Abstract
Objective:
Serum albumin (ALB) plays a vital role in maintaining oncotic pressure and contributing to hemodynamic stability, with low levels associated with adverse outcomes in critically ill patients. This study aimed to assess the association between serum ALB concentrations and poor outcomes and the possible benefits of ALB supplementation.
Methods:
A retrospective study involving 300 intensive care unit (ICU) patients. Albumin levels were recorded upon admission and throughout the stay, and patients were categorized based on a cutoff of 2.49 g/dl. The associations between low ALB levels and mortality were assessed using regression analysis. Additionally, it investigated the association of albumin supplementation with patient outcomes and mortality in specific patient populations.
Results:
The mean age was 54.9 years, with 68% having sepsis. Patients with low baseline ALB concentrations exhibited higher overall mortality (71% vs. 52%) and 28-day mortality (50% vs. 39%). Adjusted analyses confirmed associations with mortality. Albumin supplementation was administered to 53% of the patients. Its use demonstrated potential benefits, particularly in reducing mortality, when given to specific groups, such as sepsis and hypoalbuminemia patients.
Discussion:
The study confirms that low serum albumin levels are strongly associated with higher mortality rates in ICU patients. Albumin supplementation showed potential benefits, particularly in patients with sepsis and low albumin levels. Further analyses explored the dosage–response relationship and identified potential groups that would benefit from albumin supplementation.
Conclusion:
Albumin can play a major role in predicting mortality in critically ill patients. Moreover, ALB supplementation may improve survival, especially in resource-limited settings. Future research should refine protocols through clinical trials for optimal survival in critically ill patients.
Introduction
Albumin (ALB) is a crucial protein with various important functions in the human body. It is responsible for maintaining oncotic pressure, as it affects microvascular permeability and subsequently contributes to hemodynamic stability. 1 Moreover, ALB exhibits antioxidant and antiinflammatory properties, maintaining endothelial stability, as it can bind with free fatty acids, preventing lipid peroxidation damage and eliminating external toxic materials by combining them, ultimately reducing endothelial damage.2,3
Additionally, ALB levels reflect patients’ nutritional state and organ function. 4 Moreover, it is considered a biomarker of severity in acute stress, such as sepsis, 1 and monitoring its levels can serve as a predictor of outcomes in critically ill patients. Numerous studies have noted a correlation between low ALB levels and unfavorable outcomes in critically ill patients, including higher mortality rates. 4 The pathophysiology of low ALB levels in critical illness patients is attributed to impairment of the vascular endothelium and increased capillary permeability, which leads to ALB redistribution between vascular and interstitial spaces. This, along with nutritional deficiencies, eventually causes a decrease in the serum ALB concentration. Furthermore, hepatic ALB synthesis is impaired during acute stress. 5
Despite the recognized importance of ALB, several aspects, such as the specific cutoff for low ALB levels, that is associated with mortality and other poor outcomes in different groups of critically ill patients, remain unclear, as is the role of ALB supplementation in these patients across different conditions. Although many previous studies have shown a negative correlation between the serum ALB concentration and clinical outcomes in critically ill patients, most of these studies have relied on a single ALB reading, 5 since ALB levels are influenced by acute stress, nutritional deficiencies, and other factors, such as fluid resuscitation, it is increasing the reliability of continuous ALB monitoring in predicting outcomes. 1 Moreover, there is uncertainty about which patient groups, if any, would benefit from ALB therapy and at what cutoff of ALB levels.
Previous studies have assessed the effect of human ALB in treating liver cirrhosis patients, and fewer studies have assessed the effect of human ALB in treating sepsis and septic shock patients. However, very few studies have assessed the use of human ALB in other critically ill groups. 6 Randomized clinical trials evaluating the therapeutic effects of various fluid resuscitation therapies on sepsis have consistently concluded that crystalloids and human ALB are the most beneficial agents. 7 A meta-analysis showed that ALB treatment, particularly 20% ALB, significantly reduced 90-day mortality in septic shock patients. 7 However, whether mortality beyond fluid resuscitation, as in critically ill patients and patients with hypoalbuminemia, may reduce mortality is still unclear. Although ALB supplementation has no particular side effects if given to any particular group, 6 patients receiving ALB therapy in resource-limited settings are hindered by its high costs. It is necessary to determine what patient groups could benefit from ALB therapy.
