Abstract
Objective
In this feasibility study, firstly, we aimed to assess whether intramuscular pH (IMpH) could be measured with a glass-tipped electrode. Secondly, we aimed to explore whether we could monitor changes in skeletal muscle pH before and throughout static (isometric) and after dynamic (treadmill) exercise, comparing individuals with intermittent claudication (IC) to a group of healthy controls.
Methods
Forty participants with IC (mean age ± SD) 68 ± 8) and 40 healthy volunteers (63 ± 10) were recruited. Participants in the IC group had an ankle brachial pressure index of <0.7 (mean: 0.62 ± 0.13). The first 20 participants in each group underwent isometric ankle dorsiflexion for 120 s at 30% of maximum voluntary contraction and the remainder underwent treadmill exercise (3.6 km/h, 5° incline) to maximal walking distance. A sterilised reusable 1.5 mm shatterproof, heat strengthened glass tipped pH probe inserted using a peripheral venous cannula and in combination with a pH meter was used to measure IMpH of the tibialis anterior muscle prior to, during and continuously for 1 h after exercise.
Results
Resting IMpH was similar between the groups (median 6.8 (interquartile range (IQR) 6.7-6.9)) for IC individuals and 6.7 (6.6 to 6.8) for controls). Following isometric exercise, the magnitude of the decrease in IMpH was greatest in the IC group (median ΔpH max. in patients −0.2 (IQR: −0.2 to −0.1) versus −0.1 (IQR: −0.2 to −0.1) in the control group (p = 0.002). After treadmill exercise, the magnitude of the decrease in IMpH (ΔpH max) was greatest in individuals with IC (median ΔpH max. in patients −0.3 (IQR: −0.3 to −0.3) versus 0 (IQR: −0.2 to 0) in the control group, p = 0.04.
Conclusions
IMpH monitoring with a flexible glass probe inserted into the tibialis anterior muscle is viable and changes in IMpH during static and dynamic exercises are detected.
Introduction
Tissue pH has been suggested to be an indicator of the cell's metabolic state and has been described as a useful biomarker of skeletal muscle perfusion and the severity of peripheral arterial disease (PAD).1,2 Skeletal muscle pH is maintained at around seven, primarily through adenosine triphosphate (ATP) utilisation and ATP resynthesis and the buffering capacity, in part, provided through proton transfer across the sarcolemma.
Although tissue oxygen levels are rapidly depleted after the onset of muscle ischaemia, tissue pH continues to decline over a more prolonged period of time.3,4 Monitoring muscle pH could therefore be a clinically useful biomarker regarding the metabolic status of a muscle group under observation. 1 Indwelling fibre-optic pH probes and glass-tipped pH electrodes allow continuous monitoring of tissue pH giving valuable information regarding temporal changes in intramuscular pH (IMpH) and ultimately tissue viability,5,6 and maybe a viable alternative to 31-Phosphorus Magnetic Resonance Spectroscopy, which is both costly and at times practically difficult to arrange. Furthermore, the accuracy of near-infrared spectroscopy readings is affected by large vessels and does not provide a direct measure of IMpH.7,8
In this feasibility study, firstly, we aimed to assess whether IMpH could be measured with a glass-tipped electrode. Secondly, we aimed to explore whether we could monitor changes in skeletal muscle pH before and throughout static (isometric) and after dynamic (treadmill) exercise, comparing individuals with intermittent claudication (IC) to a group of healthy controls. We hypothesised that in patients with IC, there would be a greater decline in IMpH following either dynamic or static exercise than that observed in similarly aged controls.
