Abstract
Introduction
Healthcare workers (HCWs) are at risk for developing sleep problems compared to the general population due to their heavy workload and high-stress levels, which can be exacerbated during a pandemic or other health crisis.1–4 A recent meta-analysis found that HCWs had a significantly higher prevalence of insomnia compared to non-HCWs throughout the pandemic. 5 Concerningly, compared to the initial months of the pandemic, HCWs’ sleep quality has progressively worsened and has been associated with psychiatric symptoms such as acute stress, depression, and anxiety symptoms.6,7
In addition to increasing the risk for psychiatric disorder,8,9 impaired sleep is an occupational hazard in HCWs, adversely affecting multiple domains of performance and increasing risk of medical errors.10–12 As such, improving sleep in HCWs plays a critical role in both individual and population health and represents an urgent public health need.
Many nonpharmacological interventions exist to improve sleep quality, 13 such as music-assisted relaxation, cognitive behavioral therapy, and mindfulness training. Both Tai Chi and meditation improved sleep in HCWs during the pandemic.14,15 While promising, a limitation of interventions like Tai Chi and meditation is that they require significant involvement on the part of the individual to be effective. Easily implementable nonpharmacological interventions may have an important role for busy HCWs who already feel a sense of burnout and who are accordingly reluctant to engage in time-intensive interventions.
In order to address the need for acceptable and efficacious nonpharmacological sleep interventions in HCWs, the present study evaluated the effect of electronic noise-masking earbuds on both subjective sleep perception and objective sleep parameters in HCWs who self-reported sleep difficulties. We hypothesized that HCWs using the noise-masking earbuds would demonstrate an improvement in sleep quality through both subjective and objective outcome measures.
Methods
Study design
This interventional study employed a pre–post study design over the course of a 10-day study period (Figure 1). Baseline sleep measurements were collected during nights 1–3 from bedtime to wake-up time via the Dreem headband (see “Devices” below). During nights 4–7, participants wore a pair of Bose Noise-Masking SleepbudsTM without using the Dreem headband to allow for habituation to the intervention. Intervention sleep measurements occurred during nights 8–10 during which participants used the Bose Noise-Masking SleepbudsTM and the Dreem headband.

Study timeline. During nights 1–3, participants wore the Dreem headband to collect baseline objective sleep measures, and baseline subjective sleep measures were collected through daily surveys. During nights 8–10, participants wore the Dreem headband and Bose Noise-Masking SleepbudsTM and collected objective sleep measures, while subjective sleep measures were collected through daily surveys. The Insomnia Severity Index (ISI) was administered prior to night 1 and after night 10 to assess change in self-reported insomnia symptoms. Image created with biorender.com
Participants
HCWs who worked primarily in a hospital setting and had face-to-face patient contact were recruited to participate in the study through several methods, such as word of mouth, the internet, electronic mailing lists, and flyers. Recruitment and enrollment occurred from May 2021 to May 2022, where potential participants were primarily recruited from the University of Colorado Anschutz Medical Campus. Of the 901 individuals who completed the prescreener online survey, 333 met preliminary inclusion criteria, of which 93 were reachable by phone and agreed to full assessment of eligibility. Basic eligibility criteria included working as a HCW in an in-person setting, self-reported difficulties falling or staying asleep due to environmental stimuli, self-reported light or moderate sleepers, and a score of ≥ 8 on the Insomnia Severity Index (ISI). Exclusion criteria included factors that could independently interfere with sleep, such as any currently known sleep disorder, psychiatric disorder, or substance use disorder. Furthermore, participants were excluded for irregular caffeine, tobacco, alcohol, or cannabis consumption, as well as irregular sleep schedules. Given the unique consequences of night shift work, we limited our sample to individuals who worked day shifts.16,17 Eighty-nine participants were considered eligible to participate and were assigned to the study protocol. Seventy-seven participants completed the protocol, and 61 participants had high-quality, usable objective data for at least one night at baseline and intervention during the study period (see below under Devices for discussion of the determination of quality data). Review and approval for this study was obtained from the Colorado Multiple Institutional Review Board. All participants provided consent to participation via an electronic consent form approved by the Institutional Review Board. A CONSORT diagram of study participants is shown in Figure 2.
