Abstract
Background:
Ulinastatin has been applied in a series of diseases associated with inflammation but its clinical effects remain somewhat elusive.
Objective:
We aimed to investigate the potential effects of ulinastatin on organ failure patients admitted to the intensive care unit (ICU).
Methods:
This is a single-center retrospective study on organ failure patients from 2013 to 2019. Patients were divided into two groups according to using ulinastatin or not during hospitalization. Propensity score matching was applied to reduce bias. The outcomes of interest were 28-day all-cause mortality, length of ICU stay, and mechanical ventilation duration.
Results:
Of the 841 patients who fulfilled the entry criteria, 247 received ulinastatin. A propensity-matched cohort of 608 patients was created. No significant differences in 28-day mortality between the two groups. Sequential organ failure assessment (SOFA) was identified as the independent risk factor associated with mortality. In the subgroup with SOFA ≤ 10, patients received ulinastatin experienced significantly shorter time in ICU (10.0 d [interquartile range, IQR: 7.0∼20.0] vs 15.0 d [IQR: 7.0∼25.0];
Conclusion:
Ulinastatin was beneficial in treating patients in ICU with organ failure, mainly by reducing the length of ICU stay and duration of mechanical ventilation.
Keywords
Introduction
Multiple organ dysfunction syndrome (MODS) is a clinical syndrome caused by different etiologies. Severe infection, trauma, major surgery, and secondary damage such as hypovolemic shock, sepsis shock, and ischemia-reperfusion injury are recognized as the common factors accounting for the occurrence of MODS. Despite great progress in pharmacological interventions and organ support technology for critical patients, MODS remains the leading cause of death in current intensive care unit (ICU) practice. 1 The pathogenesis of MODS is complex, while an excessive and uncontrolled inflammatory response of the body along with the immunologic derangement is considered to be crucial in MODS progression.2,3 Exaggerated inflammatory responses cause tissue injury while the injured tissue becomes a new initiator to further inflammation response, which results in a vicious recycle to aggravate the tissue and organ damage. The past decades have witnessed some solutions that were developed to ameliorate inflammation disturbance, but no significant clinical benefits have been confirmed yet. 4
Ulinastatin is a broad-spectrum hydrolase inhibitor isolated and purified from the urine of healthy humans. It is a glycoprotein that can inhibit the activity of a variety of proteases, including trypsin, kallikrein, chymotrypsin, plasmin, thrombin, and neutrophil elastase.5,6 The mechanism accounting for the therapeutic benefits of ulinastatin mainly depends on the inflammatory microenvironment rebalancing process. A series of studies suggested that ulinastatin can activate polymorpho-nuclear leukocyte (PMN), macrophages, and platelets, inhibit the release of (PMN)-derived elastase and other proinflammatory cytokines or interleukins (TNF-α, IL-1, IL-6, and IL-8), upregulate the antiinflammatory cytokines, and rebalances the inflammatory microenvironment ultimately.7–9 Moreover, ulinastatin exerts the organ protective effect by suppressing reactive oxygen species and maintaining the integrity of vascular endothelium, by targeting different signal pathways.9,10
Recent years, ulinastatin has been widely used in China, Japan and some other Asian countries for organ protective therapy in different kinds of diseases. A meta-analysis from China included 13 randomized controlled trial (RCT) and two prospective studies demonstrated that ulinastatin reduced the incidence of MODS and improved all-cause mortality in sepsis patients. 11 Another prospective multicentral cohort study targeting ARDS patients manifested respiration protective effect by reducing mechanical ventilation time and ICU treatment time. 12 Similarly, high dose of ulinastatin exhibited potential beneficial effect in preventing COVID-19 deterioration. 13 Moreover, ulinastatin has been administrated in hepatic disease, renal disease, brain trauma, and vasculitis syndromes such as Kawasaki disease, as a potentially beneficial immunomodulator drug for organ protective thearapy.14–17 The clinical research mentioned above indicated clinical benefits acquired by the patients treated with ulinastatin. However, most of these studies targeted some special group of diseases or surrogated endpoint instead of the outcome. The therapeutic efficacy and prognostic effect of ulinastatin in critical patients with organ failure remain poorly understood, especially lacking in real-world data. In recent years, data from the real world have attracted increasing attention in medical research. Multiple real-world studies of drug evaluation have been conducted to provide valuable guidance in clinical medicine, such as the effectiveness and safety evaluation of mepolizumab in asthma patients, 18 the study of comparative antidepressants effectiveness on geriatric depression, 19 and the effectiveness and economic evaluation of polyene phosphatidylcholine in patients with liver diseases. 20 In brief, the real-world study, which is mutually complementary to RCT, more closely reflects the clinical practice with good external validity and provides a reference for the diagnosis and treatment of patients to the greatest extent through the analysis of real-world data. Therefore, we conducted a study based on real-world data to investigate the therapeutic effect of ulinastatin on patients with organ failure that is attributed to different etiology.
