Abstract
Purpose:
Programmed death-1 antibody plus chemotherapy has gained approval for the treatment for (human epidermal growth factor receptor 2 negative locally advanced or metastatic gastric or gastroesophageal junction cancer. This study aims to analyze the efficacy and safety of anti-programmed death-1 antibody combined with chemo- or anti-angiogenesis therapy in Chinese patients with advanced or metastatic gastric or gastroesophageal junction cancer in a real-world setting.
Methods:
In total, 122 patients treated with anti-programmed death-1 antibody-based combination therapy between April 2019 and December 2021 were encompassed. Clinical outcomes and safety profile were measured and analyzed.
Results:
In the whole cohort, median overall survival was 17.2 months, median progression-free survival was 10.9 months, and median duration of response was 9.4 months. Notably, in the first-line patients, the median overall survival was not reached, median progression-free survival was 14.8 months, objective response rate was 68.4%. In the second-line group, median overall survival, median progression-free survival, median duration of response, and objective response rate were 10.9 months, 5.9 months, 4.5 months, and 41.5%, respectively. Treatment-related adverse events of any grade were observed in 28.2% of the overall cohort, primarily affecting the hematological and liver function. Grade 3 or 4 adverse events were mainly characterized by increased levels of aspartate aminotransferase, alanine aminotransferase, along with decreased lymphocyte and white blood cells, as well as anemia.
Conclusions:
Patients in our cohort experienced a clinical benefit from anti-programmed death-1 antibody-combined treatment in first-line treatment settings, with acceptable treatment-related adverse events. The benefit of anti-programmed death-1 antibody combined with chemo- or anti-angiogenesis treatment to the second-line patients should be further confirmed by large multi-center randomized, controlled clinical trials.
Introduction
The staggering number of new cases of gastric cancer (GC) was 1,089,103 in 2020, while the number of new death cases was 768,793 worldwide. This unfortunate statistics firmly positioned GC as the fourth both in incidence and mortality in world cancer statistics. 1 In the era of chemotherapy, overall survival (OS) of metastatic GC was limited to a mere 10 months. However, the landscape changed dramatically with the advent of immunotherapy, a revolutionary treatment strategy for advanced cancers, while GC is no exception to this transformative trend. The pivotal CHECKMATE649 has changed the first-line treatment paradigm of GC into the era of immune-combination chemotherapy. 2 Subsequently, two Chinese multi-centers, phase III randomized controlled studies, ORIENT-16 and RATIONALE 305 reinforced the survival benefit of Chinese-produced anti-programmed death-1 (PD-1) monoclonal antibodies, sintilimab and tislelizumab, when combined with chemotherapy for advanced GC patients.3,4 Notably, in the Chinese cohort participating in the CHECKMATE649 study, the results mirrored the global primary analysis, showing that nivolumab in combination with chemotherapy led to a clinically significant improvement in OS, progression-free survival (PFS), and response rate compared to chemotherapy alone, all while maintaining an acceptable safety profile for Chinese patients. 5 The result from the phase III ATTRACTION-2 trial showed that the anti-PD-1 antibody nivolumab improved survival outcomes for Asian patients with metastatic, chemotherapy-refractory gastric and gastro-esophageal junction (GEJ) cancer regardless of patients' programmed death-ligand 1 (PD-L1) expression. 6 Additionally, the KEYNOTE-059 study demonstrated an objective response rate (ORR) of pembrolizumab as 11.6%, and the duration of response (DOR) was 8.4 months in previously treated GC patients, and with a longer DOR of 16.3 months in PD-L1-positive patients. 7
Based on these above clinical trials, immunotherapy has garnered approval for employment in both first-line and third-line treatments for advanced gastric and GEJ cancer by reputable institutions such as the National Comprehensive Cancer Network (NCCN), Japanese Pharmaceuticals and Medical Devices Agency, as well as Chinese Society of Clinical Oncology. Furthermore, the NCCN guidelines advocate for pembrolizumab as a second-line or subsequent therapy in patients with microsatellite instability-high (MSI-H) or deficient mismatch repair (dMMR) solid tumors. However, there is a conspicuous absence of evidence to substantiate the benefits of immunotherapy in the second-line treatment of advanced GC. The KEYNOTE-061 study was a randomized, open-label, phase III study, showing that pembrolizumab failed to yield a statistically significant improvement in OS compared to paclitaxel as a second-line therapy for advanced gastric or GEJ cancer in patients with PD-L1 combined positive score (CPS) of 1 or higher. 8 A similar outcome was observed in the phase III KEYNOTE-063, conducted in Asian countries. 9
The primary aim of this study was to collect and analyze the efficacy and safety data in a real-world setting about patients with advanced or metastatic gastric and GEJ cancer treated with immune checkpoint inhibitors alone or in combination in first-line and second-line treatment cohorts in our hospital.
Materials and methods
Study design and data collection
It is a single-institutional retrospective study to review clinical data in a Chinese cohort diagnosed with advanced or metastatic gastric or GEJ cancer who received the PD-1 inhibitor nivolumab or Chinese-produced anti-PD-1 antibody alone or combination therapy. It was conducted from April 1, to 2019 December 31, 2021 in Shandong Cancer Hospital and Institution.
Eligible patients were (1) aged 18 years or older; (2) had histologically confirmed gastric or GEJ adenocarcinoma assessed by pathology and (3) assessed by clinicians in advanced or late-stage disease; (4) and received nivolumab or Chinese-produced anti-PD-1 antibody alone or combination therapy; (5) patients who received anti-PD-1 antibody in first-line or second-line treatment.
Exclusions: patients who were (1) neuroendocrine carcinoma (2) receiving perioperative or third-line treatment (3) combined with second primary carcinoma within the last 5 years.
