Abstract
Background:
We sought to screen for clinical and laboratory features of hemophagocytic lymphohistiocytosis among pediatric patients with severe sepsis.
Methods:
We conducted a retrospective study that analyzed the clinical and laboratory data of 70 pediatric patients who died of severe sepsis. Medical records were revised for the presence of fever, splenomegaly, pancytopenia, hyperferritinemia, hypertriglyceridemia, and hypofibrinogenemia. Soluble CD25 was measured in stored samples.
Results:
Patients’ ages ranged between 0.5 and 11 years with median (interquartile range) 2 (1–5). All patients had fever (≥38.5 °C) and pancytopenia, 58 (82.9%) hepatosplenomegaly, 36 (51.4%) lymphadenopathy, 37 (52.9%) had ferritin >500 ng/ml, 20 (28.6%) had fibrinogen <1.5 mg/ml, 14 (20%) had fasting triglycerides >264 mg/dl while 5 (7.1%) had soluble CD25 >2400 U/ml. Twenty-five (35.7%) patients fulfilled at least 5/6 of the hemophagocytic lymphohistiocytosis-2004 diagnostic criteria. Multivariate backward binary logistic regression analysis revealed lymphadenopathy as an independent predictor for hemophagocytic lymphohistiocytosis criteria fulfilment with odds ratio of 23.9. Fibrinogen had the best performance in discriminating hemophagocytic lymphohistiocytosis fulfilling from non-fulfilling groups (cut-off value: <1.8 mg/ml), followed by ferritin/erythrocyte sedimentation rate ratio (cut-off value: >17).
Conclusion:
There is a significant clinical and laboratory overlap between hemophagocytic lymphohistiocytosis and severe sepsis, making the syndromes difficult to distinguish. The use of current hemophagocytic lymphohistiocytosis-2004 diagnostic criteria should be applied cautiously in those patients.
Introduction
Hemophagocytic lymphohistiocytosis (HLH) is an inflammatory syndrome consisting of fever, pancytopenia, hyperferritinemia, multiorgan dysfunction, and the pathologic finding of hemophagocytosis. 1 Classically, primary HLH is defined as an inherited genetic disorder manifesting in the first few years of life, while secondary HLH, which presents in older children and adults, occurs in the context of infection, autoimmune disease, or malignancy. 2 Due to the relatively non-specific nature of the clinical signs and symptoms, diagnosis is often delayed. 3 Severe sepsis is a leading cause of death for critically ill children treated in intensive care units (ICUs). Refractory shock, multisystem organ failure, and respiratory failure are the main causes of mortality among children with severe sepsis, with more than half of the deaths occurring within the first 7 days after sepsis recognition. 4
HLH, systemic inflammatory response syndrome (SIRS), sepsis, and severe sepsis share clinical and laboratory features. All these disorders have high rates of mortality, yet the therapeutic options are radically different. Chemotherapy and bone marrow transplantation have been used for the treatment of HLH, whereas antibiotics and supportive treatment are used in severe sepsis/SIRS.5–7
The treatment protocols HLH-94 and HLH-2004 have been established as scientific cornerstones for diagnosis, classification, and treatment of HLH in patients younger than 18 years. Therapeutic options for HLH include dexamethasone, Etoposide, cyclosporin A, and anti-cytokine therapy aiming to delete activated T cells and suppress inflammatory cytokine production. Rituximab might be used for Epstein-Barr virus (EBV) associated HLH.3,8 Plasmapheresis or the use of cytokine adsorption columns may aid in rescuing critically ill patients from a deleterious cytokine storm. 9 Therapeutic options not available in our center include anti-CD52 antibody alemtuzumab which has been reported to be beneficial in refractory pediatric patients. 10 Other salvage options include cyclophosphamide, doxorubicin, vincristine, and prednisone like protocols plus etoposide and targeted inhibition of JAK signaling with ruxolitinib. 11 Emapalumab (anti-IFN-γ monoclonal antibody) as a second-line therapeutic agent for primary HLH in children and adults, has also been approved. 12 The lower toxicity of the recent treatment options compared to cytotoxic chemotherapy underscores the need for differentiation of HLH from sepsis, as now more treatment options might be available in some centers for the very ill HLH patients and highlights the importance of the present study.