Methods
Study design
This was a single-center retrospective study on organ failure patients from 2013 to 2019. It was performed in a general ICU at a tertiary general hospital in Shanghai, China, which was approved by the Research Ethics Committee of Shanghai Changzheng Hospital (2020SL023). Study data has been fully deidentified, and all confidential information of the patients has been deleted, in accordance with the CIOMS/WHO International Ethical Guidelines for Health-related Research Involving Humans (2016). Consequently, the requirement for informed consent was waived in this retrospective study.
Inclusion criteria and exclusion criteria
The inclusion criteria were: (1) admitted to ICU with sequential organ failure assessment (SOFA)≧2; (2) received respiratory support; (3) surviving over 24 h after ICU admission; (4) surviving over 24 h after ulinastatin initiation. The exclusion criteria were: (1) the patients with an initial time of ulinastatin over 5 days prior to ICU admission or over 5 days after ICU admission; (2) The patients with missing primary study data (such as patient ID and essential clinic index).
Date collection
A total of 841 patients were enrolled according to the inclusion and exclusion criteria after data cleansing, including 247 cases in the experimental group (using ulinastatin) and 594 cases in the control group (no use of ulinastatin). The workflow of data processing was displayed in Figure 1. Personnel information of patients was anonymized and each patient was given a unique ID to avoid duplicate records. The demographic information (age, gender, smoking history), medical history, comorbidities, laboratory parameters on the routine blood test, SOFA, and clinical outcomes including length of ventilator duration, length of ICU stay, and all-cause mortality, were acquired from the electronic medical chart system and lab assay information system.

Flowchart of data processing.
Exposure and outcomes definition
The exposure of this study was defined as intravenous administration of ulinastatin (Guangdong Tianpu Biochemical Pharmaceutical Co., Ltd, H20040506, 100,000 U) with an initial dose ≥ 900,000u daily. The patients were classified into the ulinastatin group (UTI group) and the nonulinastatin group (non-UTI group).
The primary outcome was 28-day all-cause mortality. Secondary outcomes included the following: (1) the length of ventilator duration, (2) the length of ICU stay, (3) the ratio of ICU stay to hospital stay, and (4) the decreasing amplitude of SOFA at day 14 and day 28 after admission.
Multivariate analysis
Multivariate logistic regression was employed to analyze whether ulinastatin use was an independent influencing factor (protective factor) of mortality within 28 days. Besides ulinastatin use, comorbidity, smoking, gender, age, SOFA score, and C-reactive protein (CRP) were included in the multivariate analysis based on clinical knowledge.
Subgroup analysis
In subgroup analysis, we further explored the effectiveness of ulinastatin in survival patients with SOFA ≤ 10.
Statistical analyses
Propensity scores matching (PSM) analysis using nearest match was applied to eliminate the selection bias from the potential confounding factors, which included age, gender, smoking history, baseline SOFA, and CRP between groups. Two groups were matched at a ratio of 1:2 with the caliper 0.2, and the balance between groups was recognized as the covariates’ standardized difference less than 0.1.
The continuous variable was described by the median and interquartile range (IQR), and the categorical variable was presented as frequency and percentage. The statistical differences in continuous variables were tested using the analysis of the Wilcoxon rank sum test, while the chi-squared test or Fisher exact test was employed for the analysis of categorical data.
Multivariate logistic regression was applied to calculate the risk of outcomes. Variables adjusted in the multivariate analysis included age, gender, smoking history, SOFA, CRP, comorbidities, and ulinastatin administration.
All reported probability values were two-sided, and a level of 0.05 was used to declare statistical significance. Analyses were performed using the statistical R software (version 4.1.2) and SPSS 21.0.