Neutrophil-to-lymphocyte (N/L) ratio was defined as the absolute neutrophil count divided by the entire lymphocyte count in peripheral blood, collected before the initiation of anti-PD-1 treatment. The platelet-to-lymphocyte (P/L) ratio was defined as the absolute thrombocyte count divided by the absolute lymphocyte count. Neutrophil-to-monocyte (N/M) ratio was defined as the absolute neutrophil count divided by the absolute monocyte count. Median N/L, P/L, and N/M ratios were adopted as cutoffs in our analysis.
The study has been examined and approved by the ethics committee of Shandong Cancer Hospital and Institute SDTHEC2022001017. All human studies have been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki. The requirement of obtaining informed consent was waived by the Institutional Review Board/Ethics Committee.
Treatment
The treatment with a PD-1 antibody and type of therapy were not predefined in this retrospective analysis. The decision to opt for either nivolumab or Chinese-produced anti-PD-1 antibody was left to the discretion of the attending physicians responsible for the patients. These choices were made based on the findings of CHECKMATE649, ORIENT-16, and RATIONALE 305 trial results, and according to the patient's economic status. Nivolumab was administered either at a dose of 360 mg every 3 weeks or 240 mg every 2 weeks. Similarly, Chinese-produced anti-PD-1 antibodies, such as sintilimab and tislelizumab were administered at a dose of 200 mg every 3 weeks intravenously. Table 2 displays the combined treatment approachs. Palliative treatment was classified as first-line therapy concerning evidence of metastatic disease without treatment. Second-line therapy was categorized as either when patients progressed from first-line therapy or when the interval time was less than 6 months from the last adjuvant treatment for localized disease.
Tumor tissue analyses
Analysis of tumor tissue was performed by an experienced pathologist. The expression of PD-L1 was assessed through immunohistochemistry (IHC) (DAKO, 22C3) and classified by either CPS ≥ 1 or tumor proportion score (TPS) ≥ 1%. CPS was determined by the number of tumor cells and immune cells staining for PD-L1-positive, divided by the total number of viable tumor cells, and then multiplied by 100. On the other hand, TPS was calculated by the number of PD-L1-positive tumor cells, divided by the total number of viable tumor cells. 7 Microsatellite status was assessed using IHC or polymerase chain reaction (PCR). For mismatch repair (MMR) protein expression, tumors were designated as deficient phenotype when one of the four MMR proteins (MLH1, MSH2, MSH6, or PMS2) was negative in tumor cells. 10 PCR was conducted for analysis of five tumor microsatellite loci (BAT25, BAT26, NR21, NR24, and Mono27). MSI-H was defined when at least two allelic loci size shifts among the five analyzed microsatellite markers were detected by PCR. 11 To identify human epidermal growth factor receptor 2 (HER2)-overexpressing, IHC and fluorescence in situ hybridization (FISH) methods have been validated. Gastric tumors were divided into three categories according to the IHC score: negative (0+ or 1+), equivocal (2+), or positive (3+). In the case of an equivocal score of 2+, HER2 positivity was further confirmed by detecting HER2 amplification using FISH. According to the current guidelines, FISH positivity was defined as a ratio of HER2 signal to centromere (control) CEP17 signal ≥ 2.0. 12
Statistical analyses
Baseline characteristics were analyzed descriptively by SPSS 26. PFS was calculated from the date of starting anti-PD-1 antibody therapy until radiologically confirmed progression or death from any cause. Patients without progression at the last contact were regarded as censored. OS was calculated from the date of commencement of PD-1 antibody therapy until death from any cause. Patients alive at the last contact were considered as censored. The DOR was defined as the time between the first documented evidence of complete response (CR) or partial response (PR) and the first documented sign of disease progression or death from any cause. ORR was evaluated using response evaluation criteria in solid tumors (RECIST 1.1). The median values of PFS, OS, and DOR were determined using the Kaplan–Meier method. The log-rank test was used to compare survival between different patient groups. Additionally, hazard ratios (HR) and their 95% confidence intervals (CIs) were obtained by using the Cox proportional hazards model. To analyze the association between clinical characteristics and survival outcomes, univariate and multivariate analyses of OS and PFS were conducted with the Cox regression model using SPSS 26. In the multivariable Cox regression model, variables with a P-value <0.1 in the univariate Cox regression or acting as clinically relevant factors were adjusted. All P-values are two-tailed, and P-values <0.05 were considered to indicate statistical significance.
Results
Patients baseline characteristics
In total, 158 patients were screened, according to our inclusion criteria, 122 patients diagnosed with advanced or metastatic gastric or GEJ cancer, who had been treated with nivolumab or Chinese-produced anti-PD-1 antibody combination in first- or second-line therapy between April 2019 and December 2021, were included at our cancer center. The study flow-chart is described in Figure 1. The last follow-up date was December 22, 2022. The baseline characteristics of these patients are detailed in Table 1. The median age was 58.3 years with a range from 20 to 83 years when anti-PD-1 antibody was initiated. The majority of our cohort (92.2%) exhibited a Karnofsky performance status (KPS) ≥ 80. Most of the primary tumor site was GC (92.6%). Of the 122 patients, the majority had evidence of metastatic disease at the initiation of immunotherapy, with 90 (73.8%) patients categorized as stage IVb. The leading organs of secondary dissemination were retroperitoneal lymph nodes, liver, and peritoneum. Treatment regimens are depicted in Table 2. In our cohort, 65 patients (53.3%) were undergoing the second-line treatment; 57 patients (46.7%) were receiving the first-line treatment. Notably, the patients in second-line therapies were anti-PD-1 antibody treatment-free previously. Among the patients, 17 (13.9%) patients received nivolumab therapy, the most administered Chinese-produced anti-PD-1 antibody was sintilimab in 95 (77.9%) patients, followed by tislelizumab in 10 (8.2%) patients. It was noted that only 11 (9%) patients received mono anti-PD-1 therapy, while the remaining 111 patients received anti-PD-1 combination therapy. Taking into account of combination treatment regime, 43 (35.2%) patients received anti-PD-1 combined with nano-albumin paclitaxel-based chemotherapy; 26 patients in the second-line setting, 17 patients in the first-line setting. Additionally, 40 (32.8%) patients received anti-PD-1 combined with fluorouracil- and oxaliplatin-based chemotherapy; 30 patients as first-line therapy, 10 patients as second-line therapy. Furthermore, 18 (14.8%) patients received anti-PD-1 combined with Apatinib (an anti-angiogenesis inhibitor), including 15 patients in the first-line, 3 patients in the second-line. Finally, in 15 patients whose HER2 IHC were 3+/2+ and FISH (+), six patients received anti-PD-1 combined with CAPEOX/SOX/T/TS and Herceptin, there were three patients in first-line therapy, three patients in second-line therapy.
