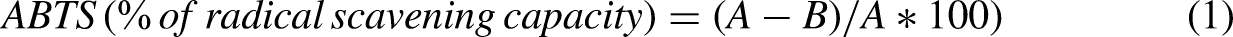Abstract
The current emphasis within the cosmetic market on sustainable ingredients has heightened the exploration of new sources for natural, active components. Actinomycetota, recognized for producing pigments with bioactive potential, offer promising functional cosmetic ingredients. This study aimed to optimize pigment and antioxidant metabolite production from the Gordonia hongkongensis strain EUFUS-Z928 by implementing the Plackett-Burman experimental design and response surface methodology. Extracts derived from this strain exhibited no cytotoxic activity against human primary dermal fibroblast (HDFa, ATCC® PCS-201-012™, Primary Dermal Fibroblast; Normal, Human, Adult). Eight variables, including inoculum concentration, carbon and nitrogen source concentration, NaCl concentration, pH, incubation time, temperature, and stirring speed, were analyzed using the Plackett-Burman experimental design. Subsequently, factors significantly influencing pigment and antioxidant metabolite production, such as temperature, inoculum concentration, and agitation speed, were further optimized using response surface methodology and Box-Behnken design. The results demonstrated a substantial increase in absorbance (from 0.091 to 0.32), DPPH radical scavenging capacity (from 27.60% to 84.61%), and ABTS radical scavenging capacity (from 17.39% to 79.77%) compared to responses obtained in the isolation medium. The validation of the mathematical model accuracy exceeded 90% for all cases. Furthermore, liquid chromatography coupled with mass spectrometry (LC-MS) facilitated the identification of compounds potentially responsible for enhanced pigment production and antioxidant capacity in extracts derived from G. hongkongensis. Specifically, six carotenoids, red-orange pigments with inherent antioxidant capacity, were identified as the main enhanced compounds. This comprehensive approach effectively optimized the culture conditions and medium of a G. hongkongensis strain, resulting in enhanced carotenoid production and antioxidant capacity. Beyond identifying bioactive compounds and their potential cosmetic applications, this study offers insights into the broader industrial applicability of these extracts. It underscores the potential of G. hongkongensis and hints at the future utilization of other untapped sources of rare actinomycetes within the industry.
Introduction
The burgeoning desire for a healthier, youthful appearance has fueled substantial demand and market opportunities, becoming a global priority. Cosmetics, designed for direct application to the human body's external surfaces, serve four primary functions: maintaining good condition, altering appearance, protecting, and addressing body odor. 1 To fulfill these functions, cosmetics employ various additives, including pigments and antioxidants, which can be sourced naturally or synthetically. Recent years have witnessed an upsurge in demand for natural pigments, primarily due to the ban on numerous synthetic pigments by reputable bodies like the World Health Organization (WHO), the US Food and Drug Administration (FDA), and the European Food Standards Authority (EFSA). This ban resulted from the synthetic pigments’ adverse effects, including toxicity, allergic reactions, carcinogenic properties, and other harmful impacts. 2 Consumers are drawn to natural pigments due to their inherent biological properties, particularly antioxidant activity, 3 which safeguards the body against oxidative damage, a key factor in developing diseases such as Alzheimer's, Parkinson's, rheumatoid arthritis, cancer, cardiovascular diseases, and skin disorders. 4
Plants, insects, and microorganisms serve as potential sources of natural products beneficial to the cosmetic industry. Microorganisms, in particular, offer distinct advantages over plants and invertebrates due to their ability to produce bioactive metabolites through fermentation, independent of seasonal influences, thereby offering a more cost-effective production alternative. 5 Moreover, owing to their stability, accessibility of culture technology, and potential for diverse industrial applications, 6 microbial pigments represent one of the most promising research fields. 7 Among these microorganisms reported as producers of natural pigments, actinomycetes stand out for their widespread distribution in diverse ecosystems, including terrestrial and aquatic environments. They are Gram-positive bacteria renowned for producing different agents, including antibiotics, enzymes, antioxidants, antimicrobials, and pigments. In this regard, the actinomycetes-derived pigments have exhibited notable bioactivities, 8 including naturally occurring compound representatives such as prodigiosins, melanins, and phenazines, among others. 9
The genus Gordonia, belonging to the class Actinomycetes, represents an emerging group encompassing Gram-positive bacteria. Initially regarded as opportunistic pathogens in humans, 10 Gordonia species have attracted considerable interest in recent years owing to their ability to biodegrade environmental pollutants and synthesize organic compounds beneficial for both environmental and industrial applications. 11 Moreover, Gordonia strains have demonstrated proficiency in synthesizing non-toxic pigments, especially carotenoids. 12 Carotenoids rank among the high-value natural pigments due to their diverse biological activities, 13 ranging from antioxidant capacities to serving as precursors to vitamins and exhibiting antitumor and antimicrobial properties. 14 These liposoluble pigments, prevalent in the plastids of photosynthetic organisms like plants, algae, and phototrophic microorganisms, 15 find utility in various industries, including pharmaceuticals, nutraceuticals, food/feed additives, textiles, cosmetics, and fine chemicals. The estimated market value for carotenoids stood at approximately $1.8 billion in 2019, reflecting substantial industrial interest. 16 Within the genus Gordonia, several carotenoids have been identified, such as carotenoid K (1′-OH-4-keto-γ-carotene) and B (1′-OH-keto-γ-carotene) from G. amicalis, 10 canthaxanthin from G. jacobaea,17,18 and lutein from G. rubripertncta. 19 Studies on carotenoid optimization have revealed relevant findings, such as glucose as the preferred carbon source and sulfate as the optimal sulfate source for maximal carotenoid production in G. alkanivorans strain 1B. 16 In fact, light exposure for large periods (e.g. 400–3000 lux and after 19 days) is also crucial to carotenoid production. 16 Similarly, echinenone production from G. terrae TWRH01 was enhanced by utilizing sucrose as the carbon source, yeast extract as the nitrogen source, and adjusting salinity concentration, initial pH, and calcium chloride supplementation. 3
Considering the economic significance of natural pigments, engineering of carotenoid overproducing strains of Gordonia species offers potential as a novel biological source for these essential compounds. 12 Therefore, finding optimal cultural conditions to produce these bioactive pigments becomes pivotal, given that carotenoid production relies on factors like temperature, light exposure, and nutrient availability, encompassing carbon and nitrogen sources and mineral salts. 20 This study seeks to optimize Gordonia's cultural conditions to produce carotenoids exhibiting antioxidant activity, serving as a sustainable source for these non-toxic, economically important compounds. This optimization was achieved through cost-effective experimental approaches such as the Plackett-Burman and Box-Behnken design.
Materials and methods
Screening of Actinomycetota for bioactive activities
Gordonia hongkongensis strain EUFUS-Z928 was isolated from the octocoral Eunicea fusca (Marine environment of Santa Marta Bay, Colombia), as described in a previous study. 21 This strain was selected for its presence of orange intracellular pigment production and antioxidant activity without cytotoxic activity against human primary dermal fibroblast (HDFa, ATCC® PCS-201-012™, Primary Dermal Fibroblast; Normal, Human, Adult) as described by Sanchez-Suarez et al. 22 The actinobacterium of interest was reactivated in the growth medium (Zobell agar) (composition g/L: yeast extract (Oxoid LP0021); 1.25; Peptone (PanReac CAS: 91079-40-2), 3.75; NaCl (Honeywell Cat. 31434), 18; MgCl2 (PanReac CAS: 7791-18-6), 2.0; KCl (PanReac CAS: 740-61), 0.525; CaCl2 (PanReac CAS: 10043-52-4), 0.075; pH, 7.4 ± 0.2) in agar), 23 and incubated for seven days at 30 °C (Friocell, USA). Once incubation time was reached, the isolates showing pigment production were cultured in liquid Zobell medium (3, 10, and 100 mL) for seven days at 30 °C and 150 rpm (New Brunswick Scientific, Connecticut, USA). The biomass obtained from this process was lyophilized at 0.22 mbar and −57°C (Labconco, Kansas, USA) and used in subsequent fermentations.
Culture media evaluation
The isolated strain was cultured (New Brunswick Scientific, Connecticut USA) in GYM 24 (g/L composition: glucose (PanReac CAS: 50-99-7), 4; yeast extract (Oxoid LP0021), 4; malt extract (Neogen NCM0207A); 10; NaCl (Honeywell Cat.31434), 24; MgCl2 (PanReac CAS: 7791-18-6), 5.3; KCl (PanReac CAS: 740-61), 0.7; CaCl2 (PanReac CAS: 10043-52-4), 0.1; pH, 7.4 ± 0.2) and Zobell medium (composition g/L: yeast extract (Oxoid LP0021); 1.25; Peptone (PanReac CAS: 91079-40-2), 3.75; NaCl (Honeywell Cat.31434), 18; MgCl2 (PanReac CAS: 7791-18-6), 2.0; KCl (PanReac CAS: 740-61), 0.525; CaCl2 (PanReac CAS: 10043-52-4), 0.075; pH, 7.4 ± 0.2), 23 for ten days, 30°C and 150 rpm. The same amount of lyophilized bacterial biomass was added to each medium, and the experiments were carried out in triplicate. Carbon and nitrogen sources were evaluated as described below. Notably, in each instance, the amount of resulting biomass was measured to assess the direct relationship between metabolite production and biomass yield.
