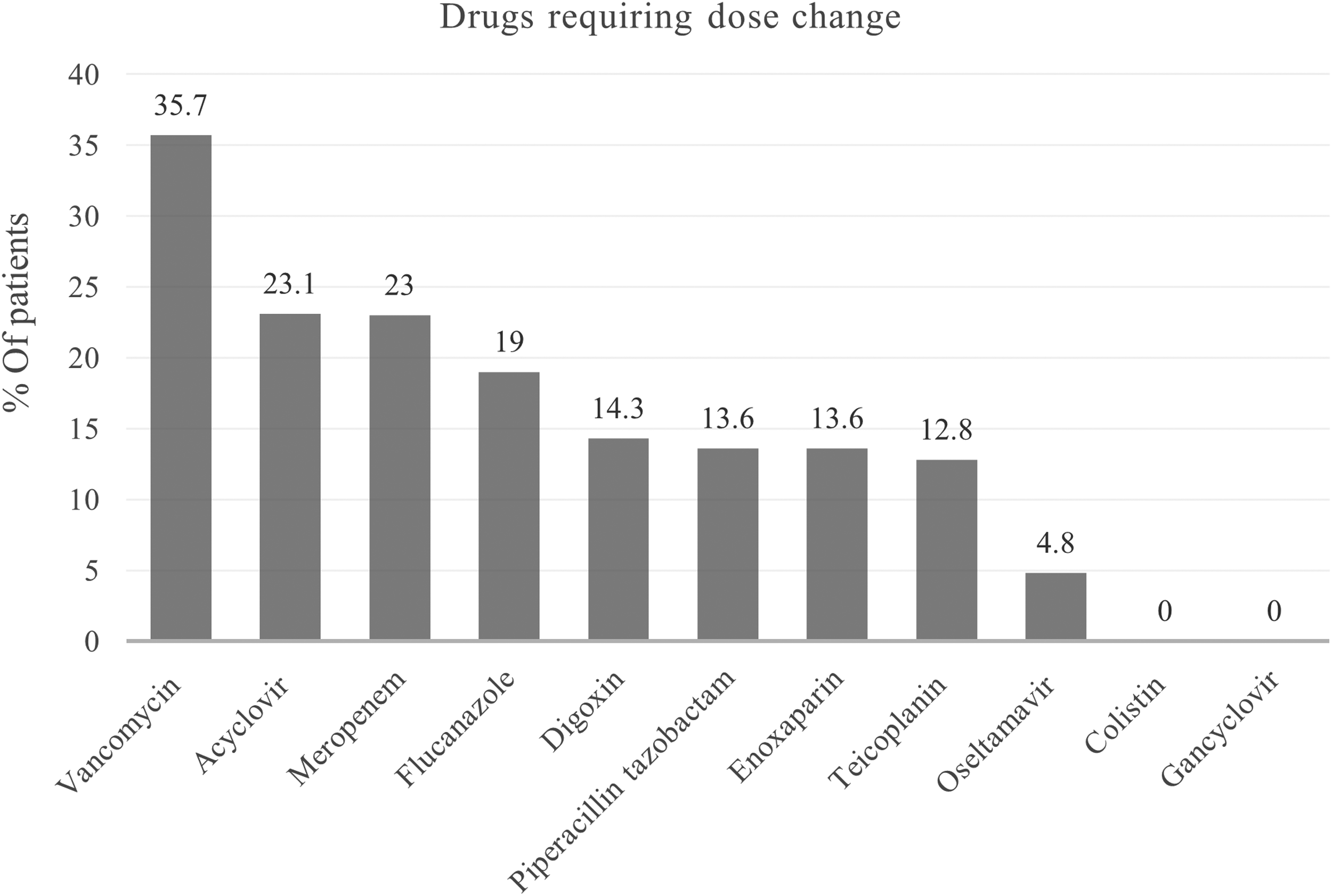
Percentage of drugs requiring dose change.
Table 4 shows the overall difference between kGFR and eGFR according to demographic variables and different comorbidities with a p-value of .03. Overall, there was a significant difference between kGFR and eGFR, but when demographic variables were checked statistical significance was present among only females, patients above 50 years and patients with normal BMI. Among comorbidities diabetes and hypertensives continued to show a significant difference between the two GFR's but this difference was not significant among CKD, CAD, CHF, and CLD patients. It is pertinent to note that the number of patients in our study who had either diabetes and/or hypertension, or both were much higher than patients with other comorbidities mentioned.