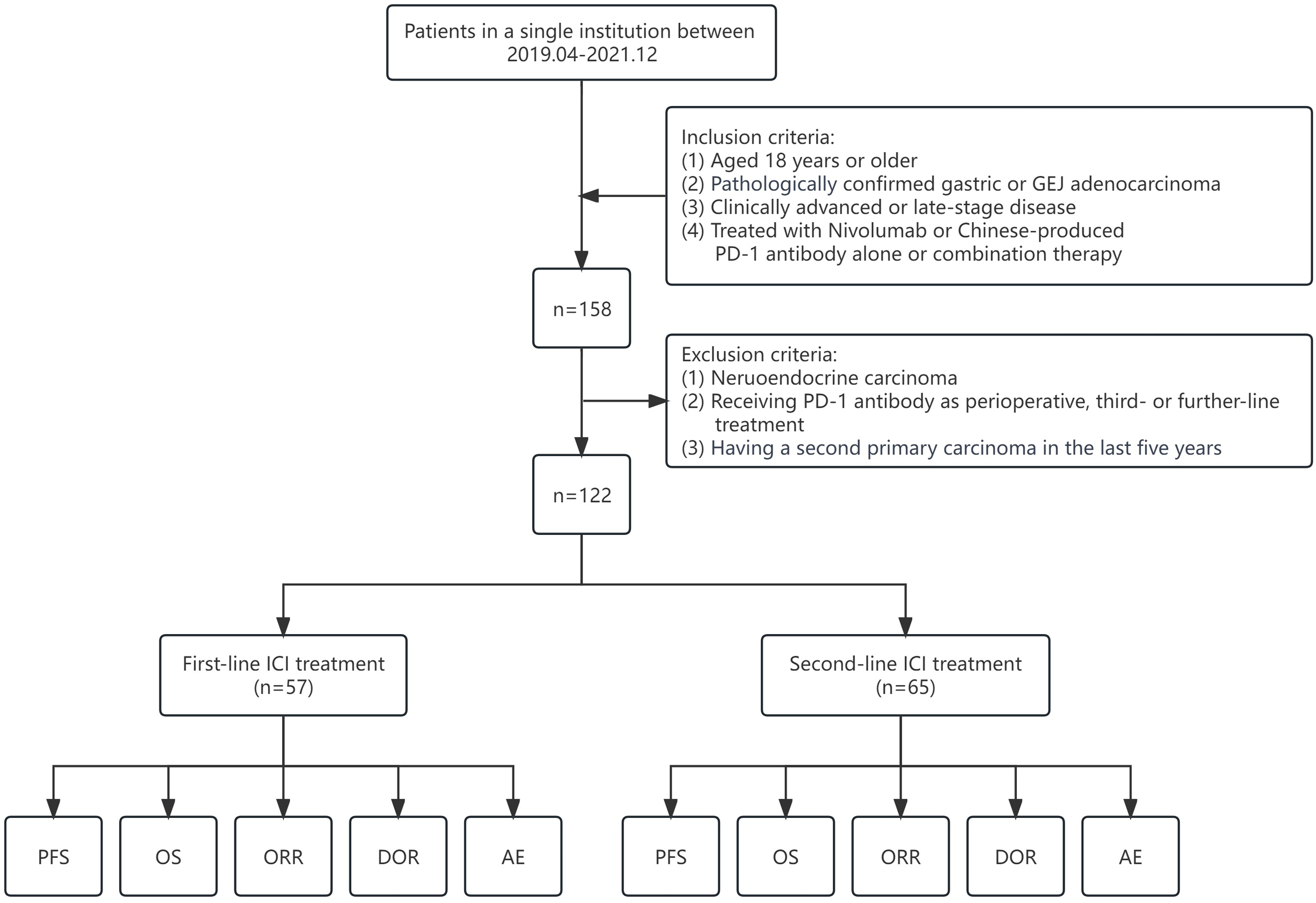
Study flow-chart, including inclusions and exclusions.
Baseline characteristics of patients.

Note. KPS: Karnofsky performance status; MMR: mismatch repair; pMMR: proficient mismatch repair; dMMR: deficient mismatch repair; GC: gastric cancer; gastroesophageal junction (GEJ); MSI: microsatellite instability; FISH: fluorescence in situ hybridization; PD-L1: programmed death-ligand 1; CPS: combined positive score; NA: not available.
Treatment-line and regimes of the patients.

Note. T: nano-albumin paclitaxel; X: capecitabine; P: cisplatin; RT: radiation therapy.