Results
Clinical characteristics of the patients
A total of 841 patients who met the inclusion criteria were enrolled, among which 247 (29.4%) received ulinastatin were assigned to the ulinastatin group (UTI group), and the other 594 (70.6%) were classified as the control group (Figure 1). The average daily dose of ulinastatin in the UTI group was 750,000 u [IQR: 570,000, 900,000] and no adverse events associated with the agent were noted in the UTI group. According to the unadjusted analysis, the patients in the ulinastatin group were younger (60.0 years [IQR: 47.0–70.5] vs 66.0 years [IQR: 52.0–75.0]), with higher CRP level (89.6 mg/L [IQR: 47.0–138.0] vs 58.2 mg/L [IQR: 27.3–108.0]), and more severe organ failure (SOFA, 9.0 [IQR: 6.0–13.0] vs 7.0 [IQR: 4.0–10.0]). No significant differences in gender, smoking history, and comorbidity between the ulinastatin and the control group (Table 1). After propensity score matching, a total of 608 patients were successfully matched, including 229 patients in the UTI group and 379 patients in the control group, all characteristics were balanced and no significant difference existed between the two groups (Table 1).
The comparison of the baseline data before and after propensity score matching.

Abbreviations: CRP: C-reactive protein; DM: diabetes mellitus; HPT: hypertension; COPD: chronic obstructive pulmonary disease; MT: malignant tumor; CKD: chronic renal disease; and IS: postorgan transplantation or immunosuppressive status; SOFA: sequential organ failure assessment.
‡Comorbidity is defined as the following diseases existed or not.
The primary illness that contributed to organ failure in the study cohort consisted of severe pneumonia (45.9%), sepsis (25.0), multitrauma (19.2%), postcardiac arrest syndrome (2.5%), acute pancreatitis (2.1%), and others diseases (5.3%) (Table 2).
Primary disease in the cohort after propensity matching.

Primary outcome and secondary outcome
No significant differences were observed in the 14-day and 28-day mortality, the length of ICU stay, duration of mechanical ventilation, and the decrease of SOFA and CRP levels between the UTI group and control group (Table 3).
The primary and secondary outcomes between the UTI group and non-UTI group.
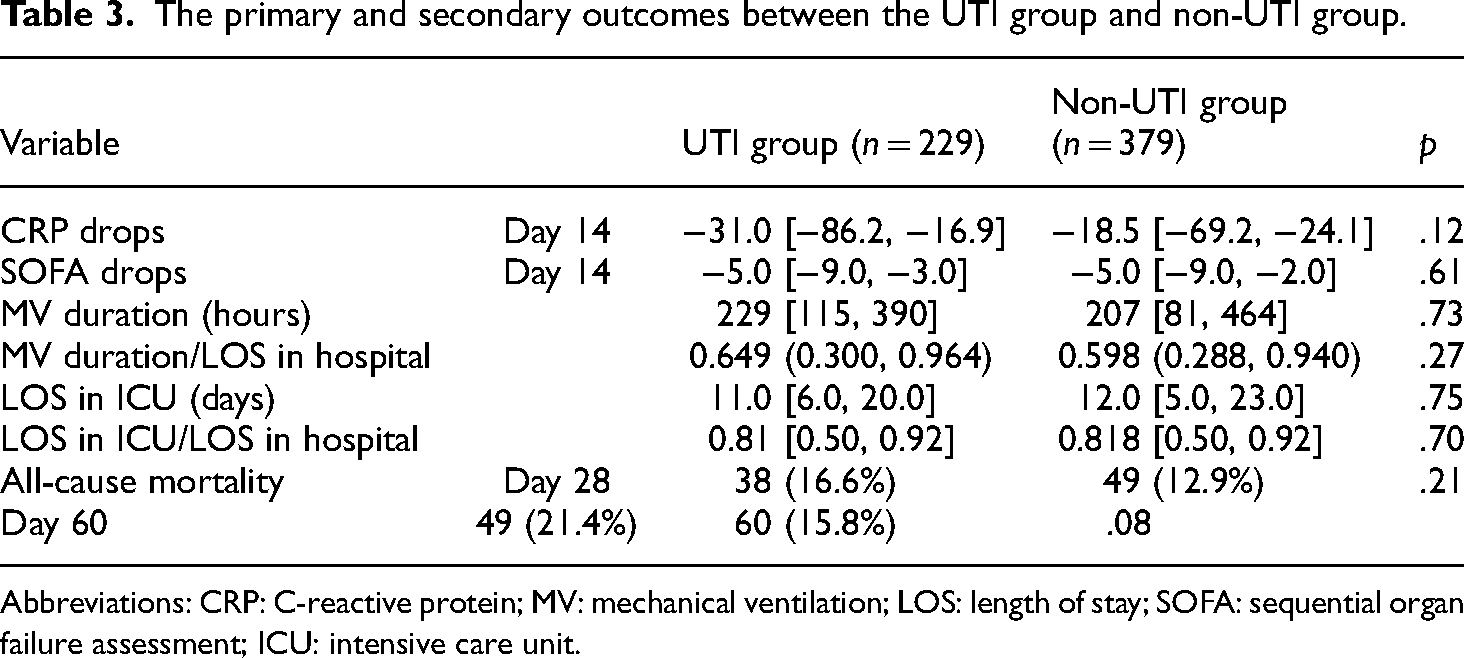
Abbreviations: CRP: C-reactive protein; MV: mechanical ventilation; LOS: length of stay; SOFA: sequential organ failure assessment; ICU: intensive care unit.