Carbon and nitrogen source selection
To enhance the bioproduction of pigments and antioxidant metabolites, the strain of interest was initially cultivated on the chosen media, with modifications made to the carbon sources by incorporating glucose (PanReac CAS: 50-99-7), sucrose (Incauca sugar), starch (Carlo Erba CAS: 9005-84-9), glycerol (Chemi CAS: 56-81-5) and lactose (PanReac CAS: 10039-26-6). Subsequently, the nitrogen source was also modified with urea (PanReac CAS: 5713-6), malt extract (Neogen NCM0207A), yeast extract (Oxoid LP0021), casein, ammonium nitrate (PanReac CAS: 7783-20-2) and peptone (PanReac CAS: 91079-40-2), maintaining steady the concentration of the other components in the medium and adding to each medium the same amount of lyophilized biomass. The experiments were performed by triplicate and incubated (New Brunswick Scientific, Connecticut USA) for seven days at 30 °C and 150 rpm. The media where the isolate did not grow or failed to produce pigments were discarded. Finally, bacterial growth was evaluated via dry biomass measurement, pigment production by absorbance readings, and antioxidant capacity determined through DPPH (2,2-diphenyl-1-picrylhydrazyl) and ABTS (2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid)) radical scavenging assays. These evaluations aimed to identify the medium that yielded the most favorable outcomes across all three response variables mentioned.
Preparation of pigmented crude extract
The biomass obtained from the bacterial cultures was previously separated from the fermented medium using a centrifuge (Thermo Scientific, Connecticut, USA) at 20°C and 3500 g for 5 min. Once the culture medium was removed, the biomass was frozen (Biomemory, France) for 12 h at −80°C and subsequently lyophilized (Labconco, Kansas, USA) for 24 h at −57°C and 0.22 mbar. Extracts were prepared from the lyophilized biomass from bacterial cultures using ethanol 96% (MilliporeSigma) and ultrasound-assisted extraction (UAE) (Heidolph, Germany) at 30 HZ for 11 min to ensure cellular disruption and pigment extraction. The solvent was removed by a rotary evaporator (Heidolph, Germany) at 30°C and 50 mbar for 30 min. The resulting extracts were used for assessing the investigated response variables (i.e. absorbance and antioxidant capacity).
Determination of pigment absorbance in the crude extract
Ultraviolet-visible (UV-Vis) spectrophotometry (ThermoScientific, Waltham, MA, USA) measured the crude extract pigmentation. The wavelength of the maximum absorbance (λmax) of the pigmented strain-derived extract was initially determined by recording the respective absorption UV-Vis spectra between 200-800 nm. The maximum absorption of the pigmented crude extracts was determined in 96-well plates (Bio-Rad, California, USA) at 5 mg/L for each case. On the λmax was determined (ca. 460 nm), the resulting absorbance was determined at maximum absorption for estimating the pigment content for comparative purposes.
Determination of the antioxidant capacity of the crude extract
ABTS radical scavenging capacity
The free-radical scavenging capacity of each extract was determined using the 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) radical cation decolorization assay (ABTS•+) (MilliporeSigma, St Louis, MO, USA). The ABTS radical cation (ABTS•+) was produced by reacting the ABTS stock solution with 2.45 mM ammonium persulfate (MilliporeSigma, St Louis, MO, USA). The determination was performed in 96-well plates by adding the strain-derived extract (10 μL) and ABTS (190 μL) solutions. Incubation was carried out at 20°C, in the dark, for 30 min, and absorbance (735 nm) was measured (Bio-Rad, California, USA). 25 Trolox (MilliporeSigma, St Louis, MO, USA) was used as the positive control from 25 to 0.39 µM. The ABTS radical scavenging capacity was calculated using Equation 1.
Where A: control absorbance; B: extract's absorbance.
DPPH radical scavenging capacity
The scavenging capacity of 2,2-diphenyl-1-picrylhydrazyl (DPPH•) radicals was measured by reacting a DDPH stock solution (MilliporeSigma, St Louis, MO, USA) with the test extract for 30 min in the dark. The determination was carried out in 96-well plates by adding the extract (100 μL) and DPPH (100 μL) solutions and measuring the absorbance at 515 nm (Bio-Rad, California, USA). 25 Trolox (MilliporeSigma, St Louis, MO, USA) was used as the positive control from 50 to 0.39 µM. The DPPH radical scavenging capacity was calculated using Equation 2.
Where A: control absorbance; B: extract's absorbance.
Optimization of media fermentation
Selection of the significant variables using Plackett- Burman design
After establishing the carbon and nitrogen sources, the primary factors influencing pigment production and antioxidant metabolites were examined. The cultural conditions under scrutiny for the strain encompassed carbon and nitrogen source concentration, NaCl concentration, pH, temperature, agitation, growth time, and inoculum. Utilizing the Plackett-Burman statistical experimental design 26 enabled the identification of physicochemical parameters significantly influencing the enhancement of pigment and antioxidant metabolite bioproduction (response variables). This method's advantage lies in pinpointing the most critical factors based on their primary effects. 27 This model was based on a first-order model with n + 1 experiments (equation 3), where n is the number of variables, Y is the response or independent variable, β0 is the model's intercept, βi is the regression coefficient, and Xi is the independent variable. 28
Each variable cited above was represented at two levels: high (+1) and low (−1). All experiments were performed in triplicate. Table S7 shows the experimental design that was carried out in Minitab 18.
Response surface methodology
This phase includes optimizing the levels and interaction effects among the notable variables that positively influenced the enhancement of pigment and antioxidant metabolite bioproduction, employing a Box-Behnken design. 29 In this experimental setup, three factors were examined at three distinct levels, i.e. low (−1), medium (0), and high (+1), conducted in triplicate. This process aims to conform the outcomes to an independent second-order polynomial model 28 (equation 4).
Where Y is the predicted response value, β0 is the coefficient of the constant term, βi is the coefficient of the primary term, βii is the coefficient of the quadratic term, and xi is the independent variable. The relationship between Xi and the real value of the independent variable Xi was calculated with equation 5.
Where X0 is the independent variable's true value at the test's central point, and δX is the step change of the independent variable. Table S8 shows the experimental design carried out, developed in Minitab 18.
Statistical analysis
In the statistical analysis of the model, results were expressed as the mean ± standard deviation (SD), and statistical significance was determined through a one-way analysis of variance (ANOVA) to assess the null hypothesis (equality among factor levels’ means). Significance was established when the p-value was less than 5% (P < 0.05). The adequacy of the polynomial model equation was evaluated using the R coefficient, and its superiority was verified via the F test for statistical significance.
LC-MS analysis
High-performance liquid chromatography coupled with mass spectrometry (LC-MS) was performed to determine the chemical composition of the pigmented bacterial extracts with antioxidant activity. The freeze-dried extracts were dissolved in ethanol and analyzed by LC-MS on an LCMS2020 system (Shimadzu Corp, Japan) equipped with a quadrupole analyzer and a dual ion source (DUIS-2020) for simultaneous electrospray (ESI) and atmospheric-pressure chemical (APCI) ionizations. A kinetex C18 column (4.5 × 250 mm, 2.6 µm) (Phenomenex, Torrance, CA, USA) was employed for metabolite separation. The mobile phase involved a mixture of 0.1% formic acid in acetonitrile (phase A) and 0.1% formic acid in water (phase B). Gradient elution was performed as follows: 0 min, 10% A; 3 min, 10% A; 14 min, 40% A; 21 min, 70% A; 25 min, 100% A; 30 min, 10% A. The DUIS interface was operated simultaneously in positive and negative ion mode (200-1200 m/z full scan). In addition, the MS parameters involved 250 °C CDL temperature, 1.4 kV detector voltage, 1.3 L/min nebulizer gas flow rate, and 13 L/min drying gas flow rate.
Results
Effect of culture media
Considering the known influence of culture media on microorganism growth and metabolite production rates, our initial assessment focused on the impact of GYM and Zobell culture media on biomass (Figure 1(a)), pigments (Figure 1(b)), and antioxidant metabolites (Figure 1(c)-(d)) over ten days, constructing a growth curve for G. hongkongensis EUFUS-Z928 in these respective media. The selection of GYM and Zobell media was based on their proven support for a diverse range of marine bacteria, 30 providing essential nutrients for the growth of heterotrophic bacteria such as actinomycetes. 23 All experiments were conducted in triplicate, and data are depicted as means with their corresponding standard deviations. Remarkably, significantly higher biomass production of G. hongkongensis was observed in the GYM medium compared to Zobell (P < 0.0001). Notably, biomass production in the Zobell medium averaged 39% lower than that in the GYM medium over the 10-day culture period. Further analysis using Tukey's test revealed a significant difference in biomass generated using GYM medium after day 2, as indicated in Figure S1a. While pigment production also exhibited significant differences when employing GYM (P < 0.0001) throughout the culture duration (Figure S1b), no noteworthy differences were observed in pigment production itself. Additionally, the antioxidant capacity, measured by DPPH and ABTS radical scavenging, was markedly higher in the GYM medium (Figure S1c-d).