This research aimed to address these uncertainties, as it sought to determine the cutoff ALB concentration that is associated with worse outcomes and subsequently be used as a predictor of mortality and outcomes in the intensive care unit (ICU) and to identify patients who may benefit from ALB supplementation, providing potential guidance for the initiation of ALB supplementation.
Methods
Study design
This study adopted a retrospective design in which the data were collected through hospital electronic records that were reviewed from 2019 to 2023.
Setting and population
All ICU patients who were treated at An-Najah National University Hospital (NNUH) were consecutively selected. Patients under 18 years of age, patients with missing ALB levels for more than 50% of the days, patients with less than 72 h of ICU stay, and patients who were referred from another ICU were excluded.
Sample size
A minimum of 276 patients needed to be recruited for 80% power, considering a two-sided significance of 5% and a 95% confidence interval (CI), with an estimated hypoalbuminemia incidence based on the literature ranging from 40% to 50%.2,5
Data extraction and variables
The data collected included demographic data (such as age, sex, and body mass index), past medical history, reason for hospital and ICU admission, duration of hospital and ICU stay, condition upon ICU admission as whether sepsis was present, and source of sepsis, if shock and type of shock occurred, respiratory failure, mechanical ventilation, disease severity score at baseline (using the Sequential Organ Failure Assessment (SOFA) score) and baseline laboratory results at ICU admission. Moreover, the ALB-related data included daily ALB levels from the day of ICU admission to the 30-day follow-up, and if the patient received ALB supplementation, given the rationale and frequency of supplementation. Primary outcomes were overall mortality and 28-day mortality. While secondary outcomes were related to the occurrence of hemodynamic instability, respiratory failure, or acute kidney injury, for which neither was present upon ICU admission.
Albumin measurement in our hospital is made from peripheral venous blood samples that were collected from patients and stored in tubes with inert separation gel and coagulant. Following centrifugation, serum was obtained and albumin levels were measured using bromocresol green colorimetry with albumin test kits from Roche Diagnostics GmbH, Mannheim, Germany.
The study was conducted in accordance with the Helsinki Declaration and approved by the An-Najah National University Ethics Committee. Reference: MED.OCT.2023/113. Furthermore, consent form is waived for this study as it is a retrospective study based on electronic records.
In accordance with ethical guidelines, all patient details in this study have been deidentified to protect confidentiality. Personal identifiers such as names, medical record numbers, and specific demographic information have been removed or altered to prevent the identification of individual patients.
The reporting of this study conforms to STROBE guidelines. 8
Definitions
A low serum ALB concentration was defined as an album in a concentration less than the prespecified cutoff based on the analysis results. ALB supplementation was categorized based on its indication: hypoalbuminemia, the presence of sepsis, or other causes not applicable to any of the former causes. The duration of ALB supplementation is expressed as a fraction of the total duration by dividing the number of days given ALB by the total duration of ICU stay.
Overall mortality was defined as mortality occurring from admission until death or up to one month postdischarge, whichever occurred first.
Septic shock was defined according to the sepsis campaign guideline as persistent hypotension despite adequate fluid resuscitation. 9 Acute kidney injury was defined according to the kidney disease: improving global outcomes definition. 10
Statistical analysis
All variables were analyzed using the Statistical Package for the Social Sciences version 29. Descriptive analysis was performed to express baseline characteristics and laboratory values as either the mean ± standard deviation (SD) or percentage. The mean, minimum, and baseline ALB concentrations were calculated, and their associations with patient outcomes were analyzed via univariate analysis, the independent t-test, or the chi-square test. Multivariate analysis was conducted based on the results from univariate analysis and the literature to determine the appropriate models using binary logistic regression to adjust for confounders. Moreover, a cutoff value for the serum ALB concentration was determined using receiver operating characteristic (ROC) curve analysis. Furthermore, the association between the serum ALB supplementation and patient outcome was assessed for each category (hypoalbuminemia, and sepsis patients). A p-value less than .05 was considered to indicate statistical significance.