Whether primary or secondary HLH, both might be triggered by infections, autoimmunity, autoinflammatory disorders, inborn errors of metabolism and malignancies, particularly T-cell lymphomas. Infections triggering HLH include viral infections, the commonest of which include EBV, Cytomegalovirus, Parvovirus, Herpes virus infections, human Herpes virus-8, and HIV infection. HLH may be caused by brucella, gram-negative bacteria, rickettsia, leptospira, tuberculosis, malaria, leishmania, and fungal infections. 13 For that reason, we were stimulated to revise the clinical and laboratory data of patients who died of severe sepsis for the possibility of underlying unrecognized HLH using the HLH-2004 criteria. 3
Methods
A retrospective analytical study was carried out at the Children's Hospital, Ain-Shams University over the period from September 2017 to February 2019. We revised the medical records of pediatric patients above the age of 1 month to 18 years with severe sepsis who were admitted to the ICU over a period of 18 months. The study included data from the medical records of 70 pediatric patients who had positive bacterial cultures and died of severe sepsis despite receiving proper antimicrobial therapy according to the culture results and the ICU protocol.
Severe sepsis was defined as the presence of SIRS in the presence of suspected or proven infection, together with cardiovascular dysfunction, acute respiratory distress syndrome, or dysfunction in two or more other organ systems. 14 The SIRS was evidenced by at least two of the following criteria: core temperature >38.5 °C or <36 °C, tachycardia with mean heart rate >2 SD above normal for age in the absence of external stimuli drugs or painful stimuli or unexplained persistent elevation over 0.5–4 h in children <1 year or persistent bradycardia over 0.5 h with a mean heart rate <10th percentile for age in the absence of vagal stimuli, β-blocker drugs, or congenital heart disease, respiratory rate >2 SD above normal for age or the acute need for mechanical ventilation not related to neuromuscular disease or general anesthesia, leukocyte count elevated or depressed for age not secondary to chemotherapy or >10% immature neutrophils. 15
Patients having known underlying autoimmune disorders, malignancy, immunodeficiency, metabolic disorders, or any other chronic illness (cardiac, pulmonary, gastrointestinal, etc.) were excluded from the study to minimize the heterogenicity of the studied population.
Medical records of enrolled patients were examined for the presence of clinical and laboratory criteria of HLH as set forward by the Pediatric HLH Study Group of the Histiocyte Society. 3 None of the included patients had a molecular diagnosis of HLH. We searched for the following features, five of the following six would fulfill HLH diagnosis: (1) fever ≥38.5 °C, (2) cytopenia of two lines, (3) hypertriglyceridemia (≥265 mg/dl) and/or hypofibrinogenemia (≤1.5 g/l, (4) hyperferritinemia (≥500 microg/L), (5) elevated soluble interleukin-2 receptor (CD25) ≥ 2400 U/ml, and (6) splenomegaly. HLH diagnosis was based on the fulfillment of 5/6 criteria, not 5/8, as the evidence of hemophagocytosis and natural killer cell dysfunction was not available in the examined data. Records were checked for the presence of fever and its duration, clinical evidence of infection, recorded vital signs, the presence of hepatosplenomegaly or lymphadenopathy, and the evidence of organ dysfunction. Worth noting that lymphadenopathy was considered significant when the diameter clinically exceeded 1 cm for cervical and axillary nodes and 1.5 cm for inguinal nodes or radiologically evident internal lymph nodes with a diameter exceeding 1 cm. Hepatosplenomegaly was documented based on both the clinical and radiological assessment according to age and sex.
Laboratory data included a complete blood count with the differential leucocytic count, and results were compared with age-related reference ranges. 16 Erythrocyte sedimentation rate (ESR) was measured using the Westergren method. Prothrombin time, international normalized ratio, partial thromboplastin time, and fibrinogen measured by the Clauss clotting method using STA Compact Max analyzer, France. Serum levels of creatinine, aspartate aminotransferase, alanine aminotransferase, albumin, bilirubin, ferritin, and serum triglycerides were measured using Synchron CX-9 autoanalyzer using Beckman Instruments Inc., Fullerton, California, USA. Stored sera at −20 °C were used to measure soluble CD25 by ELISA using Stat Fax 2100 Microplate Reader, USA. Patients with incomplete medical records or missing laboratory results were considered ineligible for enrollment.
Sequential Sepsis-Related Organ Failure Assessment Score (SOFA score) was assessed as a measure of mortality risk according to data of enrolled patients in relation to their sepsis and based on organ dysfunction. 17
The study was conducted following the ethical standards of the Helsinki Declaration of 1964 and revised in 2008. The study protocol gained approval from the local ethics committee of the Pediatric Department (approval number: 118-205-2017). Informed written consent was obtained from the caregivers/parents of patients with the preservation of patient anonymity.