Methods
This is a feasibility study conducted in Aberdeen (NHS Grampian), Scotland, from January 2002 to December 2005. The reporting of this study conforms to STROBE guidelines. 9
Study participants
Forty patients with lifestyle-limiting, short-distance IC (Fontaine grade II) 10 were recruited from the vascular outpatient clinic. All participants underwent formal ankle brachial pressure index (ABPI) testing, arterial duplex scanning and/or digital subtraction angiography to confirm the diagnosis of PAD. Patients were eligible if they had stable claudication of >6 months duration, an ABPI <0.8 and no rest pain or tissue loss. All participants were on aspirin and statin therapy. Limited participant characteristics were collected in this pilot study, as the main focus of our study was to assess whether IMpH measurement was possible and secondarily, whether there was a difference in IMpH readings when comparing IC and healthy volunteers during static and following dynamic exercise. Forty similarly aged individuals, with ABPI's >0.9 and no history of PAD were also recruited. Study exclusion criteria included: concurrent treatment with steroids, clopidogrel, warfarin or non-steroidal anti-inflammatory therapy; inability to exercise due to other systemic conditions such as angina or chronic obstructive pulmonary disease; haemoglobin < 10 g/L; platelet count < 150 × 109/L; abnormal liver or renal function; suspected drug or alcohol abuse; recent blood transfusion and recent systemic illness; patients who had percutaneous transluminal angioplasty or surgery in the preceding 6 months were excluded. The study leg was the right leg for controls, and for participants with bilateral symptoms, the most symptomatic leg was nominated. Given the small scale of this pilot study, no attempt was made to match patient co-morbidities.
Exercise regimen
Study participants attended the laboratory in the morning fasting for at least 6 h and having abstained from unaccustomed physical activity for the preceding 24 h. The first 20 participants in each group performed intermittent isometric ankle dorsiflexion for 120 s at 30% maximal voluntary contraction (MVC). The second cohort of 20 participants in each group underwent treadmill (3.6 km/h, 5° incline) exercise to maximal walking distance (MWD). The isometric (static) exercise regimen was performed with the use of a dynamometer (Biodex System II, Biodex Medical Systems, New York) and treadmill (dynamic) exercise was performed on a Technogym RunXT® (Technogym UK Ltd, Bracknell, UK) treadmill.
Method for measuring IMpH
Local anaesthetic was used to infiltrate the skin overlying the tibialis anterior muscle (2 cm lateral and 4 cm distal to the tibial tuberosity) prior to the insertion of the glass-tipped probe using a standard peripheral venous cannula (see Supplemental Figure S1). Insertion of the cannula was not reported to be a painful experience by any of the participants. There were no infections, or deep hematomas, and only superficial bruising in some individuals – the procedure was well tolerated by all participants. The insertion of the cannula was akin to peripheral venous cannula insertion but with the tip of the cannula resting within muscle tissue. The probe was then attached to the IMpH monitor. In this proof-of-concept study, the tibialis anterior muscle was chosen as the probe insertion site because it is a superficial muscle that is easily identifiable with clear anatomical landmarks which allow for reproducibility of the probe insertion site and therefore standardisation across all participants. The choice of tibialis anterior was the preferred option compared with insertion of the cannula into the larger calf muscle groups in which it would be difficult to reproduce the exact position of the cannula tip. The anterior tibialis muscle allows for ease of access for continuous pH monitoring, which was of particular importance throughout both isometric and dynamic exercises. Previous studies have used the tibialis anterior muscle as the probe insertion site to monitor muscle pH changes in response to interventions.11,12
For IMpH monitoring a calibrated Flexilog 2010 Recorder in combination with a sterilised reusable 1.5 mm heat strengthened glass-tipped pH probe with an external Ag/AgCl skin reference electrode was used (Oakfield Instruments Inc., Oxon, UK). IMpH measurements were recorded prior to, during and continuously for 1 h after exercise. Continuous IMpH measurements were found to be accurate to one decimal place. 13 A two-point calibration method using standard buffers of pH 1.07 and pH 7.01 buffer (Oakfield Instruments Ltd, Oxon, UK) was deployed. Calibration was carried out within a maximum of 30 min prior to insertion.
After calibrating the pH probe, it was submerged in an equipment treatment bath containing Tristel, a medically approved sterilising solution, for 20 min. Thereafter, the probe was removed and rinsed down with sterile N-saline prior to being inserted into the tibialis anterior muscle of the affected leg under an aseptic technique. A reference electrode (electrocardiogram-type pad) was attached to the medial aspect of the participant's knee. The glass pH sensor readings stabilised quickly (within 60 to 90 s) after they had been calibrated in the buffer solution.