Results
There were 460 adult patients in the NNUH medical ICU between 2019 and 2023. Among them, 160 patients were excluded because of a short ICU stay of less than 72 h, missing ALB values, or referral from different ICUs. Of the remaining 300 enrolled participants, the mean age was 54.9 ± 18.2 years, the majority of patients were male (60%), and approximately 47% were hemato-oncology patients. The most common causes at admission were respiratory failure (29%), mostly pneumonia; sepsis/septic shock (27%); cardiogenic shock (4%); and hemorrhagic shock (3%) (Table 1).
Baseline demographic, hospitalization-related, disease severity and biochemical data, and comparisons between patients with hypoalbuminemia and patients with normal albumin levels.

Note: SD: standard deviation; ESRD: end-stage renal disease; ICU: intensive care unit; COPD: chronic obstructive pulmonary disease; SOFA: Sequential Organ Failure Assessment; WBC: white blood cell; HGB: hemoglobin; CRP: C-reactive protein; BMI: body mass index.
Missing values in this variable were approximately 50%.
The albumin cutoff used to form the two groups was 2.49 g/dl.
Participants were categorized according to their baseline ALB levels as low or normal. The ALB cutoff was calculated through ROC curve analysis, which yielded an area under the ROC curve of 0.62 (95% CI [0.55–0.68]; p = .001). The cutoff value of the serum ALB concentration was determined to be 2.49 g/dl, with a specificity of 0.59 and a sensitivity of 0.74 (Figure 1). Baseline characteristics were compared between these two groups, with similar values observed in terms of demographic characteristics. However, an expected result was that hemato-oncology patients and those with metabolic acidosis had lower ALB levels. However, the association was not significant for the presence of chronic kidney disease, heart failure, or liver cirrhosis. Disease severity at baseline was not significantly different between the two groups, as the mean SOFA scores were 7.61 ± 3.98 and 7.20 ± 4 for hypoalbuminemia patients and patients with a normal serum ALB concentration, respectively (Table 1).

Receiver operating characteristic (ROC) curve for baseline ALB concentration and mortality.
The main outcomes of the study (mortality and 28-day mortality) were compared between patients with low and normal ALB concentrations, which yielded significantly greater mortality among hypoalbuminemia patients for both overall (71% vs. 52%) and 28-day mortality (50% vs. 39%), p-value < .05. Adjustments for mortality confounders considered were as follows: age, diabetes status, hypertension, heart failure, chronic kidney disease, end-stage renal disease, liver cirrhosis, malignancy, baseline SOFA score, and the presence of sepsis, shock, respiratory failure, acute kidney injury or need for ventilation at admission. Further outcomes were assessed, and the results showed that the incidence of acute kidney injury, metabolic acidosis, and acute liver injury was greater among patients with a low ALB concentration after adjustment for confounders (Table 2).
Mortality and other clinical outcomes in patients with low versus normal albumin levels.