Statistical methods
Data were tabulated and analyzed using IBM© SPSS© Statistics version 23 (IBM© Corp., Armonk, NY) and MedCalc© version 15.8 (MedCalc© Software bvba, Ostend, Belgium). For quantitative data with parametric distribution, an independent t-test was employed, while for non-parametric data, the Mann–Whitney test was used. Categorical variables were compared using Fisher's exact test. Correlations were tested using the Spearman rank correlation test; the correlation coefficient (Spearman rho) was interpreted according to Hauke and Kossowski. 18 The diagnostic/predictive values of HLH markers were determined using receiver-operating characteristic (ROC) curve and the area under the ROC curve (AUC) was interpreted according to Kumar and Indrayan. 19 Multivariate backward binary logistic regression analysis was used to determine predictors of HLH. Statistical significance was defined at a probability p-value of <0.05.
Results
We intended through our work to analyze the clinical and laboratory data of 70 pediatric sepsis patients who died despite receiving adequate antimicrobial therapy. Their age ranged between 0.5 and 11 years with a mean ± SD of 3 ± 3 years, median (interquartile range (IQR)) 2 (1–5), and a male to female ratio of 1:1. They all had documented infections in the form of pneumonia (n = 40; 57.1%), gastroenteritis (n = 11, 15.7%), CNS infection (n = 10; 14.3%), or postoperative sepsis (n = 9; 12.9%). Body fluid bacterial cultures demonstrated growth of gram-positive organisms in 20 patients (28.6%) (commonest of which were Staphylococcal aureus and Streptococcus pyogenes), gram-negative organisms in 29 (41.4%) (commonest of which were Klebsiella, E. coli, and Acinetobacter) and mixed infections in 21 (30%). Their SOFA score ranged between 9 and 19 and median (IQR) 15 (13–16) at the time of evaluation for HLH criteria. They all received antimicrobial therapy based on their cultures and the ICU protocol. None of them received HLH specific therapy. They died after a period of ICU admission ranging from 4 to 9 days, median (IQR) 5 (4–7) days.
Revision of clinical and laboratory features suggestive of HLH revealed that fever (≥38.5 °C) was present in all enrolled patients, 58 (82.9%) had hepatomegaly, 58 (82.9%) had splenomegaly, 36 (51.4%) had significant lymphadenopathy, while skin rashes in the form of purpuric, macular, or papular rash were found in 27 (38.6%). All patients (100%) had pancytopenia, 37 (52.9%) had high serum ferritin (>500 ng/ml) (median (IQR) in the whole sample: 625 (280–1500)); 20 (28.6%) had consumed fibrinogen (<1.5 mg/ml) (median (IQR): 2.1 (1.2–3.4)); 14 (20%) had high fasting triglycerides (>264 mg/dl) (median (IQR): 198 (160–240)) and only five (7.1%) had elevated soluble CD25 (sCD25) (>2400 U/ml) (median (IQR): 888 (500–1375)).
According to the HLH criteria, 25 patients fulfilled the diagnosis of HLH (35.7%), while 45 (64.2%) did not.
The HLH fulfilling group demonstrated a significantly higher frequency of splenomegaly (p = 0.05), lymphadenopathy (p < 0.001), elevated soluble CD25 (p = 0.004), elevated serum ferritin (p < 0.001), elevated triglycerides (p < 0.001), and consumed fibrinogen (p < 0.001) than the non-HLH group (Figure 1). The HLH group also had lower neutrophil counts, fibrinogen levels, ESR, and higher ferritin, soluble CD25, triglycerides levels, and ferritin/ESR ratio than the non-HLH group (Table 1). SOFA scores were comparable between the HLH and non-HLH groups, t = 8.558, p = 0.992.