Outcomes
OS, PFS, DOR, and ORR
As of the cut-off date of December 22, 2022, the median follow-up time was 23 months, ranging from 21.8 to 24.2 months. During this follow-up, 86 patients experienced progression events and 65 patients resulted in death events. In the overall cohort, the median OS (mOS) was 17.2 months (95% CI: 14.8–19.5), and the median PFS (mPFS) was 10.9 months (95% CI: 8.0–13.8). The mOS was not reached and 10.9 months (95% CI: 7.9–14.3) for patients in the first-line and second-line treatment, respectively. As for PFS, it was 14.8 months (95% CI: 9.5–20.0) in the first-line group and 5.9 months (95% CI: 4.7–7.1) in the second-line group. Figure 2 and Table 3 present these results.

Clinical outcomes of the cohort. OS (a), PFS (b), and DOR (c) in the First-line and Second-line patients.
Clinical outcomes of patients in various treatment-line.

Note. OS: overall survival; PFS: progression-free survival; DOR: duration of response; ORR: objective response rate; NR: Not Reached;
In the whole cohort, the median duration of response (mDOR) was 9.4 months (95% CI: 7.0–12.3). Specifically, mDOR was 13.3 months (95% CI: 8.0–18.5) in patients receiving first-line treatment, while it was 4.5 months (95% CI: 3.2–5.6) in second-line patients (Figure 2(c) and Table 3).
In the context of first-line therapy, the ORR was 68.4%, PRs were observed in 68.4% of cases, with no instances of CR, 24.6% of patients achieved stable disease (SD), resulting in a disease control rate (DCR) of 93%. While in the second-line therapy, ORR was 41.5%, characterized by PR in 41.5% of cases, and SD in 38.5%, resulting in a DCR of 80%. The objective response is described in Table 4.
Response rate of treatment.

Note. CR: complete response; PR: partial response; SD: stable disease; ORR: objective response rate; DCR: disease control rate.
Peritoneal metastasis had unfavorable OS, while DOR exceeding 10.7 months exhibited a favorable OS.
Subgroup analyses were conducted to further investigate in the whole cohort. Peritoneal metastasis and treatment-line number influenced both OS and PFS, while patients with a good KPS status exhibited a better OS but not PFS, compared to those with a worse KPS status (≤80); patients who reached a longer DOR (>10.7 m) had a better OS, compared with whose DOR <10.7 m. Notably, liver metastasis or lung metastasis did not affected PFS or OS significantly. Specific results showed that mOS was 18.2 months (95% CI: 11.0–25.4) in patients without peritoneal metastasis versus 10.2 months (95% CI: 5.2–15.3) in patients with peritoneal metastasis groups (p = .001). In terms of mPFS, it was 13.1 months (95% CI: 10.1–16.0) in the former group, while it was 7.1 months (95% CI: 3.5–10.7) in the latter group (p = .01) (Figure 3). mOS was 26 months (95% CI: 12.0–40.0) in patients with KPS of 90 versus 14.5 months (95% CI: 11.7–18.0) in patients with KPS ≤ 80 (p = .038).

Subgroup analysis of clinical outcomes of the cohort. OS (a) and PFS (b) of all comers depend on peritoneal metastasis or not; OS of all comers depends on KPS (c); OS of all comers depends on DOR > 10.7 months or DOR <10.7 months (d).
Other important clinical characteristics were treatment-line number and DOR. As expected, patients in second-line treatment exhibited worse PFS and OS compared with patients in first-line treatment, HR was 2.9 (95% CI: 1.67–5.03) for OS, and it was 2.59 (95% CI: 1.64–4.10) for PFS, the results are depicted in Tables 5 and 6. mOS of patients who reached a DOR >10.7 months was not reached, while mOS of patients who reached a DOR <10.7 months was only 7.57 months (95% CI: 5.4–9.7) (p < .001) (Figure 3(d)).
Cox analysis of OS in all comers depends on various clinical characteristics.
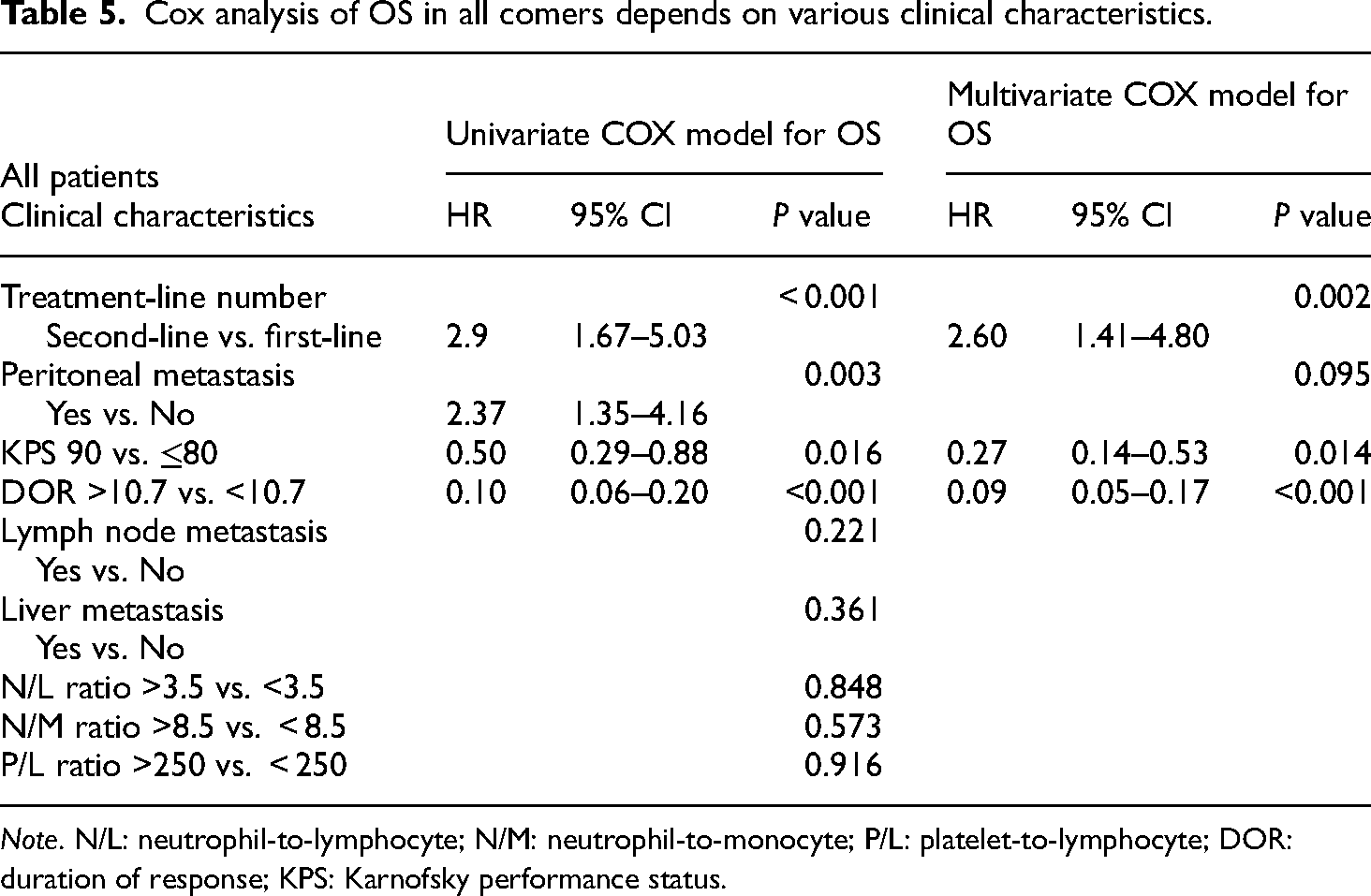
Note. N/L: neutrophil-to-lymphocyte; N/M: neutrophil-to-monocyte; P/L: platelet-to-lymphocyte; DOR: duration of response; KPS: Karnofsky performance status.