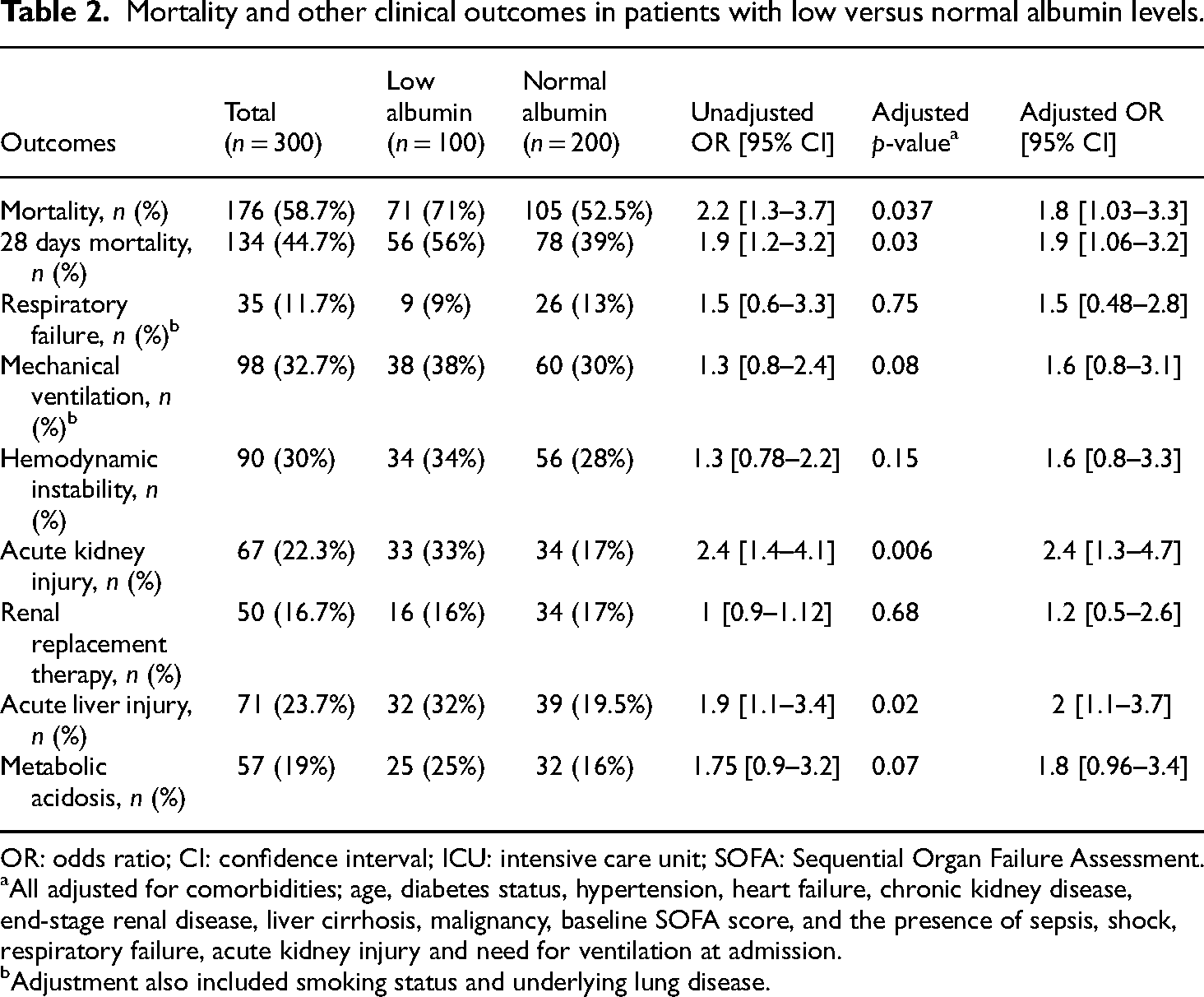
OR: odds ratio; CI: confidence interval; ICU: intensive care unit; SOFA: Sequential Organ Failure Assessment.
All adjusted for comorbidities; age, diabetes status, hypertension, heart failure, chronic kidney disease, end-stage renal disease, liver cirrhosis, malignancy, baseline SOFA score, and the presence of sepsis, shock, respiratory failure, acute kidney injury and need for ventilation at admission.
Adjustment also included smoking status and underlying lung disease.
The trends in the serum ALB concentration over time and mortality were also studied and are presented as the baseline, mean, and minimum values of the serum ALB concentration. The association with mortality was significant with inverse relation, and after adjustment for mortality confounders, the association was significant when comparing with the mean ALB level (OR, 95% CI 2.3 [1.28–4]) and the baseline value (OR, 95% CI [1.3–3.1), indicating that patients with lower ALB levels had greater mortality (Table 3).
Multivariate analysis of the factors associated with low serum albumin concentration and mortality.

OR: odds ratio; CI: confidence interval; SD: standard deviation; SOFA: Sequential Organ Failure Assessment.