Statistical methods
This is a feasibility study, and as such no power calculations were performed to determine study numbers. Statistical analysis was performed using Statistical Package for the Social Sciences (SPSS) version 27 and R Studio version 4.3.2 (USA) was used to graph data. A Biostatistician was consulted for statistical planning and analysis. Patient baseline characteristics were normally distributed. However, pH data were found not to be normally distributed and as a consequence, non-parametric statistical tests were employed. The initial analysis was conducted using the Friedman test. Between time-point comparisons were performed using the Wilcoxon signed-rank test. The difference in baseline measurements between groups was assessed using the Mann–Whitney U test. Chi-squared test was used to compare differences in participant characteristics between groups and statistical significance was expressed as a 5% probability (p < 0.05).
Results
Baseline characteristics of the study participants are reported in Table 1. There was no significant difference in resting IMpH between the groups (median baseline pH was 6.8 (interquartile range (IQR): 6.7 to 6.9) for the participants with IC and 6.7 (IQR: 6.6 to 6.8) in the control group (p = 0.50). The mean ABPI in the IC group was 0.62 (SD ±0.13) vs. 1.14 (0.10) for the control participants. We observed no correlation between baseline IMpH and resting ABPI in the IC group (correlation coefficient −0.179 (Spearman's rho), p = 0.20). After baseline measurements, we did not collect any further ABPI readings.
Participant baseline characteristics.

ABPI: ankle brachial pressure index; SD: standard deviation.
Values presented are mean ± SD for continuous data and actual number (%) for categorical data.
Isometric exercise
There was no difference in the torque produced during MVC between groups (median control group MVC 26.1 Nm (IQR: 15.1 to 29.9), median IC MVC 25.3 Nm (IQR: 16.2 to 29.4), p = 0.794) and total work done was comparable between the groups (mean total work done in volunteers vs. IC; 21.4 J vs. 20.4 J).
In the IC group, during isometric exercise, a significant decline in IMpH was observed at 180 s (p = 0.004) (Tables 2 and 3; Figure 1). Thereafter, pH decreased to a minimum at 240 s (median pH 6.6 (IQR: 6.5 to 6.8)), returning to resting values at 30 min post-exercise. Whereas, in the control group, the IMpH remained relatively stable during exercise but 2 min after exercise there was a significant reduction (p = 0.022) in pH to a minimum of 6.6 (IQR: 6.4 to 6.7). In contrast to the IC group, pH returned to resting levels 5 min after exercise and there was a further increase above baseline at 15 min (p = 0.048) (Figure 1). At 1 h post-exercise, pH was still elevated above resting values (p = 0.005). The magnitude of the decrease in IMpH was greatest (p = 0.002) in the IC group following isometric exercise (ΔpH max −0.2, IQR: −0.2 to −0.1) when compared with the healthy controls (ΔpH max −0.1, IQR: −0.2 to −0.1).

Intramuscular pH during isometric exercise in patients with intermittent claudication versus healthy volunteers.
IMpH of healthy volunteers in the static exercise group at set time points (n = 20).

IMpH of patients with IC in the static exercise group at set time points (n = 20).

IMpH: intramuscular pH; IC: intermittent claudication.
Dynamic treadmill exercise
The effect of movement during exercise resulted in significant recording artefacts of IMpH. Therefore, pH was recorded at baseline and then for 60 min immediately after the subject stepped off the treadmill. Immediately following exercising to their MWD, participants with IC had a reduction in pH from the resting value pH 6.8 to 6.5 (IQR: 6.3 to 6.6), p > 0.001), Wilcoxon signed rank test, Friedman test, p > 0.05). The IMpH remained depressed at 6.5 post-exercise for up to 30 min and thereafter returned to the baseline value (Tables 4 and 5; Figure 2).