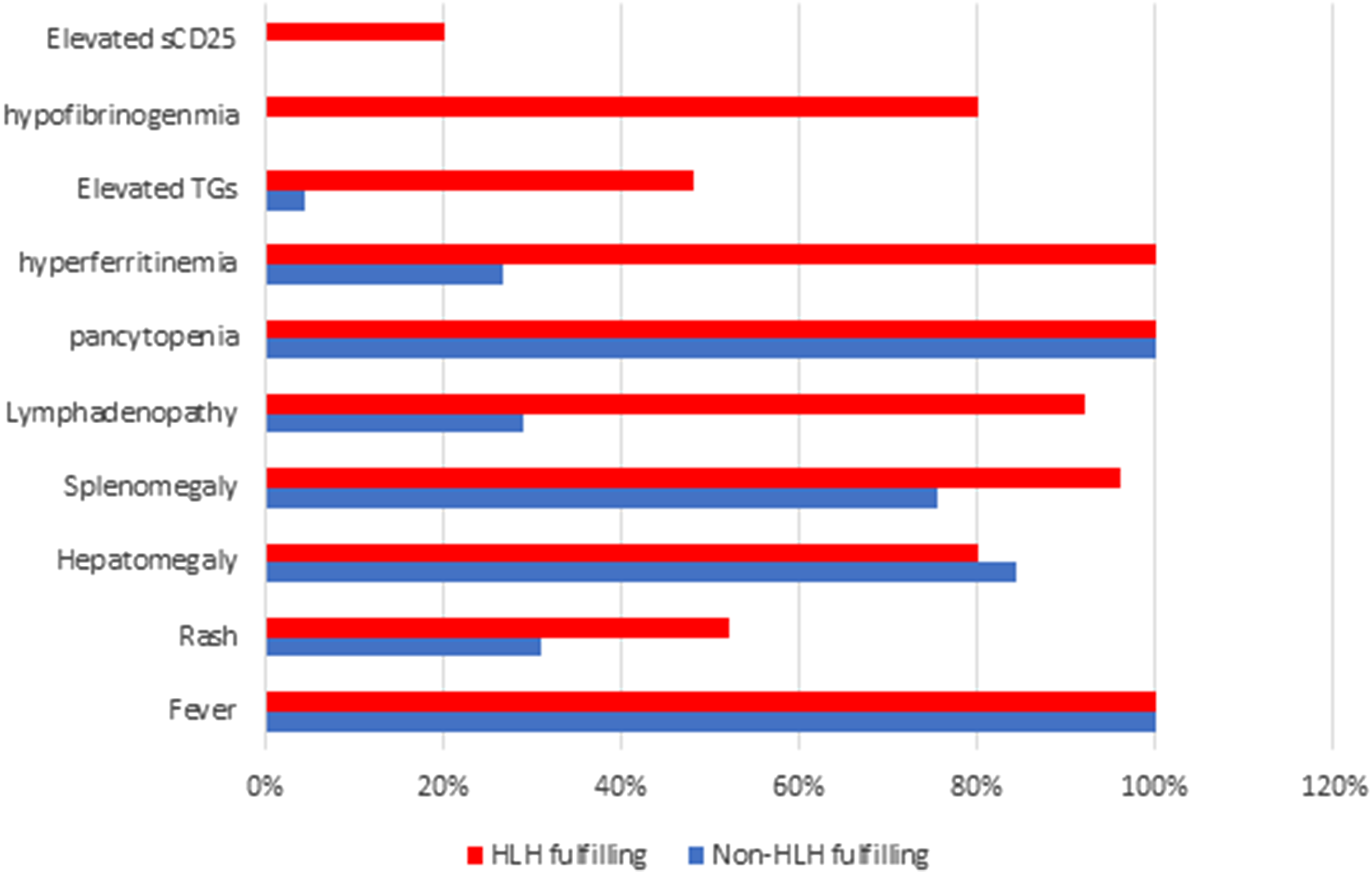
Frequency of clinical and laboratory abnormalities among hemophagocytic lymph proliferative histiocytosis (HLH) and non-HLH groups.
Comparison of laboratory results among HLH and non-HLH groups.

Data are presented as mean ± SD or median (IQR).
ALT: alanine aminotransferase; AST: aspartate aminotransferase; CRP: c-reactive protein; ESR: erythrocyte sedimentation rate; INR: international normalized ratio; HLH: hemophagocytic lymphohistiocytosis; Max.: maximum; Min.: minimum; n: number; PTT: partial thromboplastin time; PT: prothrombin time; S: serum; SD: standard deviation; TLC: total leucocytic count.
Unpaired t-test;
Mann–Whitney test.
Results of soluble CD25, ferritin, fasting triglycerides, ferritin/ESR ratio, and fibrinogen were plotted in a ROC curve, and the cut-off values that could discriminate between HLH and non HLH groups were determined. Fibrinogen and ferritin/ESR ratio were the best to discriminate between the HLH fulfilling and non-fulfilling group. This was followed by soluble CD25 and serum ferritin. Serum triglycerides levels >190 mg/dl could discriminate between the two groups with a sensitivity of 88% and a specificity of 68.9%, while levels >295 mg/dl demonstrated 100% specificity for HLH but a low sensitivity of 28%. Results of the cut-off values with sensitivity, specificity, positive predictive, and negative predictive values are shown in Table 2 and Figure 2.

Receiver-operating characteristic (ROC) curves for discrimination between HLH fulfilling and non-fulfilling groups using soluble CD25, serum ferritin, triglycerides, fibrinogen, and ferritin/ESR ratio.
Receiver-operating characteristic (ROC) curve analysis for discrimination between HLH and non-HLH groups using sCD25, ferritin, fasting triglycerides, fibrinogen, SOFA 1, and SOFA 2 scores.