Adjusted for comorbidities: age, diabetes status, hypertension, heart failure, chronic kidney disease, end-stage renal disease, liver cirrhosis, malignancy, baseline SOFA score, and the presence of sepsis, shock, respiratory failure, acute kidney injury and need for ventilation at admission.
Approximately 53% of patients were given ALB infusion at some point. The common causes for albumin supplementation were usually a low ALB concentration (18%), shock (mostly septic) (42%), or combinations of both (27%). The mean number of days of ALB supplementation was 4 ± 6 days, and the mean number of fraction of ALB supplementation days (the number of days given ALB to the total duration of ICU stay) was 0.23 ± 0.29. Patients were categorized according to the ALB supplementation fraction: those who were given more than 25% and those who were given more than 50% of their ICU days. Approximately 67% had ALB infusion at least 25% of ICU days which means almost every 2–3 days, while 35% had it for at least 50% of ICU days which is almost every other day, while less than 18% had it for more than 75% of days.
Subgroup analysis was performed to compare mortality outcomes for patients who received ALB infusion; patients were categorized based on the above fraction and based on the serum ALB concentration, and patients who were diagnosed with sepsis or malignancy, as shown in Table 4. Given ALB infusion in general was not significant in reducing mortality among hypoalbuminemia patients and sepsis patients. While once administering ALB supplementation for at least 25% of the days significantly decreased mortality among patients with malignancies and sepsis (OR, 95% CI; p-value: 8.7 [2.4–32], 0.001, and 2.6 [1.2–5.5], 0.016, respectively), but was not significantly associated with mortality reduction among hypoalbuminemia patients, but was significant in this group when given at least 50% of ICU days (OR, 95% CI, p-value = 1.8 [0.5–6.3], 0.37).
Multivariate analysis of the association between albumin infusion and patient outcomes.
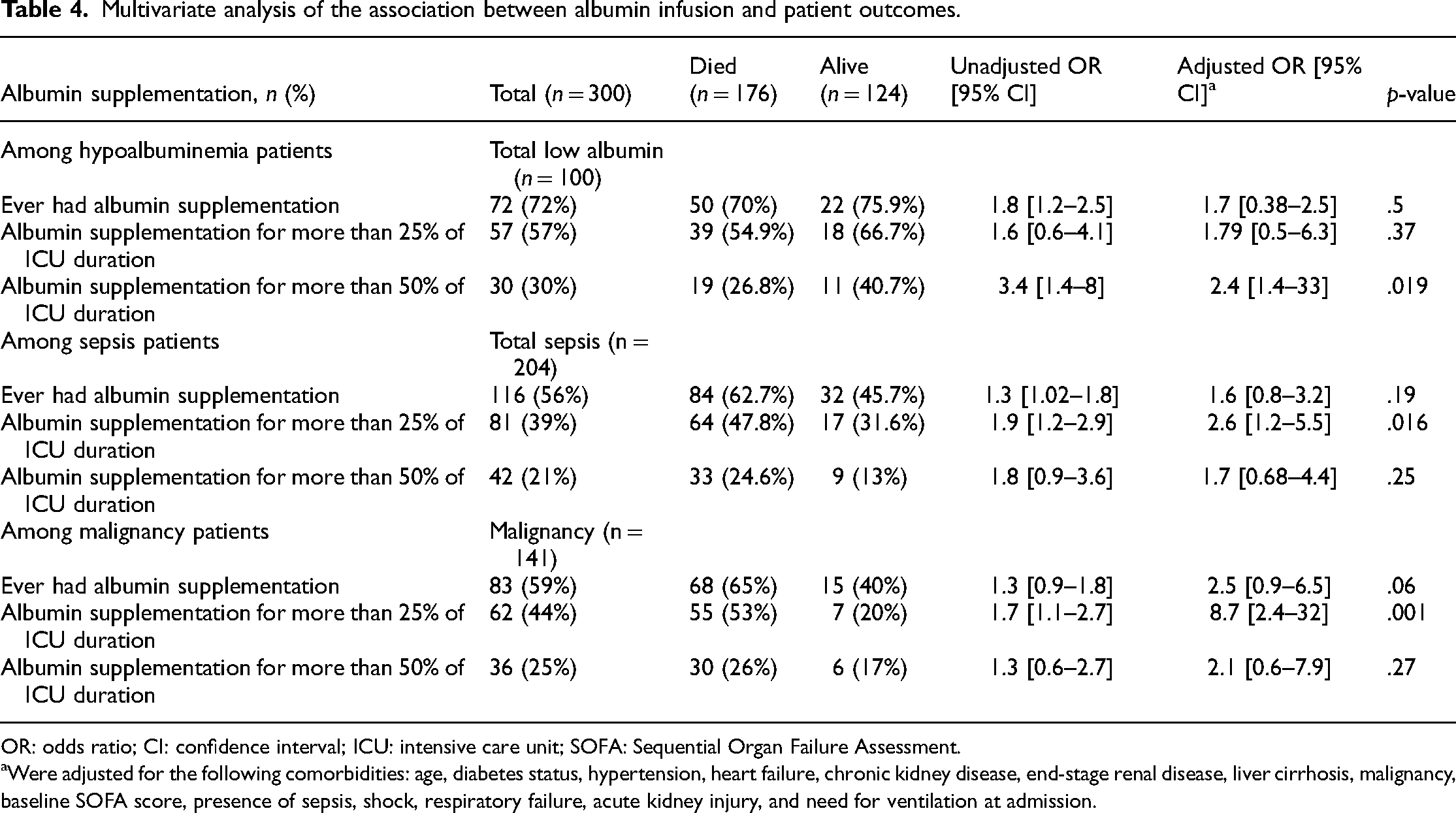
OR: odds ratio; CI: confidence interval; ICU: intensive care unit; SOFA: Sequential Organ Failure Assessment.