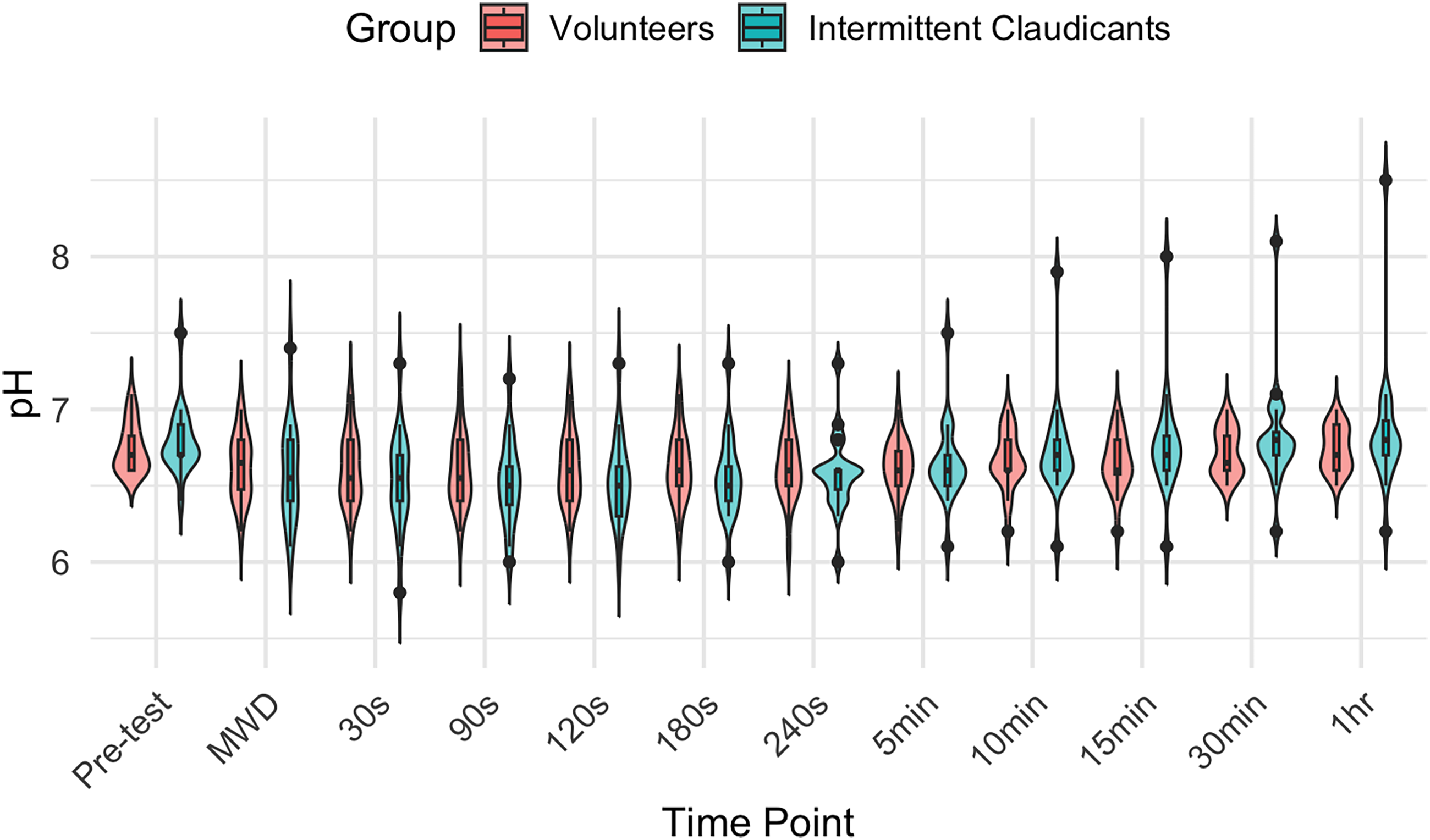
Intramuscular pH during treadmill exercise in patients with intermittent claudication versus healthy volunteers.
IMpH of healthy volunteers in the treadmill exercise group at set time points (n = 20).

IMpH: intramuscular pH; MWD: maximal walking distance.
IMpH of patients with IC in the treadmill group at set time points (n = 20).