AUC: area under the ROC curve; CI: 95% confidence interval; HLH: hemophagocytic lymphohistiocytosis; SE: standard error; 95%; SOFA score: Sequential Sepsis-Related Organ Failure Assessment Score; ESR: erythrocyte sedimentation rate.
Multivariable backward binary logistic regression analysis was used to identify predictors of HLH (Table 3). Potential predictors that were entered in the model included lymphadenopathy, splenomegaly, high soluble CD25, high serum ferritin, high triglycerides, and consumed fibrinogen. Only lymphadenopathy (odds ratio = 23.9, 95% CI = 4.22–135.43, p-value <0.001) and high triglycerides (odds ratio = 15.33, 95% CI = 2.11–111.44, p-value = 0.007) were independent predictors for HLH diagnosis fulfillment.
Multivariable backward binary logistic regression model for predictors of HLH.

CI: 95% confidence interval; HLH: hemophagocytic lymph proliferative histiocytosis; SE: standard error; SOFA score: Sequential Sepsis-Related Organ Failure Assessment Score.
High triglycerides: Fasting serum triglycerides ≥265 mg/dl.
Combined hypertriglyceridemia and lymphadenopathy showed good predictive value/logistic value for HLH diagnosis fulfillment with an AUC of 0.882. The best cut-off criterion is predicted probability >0.0387 which had a sensitivity of 96% and specificity of 68.9% (Figure 3).
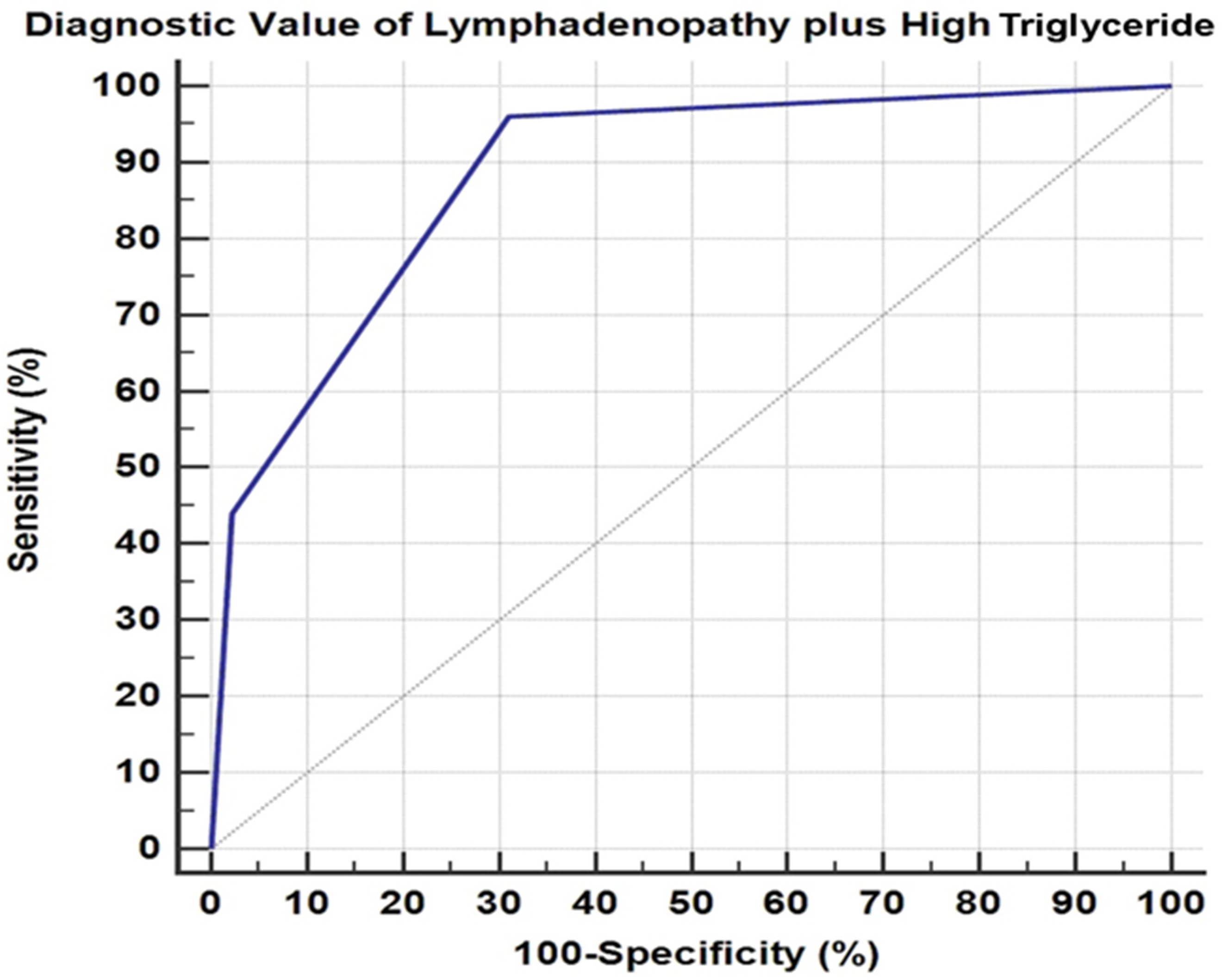
Receiver-operating characteristic (ROC) curve for discrimination between HLH and non-HLH groups using lymphadenopathy plus high triglycerides. AUC = 0.882, sensitivity = 96%, specificity = 68.9%, cut-off criterion = predicted probability >0.0387.