Were adjusted for the following comorbidities: age, diabetes status, hypertension, heart failure, chronic kidney disease, end-stage renal disease, liver cirrhosis, malignancy, baseline SOFA score, presence of sepsis, shock, respiratory failure, acute kidney injury, and need for ventilation at admission.
Generally, patients treated with albumin supplements were less likely to develop acute kidney injury and septic shock and less prone to need mechanical ventilation for both the 25% and the 50% fractions, as illustrated in Figure 2. The differences were significant for patients who developed septic shock and needed ventilation, once given albumin infusion for at least 25% of the days (p-value <.05). The remaining associations were nonsignificant (Table 5).

(a) Outcome percentages among patients with low albumin levels. (b) Outcomes among patients with low albumin levels who were not given albumin supplements compared to those who were given a more than 25% stay and those given a more than 50% stay.
Clinical outcomes of patients with low ALB levels who were given albumin supplementation.

OR: odds ratio; CI: confidence interval; ICU: intensive care unit.
All adjusted for comorbidities; age, diabetes status, hypertension, heart failure, chronic kidney disease, end-stage renal disease, liver cirrhosis, malignancy, baseline SOFA score, and the presence of sepsis, shock, respiratory failure, acute kidney injury and need for ventilation at admission.
Adjustment also included smoking status and underlying lung disease.
Discussion
In this study of 300 adult ICU patients, the findings revealed several significant associations related to albumin levels and patient outcomes. Hypoalbuminemia was notably linked with higher overall mortality rates (71% vs. 52%) and increased 28-day mortality (50% vs. 39%), even after adjusting for age, comorbidities, and disease severity. Patients with low serum albumin levels also exhibited heightened risks of acute kidney injury, metabolic acidosis, and acute liver injury. Analysis of albumin infusion, administered to 53% of patients primarily for low albumin or shock, demonstrated varied effects on mortality outcomes, showing potential benefits when used intensively in patients with malignancies and sepsis.