Effect of cultural media on the (a) dry weight of biomass production, (b) absorbance of pigmented ethanolic extracts measured at 490 nm, (c) DPPH radical scavenging capacity of pigmented ethanolic extracts measured at 515 nm, and (d) ABTS radical scavenging capacity of pigmented ethanolic extracts measured at 735 nm. G. hongkongensis EUFUS-Z928 grown on GYM and Zobell medium. The points and bars represent the mean of three independent assays with their respective standard deviation (SD).
Effect of carbon sources
The current assessment aimed to enhance pigment and antioxidant metabolite bioproduction in G. hongkongensis EUFUS-Z928 by examining various carbon sources in the selected media (GYM). Glucose, sucrose, starch, glycerol, and lactose were individually supplied at 4 g/L. Interestingly, the variation in carbon sources resulted in statistically significant differences in biomass production, as determined by the ANOVA test (P < 0.0001). Notably, starch exhibited higher biomass production compared to other carbon sources, a finding supported by Tukey's test. Regarding the pigment production of this strain, glucose demonstrated the highest absorbance value (0.14) among the cultures, followed by starch (0.12), as depicted in Figure 2. ANOVA revealed a significant effect of these carbon sources (P < 0.0001), and Tukey's test also indicated significant differences between glucose and starch (P < 0.01) concerning the production of the targeted pigments, as illustrated in Figure 2. Conversely, glycerol exhibited the lowest absorbance value (0.08), as shown in Figure 2(a), with significantly lower values across all cases (P < 0.001).

Effect of carbon sources on the (a) absorbance of pigmented ethanolic extracts measured at 490 nm, (b) DPPH radical scavenging capacity measured at 515 nm, and (c) ABTS radical scavenging capacity measured at 735 nm. G. hongkongensis EUFUS-Z928 grown on GYM medium. The bars represent the mean of three independent assays with their respective standard deviation (SD). ns: no significant differences. *, **; ***, ***,**** described significant differences.
The antioxidant capacity of G. hongkongensis EUFUS-Z928 cultures, cultivated with different carbon sources, was assessed using DPPH and ABTS radical scavenging methods. Both assays revealed significant differences (P < 0.0001), emphasizing the pivotal role of selecting an optimal carbon source to enhance antioxidant capacity. Specifically, when cultured with starch, the extracts derived from G. hongkongensis EUFUS-Z928 exhibited higher DPPH values (72.49%), followed closely by lactose (71.46%) in the culture medium, showcasing no significant differences (P = 0.9901) according to Tukey's test (Figure 2(b)). Conversely, in terms of ABTS radical scavenging capacity, glucose as the carbon source yielded the highest results (73.59%), followed by starch (67.69%), also without significant differences (P = 0.0634). Notably, glycerol consistently produced the lowest values (DPPH: 40.60%, ABTS: 43.12%) in both assays (Figure 2(b)-(c), P < 0.0001). Considering that the weight of each response variable (i.e. absorbance and antioxidant capacity of pigmented extracts) is 50%, the selected optimal carbon source for strain G. hongkongensis EUFUS-Z928 based on these findings was starch.
Effect of nitrogen source
Nitrogen sources play a critical role in cell growth and protein synthesis across all organisms. This study used organic (malt extract, yeast extract, urea, casein, and peptone) and inorganic (ammonium sulfate) nitrogen sources to assess their effects. Each source, at a concentration of 14 g/L, was introduced into the GYM media, substituting the initial components of malt and yeast extracts with starch as the carbon source. The growth and pigment production of G. hongkongensis EUFUS-Z928 exhibited variability based on the nitrogen source employed. Interestingly, these variations were only observed when the original GYM media components (malt extract and yeast extract) were used, leading to the exclusion of the other nitrogen sources from further consideration.
Evaluation of variables affecting the pigments and antioxidant metabolites using Plackett-Burman design
Eight independent variables were screened in the Plackett-Burman experimental design comprising 16 trials conducted in triplicate. Table 1 displays the design matrix to screen significant factors to produce pigments and antioxidant metabolites, ensuring appropriate responses.
Plackett–Burman experimental design used for pigments and antioxidant metabolites production. The absorbance of pigmented ethanolic extracts was measured at 490 nm, DPPH radical scavenging capacity at 515 nm, and ABTS radical scavenging capacity at 735 nm. G. hongkongensis EUFUS-Z928 grown on modified GYM medium. The results show each independent replicate performed for each condition.
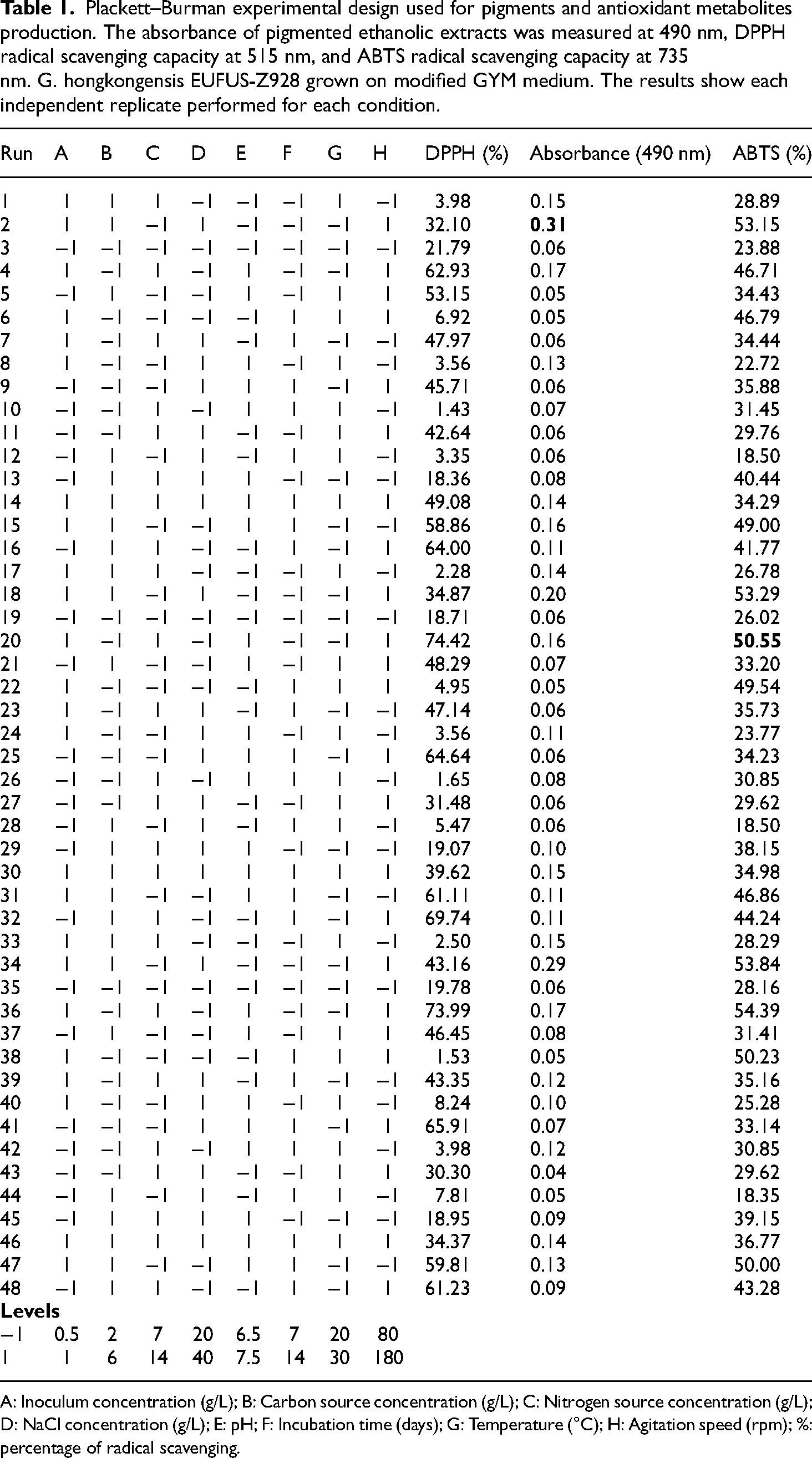
A: Inoculum concentration (g/L); B: Carbon source concentration (g/L); C: Nitrogen source concentration (g/L); D: NaCl concentration (g/L); E: pH; F: Incubation time (days); G: Temperature (°C); H: Agitation speed (rpm); %: percentage of radical scavenging.