Difference between kGFR and eGFR as per demographic variables in AKI patients.

eGFR: estimated glomerular filtration rate; kGFR: kinetic glomerular filtration rate; AKI: acute kidney injury; BMI: body mass index.
Among 107 patients with AKI, 3 patients died in ICU amounting to 2.8% mortality rate in our study. The majority (95.3%) were shifted to ward and 2 patients were discharged against medical advice. Nine (8.41%) patients received dialysis during their stay in ICU and 98(91.58%) patients did not require dialysis. The length of ICU stay among these patients was an average of <7days (88.78%).
Discussion
AKI in the critically ill is a common and challenging clinical condition found in ICUs across the world. Multinational and multicenter studies have highlighted the adverse outcome in this common condition. 10 Our study found an incidence of 12.84% of AKI when the AKIN criteria was used for diagnosis. Most of the patients in our study had an initial creatinine of less than 2 mg/dL (62.6%). Males and elderly patients above the age of 60 years had a higher incidence of AKI. Multinational and multicenter studies highlight the high incidence of AKI among ICU patients as well as the adverse outcomes with mortality going up to 60.3%. 10 Our study did not look at urine output to classify patients and this could have led to an underestimation of AKI.
As per KDIGO guidelines published in 2012, serum creatinine and urine output remain the cornerstone of renal function assessment and classification of AKI severity despite their limitations. 11 The authors acknowledged the lack of accuracy of using these criteria but specify that this will remain the cornerstone of diagnosis until use of better biomarkers for GFR measurement are shown to improve patient outcome. In our study we compared the GFR values of two formulae applied on the same patient. When we compared the GFR's on Day 1 of admission we found that mean GFR estimated by kGFR formula was higher than the mean GFR estimated using the CKD-EPI formula (42.5 versus 37.25 ml/min/1.72 m2). Most patients in our study had a mean admission creatinine of < 2 mg/dL (62.6%). This could mean that our patient cohort included more patients who had milder forms of AKI. We found that GFR estimated with the CKD-EPI formula tended to underestimate the GFR in the initial period of ICU admission. This could lead to under dosing of drugs in the crucial initial period of patient admission and treatment which could have consequences towards outcome and overall morbidity.
Drug dosing in critically ill patients with AKI has always been and remains a challenging task. In our study, we found that overall, 65.2% of patients had different drug dosing requirements and 31.7% had the same dose when the two formulas were compared. The need for drug dosing change remained high at 53/104 (50.9%), 39/81 (48.1%), 12/26 (46.1%), 2/9 (28.5%), 1/2 (50%) from Day 1 to 5 respectively, which has clinical implications. This shows that among patients with AKI admitted in ICU a significant portion of patients require drug dose modifications as per their GFR. Using the CKD-EPI formula for calculations of GFR and drug dosing could lead to over or under dosing of patients. The majority of patients who had a different drug dosing required the change on Day 1 to 3 of admission as the number of patients with AKI reduced progressively in this cohort over the days, though percentage requiring change were consistent. It is also important to note that the creatinine values are more dynamic in the initial part of their illness. With persisting AKI, creatinine levels could stabilize, and both formulae could estimate similar GFR's. It is more crucial to assess GFR accurately during dynamic changes.
A total of 11 drugs that require renal dose modification were prescribed in our study. Majority of the drugs were antimicrobials. Meropenem was the most common drug prescribed and had different drug dosing in 23% of the cases. Among all the drugs, vancomycin was the drug that required maximum drug dose change (35.7%) when the two GFR calculations were compared. Dosing of antimicrobials in critically ill is known to be problematic due to the different pharmacokinetics, hemodynamic and organ dysfunctions prevalent in the critically ill.12,13 Use of vancomycin for MRSA infections in the critically ill is a standard of care. It is well documented that vancomycin is nephrotoxic, and it is recommended to use trough levels to ensure therapeutic levels and to prevent further renal damage. Bakke et al. showed that less than 40% of the patients who were critically ill attained therapeutic trough serum concentrations during the first three days of vancomycin therapy. 14 Hanrahan et al. found that vancomycin use was an independent risk factor for nephrotoxicity among the critically ill. 15 Vancomycin under dosing can also lead to development of vancomycin resistant organisms especially in enterococcus species which could have long lasting adverse impact in the population.