Previous studies have investigated the association between mortality and serum ALB concentrations, and their results have varied due to the diversity of their populations and the variation in definitions of hypoalbuminemia. For the latter, it was suggested that a local cutoff should be employed for each population, as this would affect the interpretation. 11 In the literature, the calculated cutoff values were mostly close to the 2.5 g/dl cutoff chosen in this study1,2,4; a higher value of 3.0 g/L was reported by Jin et al. 5 ; and the lowest cutoff was used in Atrash et al.'s cohort, which was 1.85 g/dl. 11 The findings subsequently varied in the literature, but in terms of mortality, most studies have shown a significant association between low ALB levels and higher rates of mortality, as our findings suggested, supporting that the ALB concentration is a useful biomarker for predicting mortality.2,4 Studies such as Cao et al. and Qian et al. evaluated only the serum ALB concentration at ICU admission.2,4 Recognizing that analyzing the trend in the serum ALB concentration may offer greater reliability in predicting mortality, our study demonstrated a significant association with mortality for both the mean ALB concentration throughout the stay and the minimum value. Conversely, Atrash et al. did not observe a significant association when examining trends in the serum ALB concentration over 48 h. This discrepancy could be attributed to the low serum ALB concentrations at admission and limited statistical power, as approximately 40% of patients lacked follow-up data. 11 Nevertheless, McCluskey et al. and Kendall et al. revealed a significant association between baseline and serial ALB levels, aligning with our findings.1,12 For instance, in the study by Kendall et al., a similar cutoff was used (2.45 g/dl), and the sensitivity and specificity were 0.72 and 0.57, respectively. Their findings revealed that among subjects with ALB values above the cutoff, the mortality rate was 17%, while those whose serum ALB concentration trended below the cutoff had a higher mortality rate of 35%, and when the admission ALB concentration was below the cutoff, the mortality rate was 30%. Those with a minimum ALB concentration below the cutoff had the highest mortality rate at 41%, with all differences being statistically significant. 1 Their study included only sepsis patients, while we assessed general critically ill patients, including sepsis patients; moreover, a subgroup analysis was performed, and sepsis patients were found to be an independent factor if only sepsis patients were accounted for.
ALB levels have been studied in different clinical settings and proven to be a predictor of poor outcomes, such as among patients with sepsis,1,4,13,14 septic shock, 2 critically ill patients in general,5,11 and patients with solid tumors. 15 This study combined data from different populations of sepsis patients and septic shock patients, oncology patients, and patients with kidney injury, indicating that the serum ALB concentration is an independent factor for mortality even in different clinical scenarios. A similar study conducted by Ghimire et al. among critically ill patients found comparable trends. In the survivor group, 54.23% of patients had normal mean albumin levels, compared to only 36.8% in the nonsurvivor group. On day 1, the average albumin level was 3.39 ± 0.36 g/dL overall. Survivors averaged 3.42 ± 0.35 g/dL, while nonsurvivors averaged 3.30 ± 0.40 g/dL. Survivors’ albumin levels declined by 0.109 g/dL over five days, whereas nonsurvivors experienced a decline of 0.512 g/dL over the same period. 16
Secondary outcomes were also investigated in this study; the results revealed significant differences between patients with low ALB concentrations and those with normal ALB concentrations in terms of acute kidney injury, acute liver injury, and the need for ventilatory support. Similarly, for ventilatory support, a difference was observed in Qian et al.'s study among sepsis patients and another study of hospitalized patients in general that showed less ventilatory need and a lower incidence of respiratory failure.2,17 Additionally, in our study, patients who received ALB supplementation had a significantly lower incidence of ventilatory support needs, indicating a possible correlation. While in the study by Ghimire et al., no significant associations were found between SA levels and the need for mechanical ventilation, inotropes, or the mean duration of hospital stay. 16
A recent meta-analysis of ALB infusion showed diverse effects across different clinical outcomes. 6 In critically ill patients, ALB infusion outperformed crystalloids in stabilizing resuscitation; however, no significant impact on mortality was observed. However, among septic patients, there was a significant reduction in mortality. Moreover, a meta-analysis also evaluated the combination of human ALB with diuretics to enhance the effects of these agents, yet the response was heterogeneous. Since evidence about hypoalbuminemia was lacking in recent studies, this meta-analysis had no conclusions in this regard. In contrast, our study focused on ALB infusion in patients with low albumin levels and involved the use of the ALB days fraction from ICU stay, which is used for cost-related issues, considering the low-resource setting in this study, it was concluded that we need ALB infusion every other day to presumably reduce mortality among patients with low ALB levels; however, when accounting for sepsis patients, ALB was found to be a significant when given in less frequent infusion rate. However, a randomized trial is needed to verify these findings.