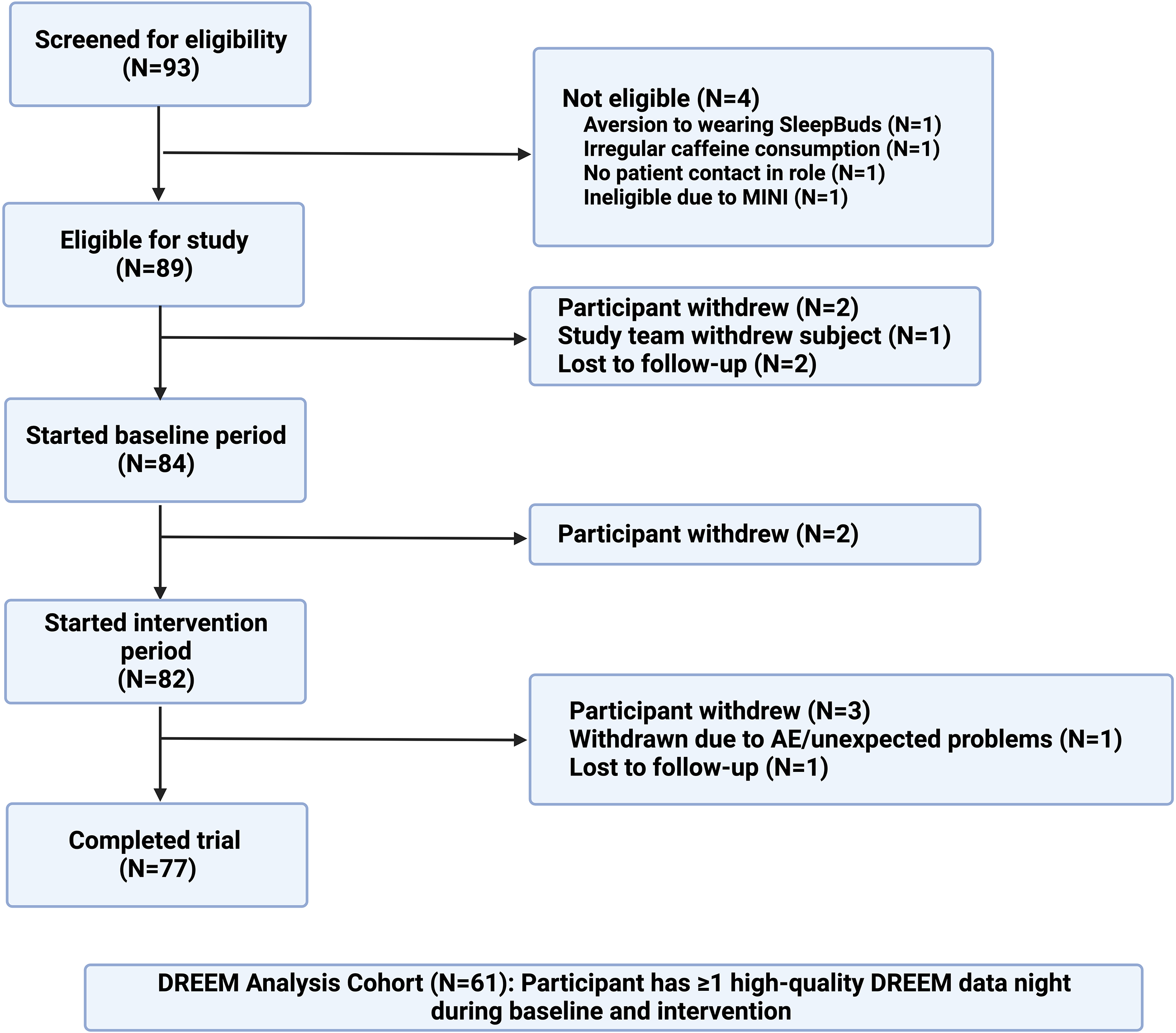
CONSORT diagram. This CONSORT diagram shows the flow of study participants in the trial and how they were chosen for inclusion in the analysis cohort. Dreem data was considered “high-quality” if the measure had at least 70 in record quality (Dreem metric), was longer than 2 hours, and had an off-head proportion of < 20%. Image created with biorender.com
Devices
Dreem Headband: The Dreem headband device is a wireless headband that can record, store, and automatically analyze physiological sleep data. Five types of physiological signals are recorded via three types of sensors embedded in the device: (1) brain cortical activity via six electroencephalogram dry electrodes yielding seven derivations (Fpz-O1, Fpz-O2, Fpz-F7, F8-F7, F7-O1, F8-O2, Fpz-F8; 250 Hz sampling frequency); (2)–(4) movements, position, and breathing frequency via a three-dimensional (3D) accelerometer located over the head; and (5) heart rate via a red-infrared pulse oximeter. From these data, an automatic algorithm derives sleep staging and calculates clinical parameters from that staging, including sleep onset latency (SOL), wake after sleep onset (WASO), number of arousals, and total sleep time. The headband has been validated to acquire physiological data and perform automatic sleep staging on par with medical-grade polysomnography. 18 At the time of writing, the Dreem headband has been used to acquire sleep data in multiple peer-reviewed studies.18–24 For each recorded night for a participant, the Dreem algorithm provides an overall “quality” score of the data from 0 to 100, indicating the percentage of epochs within a recording deemed to be scorable based on the algorithm's learning models. Recorded nights with quality scores of 70 or greater were considered high quality by the device manufacturer and eligible for inclusion in the analysis.
Bose Noise-Masking SleepbudsTM: Bose Noise-Masking SleepbudsTM are a commercially available product initially designed to help people who have difficulty sleeping due to ambient noise (e.g. snoring and traffic). They do so by playing masking and relaxing sounds at bedtime and during sleep, as well as through their passive noise blocking design.