Multivariate regression analysis
The multivariate regression model was employed to investigate the possible confounding factors related to the 28-day outcomes. The results suggested that SOFA was independently associated with 28-day mortality (Table 4).
Multi-variable logistic analysis of risk factors in 28-day mortality.

Abbreviations: CRP: C-reactive protein; SOFA: sequential organ failure assessment.
Receiver operating characteristic curves (ROCs) were used to assess the predictive value of the SOFA in prognosis after multivariate regression analysis. The optimal threshold for SOFA that discriminates between survival and nonsurvival is 10.5, with an area under the curve of 0.72 (95% CI [0.67, 0.77]) (Figure 2).
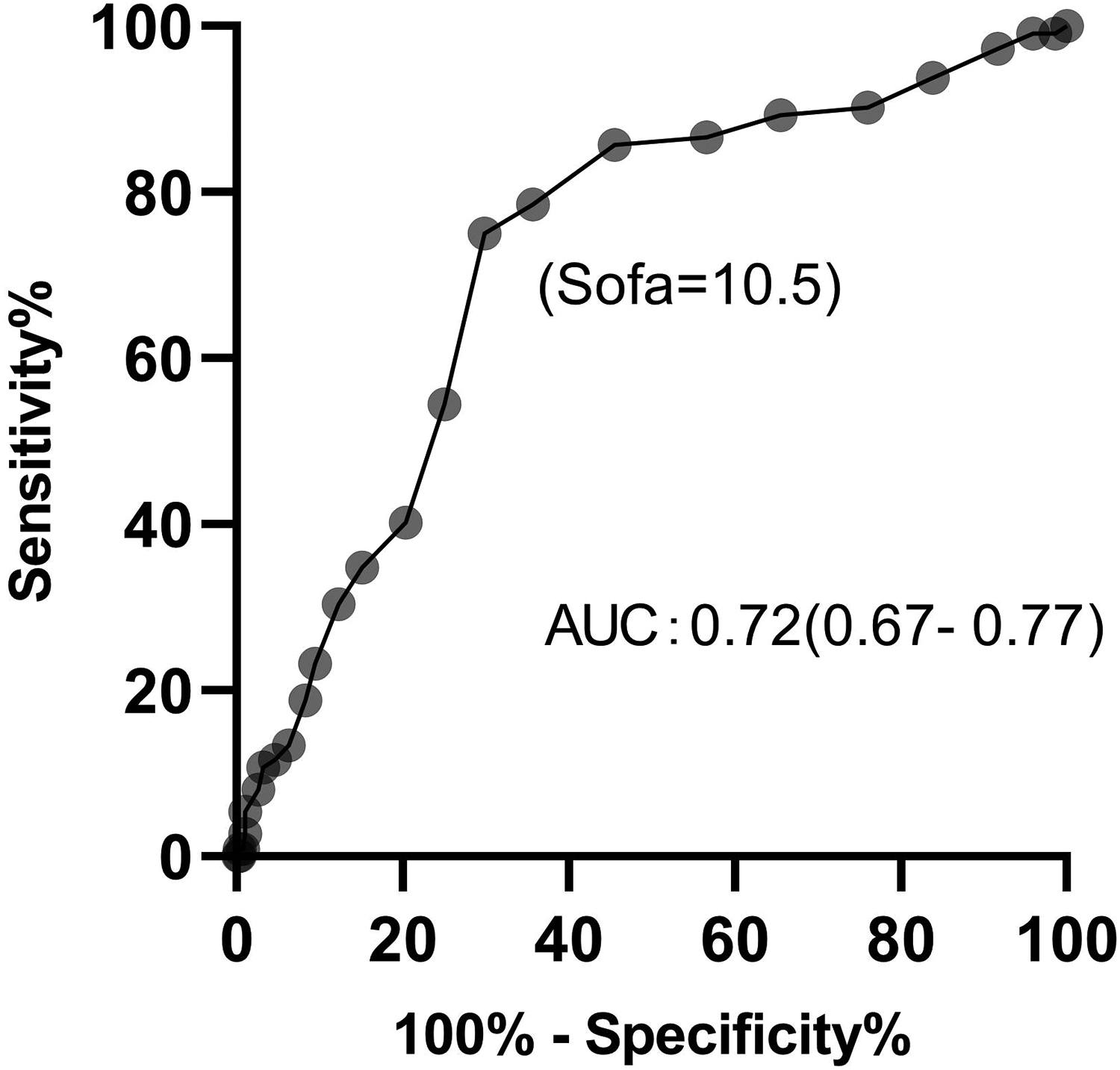
ROC of SOFA in mortality prediction. The area under the curve (AUC) is 0.72 (95% CI: 0.67–0.77). SOFA = 10.5 is the optimal threshold that discriminates against survival and nonsurvival.