The absorbance of the pigmented extract
The maximum production of pigmented extract (Table 1), quantified by absorbance (0.31), occurred in run No. 2, characterized by an inoculum concentration of 1.5 g/L, starch concentration of 6 g/L, malt extract concentration of 5 g/L, yeast extract concentration of 2 g/L, NaCl concentration of 30 g/L, pH at 7.5, a 7-day incubation period, a temperature of 20°C, and an agitation speed of 180 rpm. Conversely, the lowest value (0.04) was observed in run No. 43, with an inoculum concentration of 0.5 g/L, starch concentration of 2 g/L, malt extract concentration of 10 g/L, yeast extract concentration of 4 g/L, NaCl concentration of 30 g/L, pH at 6.5, a 7-day incubation period, a temperature of 30°C, and an agitation speed of 180 rpm. Analyzing the main effects of each variable (Figure S5), six factors, i.e. inoculum concentration, carbon source concentration, nitrogen source concentration, NaCl concentration, pH, and agitation speed, displayed positive impacts on absorbance. At the same time, temperature and incubation time negatively influenced the same response variable. The statistical analysis (Table S1) of the absorbance confirmed the statistically significant variables influencing pigment production, considering a confidence level of 95%. Specifically, inoculum concentration, carbon source concentration, incubation time, temperature, and agitation speed were found to be statistically significant (P < 0.05). This information is depicted in the Pareto chart (Figure 3), illustrating absolute values of standardized effects from the most substantial to the smallest effects, considering a reference line indicative of significance level adherence.

The pareto chart illustrates the standardized effects of various factors on the absorbance measurements (at 490 nm) of pigmented ethanolic extracts from G. hongkongensis EUFUS-Z928 cultivated on GYM medium. These factors were scrutinized using the Plackett-Burman design. The bars depict the t-value derived from three independent assays along with their corresponding standard deviations. Factors displaying a negative effect are highlighted in red, while those positively impacting the absorbance of pigmented extracts are depicted in blue.
The first-order polynomial equation (6) was formulated to represent the relationship between the absorbance in pigmented extract production and the independent variables:

Where A: Inoculum concentration (g/L); B: Carbon source concentration (g/L); C: Nitrogen source concentration (g/L); D: NaCl concentration (g/L); E: pH; F: Incubation time (days); G: Temperature (°C); H: Agitation speed (rpm).
The R2 value of 0.9074 signifies that the variations in the response are largely influenced by the independent variables, leaving only 9.26% of the total variations unexplained by these factors. The adjusted determination coefficient, at 0.8640, further emphasizes the model's significant relevance.
DPPH radical scavenging capacity
The highest DPPH radical scavenging activity, at 74.42%, was observed in run No.20 (see Table 1). This run employed an inoculum concentration of 1.5 g/L, starch concentration of 2 g/L, malt extract concentration of 10 g/L, yeast extract concentration of 4 g/L, NaCl concentration of 20 g/L, pH of 7.5, 7 days of incubation time, temperature of 20 °C, and agitation speed of 180 rpm. Conversely, the lowest value recorded, reaching 1.43%, occurred in run No.10, with an inoculum concentration of 0.5 g/L, starch concentration of 2 g/L, malt extract concentration of 10 g/L, yeast extract concentration of 4 g/L, NaCl concentration of 20 g/L, pH of 7.5, 14 days of incubation time, temperature of 30 °C, and agitation speed of 80 rpm. Regarding the main effects of each variable (illustrated in Figure S6), seven variables, i.e. inoculum concentration, carbon source concentration, nitrogen source concentration, pH, incubation time, and agitation speed, positively impacted the antioxidant capacity outcome. Meanwhile, two variables—NaCl concentration and temperature—negatively affected the same response variable. Scrutinizing the respective ANOVA (Table S2), we can confidently determine the statistically significant variables (i.e. carbon source concentration, nitrogen source concentration, NaCl concentration, pH, incubation time, temperature, and agitation speed) affecting the DPPH scavenging capacity, considering a confidence level of 95%. This information is visually depicted in the Pareto chart (Figure 4), elucidating the order of significance among the variables and highlighting the insignificance of inoculum concentration.

The pareto chart illustrates the standardized effects of various factors on the DPPH radical scavenging (measured at 515 nm) of pigmented ethanolic extracts from G. hongkongensis EUFUS-Z928 cultivated on GYM medium. These factors were scrutinized using the Plackett-Burman design. The bars depict the t-value derived from three independent assays along with their corresponding standard deviations. Factors displaying a negative effect are highlighted in red, while those positively impacting the absorbance of pigmented extracts are depicted in blue.
The first-order polynomial equation (7) was formulated to represent the relationship between the DPPH radical scavenging capacity of pigmented extracts and the independent variables:

Where A: Inoculum concentration (g/L); B: Carbon source concentration (g/L); C: Nitrogen source concentration (g/L); D: NaCl concentration (g/L); E: pH; F: Incubation time (days); G: Temperature (°C); H: Agitation speed (rpm).
The model's R2 value of 0.9731 indicates that the variability in the response can be attributed to independent variables, with only 2.79% of the total variations unexplained by these factors. Additionally, the adjusted determination coefficient, at 0.9605, underscores the significant relevance of the model.
ABTS radical scavenging activity
The highest ABTS radical scavenging capacity (54.39%) was observed in run No. 36 with specific parameters (i.e. inoculum concentration: 1.5 g/L, starch concentration: 2 g/L, malt extract concentration: 10 g/L, yeast extract concentration: 4 g/L, NaCl concentration: 20 g/L, pH: 7.5, incubation time: 7 days, temperature: 20°C, and agitation speed: 180 rpm), while the lowest value (18.35%) was obtained in run No. 44 (inoculum concentration: 0.5 g/L, starch concentration: 6 g/L, malt extract concentration: 5 g/L, yeast extract concentration: 2 g/L, NaCl concentration: 30 g/L, pH: 6.5, incubation time: 14 days, temperature: 30°C, and agitation speed: 80 rpm). The main effects of each variable (Figure S7) revealed that six variables (i.e. inoculum concentration, carbon source concentration, nitrogen source concentration, pH, incubation time, and agitation speed) positively influenced the ABTS antioxidant activity. In comparison, two variables (NaCl concentration and temperature) had a negative impact on the same response variable. ANOVA (Table S3) enabled us to identify, with a 95% confidence level, the variables significantly affecting the antioxidant capacity (ABTS), including inoculum concentration, carbon source concentration, NaCl concentration, pH, incubation time, temperature, and agitation speed. This information is graphically represented in the Pareto chart (Figure 5), illustrating the significance order and insignificance of certain variables (e.g. nitrogen source concentration).

The pareto chart illustrates the standardized effects of various factors on the ABTS radical scavenging (measured at 735 nm) of pigmented ethanolic extracts from G. hongkongensis EUFUS-Z928 cultivated on GYM medium. These factors were scrutinized using the Plackett-Burman design. The bars depict the t-value derived from three independent assays along with their corresponding standard deviations. Factors displaying a negative effect are highlighted in red, while those positively impacting the absorbance of pigmented extracts are depicted in blue.
The first-order polynomial equation (8) represents the ABTS radical scavenging capacity in pigmented extract production as a function of the independent variables:
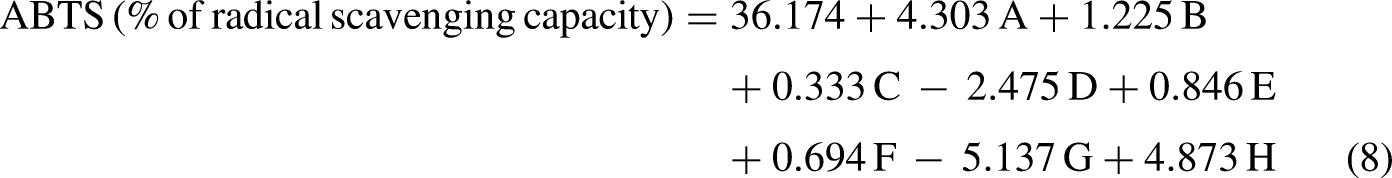
Where A: Inoculum concentration (g/L); B: Carbon source concentration (g/L); C: Nitrogen source concentration (g/L); D: NaCl concentration (g/L); E: pH; F: Incubation time (days); G: Temperature (°C); H: Agitation speed (rpm).
The model's R2 value of 0.9846 indicates that the variability in the response can be largely attributed to the independent variables. A mere 1.64% of the total variations are unaccounted for by these independent variables. Moreover, the adjusted determination coefficient, at 0.9774, emphasizes the high significance of the model.
The main effects were weighted in efforts to optimize pigment and antioxidant metabolites’ production, considering the absorbance of pigmented extract and DPPH/ABTS radical scavenging capacities as response variables. Both absorbance and antioxidant activity were equally weighted at 50%. Figure 6 illustrates this selection process, employing a response surface design, which indicates temperature, agitation speed, and inoculum concentration as the three most significant variables for maximizing pigment and antioxidant metabolites’ production.

The pareto chart illustrates the standardized effects of various factors on the absorbance and DPPH/ABTS radical scavenging capacities of pigmented ethanolic extracts from G. hongkongensis EUFUS-Z928 cultivated on GYM medium. Absorbance was measured at 490 nm, DPPH radical scavenging at 515 nm, and ABTS radical scavenging at 735 nm. These factors were scrutinized using the Plackett-Burman design. The bars depict the t-value derived from three independent assays along with their corresponding standard deviations.