Assessment outcomes
The primary outcomes for this study were the change from baseline on self-reported ISI, perceived change in SOL, objective average SOL, WASO, and the number of awakenings as measured by the Dreem headband. Additional secondary objective outcomes include changes in total sleep time, sleep stage percentages, and sleep efficiency as measured by the Dreem headband. Secondary subjective outcomes were obtained through standardized self-report measures and daily morning surveys.
Subjective Sleep Quality Measures (ISI, ESS, PSQI): The ISI is a seven-question measure to assess the individual's perception of their sleep difficulties and determine the severity of insomnia. 25 The Epworth Sleepiness Scale (ESS) is an eight-item measure to assess levels of daytime sleepiness. 26 The Pittsburgh Sleep Quality Index (PSQI) is a 10-question instrument to assess sleep quality, providing measures of sleep duration, disturbance, sleep latency, daytime dysfunction, sleep efficiency, and subjective sleep quality. These measures are summed to provide an overall sleep quality index score, in which scores >5 are interpreted as poor quality. 27 The ISI, ESS, and PSQI were measured at baseline and postintervention.
Depression, Anxiety, and Stress Measures (CES-D, GAD-7, and PSS): The Center for Epidemiologic Studies Depression Scale (CES-D) is a widely used 10-item self-report questionnaire to assess symptoms of Major Depressive Disorder. 28 The Generalized Anxiety Disorder Scale 7 (GAD-7) is a widely used scale with seven questions to assess symptoms of GAD. 29 The Perceived Stress Scale (PSS) is the used scale to measure the degree to which events in one life are perceived as stressful. 30 The CES-D, GAD-7, and PSS were measured at baseline and postintervention.
Daily Morning Surveys: After each night during the study period, participants completed a daily morning survey. This morning survey collected information about use of the earbuds, the time participants attempted to fall asleep, their perception of their sleep, factors disrupting their sleep, and their morning wake-up time.