Cox analysis of PFS in all comers depends on various clinical characteristics.

Note. N/L: neutrophil-to-lymphocyte; N/M: neutrophil-to-monocyte; P/L: platelet-to-lymphocyte; KPS: Karnofsky performance status.
In the whole cohort, we did not see significant difference in OS and PFS between patients who received regimens containing fluorouracil-based combined with oxaliplatin and those who were administered nano-albumin paclitaxel (Supplement Figures 1(a) and 1(b)). This similar outcomes is independent of treatment-line numbers (Supplement Figures 1(c)–1(f)). In summary, it appears that the choice of chemotherapy regime in combination with anti-PD-1 antibody does not significantly affect OS and PFS of patients whether they are in the first-line or second-line setting.
Association between clinical characteristics and survival outcomes
To investigate the association between clinical characteristics and survival outcomes, we conducted a comprehensive analysis using the Cox regression model. Initially, we analyzed clinical characteristics with the OS and PFS in all the patients. Subsequently, we performed separate analyses for patients receiving first-line and second-line treatments.
Patients with good KPS, longer DOR, and receiving the first-line therapy had a better OS, while peritoneal metastasis was associated with a worse OS and PFS
In the whole cohort, we identified several clinical factors that were significantly correlated with OS. Notably, treatment-line number, KPS, DOR, and peritoneal metastasis demonstrated substantial associations with OS outcome. Of particular significance were treatment-line number and DOR, which exhibited consistency with the clinical pattern. In multivariate analyses using the Cox regression model, patients with a KPS of 90 had a better OS outcome (HR = 0.27, 95% CI: 0.14–0.53), compared with those whose KPS ≤80. Additionally, patients who gained DOR >10.7 months were strongly associated with superior OS outcome (HR = 0.09, 95% CI: 0.05–0.17). Conversely, peritoneal metastasis was associated with a worse OS outcome (HR = 2.37, 95% CI: 1.35–4.16) in univariate COX model, but not in multivariate COX model. Furthermore, peritoneal metastasis was associated with a worse PFS outcome (HR = 1.83, 95% CI: 1.10–3.02). But KPS with 90 was not shown a superior PFS outcome in Cox regression. Tables 5 and 6 show detailed summary of these findings.
Furthermore, in multivariate analyses using the Cox regression model, specifically in the subset of patients receiving their first-line treatment, a KPS of 90 indicated a better OS outcome (HR = 0.28, 95% CI: 0.08–0.97). Similarly, DOR >10.7 months was strongly associated with a better OS outcome (HR = 0.11, 95% CI: 0.04–0.28). Conversely, peritoneal metastasis was associated with a poorer OS outcome (HR = 4.21, 95% CI: 1.63–10.90), as presented in Supplement Table 1.
In the multivariate analyses of PFS using the Cox regression model, which included the first-line treatment patients, peritoneal metastasis was found to be associated with a worse PFS outcome (HR = 2.67, 95% CI: 1.20–5.96). Additionally, P/L ratio >250 was associated with a worse PFS outcome (HR = 2.43, 95% CI: 1.07–5.55). Detailed results are depicted in Supplement Table 2.
In multivariate analyses using the Cox regression model for the second-line treatment patients, both KPS with 90 and DOR >10.7 months were found to be associated with a better OS outcome. These results are described in Supplement Tables 3 and 4. It was worth noting that we did not identify any correlation between N/L or N/M ratios and either OS or PFS in either all patients, patients receiving first-line treatment, or patients receiving second-line treatment (Tables 5, 6 and Supplement Tables 1–3, Supplement Figure 2).