Optimization by response surface methodology
The Box-Behnken design was employed to investigate the optimal levels and interactions among the selected significant factors listed in Table 2. These factors were determined based on their positive or negative impact on pigment and antioxidant metabolite production. Other variables were held constant at levels that had previously yielded maximal output in the Plackett-Burman experiments. This study comprised a total of 15 experiments (conducted in triplicate), exploring various combinations of temperature, inoculum concentration, and agitation speed. The outcomes of these experiments aimed to analyze the effects of the three independent variables on the absorbance of pigmented extract and the radical scavenging capacity (DPPH• and ABTS•+).
Box-Behnken experimental design utilized for pigment and antioxidant metabolite production. The absorbance of pigmented ethanolic extracts was measured at 490 nm, while the DPPH radical scavenging capacity was evaluated at 515 nm, and the ABTS radical scavenging capacity at 735 nm. The experiments were conducted using G. hongkongensis EUFUS-Z928 grown on GYM medium. The table exhibits the outcomes of each independent replicate conducted for every condition.

The absorbance of the pigmented extract
The peak absorbance of pigmented extract (0.40) was recorded during run No. 34, characterized by an inoculum concentration of 2 g/L, a temperature of 15°C, and an agitation speed of 180 rpm, as outlined in Table 2. Conversely, the lowest absorbance value (0.05) was observed in run No. 8, under the following conditions: 2 g/L inoculum concentration, 25°C temperature, and 180 rpm agitation speed. Through ANOVA (Table S4), statistically significant variables (P < 0.05) influencing pigment production, as measured by absorbance, were confidently identified at a 95% confidence level. Notably, variables such as inoculum concentration, temperature, agitation speed, and their respective interactions remained significant, except for the double interaction involving the inoculum concentration. The second-order polynomial equation (9) representing pigment production (absorbance) as a function of the independent variables was derived as follows:

Where A: Inoculum concentration (g/L); G: Temperature (°C); H: Agitation speed (rpm)
The ANOVA data (Table S4) was subjected to multiple regression analysis, resulting in a coefficient of determination (R²) of 0.8866. This value indicates that approximately 88.66% of the sample's variation can be attributed to the selected variables. Additionally, the residuals’ normal probability plot (Figure S8) exhibits points close to the line and a p-value of 0.008, indicating normal distribution and confirming the model's fit to the experimental results. Consequently, the model's major assumptions were validated. Furthermore, contour plots were employed to determine interaction effects and optimal variable levels. Figure 7 (a, b, c) showcases the behavior when two variables interact while the third variable remains at the optimum value. It illustrates that the highest pigment production occurs with a high inoculum concentration interacting with low temperature or agitation speed or a low temperature interacting with moderate agitation. Other combinations of the studied factors yield medium or low absorbances of the pigmented extract.

Contour response surface plots depict the impact on pigmented extract absorbance due to the interaction between various factors: (a) temperature and inoculum concentration at an optimal agitation speed, (b) agitation speed and inoculum concentration at an optimal temperature, and (c) temperature and agitation speed, showcasing their combined effect on the absorbance with the inoculum concentration set at an optimal level. The absorbance measurement in the pigmented extract was conducted at 490 nm, utilizing G. hongkongensis EUFUS-Z928 cultivated on a modified GYM medium. The factors were analyzed by a Box-Behnken design. Each contour plot represents the mean of three independent assays along with their respective standard deviation.
DPPH radical scavenging capacity
The highest observed DPPH radical scavenging activity (88.74%) was recorded during run No. 34, characterized by an inoculum concentration of 2 g/L, a temperature of 15°C, and an agitation speed of 180 rpm. Conversely, the lowest recorded value (35.18%) was noted in run No. 8, with an identical inoculum concentration but a temperature of 25°C and the same agitation speed. The respective ANOVA (Table S5) allowed us to determine, with a confidence level of 95%, the statistically significant variables influencing the antioxidant capacity when the p-value is below 0.05. Notably, the inoculum concentration, temperature, and agitation speed remained significant factors, except for the double interaction of agitation speed and the interaction between inoculum concentration and agitation speed. The derived second-order polynomial equation (10) that characterizes the DPPH radical scavenging capacity as a function of the independent variables is as follows:

Where A: Inoculum concentration (g/L); G: Temperature (°C); H: Agitation speed (rpm).
The ANOVA data from Table S5 underwent multiple regression analysis, revealing a coefficient of determination (R²) of 0.8280. Additionally, examination of the normal probability plot (Figure S9) for residuals indicates a close alignment to the line with a p-value of 0.04, suggesting a normal distribution of residuals. This alignment confirms that the model effectively fits the experimental results, validating all significant assumptions. Figure 8 illustrates the DPPH radical scavenging capacity resulting from the interaction of two studied variables while maintaining the remaining variable at its optimal level. It demonstrates that the highest antioxidant capacity was observed when there was an interaction between the highest inoculum concentration and a medium-low temperature value (Figure 8(a)). Furthermore, when the inoculum concentration was high, varied levels of agitation speed yielded either the highest or lowest antioxidant capacity (Figure 8(b)). Additionally, a notably high antioxidant capacity value was achieved when lower levels of temperature and agitation interacted (Figure 8(c)). Other combinations of factors resulted in a decrease in the DPPH radical scavenging capacity.
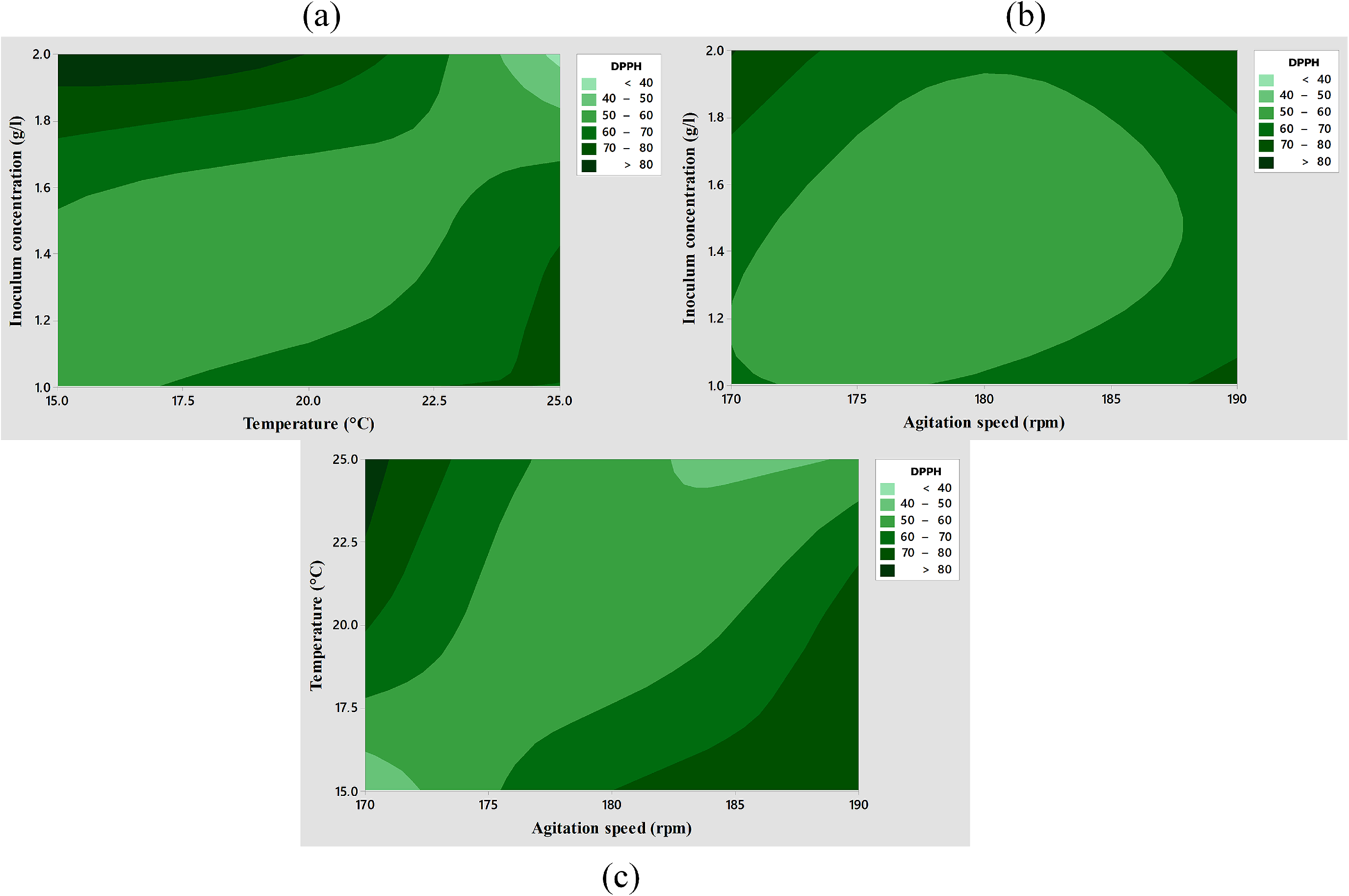
Contour response surface plots depict the impact on pigmented extract DPPH radical scavenging capacity due to the interaction between various factors: (a) temperature and inoculum concentration at an optimal agitation speed, (b) agitation speed and inoculum concentration at an optimal temperature, and (c) temperature and agitation speed, showcasing their combined effect on the absorbance with the inoculum concentration set at an optimal level. DPPH radical scavenging capacity of pigmented extract was conducted at 515 nm, utilizing G. hongkongensis EUFUS-Z928 cultivated on a modified GYM medium. The factors were analyzed by a Box-Behnken design. Each contour plot represents the mean of three independent assays along with their respective standard deviation.