Kinetic GFR performs well when compared to GFR measured using plasma markers as shown by Dewitte et al. (15). Among the 57 patients with AKI in their study, both kinetic GFR and plasma marker NGAL and [TIMP-2]*[IGFBP7] were able to predict renal recovery. Since the use of plasma markers has not been shown to improve patient outcomes and is limited by both cost and expertise, kinetic GFR could be a feasible and inexpensive method of estimating GFR. In our study, we found that when the total 107 patients were compared there was a significant difference between the two GFR estimation formulae (p = .003). By categorizing patients into various subgroups, sampling number reduces, number for comparison decreases, and hence the analysis is not sufficiently powered to detect a difference.
Among different comorbidities, there was a significant difference between the two GFR formulae among patients with diabetes and hypertension (p = .001 and p = .003). These two comorbidities were the most frequent in our study. As the difference between the two estimation formulae are consistent in these two disease conditions, kinetic GFR can be used to estimate GFR in these patients. Both diabetes and hypertension have a direct association between renal failure and ICU admission especially in the Indian subcontinent, 16 highlighting the importance of accurate diagnosis in this cohort.
Among 107 patients with AKI, three patients died in ICU amounting to 2.8% mortality rate in our study. This is a lower mortality compared to most multicenter and multinational studies. 10 Eswarappa et al. published data from an ICU in south India and found mortality among AKI patients being as high as 37.6%. 16 As we did not include patients who were sick enough to require dialysis on day 0, which was the day of admission, sicker patients were not included in our analysis. Only patients who had at least two values of creatinine after admission were included in our study due to the nature of GFR calculations using kinetic GFR.
Multiple studies and recommendations have criticized the use of estimation formulae in AKI both for diagnosis and for drug dosing.17–19 A position paper was published in 2020 in Intensive Care Med reviewing all data available to give recommendations on the best course of action for drug dosing in the critically ill. They concluded that although therapeutic drug monitoring was useful, the limitations it poses like cost and availability of laboratory services does not justify its use in routine critical care management. 20 In India, as of 2021, TDM is used very sparingly and is being recommended for use in selected antivirals for HIV and antitubercular treatment only. 21
There is no published data to date studying the drug dosing based on kinetic GFR in critically ill patients with AKI. Our study showed that there is a high percentage of patients that require a change in drug dosing when kinetic GFR is used for GFR estimation compared to conventional GFR formula recommended currently. This is especially true during the first three critical days of ICU stay. Since this is a small study where drug levels, plasma markers of AKI and actual GFR measurement were not done, the authors cannot recommend change of practices. This study only highlights the current drug dose inaccuracies that are prevalent and the difference in drug dosing when we account for change in creatinine values over time instead of static measures. By replacing conventional GFR estimation formulas with kinetic GFR calculations we may be able to reduce the drug dosing inaccuracies that are currently prevalent in the treatment of this cohort of patients.
Conclusion
Drug dosing changes using kinetic GFR were required in 70/104 (65.4%) of critically ill patients admitted with AKI during the first five days of ICU stay. Dosing changes from Day 1 to Day 5 were required in 53/104(50.9%), 39/81(48.1%), 12/26(46.1%), 2/9(28.5%), 1/2(50%). Majority of drugs requiring dose change was antimicrobials with vancomycin (35.7%) being the most common followed by acyclovir (23.1%) and meropenem (23%). There is a statistically significant difference between kinetic GFR and estimated GFR using CKD-EPI formula (p-value = .03). Drug dosing changes using kinetic GFR may lead to a high proportion of change in dosages of drugs especially antimicrobials, compared to the current practice of using estimation formulae in critically ill patients with AKI.
Footnotes
Acknowledgments
We would like to thank our statistician Dr Amrutha.
Authors’ contributions
Divya Dinakar: conceptualization, data curation, formal analysis, investigation, methodology, validation, visualization, writing—original draft preparation, and writing—review and editing; Garud Chandan: investigation, methodology, supervision, validation, and writing—review and editing; Rajanna Sreedhara: conceptualization, investigation, methodology, software, supervision, validation, visualization, and writing—review and editing; Aashish Parekh: conceptualization, investigation, methodology, software, supervision, validation, visualization, and writing—review and editing. Padmakumar Aryamparambil: investigation, methodology, supervision, validation, and writing—review and editing; Pooja Prathapan Sarada: investigation, methodology, supervision, validation, and writing—review and editing; Ganesh: investigation, methodology, supervision, validation, writing—review and editing.
Data availability
Data for the study is available with the authors.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