Safety and adverse events
Treatment-related adverse events (TRAEs) of any grade were observed in 28.2% of the overall cohort. There is no treatment-related death. The most commonly reported adverse events (AEs) occurred in the hematological system, including lymphopenia (85.2%), anemia (86%), and white blood cell (WBC) decrease (62.3%). The secondary AEs involved liver dysfunction, which manifested with alanine aminotransferase (ALT) and aspartate aminotransferase (AST) elevation (29.5% and 32%, respectively). It was worth noting that proteinuria occurred in 24.6% of cases, and coexisted with a hypoalbuminemia rate (72.1%), followed by a hyponatremia rate (27%), hypokalemia rate (23.8%), although these were predominantly grades 1–2, with grade 3 occurrences being rare. Additionally, 31.1% of patients manifested with elevated BNP without cardiac symptoms. Grade 3 or 4 AEs were characterized by increased levels of AST, ALT, or gamma-glutamyl transferase (γ-GGT), as well as decreased lymphocyte, WBCs, anemia and hyponatremia, and hypokalemia, and hypoalbuminemia. Pneumonitis was reported in 9.0% patients, all of which were grades 1–2 in severity. Pneumonitis, grade 3 AST, ALT elevation, and B-type natriuretic peptide (BNP) elevation were potentially immunotherapy-related AEs. Table 7 shows details of the specific AEs. An analysis was conducted for treatment-related adverse reactions and their impact on patient survival. Remarkably, we did not find a significant correlation between TRAEs, including those affecting the hematological system and liver dysfunction, and OS or PFS (Supplement Figure 3).
Incidence and grade of treatment-related adverse events.

Note. AEs: adverse events; ALT: alanine aminotransferase; AST: aspartate aminotransferase; BNP: B-type natriuretic peptide; LDH: lactate dehydrogenase; WBC: white blood cell; γ-GGT:gamma-glutamyl transferase.
Discussion
This is a retrospective study to analyze clinical data in a Chinese cohort comprising patients with locally advanced or metastatic gastric or GEJ cancer who underwent treatment with the anti-PD-1 antibody nivolumab or Chinese-produced anti-PD-1 antibody-based therapy. Surprisingly, the observed OS and PFS outcomes in our cohort far surpassed those observed in clinical trials. According to 24-month follow-up results, the OS of nivolumab plus chemotherapy group in CHECKMATE649 was 14.4 and 13.8 months in patients with PD-L1 CPS ≥ 5 and in all randomized patients, respectively. 13 Meanwhile, patients in the ORIENT-16 study, treated with sintilimab plus chemotherapy exhibited an OS of 18.4 and 15.2 months in patients with PD-L1 CPS ≥ 5 and in all randomized patients, respectively. 3 In the CHECKMATE649 Chinese cohort, patients receiving nivolumab plus chemotherapy group experienced an OS of 15.5 and 14.3 months for those with PD-L1 CPS ≥ 5 and for all randomized patients, respectively. 5 Furthermore, patients who received treatment of tislelizumab + chemotherapy achieved a 17.2 months OS for those with PD-L1 tumor area percentage (TAP) ≥ 5%. 4 In summary, the PFS of anti-PD-1 plus chemotherapy was about 7.1–8.5 months in the above clinical studies. While after a median of 23 months follow-up, OS in our first-line treatment patients who received anti-PD-1 antibody and chemotherapy was not reached, the PFS reached 14.8 months in the first-line therapy. In a word, these outcomes far exceeded those reported in the previous studies. We also found that a much longer-lasting DOR of 13.3 months was reached in our first-line therapy, as compared to the median DOR of 8.4–12.2 months reported in the first-line clinical trials.3–5,13 In summary, the Chinese-produced anti-PD-1 monoclonal antibody could bring good clinical outcomes to Chinese patients. A summary of comparative results is provided in Supplement Table 5.
KEYNOTE-061 study indicated that pembrolizumab did not significantly improve OS compared with paclitaxel as a second-line therapy for advanced gastric or GEJ cancer with PD-L1 CPS ≥1, but exhibited a better safety profile than paclitaxel. 8 In another similar clinical trial conducted in Asia, KEYNOTE-063 found the same results, which indicated that the efficacy of pembrolizumab in the second-line Asian patients with advanced PD-L1-positive gastric/GEJ cancer is limited. 9 The mOS, mPFS, and ORR were 8 months, 2 months, and 13% for pembrolizumab. In a retrospective study enrolled 129 Chinese patients receiving the second-line combination therapy of PD-1 antibody and chemo or chemo-anti-angiogenic agents, the ORR was 16.3%, and the DCR was 79.1%. The mPFS was 4.10 months, mOS was 7.60 months. 14 In our second-line therapy group, our patients achieved a 10.9 mOS, 5.9 mPFS, and 41.5% ORR, all above surpassed the results reported in the clinical trial. Fifty-nine patients (91%) underwent anti-PD-1 antibody combined with chemotherapy or Apatinib therapy in our second-line patients. Forty-six patients (76%) received anti-PD-1 combined with chemotherapy containing 27 (58.7%) patients receiving nano-albumin paclitaxel, 14 (30.4%) patients receiving oxaliplatin-based chemotherapy and four patients undergoing irinotecan-based therapy. It was worth stating that all our patients were free of anti-PD-1 treatment in the first-line and adjuvant therapy. A clinical study of the efficacy and safety of anti-PD-1 in combination with nano-albumin paclitaxel in second-line patients who progressed from first-line anti-PD-1 treatment or not is still ongoing at our cancer center.