ABTS radical scavenging capacity
Regarding the ABTS radical scavenging capacity, the highest observed value (86.44%) occurred in run No. 33 (inoculum concentration (2 g/L); temperature (20 °C); agitation speed (170 rpm)), while the lowest value (21.70%) was recorded in run No. 40 (inoculum concentration (1 g/L); temperature (25 °C); agitation speed (180 rpm)). The respective ANOVA (Table S6) allowed us to infer, with a 95% confidence level, the variables that significantly impact the antioxidant capacity when their p-value is lower than 0.05. Notably, the variables influencing the antioxidant capacity included the inoculum concentration, agitation speed, their double interactions, and the interaction between agitation and temperature. The derived second-order polynomial equation (11) representing the ABTS radical scavenging capacity as a function of the independent variables is:
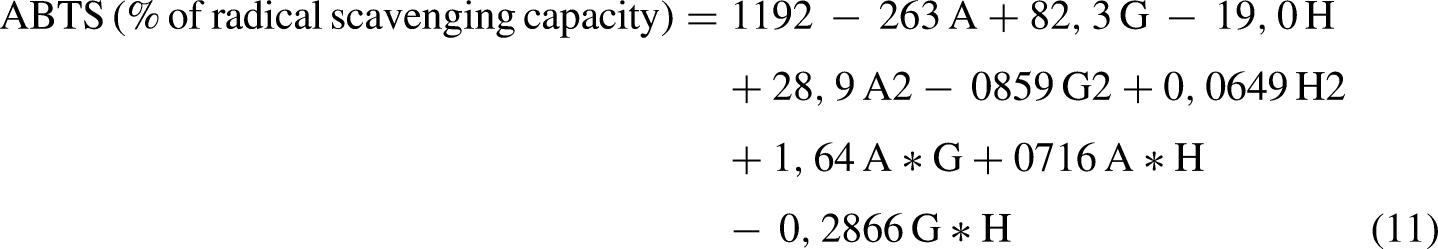
Where A: Inoculum concentration (g/L); G: Temperature (°C); H: Agitation speed (rpm).
Through multiple regression analysis of the ANOVA data (Table S6), the model yielded a coefficient of determination (R²) of 0.8555. Additionally, the normal probability plot (Figure S10) for residuals demonstrated a close alignment to the line with a p-value of 0.006, indicating a normal distribution of residuals. This alignment confirms the model's effective fit to the experimental results, validating all significant assumptions of the model. Figure 9 displays the ABTS radical scavenging capacity resulting from interactions between two studied variables while maintaining the remaining one at its optimal level. It illustrates that the highest antioxidant capacity values were observed when interactions occurred between high inoculum concentrations and medium temperature levels (Figure 9(a)) or when varying levels of agitation speed were implemented (Figure 9(b)). The highest antioxidant capacity was also observed when the lowest agitation speed and the highest temperature levels interacted (Figure 9(c)). Conversely, other variable combinations resulted in decreased ABTS radical scavenging capacity. Contour response surface plots demonstrate the effects of temperature, inoculum concentration, and agitation speed and their combined impact on ABTS-related antioxidant capacity.

Contour response surface plots depict the impact on pigmented extract ABTS radical scavenging capacity due to the interaction between various factors: (a) temperature and inoculum concentration at an optimal agitation speed, (b) agitation speed and inoculum concentration at an optimal temperature, and (c) temperature and agitation speed, showcasing their combined effect on the absorbance with the inoculum concentration set at an optimal level. ABTS radical scavenging capacity of pigmented extract was conducted at 735 nm, utilizing G. hongkongensis EUFUS-Z928 cultivated on a modified GYM medium. The factors were analyzed by a Box-Behnken design. Each contour plot represents the mean of three independent assays along with their respective standard deviation.
Optimal culture conditions and validation of the model
The earlier modeling process identified the optimal culture conditions for temperature, agitation speed, and inoculum concentration (specifically, 16°C, 185 rpm, and 2 g/L, respectively). These settings aimed to maximize the production of pigments (evaluated by the absorbance of pigmented extract) and antioxidant metabolites (assessed through DPPH and ABTS radical scavenging capacities). Thus, the experimentally verified optimal values of the tested variables were compared with the predicted data from the model. The measured absorbance of the pigmented extract was 0.32, while the model predicted 0.31. The measured DPPH antioxidant capacity was 84.61%, slightly below the model's prediction of 88.67%. Similarly, the measured ABTS antioxidant capacity was 79.77%, slightly higher than the model's prediction of 71.35%. This validation outcome demonstrates a high degree of accuracy of the models, with a precision rate exceeding 96.77%, 95.42%, and 89.44%, respectively. This robust validation confirms the reliability of the models under the tested conditions.
LC-MS-based chemical composition of the optimized G. hongkongensis-derived extract
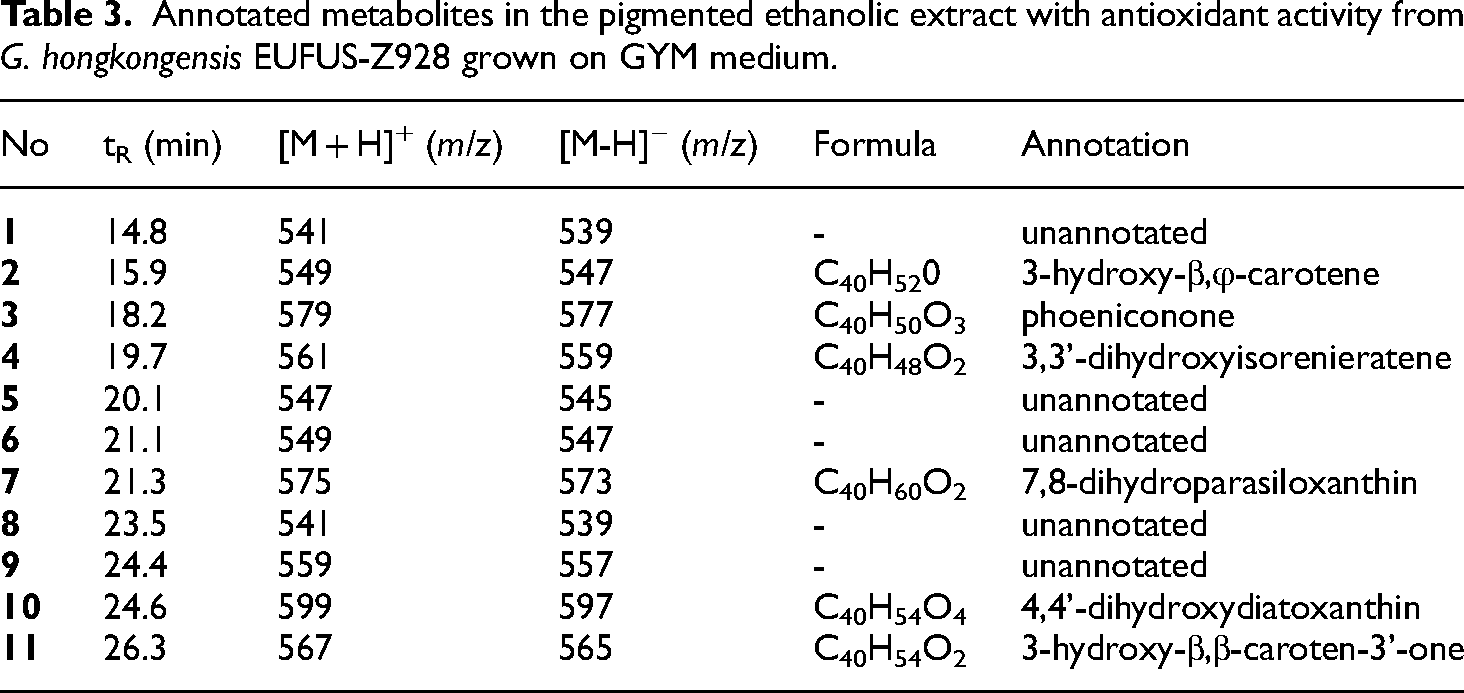
Liquid chromatography coupled with mass spectrometry (LC-MS) using DUIS (i.e. ESI + APCI) ionization was used to determine the chemical composition and the compounds plausibly responsible for the assessed responses (pigment production and antioxidant activity) of the test extract of the G. hongkongensis EUFUS-Z928. The total ion chromatogram (TIC) of the pigmented extract with the highest antioxidant capacity was scrutinized based on the mass-to-charge ratio (m/z) and retention times (tR). Hence, Figure 10 shows the TIC containing eleven main peaks related to the detected compounds in the investigated extract, whose tR and m/z values are listed in Table 3. Spectral and data comparison with the CEUMASS and KNApSAcK web databases and phylogenetic filtering led to the putative identification of six compounds (

Total ion chromatogram of the pigmented ethanolic extract with antioxidant activity from G. hongkongensis EUFUS-Z928 grown on GYM medium.