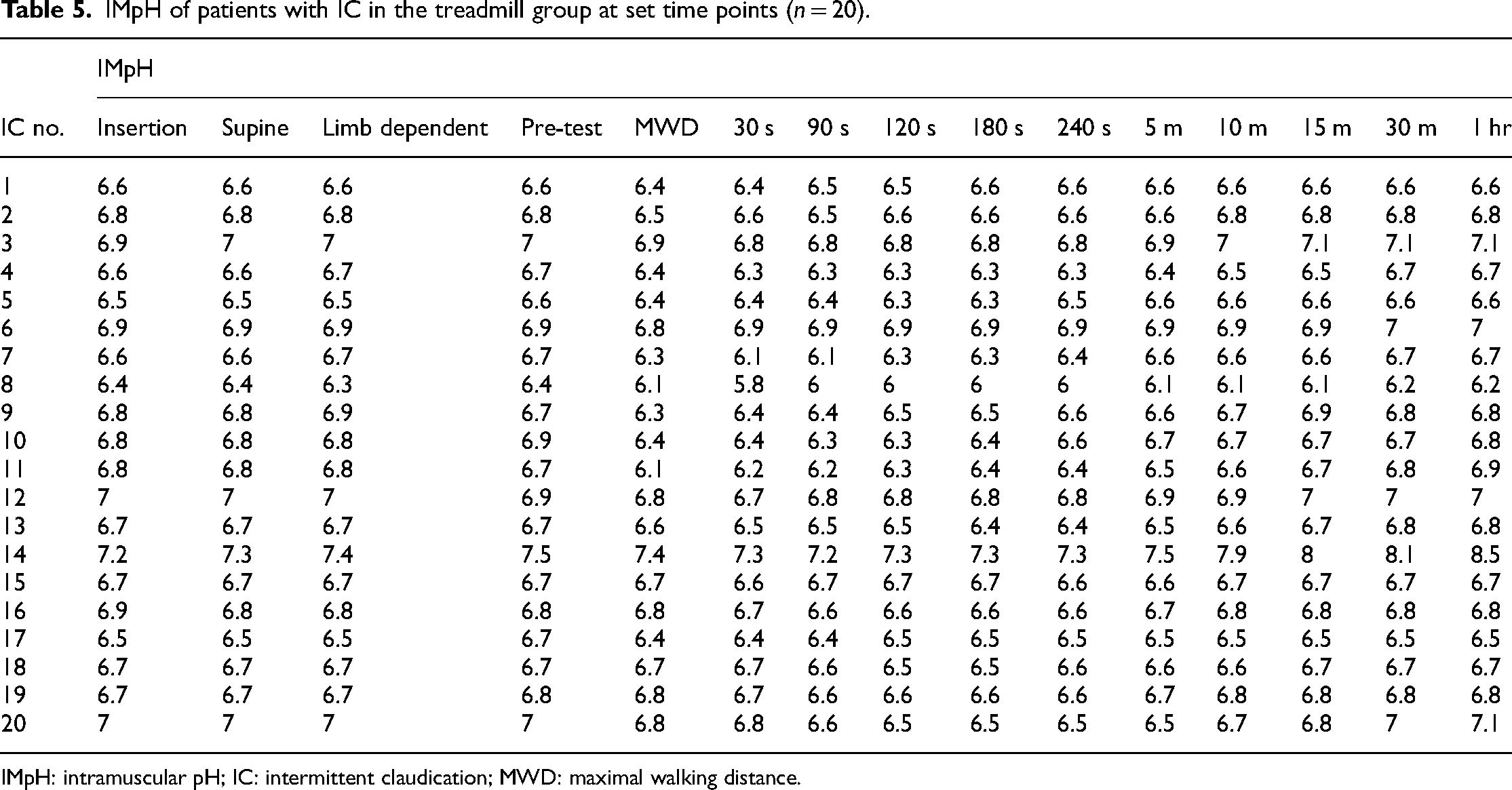
IMpH: intramuscular pH; IC: intermittent claudication; MWD: maximal walking distance.
Post-exercise the control group was also observed to experience a decline in IMpH (pH 6.6 (IQR: 6.4 to 6.8), p < 0.001) (Figure 2). When comparing the magnitude of the change in IMpH (ΔpH max) it was found that this was greatest in the participants with IC when compared with the control volunteers (ΔpH max. in patients −0.3 (IQR: −0.3 to −0.3) vs. 0 (IQR: −0.2 to 0) in the control group, p = 0.04).