Statistical analysis plan
Participant demographics were summarized with means (M) and standard deviations (SD) for continuous variables and with frequencies and percentages for categorical variables. Demographics were compared between completers (N = 77) and noncompleters that were not included in the cohort (N = 16), and there were no significant differences, so no additional missing data approaches were utilized. Differences in self-reported sleep scales (e.g. ISI) from baseline to the end of the intervention were tested with paired t-tests. In the cohort of N = 61 with at least one night of high-quality Dreem data in both baseline and intervention periods, objective, and subjective sleep measurements were modeled in linear mixed-effect models with condition (intervention vs baseline) as the primary predictor; up to three nights of high-quality data were included for each time period. A random intercept was included in the model for each participant to account for repeated measures, and since there were no differences in missingness across the night (1, 2, and 3) of each time period, night was not included in the models. Outcomes were log-transformed to improve model fit due to skew in outcome distributions and to allow for effects to interpreted on a multiplicative scale when back-transformed; these are presented as mean ratios. To test whether the intervention had differential effects on HCWs with higher objective SOL, we tested whether there was an interaction between baseline average SOL and condition on self-reported scales and both objective and subjective Dreem outcome measures in the same modeling framework. Study data was collected and managed using REDCap electronic data capture tools at University of Colorado Anschutz Medical Campus. 31 Statistical analyses used α = 0.05 for two-sided significance testing and were all conducted in R version 4.2.0. 32
Results
Study population and standardized self-report measures
All study participants worked as HCWs in an in-person setting. As seen in Table 1, the most prevalent job types were physicians (29.9%), nurses (19.5%), advanced practice practitioners (13.0%), and healthcare students (13.0%). The final study population consisted of 31 males (40.3%) and 46 females (59.7%), with a mean age of 35.6 years (SD = 7.8). Most participants had graduate or professional degrees (62.3%). Other characteristics of study participants are shown in Table 1.
Participant demographics.

n = 77 for all values. Demographics are summarized with means (standard deviations) for continuous variables and frequencies (percentages) for study completers.
SD: standard deviation.
According to daily morning surveys, 72 of 77 study completers (93.5%) reported using the earbuds all seven nights of the intervention period. Three out of 77 (3.9%) reported use for 6 of the 7 nights, and 2 of 77 (2.6%) reported use for 5 of the 7 nights.
HCWs reported significant improvements in subjective sleep quality after the intervention as measured by the ISI (M = 8.0, SD = 3.7) and PSQI (M = 4.4, SD = 2.2), compared to their baseline assessments (ISI: M = 15.7, SD = 3.7, Cohen's d = 1.74, p < 0.001; PSQI: M = 7.8, SD = 2.9, Cohen's d = 1.14, p < 0.001; Table 2). HCWs also reported statistically significant decreases from baseline on the PSS (mean decrease of 2.6 [SD: 5.6], Cohen's d = 0.46, p < 0.001), the ESS (mean decrease of 2.1 [SD: 3.6], Cohen's d = 0.58, p < 0.001; Table 2), and the CES-D (mean decrease of 1.1 [SD 4.7], Cohen's d = 0.23, p = 0. 047; Table 2), indicating less perceived stress, sleepiness, and depression. There were no significant differences in scores on the GAD-7 (Cohen's d = 0.21, p = 0.07; Table 2).
Standardized self-report measures.