Annotated metabolites in the pigmented ethanolic extract with antioxidant activity from G. hongkongensis EUFUS-Z928 grown on GYM medium.
Discussion
The Gordonia genus is characterized by mycelial growth, forming rod-shaped elements or cocci, and distinctive pigmented colonies, often displaying colors like yellow, orange, or red. 31 This characteristic makes them a significant source of carotenoids, prompting the evaluation and optimization of their production. In terms of the Gordonian strains’ origin, GYM and Zobell media were chosen for their capacity to support various marine bacteria 30 and provide essential nutrients for the growth of heterotrophic bacteria like actinomycetes. 23
Analyzing the growth pattern of this strain and observing the absorbance and antioxidant capacity (DPPH and ABTS) over time suggested a selective preference for glucose as a primary carbon source, particularly in the GYM medium compared to the complex protein mixture found in yeast extract (Zobell medium). This inclination towards glucose aligns with its quick metabolism, allowing rapid growth, development, and the production of specialized metabolites. 32 This preference for specific carbon sources like glucose within the Gordonia genus isn't uncommon. Alves & Paixão (2014) discovered that a combination of glucose and fructose served as the most favorable carbon source for cell growth and desulfurization capacity in the G. alkanivorans strain 1B, 33 emphasizing how nutrient composition in the culture medium significantly affects these organisms. Similarly, G. terrae strain 3612, isolated from soil, exhibited a distinct preference for glucose and yeast extract agar, evident through its production of rough, pink to orange colonies. 34 Moreover, it is essential to note that the bioproduction of these metabolites of interest directly relates to the culture media, which may differ from the isolation medium. For instance, strain G. hongkongensis EUFUS-Z928 was isolated in Zobell medium, yet the GYM medium notably enhanced biomass, pigment production, and antioxidant capacity. This improvement in specialized metabolite production seems to stem from increased biomass, a finding consistent with Wink et al.'s work (2017), establishing glucose as an excellent carbon source that encourages growth and biomass production in actinomycetes. 8
However, when considering the nitrogen source, it becomes evident that bacterial cell growth, specifically in terms of biomass production, does not necessarily correlate directly with the biosynthesis of specialized metabolites. This pattern echoes findings from the fermentation of G. terrae TWRH01, wherein higher biomass concentrations resulting from glucose and fructose did not proportionately impact pigment production. 3 This observation infers that in certain Gordonia strains, the production of biomass and accumulation of specialized metabolites may not exhibit a direct relationship. Based on this premise, the initial aim of this study was to establish whether there exists a connection between biomass and pigment production in this Gordonia strain. Therefore, the responses were assessed independently. However, upon reviewing the outcomes depicted in Figure 1, a correlation between these factors in the standard medium became apparent. This suggests that understanding the relationship between absorbance and cell biomass would be crucial in a subsequent investigation focused on the pigment production of G. hongkongensis EUFUS-Z928. Consequently, this study intends to proceed with the optimization of the culture medium.
The distinct ABTS and DPPH radical scavenging capacities of G. hongkongensis EUFUS-Z928-derived extracts suggest a distinct performance for their antioxidant. Consequently, the pigmented extract obtained in a glucose-amended medium appeared to stimulate the accumulation of metabolites capable of inhibiting larger quantities of DPPH free radicals. 35 Conversely, the pigmented extract produced using starch as a carbon source yielded a higher concentration of metabolites adept at inhibiting greater amounts of ABTS free radicals. 36 The variation in absorbance and antioxidant capacity across different carbon sources affirms that the production of specialized metabolites in Actinomycetota is influenced by the specific type of carbon source used. 8 Notably, glycerol, recognized as a poor energy carbon source, tends not to be metabolized by various Gordonia species and generally does not contribute significantly to producing specialized metabolites, such as carotenoid-related pigments. 3 Moreover, previous studies indicate that disaccharides (like sucrose and lactose) and polysaccharides (such as starch) 37 are among the most commonly employed carbon sources in Actinomycetota to stimulate increased production of pigments and antioxidants. This is attributed to the actinomycetes' ability to hydrolyze compounds like starch and lipids. 38 For instance, studies by Dastager et al. (2006) identified starch as the optimal carbon source for enhancing pigment production with antioxidant activity in Actinomycetota. 39 Additionally, Barczynska et al. (2015) observed that Actinomycetota demonstrated increased biomass production in the presence of starch as a carbon source. 40 Furthermore, glucose, as a rapidly assimilated carbon source, typically contributes to producing primary metabolites. It tends to induce carbon catabolic repression, delaying the synthesis of specific specialized metabolites until glucose is depleted, subsequently slowing strain growth. 8 In addition, fermenting Gordonia with organic nitrogen sources, such as yeast extract, typically leads to increased production of specialized metabolites, particularly carotenoid-like pigments. 3 According to Hakobyan et al. (2012), this regulation can be attributed to yeast extract's vitamin richness, especially B complex, amino acids, and other growth-stimulating compounds. It serves as an amino acid mixture, offering advantages over individual amino acid. 41 Malt extract, another complex organic source, is utilized for its ability to enhance Actinomycetota growth without suppressing the use of alternative nitrogen sources. 8 Its higher sugar, protein, and mineral composition than other nitrogen sources supports this role. 42 Actinomycetota rely on nitrogen not only for biomass production but also for synthesizing a diverse range of specialized metabolites from amino acids, crucial as key precursors. 8
Apart from the culture medium features, the ability of microorganisms to biosynthesize pigments and antioxidants heavily relies on environmental and culturing conditions. These variables directly impact the maximization or limitation of desired metabolites, emphasizing the importance of identifying and optimizing the right combination of culture conditions. 43 As a result, the fermentation process of actinomycetes for pigment and antioxidant production is intricate. It greatly depends on yield, culture medium, and inoculum and necessitates specific environmental conditions pH, temperature, and stirring speed. These factors significantly influence both cell growth and specialized metabolite production. 44 In this study, the variables affecting metabolite production were analyzed, highlighting the significant role of stirring in producing pigments and antioxidant substances. Adequate stirring ensures sufficient dissolved oxygen, which is crucial for organism growth and desired metabolite yield. Moreover, oxidation-reduction mechanisms within the fermentation mixture and increased nutrient uptake further influence biosynthetic products. 27 However, deviations in aeration levels from the optimum can create unsuitable conditions and affect accurate measurements. Hence, precise control of this variable is crucial. In addition, optimal inoculum concentration plays a key role, initiating rapid mycelial growth and minimizing contamination by microorganisms. However, an excessively high inoculum density might inhibit enzyme production due to nutrient depletion or accumulation of inhibitory compounds, while a lower density could lead to insufficient biomass, affecting product formation. 45 Temperature stands out as a critical environmental parameter affecting growth rate, microbial survival, and specialized metabolite production. Maintaining the optimal temperature ensures enzyme-catalyzed metabolic reactions function at their peak. Deviations from this range can lead to decreased growth rates due to enzymatic protein denaturation, membrane collapse, and thermal lysis of bacteria. 8
Supporting findings by Yolmeh et al. (2017) align with our optimization results. 46 They investigated natural pigment production with antioxidant properties by Micrococcus roseus strain, identifying an intracellular carotenoid through spectrophotometric analysis. They noted a positive correlation between agitation speed and pigment production, observing an increase at 180 rpm. 46 This observation suggests that, in certain actinomycetes, agitation levels close to 180 rpm facilitate the necessary dissolved oxygen for synthesizing these specialized metabolites with antioxidant activity. 27 Furthermore, studies on pigment production from Micrococcus sp. indicated a favorable effect of inoculum concentration on pigment generation, peaking at 2 g/L. 43 This aligns with our optimization findings, reinforcing the notion that this specific inoculum concentration supports rapid mycelial growth, ultimately enhancing pigment and antioxidant metabolite production. 45 In the case of G. alkanivorans strain 1B, Fernandes et al. (2018) found that the stress experienced by the strain was pivotal for carotenoid production, particularly under prolonged exposure to light at varying lux levels (400–3000 lux after 19 days). 16 Although many studies on actinomycetes emphasize optimal metabolite production between 30°C and 40°C, 43 it's noteworthy that bacterial pigment synthesis often occurs under conditions that protect microbial cells from extreme UV rays, photodamage, fluctuating salinity, freeze-thaw cycles, and low temperatures. 47 Carotenoid-producing actinomycetes, in particular, are frequently reported in diverse cold habitats such as glacial fjords, the Caspian Sea, Antarctica, Italian alpine glaciers, and the Himalayas.47,48 These findings agree with our study's results, indicating a negative impact of temperature on carotenoid production in G. hongkongensis EUFUS-Z928, affecting both pigment production and antioxidant capacity.