The saline versus Albumin Fluid Evaluation study showed that albumin had a stronger volume effect compared to normal saline (0.9%) in critically ill patients, especially those with hypoalbuminemia. 18 The Volume Replacement with ALB in Severe Sepsis study also emphasized this, as ALB administration, according to ALB levels, resulted in higher mean arterial pressure and lower net fluid balance than did normal saline in sepsis patients. 19 However, these findings were related to hemodynamic responses, and neither study showed a significant association between ALB infusion use and mortality in critically ill patients, contrary to our study in which the main outcome was mortality efficacy, which was significant according to the serum ALB concentration. Although the hemodynamic response was not studied exclusively, the incidence of septic shock appeared to be indifferent among the general population or sepsis patients when given ALB, but was significantly lower among patients with hypoalbuminemia, indicating that this effect relies on the serum ALB concentration, necessitating further randomized controlled trials to assess both mortality and fluid resuscitation therapy using human ALB with ALB level stratifications, focusing on the use of ALB infusion for the purpose of substitution, as no previous studies had data about this topic.
This study has several limitations. First, the retrospective design was based on electronic data, which can lead to confounders and bias. Secondly, because of the small sample size, although statistical power was achieved, a larger sample size would minimize type two errors. Moreover, the chosen cutoff value of 2.49 g/dl had high specificity but low sensitivity. Additionally, the serum ALB concentration was not measured for all of the patients on a daily basis; for that purpose, we excluded patients whose values were missing for less than 50% of the days. ALB levels and supplementation before admission to the ICU were not reported. Due to the cost of ALB supplementation, the hospital policy is restricted by infusion, as mostly being given to patients with severe hypoalbuminemia and patients with shock, but not routinely, which makes the analysis prone to bias, we tried to categorize patients according to these causes with adjustment to minimize bias and confounders. Furthermore, secondary outcomes, such as acute kidney injury, were not studied in depth for potential confounders, such as nephrotoxic medications. Additionally, the use of ALB for resuscitation was not assessed, as we focused on mortality as the main outcome and considered all potential confounders. For mortality outcome, as follow-up time considered to month follow up, for having most patients had outpatient follow-ups one month after discharge, challenges in follow-up compliance led to a significant number being lost to follow-up upon discharge. This poses a risk of follow-up bias, as mortality outside the hospital may go unrecorded, which underestimates mortality rates. To minimize this, we implemented rigorous documentation for discharged patients and adjusted our analysis to account for potential loss of follow-up. These steps were necessary to ensure the accuracy and reliability of our findings regarding mortality duration.
Conclusion
Low serum ALB levels, with a cutoff of 2.49 g/dl, were associated with poor clinical outcomes, particularly increased mortality, among patients with different critical illnesses. This finding delineates its significance as an outcome predictor, especially in resource-limited centers. Furthermore, supplementing with ALB may positively affect mortality, especially when it is administered to patients with low ALB levels and those with sepsis. However, the clinical use of ALB in the ICU remains controversial due to the advantages and disadvantages observed in different patient populations. This highlights the need for future research to assess these associations and guide further investigations in this area.
Footnotes
Acknowledgments
We gratefully thank Dr Sa'ed H Zyoud, Department of Clinical and Community Pharmacy, An-Najah National University, for his contributions in reviewing the manuscript, along with his insightful comments. Also, we thank the intensive care team at An-Najah National University Hospital for helping in the data collection and the needed data.
Author contributions
All authors made a substantial contribution to the work, in study design, data acquisition, analysis, and interpretation of the work. Additionally, they actively participated in drafting, revising, and critically reviewing the article. All agree to take responsibility and be accountable for the contents of the article.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Consent for publication
Given the utilization of secondary data, informed consent was waived by the Institutional Review Board (IRB). AL-Najah National University Hospital granted the necessary permissions. Participants’ data confidentiality was conscientiously maintained.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
The study was approved by the An-Najah National University Ethics Committee.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