In real-world molecular biomarker testing patterns and results for advanced gastroesophageal cancers in the USA between 2017 and 2020, PD-L1-positive rate was 20%, while the rate for dMMR/MSI-H was 5%. Intriguingly, only 1% of patients fell into the category of being both PD-L1 and dMMR/MSI-H positive. In total, 4% patients tested for PD-L1 and HER2 positive for both. Only one patient (<1%) in the cohort was classified as HER2-positive and dMMR/MSI-H. 15 However, in the CHECKMATE649 study, more than 80% of patients were CPS ≥1 [2]. On the other hand, MSI-H exhibited a range of 3–7% in the CHECKMATE649, KEYNOTE-062, and KEYNOTE-059 trials.2,7,16
In our cohort, 70.5% of patients underwent HER2 testing, with 18.8% of patients having undergone a test of PD-L1 before they received anti-PD-1 antibody therapy between April 2019 and December 2021. Moreover, merely 12.3% had the MSI status results before they initiated the anti-PD-1 antibody treatment. The PD-L1 CPS positive (CPS > 1) rate was 13.9%, notably lower than that was reported in the USA, potentially due to the lower detection rate in our cohort. Furthermore, the MSI-H proportion was 3.3%, similar as the level reported in the US real-world study and the clinical trials. Another real-world study on gastric biomarkers is still ongoing at our center.
A series of studies have been conducted to explore the correlation between biomarkers and the effectiveness of immunotherapy. A systematic review and meta-analysis encompassed in total of 6488 patients, and found that efficacy and survival advantages improved with a higher PD-L1 CPS. CPS ≥1 was the cutoff value for gaining survival benefit from ICI monotherapy. 17 Additionally, a meta-analysis of randomized clinical trials including phase III KEYNOTE-062, CHECKMATE649, JAVELIN Gastric 100, and KEYNOTE-061 trials, involved 2545 patients, and it was indicated that patients with MSI-H GC should be regarded as a specific and highly immunosensitive population deserving of dedicated clinical trials. 18 Furthermore, a post-hoc analysis of the phase II KEYNOTE-059 single-arm trial (174 patients) and the phase III KEYNOTE-061 (514 patients) and KEYNOTE-062 randomized trials (682 patients). Specifically, the MSI-H rate was 4.0%, 5.3%, and 7.3 in KEYNOTE-059, KEYNOTE-061, and KEYNOTE-062, respectively. Analysis of OS, PFS, and ORR in these three trials, indicated that MSI-H status may be a biomarker for pembrolizumab therapy among patients with advanced G/GEJ cancer regardless of the line of therapy. 19 Additionally, a study was conducted on the relationship between PD-L1 expression and the clinical features of GC. 20 They found that PD-L1 was expressed in 59.3% of GC patients and was associated with MSI and Epstein-Barr virus (EBV) positive, and OS was longer in patients with PD-L1 CPS ≥ 1 tumors than in those with PD-L1 CPS < 1 tumors. While a review focusing on the outcomes of low PD-L1 expressing subgroup in the first-line ICI in gastric or esophageal adenocarcinoma (EAC) included patients in CHECKMATE649, KEYNOTE-062, and KEYNOTE-590 showed that patients with PD-L1 CPS 1–4 from CHECKMATE649 and PD-L1 CPS 1–9 from KEYNOTE-062 did not benefit from the addition of ICI to chemotherapy. 21 Unfortunately, due to the small number of patients and most of them with unknown MSI and PD-L1 results, it is not feasible to conduct subgroup analysis on the relationship between molecular biomarkers and prognosis. A real-world study conducted in Germany showed that the third-line anti-PD-1 treatment of the PFS and OS was 2.1 and 6.3 months, respectively. Good performance was associated with better outcomes. 22 Nong Xu et al. found that N/L ratio was a strong predictor of PFS and OS in patients with advanced GC receiving immune checkpoint inhibitors combined with chemotherapy. 23 A lower N/L ratio was correlated with a favorable PFS and OS and was validated as an independent prognostic factor in another real-world cohort. 23 However, we did not find a correlation relationship between the N/L ratio and OS or PFS outcomes in our cohort.
The most common side effects of treatment were liver dysfunction, and abnormality in the hematological system, including lymphopenia, anemia, WBC decline. A post-hoc trial analysis showed that nivolumab plus chemotherapy was associated with a statistically significant and clinically important gain in quality-adjusted survival compared with chemotherapy alone among previously untreated patients with advanced GC/GEJC/EAC. 24 A study on AEs showed that early AE (event occurrence at or before day 30 from the initial treatment date) biomarkers were associated with clinical outcomes. Patients experienced with low-grade AEs (1 or 2) had improved PFS and OS, and were associated with disease control. In contrast, patients with early development of high-grade AEs tended to have poorer PFS and OS, and correlated with progressive disease. 25 The Spinnaker study demonstrated that a better OS and PFS survival outcome benefits in patients with immune-related adverse event (irAEs) in the first-line combination of immuno-chemotherapy in lung cancer patients. 26 Furthermore, in a Chinese real-world setting, irAEs in lung cancer patients were commonly observed, with pneumonitis being the most common fatal irAEs, patients who presented any grade irAEs may tend to achieve a longer PFS. 27 A comprehensive meta-analysis of 54 studies revealed that patients who experienced irAEs achieved a significantly higher ORR and longer PFS and OS than those who did not experience irAEs. Subgroup analysis of irAE types indicated that specific irAEs, such as thyroid dysfunction and gastrointestinal, skin, or endocrine irAEs, were associated with better PFS and OS. However, no significant differences were observed between patients with pneumonitis or hepatobiliary irAEs. 28 In our cohort, we also analyzed the clinical outcome based on the presence and grade of irAEs. However, we did not find the difference in OS between patients who had liver dysfunction or not, between those with grade 1–2 or grade 3–4 WBC decline, between patients who demonstrated lymphocyte decline or not, between those with neutrophil decline or not, or between those with or without platelet decline. The main reason for the inconsistency between our results and those previously reported may be that our study was retrospective and there was a data bias in the collection of adverse reactions.