Discussion
In this feasibility study, we observed that the resting IMpH was not different in patients with IC when compared with healthy age-matched controls and no correlation was observed between resting ABPI and IMpH. Following both isometric and treadmill exercise, the rate and extent of decline in IMpH was greater in participants with IC. A reduction in muscle perfusion and thus greater reliance on substrate-level phosphorylation may account for the greater post-exercise acidosis. The magnitude of reduction in IMpH was similar following both static and dynamic exercise. Furthermore, recovery of IMpH to baseline values took longer in participants with IC.
The differences in metabolic response to exercise between the IC and control group are indicative of an increased reliance on anaerobic metabolism due to an impairment in oxygen delivery. Based on circumstantial evidence, occlusion of the circulation to an active muscle group has been demonstrated to exert the greatest effect on type I fibres. 14 Using an isometric mode of exercise these authors reported that while the glycogenolytic rate in type II fibres remained the same, irrespective of whether there was intact or occluded circulation, a 10-fold increase in glycolytic rate (0.18 mmol/kg/s to 2.05 mm/kg/s) was observed in the type I fibre population. A similar metabolic response to dynamic exercise has been reported by Krustrup et al., 15 when examining the effect of circulatory occlusion during moderate/low-intensity exercise on muscle fibre recruitment. These authors reported that moderate exercise with unrestricted circulation resulted in an 18% decrease in phosphocreatine concentration, however, with circulatory arrest the phosphocreatine concentration was reduced by 85% in type I fibres. Collectively, the results from these two studies indicate that circulatory occlusion increases the metabolic stress in muscle by an increased reliance on phosphocreatine and carbohydrate metabolism. Occlusion also results in a shift in fibre-type recruitment towards the increasing activity of slow twitch fibres. 15 In the current study maintaining the equivalent power/force output produced by the control group, the patients with IC appear to have a greater activity of oxidative muscle fibres and have a greater reliance on anaerobic metabolism resulting in a greater degree of acidosis.
The current study is one of only a few to utilise a glass-tipped flexible pH probe to measure serial changes in IMpH following static and dynamic exercises. These findings support the results of other studies,16–20 which have shown that inadequate tissue oxygenation even at a relatively low workload results in an increased reliance on substrate-level phosphorylation. 21 However, unlike previous studies that have estimated IMpH utilising spectra shift during nuclear magnetic resonance/magnetic resonance spectroscopy22–25 or based on the changes in lactate and pyruvate, 26 we were able to directly and continuously record pH changes immediately following exercise. Changes in pH of 0.1 unit represented a well-defined change in the concentration of H+ ions due to how pH was calculated (pH = −log [H+]).
The recovery in pH post-exercise is a well-documented process and an increased recovery time for pH in participants with IC may be a consequence of an impaired perfusion rate post-exercise, thus reducing the rate of removal of lactate and H+ by the venous circulation.27,28 An interesting observation was that the rate of recovery of pH was faster in the IC participant group following treadmill exercise compared with the isometric exercise. The opposite effect was seen in the controls with post-exercise pH recovery taking longer following treadmill exercise. The explanation for these results may be due to the magnitude of post-exercise hyperaemia between exercise modalities and the overall lower venous compliance in the IC patient population. Circumstantial evidence using flow-mediated dilatation in patients with PAD supports the proposal that endothelial dysfunction in IC patients will attenuate the vasodilatory response to shear stress as a consequence of exercise. 29 A further interesting observation is the increase in muscle pH above resting values at 15 min post-isometric (static) exercise in healthy volunteers, which may be explained by post-exercise hyperaemia leading to a greater rate of hydrogen ion clearance and higher pH. 30
Perspectives
The use of IMpH as an indicator of adaptation to a training stimulus in IC patients is supported by data from studies examining the effects of training programmes and the metabolic response to an exercise challenge.31–34 The universal finding from these studies is that following training there is a reduced glycolytic flux, pyruvate and lactate accumulation and consequently maintenance of intracellular pH. Supervised exercise therapy remains a central component in the treatment of IC, which has been shown to improve symptoms and slow disease progression.34,35 Currently, progress during these programmes is typically measured as a combination of a patient's subjective pain reports in conjunction with their achieved walking distances. 36 However, currently the paucity of a standard objective measure of skeletal muscle ischaemia in clinical practice makes it difficult to evaluate disease progression and response to interventions such as medications and physiotherapy. We propose that IMpH could be used at fixed time points; initial assessment, following a course of therapy and exercise stress testing due to the similarity in resting pH between the IC and control group. The use of continuous IMpH measurement at fixed time points would allow for meaningful comparisons over time whilst also allowing clinicians to better tailor patient-centred therapies.