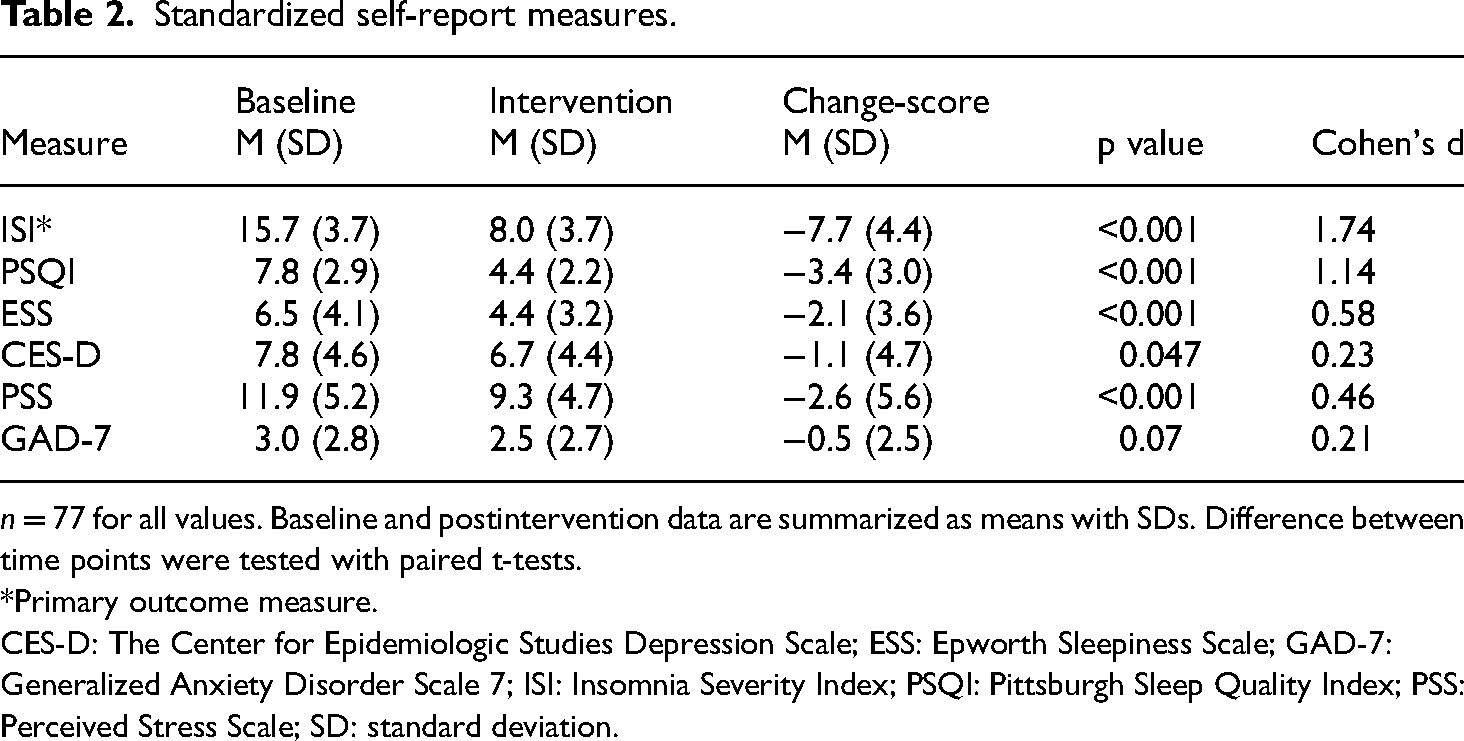
n = 77 for all values. Baseline and postintervention data are summarized as means with SDs. Difference between time points were tested with paired t-tests.
Primary outcome measure.
CES-D: The Center for Epidemiologic Studies Depression Scale; ESS: Epworth Sleepiness Scale; GAD-7: Generalized Anxiety Disorder Scale 7; ISI: Insomnia Severity Index; PSQI: Pittsburgh Sleep Quality Index; PSS: Perceived Stress Scale; SD: standard deviation.
Perceived and objective sleep measures
For primary outcomes, there was a significant reduction in perceived sleep onset latency during the intervention (M = 17.2, SD = 7.7) compared to baseline (M = 24.7, SD = 16.1), where perceived sleep onset latency was 21% faster (95% confidence interval (CI): 9% to 31% faster, Cohen's d = −0.42, p = 0.001) (Tables 3 and 4). There were no significant changes in objective SOL during the intervention (M = 15.0, SD = 11.0) compared to baseline (M = 16.3, SD = 13.9, d = 0.05, p = 0.703). Additionally, there were no significant changes in both objective WASO (d = −0.07, p = 0.606) and objective number of arousals (d = −0.14, p = 0.264) (Tables 3 and 4).
Sleep variables at baseline and intervention.

n = 61 for all values. Sleep variables are summarized as objective data (objective sleep measures) and subjective data (perceived sleep measures) at baseline and intervention. Baseline and intervention data are summarized as means with SDs.
Primary outcome measure.