Ferritin/ESR ratio correlated positively with HLH markers namely soluble CD25, ferritin, and fasting triglycerides, and negatively with fibrinogen levels (p < 0.001 for all).
Discussion
Sepsis with its different grades is known for its high mortality rate, with an estimated affection of more than 30 million people every year, and up to 6 million deaths. 20 The United States reports have shown a mortality rate of 21.2% in children with severe sepsis. 21 Results from a systematic review and meta-analysis including 170 studies from Europe, North America, and Australia between 2009 and 2019 showed an average 30-day septic shock mortality of 34.7% and 90-day septic shock mortality of 38.5%. 22 The developing world, on the other hand, carries the greatest burden of sepsis-related mortality with rates ranging between 43% and 87%. 23 Distinguishing HLH from severe sepsis is of utmost importance as both disorders have high mortality and have different plans of therapy. HLH remains an unrecognized cause of death in patients with sepsis and septic shock.5,24 HLH diagnosis is based on a set of nonspecific clinical and laboratory features, and despite the validation of the HLH-2004 criteria, early diagnosis especially when the picture is incomplete remains challenging.5,7
In our study, 35.7% of the enrolled patients fulfilled 5/6 criteria for HLH diagnosis. Those patients could have either only sepsis with an overlap of their clinical and laboratory features with HLH or they might have true HLH that was triggered by infection and severe sepsis. Another possibility is that they might have started as HLH and developed sepsis secondary to neutropenia. Inversely, macrophage activation syndrome (MAS) like phenotype, a newly evolving entity in patients with sepsis, is another possibility. However, the criteria for the diagnosis of those patients and their benefit from the use of immunosuppressive therapy are still under investigation.25–27
The possibility of HLH among patients who did not fulfill the criteria still cannot be excluded. Two of the eight diagnostic criteria were not evaluated in our study population, namely hemophagocytosis and natural killer cell dysfunction, and despite their low specificity for HLH,28,29 yet, the lack of their results might point to a possibility of an underestimate of HLH diagnosis. Molecular diagnosis in those patients could just help to define HLH recurrence risk, not the presence of an active disease state. 25 HLH is not uncommon in Egyptian children, yet there are no published nationwide incidence rates or registries. Few reports have described HLH patients as center experiences whether in our institution or other institutions in Egypt, describing 37, 18, and 101 HLH patients, respectively.30–32
Internationally, the true incidence of HLH is difficult to estimate but figures have slightly increased over time, most probably because of improved detection. The Swedish national registry estimated the incidence of primary HLH to be 1.5 cases/million births in Sweden 2007–2011, 33 while in Texas Children's Hospital, diagnosis of HLH was estimated to be 1/3000 inpatient admissions. 34 Similar to autosomal recessive inborn errors of immunity, primary HLH is expected to be higher in populations with high consanguineous marriages which exceed 50% in many countries of the MENA region. 35
According to clinical evaluation of our patients, the group fulfilling HLH diagnosis showed a higher frequency of splenomegaly (96%) and lymphadenopathy (92%) compared to the non-HLH group (75.6% and 28.9% respectively). Previous studies reported splenomegaly to occur in 79% of children with severe sepsis whereas, in HLH, it ranged from 83% to 95%. Lymphadenopathy may associate infections and can be either localized or generalized according to the infectious agent and site of infection.36–38 Although the presence of tender lymphadenopathy points to infection, yet, none of the previous studies have defined a differentiating pattern of lymphadenopathy in sepsis versus HLH. In our study, multivariate regression analysis outlined the importance of lymphadenopathy as an independent predictor for HLH with an odds ratio of 23.9.