In a cohort of 43 renal cell cancer patients, higher plasma CXCL10 levels both at baseline and post-first dose were found to be significantly associated with the occurrence of grade 2 or higher irAEs. 29 Therefore, it is crucial to monitor dynamic changes in cytokine levels, such as CXCL10, interleukin (IL)-6, and IL-10 in the peripheral blood of patients receiving immunotherapy. In another real-world study of 81 cancer patients who had undergone ICI-associated myocarditis, it was found that prior chemotherapy, classical serum cardiac markers, as well as a higher neutrophil ratio, were related to poorer cardiac outcomes, whereas higher lymphocyte and monocyte ratios were predictors of favorable cardiac outcomes. Moreover, the CD4+ T cell ratio and CD4/CD8 ratio were negatively related to Common Terminology Criteria for Adverse Events (CTCAE) grades 3–5. 30 Additionally, another meta-analysis of studies suggests that absolute eosinophil count, N/L ratio, and P/L ratio may serve as predictors of the emergence of irAEs. 31 These prognostic factors suggest that clinicians should be highly concerned about immune-related adverse cardiac events during their patients receiving immunotherapy since the incidence of grade 3–5 is more than 50% once a cardiac-related AE occurs. Although the clinical role of AEs after immunotherapy has not been fully determined, we believe that it may be a double-edged sword, on the one hand, it can react to the AEs brought about by the treatment, and on the other hand, it can be a predictor of the efficacy of immunotherapy, which also requires clinicians to pay great attention to patient's AEs, and to closely follow-up the patient, even after the patient is discharged from the hospital. Doctors should do regular follow-up, make diagnosis at the earliest time, and make intervention as early as possible, in order to avoid the occurrence of fatal AEs.
One limitation of our cohort is that our patient number is much low, and may manifest some heterogeneous, especially on the tumor genetics or other bio-characteristics, and some patients missed the information of PD-L1 and MSI detection. Only a small number of patients with detected PD-L1 and MSI status, which is insufficient to support the conduct of a biomarker subgroup analysis for outcomes. Another is that thisretrospective study has data bias in the collection of adverse reactions. Our data concerning on first-line and second-line therapies only included patients who received anti-PD-1 combination therapy, we did not include the control group not receiving anti-PD-1 treatment at the same treatment period.
Conclusion
In conclusion, this is a retrospective analysis on the efficacy and safety of anti-PD-1 antibody-based treatment in the first-line and second-line GC patients in our single center. In this study, the long-term OS and PFS benefit from a combination of immunotherapy and chemotherapy, and an acceptable safety profile, providing strong support for the adoption of chemotherapy plus anti-PD-1 antibody treatment as the standard first-line approach for patients with advanced gastric, GEJ adenocarcinoma. However, further multi-center, phase III, randomized controlled studies must be conducted to detect the efficacy and safety of anti-PD-1 and chemotherapy or anti-angiogenesis therapy in the second-line treatment.
Supplemental Material
sj-docx-1-sci-10.1177_00368504241272703 - Supplemental material for Efficacy and safety of anti-programmed death-1 antibody-based combination therapy in advanced or metastatic gastric or gastroesophageal junction cancer in Chinese patients: A real-world study
Supplemental material, sj-docx-1-sci-10.1177_00368504241272703 for Efficacy and safety of anti-programmed death-1 antibody-based combination therapy in advanced or metastatic gastric or gastroesophageal junction cancer in Chinese patients: A real-world study by Yifan Gao, Haoqian Li, Lei Qiu, Hongtu Yuan, Qing Fan, Zuoxing Niu, Ligang Xing, Mingxing Li and Dandan Yuan in Science Progress
Footnotes
Consent for publication
All authors have gone through the final draft of the manuscript and agreed to the submission/publication.
Contributorship
Yifan Gao: conceptualization, follow-up of patients, investigation, formal analysis of patients PFS and OS, drafting of the outcome part, and revision draft. Haoqian Li: conceptualization, patients’ data collection, investigation, formal analysis, and manuscript drafting. Lei Qiu: conceptualization, formal analysis of the patient treatment regime and treatment line, and did analysis of Cox regression between PFS, OS, and clinical characteristics. Hongtu Yuan: formal analysis patients pathology and biomarkers. Qing Fan: formal analysis the treatment-related adverse events, investigation, and methodology. Zuoxing Niu: conceptualization, supervision, and writing–review and editing. Ligang Xing: funding acquisition, supervision, and writing–review and editing. Mingxing Li: conceptualization, funding acquisition, supervision, and writing–review and editing. Dandan Yuan: conceptualization, funding acquisition, methodology, visualization, and writing–review and editing.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the National Natural Science Foundation of China supported to Ligang Xing (82030082), Natural Science Foundation of Shandong Province, China (support to Dandan Yuan, ZR2021MH191, Qing Fan, ZR2020QH205), and Natural Science Foundation of Sichuan Province, China (support to Mingxing Li (No. 2023NSFSC1848).
Ethics approval and consent to participate
This research is conducted according to the World Medical Association Declaration of Helsinki. The study was approved to be conducted and published by the ethics committee of Shandong Cancer Hospital (SDTHEC2022001017). In this retrospective study, formal written informed consent from patients was not required, patient information was anonymized, and all data were kept confidential.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