SD: standard deviation; SOL: sleep onset latency; TST: total sleep time; WASO: wake after sleep onset.
Modeling effect of intervention on primary outcome sleep variables.

n = 61 for all values. Missingness was not associated with time, so night was not included as a covariate.
SOL: sleep onset latency; WASO: wake after sleep onset.
Stratification by objective sleep onset latency
We next considered the possibility that differences in perceived SOL without significant changes in objective SOL may have been related to the sample's average baseline objective sleep onset latency being less than 20 minutes (M = 16.3, SD = 13.9; Table 5). Given that a mean SOL less than 20 minutes would not be considered clinically abnormal,33,34 it's possible that there was minimal potential for improvement.
Sleep variables stratified by objective SOL at baseline.

n = 61 for Dreem variables and self-reported sleep variables. Sleep variables are summarized as objective data (objective sleep measures) and subjective data (perceived sleep measures) at baseline and intervention. n = 77 for standardized self-report measures. Baseline and postintervention data are summarized as means with SDs. n = 43 for mean SOL < 20 at baseline, and n = 18 for mean SOL > 20 at baseline. See Supplemental Tables 1 and 2 for number of observations for each measure.
CES-D: Center for Epidemiologic Studies Depression Scale; ESS: Epworth Sleepiness Scale; GAD-7: Generalized Anxiety Disorder Scale 7; ISI: Insomnia Severity Index; PSQI: Pittsburgh Sleep Quality Index; PSS: Perceived Stress Scale; SD: standard deviation; SOL: sleep onset latency; WASO: wake after sleep onset.
To test whether the intervention had differential effects on HCWs with higher objective SOL at baseline, we stratified the sample according to whether average baseline SOL was less than or greater than 20 minutes. We then tested whether there was an interaction between baseline SOL group and condition (intervention vs baseline) on outcome measures. There was a significant interaction between condition and baseline SOL (p = 0.002; Table 6) such that in those with a mean SOL > 20 minutes at baseline, objective sleep onset latency was 30% faster (95% CI: 7% to 48% faster; p = 0.015; Tables 5 and 6) with the intervention (M = 22.4, SD = 14.8) compared to baseline (M = 32.1, SD = 15.8). In those with a baseline mean SOL < 20 minutes, there was no significant change in objective SOL (p = 0.054).