Strengths and limitations
The first 20 participants from each group either completed static or dynamic exercise, therefore, we were unable to compare pH changes across exercise regimens. While it would have been ideal to have participants complete both exercise modalities, experimental and ethical requirements prevented such a design. However, in order to limit the biological variability, participants were similarly aged. In order to avoid the confounding effects of an earlier onset of fatigue in IC patients, efforts were made to standardise the exercise intensity and duration in both participant groups. We were unable to measure IMpH during dynamic treadmill exercise due to the interference produced by movement. However, we were able to measure pH changes immediately after the participant had stepped off of the treadmill. While it is likely that our readings are measuring extracellular pH, we cannot be certain that other tissue compartments, including interstitial and intracellular were not being measured. Measures were taken to account for limb position and the impact of blood flow and subsequent tissue perfusion.37,38 Resting pH values measured with the subject supine were no different from resting values measured with the limb in a standard dependent position prior to isometric exercise. Data on the distribution of the arterial lesions in the claudicants was not collected, future studies should include this data. Strengths of the study include the sensitivity of the IMpH glass probe, which was able to detect pH changes to one decimal place. 13 A power analysis was not completed for this study, however, given that this is a feasibility study, our aim was a proof-of-concept.
Conclusion
This current study demonstrated that serial IMpH monitoring is feasible and changes in pH, as a consequence of exercise and recovery, can be monitored in real time. IMpH measurement may provide an objective measurement of the severity of exercise-induced ischaemia during supervised exercise programmes. In turn, the method also provides an objective measurement that may be used to chart progression and allow for better-tailored patient-centred exercise programmes. However, future research is required to further investigate its utility in the clinical environment.
Supplemental Material
sj-docx-1-sci-10.1177_00368504241292126 - Supplemental material for Intramuscular pH monitoring before and during exercise in individuals with intermittent claudication compared to healthy controls: A feasibility study
Supplemental material, sj-docx-1-sci-10.1177_00368504241292126 for Intramuscular pH monitoring before and during exercise in individuals with intermittent claudication compared to healthy controls: A feasibility study by Andrew D. Ablett, Patrick Collins, Derek Ball, Julie Brittenden and Alan J. Johnstone in Science Progress
Supplemental Material
sj-docx-2-sci-10.1177_00368504241292126 - Supplemental material for Intramuscular pH monitoring before and during exercise in individuals with intermittent claudication compared to healthy controls: A feasibility study
Supplemental material, sj-docx-2-sci-10.1177_00368504241292126 for Intramuscular pH monitoring before and during exercise in individuals with intermittent claudication compared to healthy controls: A feasibility study by Andrew D. Ablett, Patrick Collins, Derek Ball, Julie Brittenden and Alan J. Johnstone in Science Progress
Footnotes
Acknowledgements
We would like to thank Professor J. Brittenden and Professor A. Johnstone for their dedication to this project. Mr E. Macauley, Mr G.G. Cooper, Mr K. Cassar and Mr P. Bachoo, Consultant Vascular Surgeons. Dr I. Ford, Department of Medicine and Therapeutics, Dr B. Croal, Clinical Biochemist and Dr D. Ball, Senior Lecturer in Human Physiology who co-authored the grant proposals, organised and supervised my laboratory efforts.
Author contributions
ADA was primarily involved with manuscript writing and data analysis. PC and AJ designed the study, data analysis and aided with manuscript writing. DB collected data and was involved with manuscript writing. JB was involved with manuscript writing and data collection.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval was obtained from the Grampian Research Ethics Committee (No. 00/0028). All potential individuals were given written information about the study and those who agreed to participate provided written informed consent.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Scottish Enterprise (grant number 00/0028).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