Subgroup analysis in patients with SOFA 10
According to the results of multivariate regression analysis and ROC assessment, SOFA was identified as the unique independent prognostic risk factor with an optimal threshold of 10.5. In subgroup analysis, we focused on the potential efficacy of ulinastatin in the patients with SOFA ≤ 10 instead. Some results deserving attention were acquired in this subgroup analysis: the length of ICU stay (10.0 d [IQR: 7.0∼20.0] vs 15.0 d [IQR 7.0∼25.0]) and mechanical ventilation (222 h [IQR: 114∼349] vs 251 h [IQR 123∼499]) were significantly decreased in the UTI group comparing with the control group. In addition, the patients in the UTI group demonstrated a larger decrease in SOFA value and CRP level on the day7 after ICU admission. However, no significant difference was observed in 28-day mortality between the two groups (Table 5).
Subgroup comparison of the patients with SOFA≦10.

Abbreviations: CRP: C-reactive protein; MV: mechanical ventilation; LOS: length of stay; SOFA: sequential organ failure assessment; ICU: intensive care unit.
Discussion
This is a retrospective study based on real-world data aimed at investigating the impact of ulinastatin on patients with organ failure. The effect of ulinastatin introduction in critical patients was inconsistent in previous research. Some studies provided evidence for the effect of ulinastatin in clinical. For instance, a multicenter RCT conducted by Karnad et al. found that ulinastatin injection decreased the mortality of patients with severe sepsis (7.3% vs 20.3%,
The primary result of our study cohort did not show prognostic benefit from ulinastatin therapy for organ failure patients, but it is noted that the SOFA score, by which organ failure was defined, was identified as a unique independent risk factor of 28-day mortality in further multivariable regression analysis. Existing studies have suggested that SOFA had a strong predictive value of prognosis for critical patients.28,29 Ferreira et al. carried out a study of ICU patients and illustrated that the initial SOFA>8 on ICU admission is the optimal threshold of discrimination between survival and nonsurvival, while the highest SOFA>11 during ICU stay corresponded to a high mortality of more than 80%. 30 Extremely elevated SOFA score often suggests severe or irreversible organ damage of patients, who may respond poorly to any pharmacotherapy. In addition, severe organ failure could have a decisive influence on the length of ICU stay, intensity of respiratory support, and the final outcome. Considering these factors above may in part explain why ulinastatin therapy did not exhibit obvious benefits for the patients with severe organ failure in our study cohort, in subgroup analysis, we attempt to focus on the patients with SOFA ≤ 10, which is defined as the threshold value of mortality prediction. The subgroup analysis indicated some inspiring results that patients who received ulinastatin therapy experienced a decreased time of mechanical ventilation and shorter length of ICU stays. Given the substantial influence of mechanical ventilation application on the length of ICU stay, the reduction of ICU stays in the ulinastatin group is probably attributed to the shortening term of mechanical ventilation. Previous animal studies have shown that ulinastatin could exert a pulmonary protective effect via alleviating inflammatory response, ameliorating permeability of capillary endothelium, and suppressing ROC production.10,31 Besides, A meta-analysis involving 33 RCTs from China revealed the positive effect of ulinastatin on pulmonary protection, the patients with ARDS using ulinastatin acquired significant benefits by reducing the duration of mechanical ventilation (SMD = −1.29, 95% CI: [−1.76∼−0.83]), shorter ICU stay (SMD = −1.38, 95% CI [−1.95∼−0.80]) and lower mortality (RR = 0.51, 95% CI [0.43∼0.61]). 32 Another study targeting COVID-19 patients in the early stage of the pandemic showed that ulinastatin treatment significantly prevented respiration deterioration and exhibited a potential beneficial effect. 13 Nevertheless, ulinastatin administration remains insufficient to improve the 28-day survival rate of these patients with SOFA ≤ 10 in subgroup analysis. It may be difficult for a single drug to have a decisive impact on the overall prognosis of critical patients, the results of our study did not suggest the role of ulinastatin in improving the survival of patients with organ failure, but the results of subgroup analysis suggested that the respiratory protective effect attributed to the decreased duration of mechanical ventilation and ICU stay. Shorter time of mechanical ventilation and ICU stay often means fewer associated complications, lower healthcare cost and less pain of the patients. It instructs that the role of ulinastatin in the protection of organ function, especially in respiratory function, is a promising field, deserved further clinical and basic medical research.