The finding that lymphadenopathy is associated with the highest OR of meeting HLH 2004 criteria in our study is quite surprising, as lymphadenopathy is not a typical feature of HLH. 25 This might point to the possibility of the presence of underlying unrecognized viral infection, hematological malignancy, or lymphoproliferative disorder in those patients. 39
All studied patients had pancytopenia with comparable mean hemoglobin levels and platelet counts between the two groups. Thrombocytopenia might occur in deteriorating sepsis patients because of disseminated intravascular coagulation (DIC) together with hypofibrinogenemia. However, profound cytopenias are reported to be more suggestive of HLH rather than sepsis, which can be attributed primarily to high concentrations of TNF-α, INF-γ, and hemophagocytosis. 40 On the other hand, leucocytosis with neutrophilia is more common to occur with sepsis; moderate neutrophilic leucocytosis prevailed among septic patients with a mean absolute neutrophil count of 8200/mm3. 41 Although the association of HLH with leukocytosis is very unusual where leukopenia is the norm, yet, the presence of a leukocytosis should not distract the physician from a diagnosis of HLH. According to Sato et al. 42 , CD8 + lymphocytic leucocytosis was noticed with the onset of EBV viremia early in the course of HLH disease. Ramachandran et al. reported that about one-fourth of their 33 HLH children had leucocytosis and neutrophilic leukocytosis has also been reported with scrub typhus- and mycobacterial-associated HLH.43–45
The elevated serum ferritin, fasting triglycerides, sCD25, and the consumed fibrinogen were more frequently encountered, and their abnormalities were more profound among the HLH fulfilling group. Hyperferritinemia, at a cut-off serum level >700 ng/ml, could discriminate between the HLH fulfilling and the non-fulfilling septic patients with both sensitivity and specificity above 90%. Ferritin, which is secreted by activated macrophages, is increased as an acute phase reactant in response to inflammation including sepsis, with levels reported in children with sepsis to range 21–2210, with a median of 303 ng/ml. 46 On the other hand, ferritin is a well-established marker for HLH activity and ferritin levels of 500 ng/ml had 84% sensitivity for HLH in familial causes versus 69% sensitivity in the general pediatric population with febrile disease.3,47 This is while concentrations above 7500–10,000 ng/ml were associated with 96% specificity but limited sensitivity for HLH. Progressive hyperferritinemia over 2000 ng/ml in a febrile patient without defect of iron metabolism and transfusion history should urge the analysis of other HLH criteria. 40 However, even extreme ferritin concentrations are not pathognomonic for HLH, and hyperferritinemia should be interpreted in the context of the other HLH criteria. 48
Fasting triglyceride level of ≥265 mg/dl, one of the criteria for HLH diagnosis, was present in 14 of our patients, 12 of whom fulfilled HLH diagnosis, with a discriminatory cut-off level of 190 mg/dl. We also report a level of ≥265 mg/dl to be an independent predictor for HLH in patients with severe sepsis. On the other hand, Fitrolaki et al. 49 observed increased triglyceride and decreased high- and low-density lipoproteins with systemic inflammation and sepsis and reported that children with severe sepsis can have triglyceride concentrations similar to HLH, however, this might be attributed to the presence of underlying undiagnosed HLH. In the same context, Cetinkaya et al. 50 reported hypertriglyceridemia (levels >150 mg/dl) in 50% of patients with severe sepsis and was associated with increased sepsis mortality. Several mechanisms underlie hypertriglyceridemia in sepsis and SIRS including increased hepatic production of triglyceride (TGs) and very-low-density lipoproteins, increased hepatic free fatty acids synthesis, and their release from lipolysis in response to stress hormones and inflammatory cytokines, in addition to decreased activity of lipoprotein lipase. 51 On the other hand, Lekkou et al. observed that TGs levels did not exceed 180 mg/dl in their studied 50 pediatric patients with severe sepsis, 52 while Janka and Schneider observed through their work that 35% of children diagnosed with HLH had TGs levels surpassing 440 mg/dl. 47 Thus, one might conclude that although hypertriglyceridemia can occur in both sepsis and HLH, yet, higher values are more common in HLH patients without previously known hyperlipidaemic disorders.