β-carotene and lycopene are fat-soluble carotenoids known for their orange and red hues, possessing diverse chemical properties. These compounds interact and neutralize reactive oxygen species (ROS), contributing to their high antioxidant activity. 16 These characteristics support the idea that pigmented extracts with antioxidant properties often exhibit orange coloration and demonstrate notably superior values for ABTS radical scavenging activity. It is worth noting that various carotenoids with distinct maximum absorption wavelengths, akin to those found in extracts of G. hongkongensis EUFUS-Z928, are documented in the literature. For instance, Shatila et al. (2013) reported a carotenoid with a maximum absorbance at 460 nm, 49 while Chaudhari et al. (2013) found carotenoids from Planococcus maritimus AHJ 2 that absorb light at 466 nm in the visible spectrum. 50 Additionally, Sasidharan et al. (2013) suggested that most carotenoids have a maximum absorption at 467 nm.51,52 Various types of carotenoids produced by Gordonia species have been identified in prior studies. For example, Loh et al. (2020) reported the presence of echinenone in G. terrae TWRH01 through GC-MS analysis, 3 Jeon et al. (2012) discussed the metabolic role of a carotenoid produced by G. alkanivorans SKF12010, 53 and Fernandes et al. (2018) explored the impact of culture conditions on carotenoid production from G. alkanivorans 1B (44). 16 To quantify total carotenoids precisely, a calibration curve using these known standards should be established. While the specific metabolites in this strain and their nature remain unknown, this assessment was not conducted but is deemed essential for future investigations. However, LC-MS was employed to determine the chemical composition and potential compounds responsible for the pigment production and antioxidant activity within the G. hongkongensis EUFUS-Z928 extract. Detection of target compounds was made possible through ionization utilizing a dual ion source (DUIS) operating simultaneously in positive and negative ion modes. DUIS ionization allows for the simultaneous measurement of polar and non-polar compounds by combining ESI and APCI interfaces, efficiently employed in the analysis of carotenoids. 54 The analysis revealed that this strain predominantly produces C40 carotenoids. This fact agrees with a previous comprehensive survey conducting a chemodiversity assessment and a comparative genomics-based analysis, indicating the intriguing specialized metabolic potential within Gordonia species. While diverse metabolites were discovered, the focus on carotenoid production specifically highlighted the presence of C40 and C46 carotenoids. 31
These findings underscore Gordonia's potential in specialized metabolite production, specifically carotenoids, which present a diverse color palette and possess lower media requirements compared to other sources. However, the higher production costs of bacterial carotenoids compared to synthetics hinder their widespread commercial applications. 55 Further research focusing on optimizing carotenoid-producing bacteria, exploring new carotenoids, and enhancing production strategies is essential to lower production costs and expand industrial applications. 56 Future studies should delve into quantifying total carotenoids based on the compounds found in the extract, establishing a link between biomass and carotenoid production, and optimizing the culture medium accordingly, considering these variables and their potential bioactivities.
Conclusions
The species G. hongkongensis, a member of the Actinomycetota phylum, has garnered increased attention due to its proficiency in biodegrading environmental pollutants and synthesizing organic compounds. These attributes render these bacteria potentially valuable for both environmental remediation and industrial applications. This study aimed to enhance the production of pigments and antioxidant metabolites in G. hongkongensis strains through optimization methodologies. The achieved results demonstrated an increase in pigment production, exceeding 0.22 absorbance units, and a rise in ABTS and DPPH free radical scavenging capacities, surpassing 25%. The validated models for each response variable exhibited less than 10% differences, indicating their capability to reliably predict pigment production and antioxidant capacity in the studied G. hongkongensis strain within the selected experimental parameters. Through compound analysis, six C40 carotenoid-like compounds were identified in the G. hongkongensis strain EUFUS Z9-28 extracts. These compounds are recognized as red-orange pigments with both antioxidant and inhibitory capacities. The findings of this study suggest that these extracts hold promise as potential additives in the cosmeceutical industry, particularly in topical products featuring free-radical scavenging pigments.
Supplemental Material
sj-docx-1-sci-10.1177_00368504241253695 - Supplemental material for Optimized carotenoid production and antioxidant capacity of Gordonia hongkongensis
Supplemental material, sj-docx-1-sci-10.1177_00368504241253695 for Optimized carotenoid production and antioxidant capacity of Gordonia hongkongensis by Paula Rhenals-Montoya, Luisa Villamil, Jeysson Sánchez-Suárez, Luis Díaz and Ericsson Coy-Barrera in Science Progress
Footnotes
Acknowledgments
The authors thank Universidad de La Sabana for the financial support. In addition, the Ministry of Science, Technology, and Innovation for the research resources through project 123080864187 is also acknowledged. P.R.-M. also thank Remberto Rhenals and Omaira Montoya for the resources and support to pursue the master's degree. The gratitude is extended to GIBP and Actinos Group for their support, especially to Aixa Sarmiento, Maria Clara De La Hoz, Jeysson Sánchez, and Marcelo Valle. The bacterial strain used in this study originates from Colombia and was obtained in accordance with Amendment No. 5 to ARG Master Agreement No. 117 dated May 26, 2015, issued by the Ministry of Environment and Sustainable Development, Colombia.
Data availability statement
Data supporting reported results can be found in this document and the Supplementary Materials.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and informed consent
This study did not involve any local people or patients. Thus, the authors did not have ethics approval nor informed consent.
Funding
This research was funded by Minciencias (Ministerio de Ciencia, Tecnología e Innovación— Colombia, project code 123080864187 contract 80740-168-2019) and Universidad de La Sabana (project code ING-175-2016).
Supplementary Materials
Table S1. Statistical analysis of Plackett-Burman design for absorbance pigmented extract production, Table S2: Statistical analysis of Plackett-Burman design for DPPH radical scavenging capacity, Table S3: Statistical analysis of Plackett-Burman design for ABTS radical scavenging capacity, Table S4: Statistical analysis of Box-Behnken design for absorbance pigmented extract production, Table S5: Statistical analysis of Box-Behnken design for DPPH radical scavenging capacity, Table S6: Statistical analysis of Box-Behnken design for ABTS radical scavenging capacity, Table S7: Plackett–Burman experimental design used for pigments and antioxidant metabolites production, Table S8: Box-Behnken experimental design used for pigments and antioxidant metabolites production, Figure S1: Tukey's test of cultural media effect, Figure S2: Media comparison of carbon sources effect on the absorbance of pigmented ethanolic extracts, Figure S3: Media comparison of carbon sources effect on the DPPH radical scavenging capacity on pigmented ethanolic extracts, Figure S4: Media comparison of carbon sources effect on the ABTS radical scavenging capacity of pigmented ethanolic extracts, Figure S5: Main effects for absorbance of pigmented extract according Plackett-Burman Design, Figure S6: Main effects for DPPH radical scavenging capacity according Plackett-Burman Design, Figure S7: Main effects for ABTS radical scavenging capacity according Plackett-Burman Design, Figure S8: Normal probability plot of the residuals for absorbance of pigmented extract according Box-Behnken Design, Figure S9: Normal probability plot of the residuals for DPPH radical scavenging capacity according Box-Behnken Design. ![]() : Normal probability plot of the residuals for ABTS radical scavenging capacity according to Box-Behnken Design.
: Normal probability plot of the residuals for ABTS radical scavenging capacity according to Box-Behnken Design.
Author biographies
Paula Rhenals-Montoya was born in Medellín, Antioquia, Colombia. She is a chemical engineer specialized in environmental management from Universidad Pontificia Bolivariana, Colombia. She received her master's degree in process design and management from Universidad de La Sabana, Colombia. Currently, she is dedicated to working on the formulation of natural and environmentally friendly products. Her research interests encompass natural product exploration, sustainable products and processes, and the cosmetic industry.
Luisa Villamil is a Marine Biologist from Universidad Jorge Tadeo Lozano in Colombia. She has a PhD in Biological Sciences and Aquaculture from the Universidad de Vigo in Spain. She was a postdoctoral researcher at the University of Rhode Island and a visiting Professor at the Woods Hole Oceanographic Institute. She is currently the director of the PhD program in Biosciences at Universidad de La Sabana.
Jeysson Sánchez-Suárez was born in Cucuta, Norte de Santander, Colombia. He received his master's degree from Universidad Nacional de Colombia and his doctorate from Universidad de La Sabana, Colombia. Currently, he is employed at the Schepens Eye Research Institute, Harvard Medical School, and also serves as a researcher in the Research Group in Ecology and Biogeography at Universidad de Pamplona, Colombia. His research interests encompass natural product exploration, pharmacology, microbiology, and bioinformatics.
Luis Díaz was born in Bogotá, Colombia. He received his undergraduate degree in chemistry and doctorate in chemistry from Universidad Nacional de Colombia. He is currently an Associate Professor at the Universidad de La Sabana, Faculty of Engineering, and the leader of the Bioprospecting Research Group. Senior Research of the Minister of Science and Technologies. His research interests include exploring natural products, pharmacology, microbiology, and biochemistry.
Ericsson Coy-Barrera, born in Bogotá, Colombia, completed his undergraduate studies in Chemistry at Universidad Nacional de Colombia. Subsequently, he pursued his master's and PhD in Organic Chemistry at the same university. Additionally, he worked on organic synthesis during a research stay at the University of Potsdam, Germany. He was subsequently appointed as a professor of Organic Chemistry at MU Nueva Granada, Colombia, where he founded the Bioorganic Chemistry Laboratory. Here, he initiated his own research program focused on lead finding from nature, employing holistic and rational approaches based on metabolomics, chemoinformatics, molecular modeling, and organic synthesis.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.