It is worth mentioning that, due to the lack of a robust guideline in dose for ulinastatin for critical patients so far, the therapeutic dose of the agent mainly depended on the physician's discretion. In our study, no preset restrict range was set for the ulinastatin dose in the entry criteria. The average dose in the ulinastatin group is equal to the similar study, while no adverse event was observed. 33 A large-scale assessment aimed at investigating the real-world safety of ulinastatin in China with 11,252 patients demonstrated a safety and tolerance profile as well. 34
As a retrospective study based on a real-world database, the current study investigated the clinical data up to nearly 7 years without presetting upper limits of age, specific etiologies, or other conditions, that may be more representative of “real-world” practice compared to the results from the restricted populations in randomized control trials.
Limitation
There are several limitations to our study. Firstly, owning to the inherent characteristic of retrospective observational study, we could not assess the changes of inflammatory and immune factors which aid in exploring the potential mechanism involved in ulinastatin therapy, therefore no definite causal inferences can be made from the current study. Secondly, since it is nonexperimental, the outcome is inevitably interfered with by various confounding factors. Herein, we applied PSM to correct for bias, pursuing the maximum homogeneity of the characteristic variables between two groups of samples. However, after PSM, the available sample size was reduced substantially. In addition, some cases had to be removed in the early stage due to the important variables missing, such as the long-term indexes after admission, according to the exclusion criteria. It follows that it is essential to train the clinicians to record clinic data consistently over as long a period as possible. Also, power analysis was not performed in this study, which should be added in further research to estimate the sample size. Besides, we had some uncommon findings, such as the inverse results of ulinastatin effectiveness in long-term respiratory support, for which we could not find a rational explanation based on current information. Thus, further research with large sample size and a longer observational period is warranted to verify the results of this study and assess the effect of ulinastatin on long-term prognosis.
Conclusion
This is a 7-year real-world study on the therapeutic effect of ulinastatin in critical patients with organ failure. We found that ulinastatin was beneficial in treating patients in ICU with organ failure, mainly by reducing the length of ICU stay and duration of mechanical ventilation. This study provides the evidence of therapeutic effect of ulinastatin in the clinical setting.
Footnotes
Authors' Note
Lixue Wu, Deduo Xu, and Yanru Liu have contributed equally to this work.
Authors’ contribution
LW, DX, and YL conceived, designed, carried out the study and wrote the draft of the paper, YS, ZL, and WC searched literature, proposed the study, guided the researching process and provided funding support. WJ, JZ, ZY, and FG contributed to data collection, data analysis, and drafting. WL and XT took part in data interpretation as well as manuscript revision. All authors approved the final version.
Availability of data and materials
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
This research has been subjected to ethical review and approved by the Research Ethics Committee of Shanghai Changzheng Hospital (No. 2020SL023). All data involved in this retrospective study has been fully deidentified, and all confidential information of the patients has been deleted, in accordance with the CIOMS/WHO International Ethical Guidelines for Health-related Research Involving Humans (2016), requirement for informed consent was waived.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shanghai public health academic leader project, Shanghai Municipal Planning Commission of Clinical Research Fund, Shanghai Yiyuanxinxing Youth Medical Talents Development Program, Important and special project of National Science and Technique (Grant Nos. GWVI-11.2-XD35, 202040238, 202240279, SHWSRS(2020)087, and 2022YFC3103001-004).