In our study, a fibrinogen level of 1.8 mg/ml showed the best performance in discriminating HLH from non HLH among patients with severe sepsis. A recently published retrospective study showed that hypofibrinogenemia was independently associated with poor outcome HLH/MAS in pediatric sepsis and that it might help identify children with possible sepsis/HLH overlap for whom further diagnostic testing should be considered. 53 Hypofibrinogenemia could be reliably applied as a differentiating criterion between sepsis and HLH in patients without DIC and thus, should be taken into consideration together with the platelet count. 40
In our study, a sCD25 (Soluble Interleukin 2 receptor) level of 900 IU/ml was a good predictor and could discriminate between the HLH and non-HLH groups with a sensitivity of 100%, and a specificity of 82.2%. sCD25 also reflects the immune activation in autoimmune diseases, neoplasms (notably lymphoproliferation), and infections
In the 25 patients meeting HLH 2004 criteria in our study, the relatively low levels of sCD25/sIL-2r (mean 1550; SD 1250–2187) and serum ferritin (mean 2145; SD 1800–3000) suggest that many of the 25 patients meeting the HLH 2004 criteria might not in fact have HLH. Hayden et al. 59 observed sCD25 > 2400 U/ml in 37.5% of the patients with non-HLH conditions, including sepsis, and they recommended that 2515 U/ml might be a better cut-off for sCD25 in HLH patients (72.5% specificity) while specificity for HLH reaches 93% with sCD25 > 10,000 U/ml. This is while Chen et al. considered hyperferritinemia, although sensitive for HLH in children, yet, not specific and might occur in liver diseases, infections, hematological malignancies, and renal failure. 60
Thus, while HLH can be very difficult to discern from typical sepsis or SIRS, sharing common laboratory abnormalities, yet these abnormalities seem to be more profound in HLH with clues that the severity of inflammation may be more than expected. Lack of response to antimicrobials and different cytokine patterns might help distinguish between these different inflammatory conditions with a pattern of increased IFN-γ and IL-10 among HLH patients versus predominantly increased IL-6 with normal or slightly elevated IFN-γ in sepsis. 61 Furthermore, HLH, unlike sepsis, being characterized by distinctive T-cell activation was found to be associated with activation of CD8+ T cells being most pronounced and associated with increased CD38high/HLA-DR+ effector cells. A threshold of >7% CD38high/HLA-DR+ cells among CD8+ T cells was reported to have strong positive and negative predictive value for distinguishing HLH from early sepsis or healthy controls. 62 Also perforin and degranulation assays are observed to be able to differentiate between HLH and its mimickers and differentiate between primary and secondary HLH and suggested to be used as a screening tool for patients for primary HLH diseases. 63
Ferritin/ESR ratio has been reported to be of value in predicting MAS in patients with juvenile idiopathic arthritis at a cut-off level of 80. 64 In our study, ferritin/ESR showed the second-best performance, next to fibrinogen, in differentiating between patients fulfilling and non-fulfilling HLH diagnosis, at a cut-off level of 17, and its levels correlated significantly with other HLH markers. In HLH, the assumed increased ferritin and decreased ESR are expected to cause an increase in the ferritin/ESR ratio. This elevation is not expected to be the same as that in MAS due to different disease characteristics in both illnesses. However, the higher the ferritin/ESR ratio, the more probable it is to have an HLH diagnosis. Yet, no definite cut-off levels are identified in this aspect. Also, several factors may alter the ESR levels which make accurate cut-off levels difficult to be estimated. The implementation of the ferritin/ESR ratio for HLH diagnosis requires further validation in controlled prospective studies with adequate sample size to be able to set a cut-off level that might help HLH diagnosis.
The results of the current study are limited by its retrospective design, small sample size, the lack of some work up including bone marrow examination and natural killer cell function assay, the lack of cytokine profile, and the lack of implementation of HLH therapy among the HLH diagnosis-fulfilling group. A quantitative functional estimate of cytotoxic lymphocyte function may be a more precise approach to define the overlap of these conditions. The value of ferritin/ESR ratio in discriminating HLH from sepsis requires further investigation through wider scale studies to validate the value of its use.
Conclusion
HLH should be considered by intensivists as a differential diagnosis for cases presenting with sepsis and multisystem organ failure. However, the use of HLH-2004 diagnostic criteria for HLH diagnosis among those patients is hindered by the poor specificity of the currently used tests in critically ill patients and should be cautiously interpreted owing to their great overlap in children with severe sepsis. In our study, more than a third of pediatric patients who died of severe sepsis fulfilled 5/6 of the criteria required for HLH diagnosis, pointing to a possible—yet unproven—HLH diagnosis in those patients, probably secondary to their sepsis, and might be one of the reasons for their death. However, we recommend extreme caution when considering applying HLH therapies to those children. Still, we should consider the possibility of infection-related cytokine storm mimicking HLH. Judicious use of immunomodulatory therapies (IVIG, steroids, plasma exchange, cytokine blockade) might be considered as therapeutic options in those overlap patients.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