Interaction between baseline objective sleep onset latency (SOL; <20 minutes vs >20 minutes) and condition (baseline vs intervention) on sleep variables.
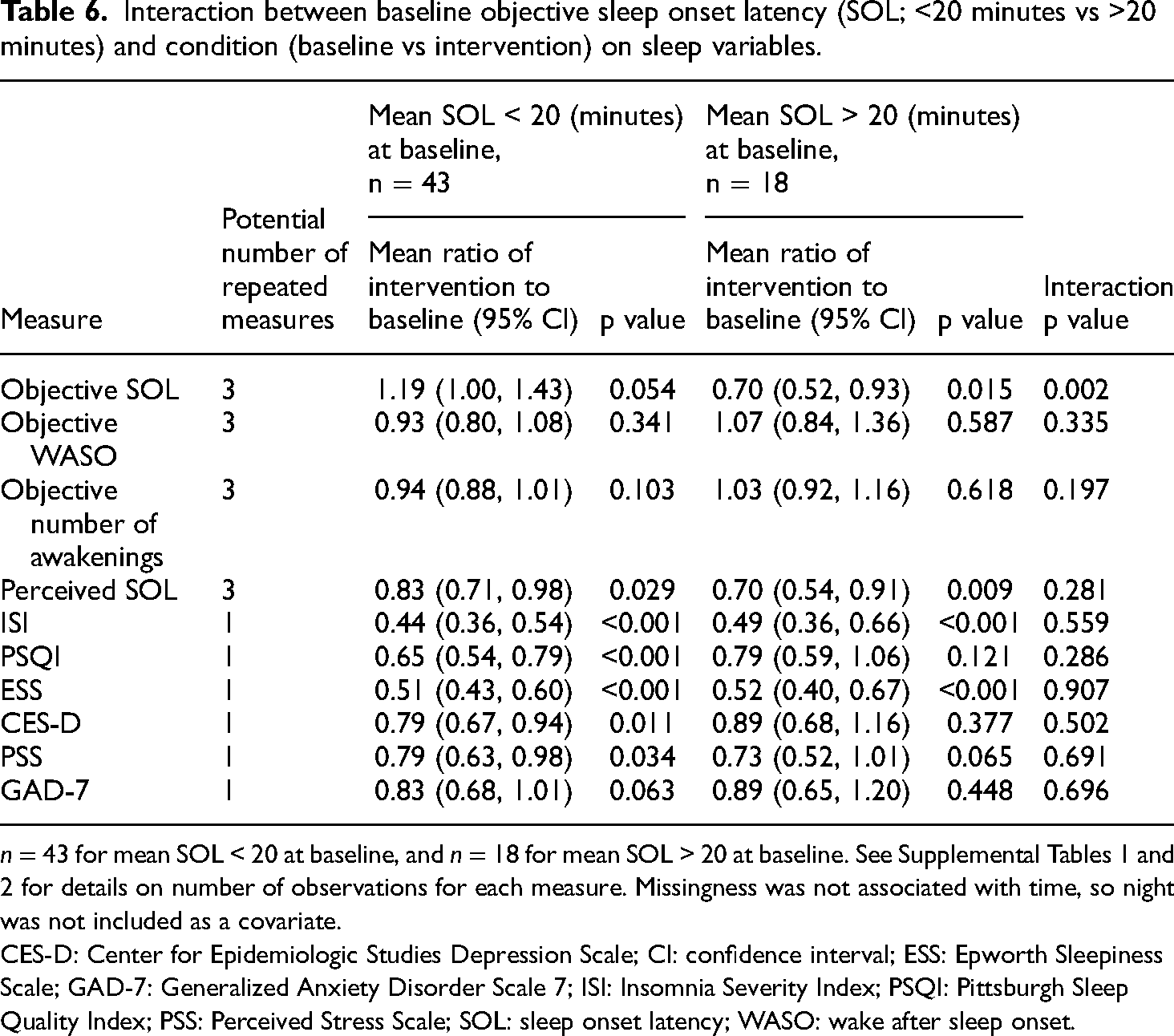
n = 43 for mean SOL < 20 at baseline, and n = 18 for mean SOL > 20 at baseline. See Supplemental Tables 1 and 2 for details on number of observations for each measure. Missingness was not associated with time, so night was not included as a covariate.
CES-D: Center for Epidemiologic Studies Depression Scale; CI: confidence interval; ESS: Epworth Sleepiness Scale; GAD-7: Generalized Anxiety Disorder Scale 7; ISI: Insomnia Severity Index; PSQI: Pittsburgh Sleep Quality Index; PSS: Perceived Stress Scale; SOL: sleep onset latency; WASO: wake after sleep onset.
Discussion
In this pilot study, we evaluated the efficacy of electronic noise-masking earbuds in improving sleep quality for HCWs reporting sleep difficulties during the COVID-19 pandemic. Our findings provide preliminary data that, as hypothesized, the noise-masking earbuds significantly improved subjective sleep quality, as evidenced by a reduction in ISI scores and decreases in perceived SOL. Contrary to our hypothesis, there was a lack of improvement in objective sleep measures. However, individuals who started with objective SOL > 20 minutes experienced a significant reduction in objective SOL during the intervention period. Electronic noise-masking earbuds could thus represent an effective nonpharmacological sleep intervention in HCWs, deserving additional controlled trials and comparisons to other modalities.
To be eligible for the study, HCWs had to report difficulty falling or staying asleep and, at minimum, subthreshold insomnia symptoms demonstrated by a score of ≥8 on the ISI. Although participants did not have a formal diagnosis of insomnia prior to enrollment, the mean baseline ISI score was 15.7 (SD 3.7), signifying moderate-severe clinical insomnia based on the ISI scaling system. 25 After the 10-day study period, we found a statistically significant reduction in ISI scores from a baseline of 15.7 to 8.0, such that the sample average met the cutoff score for subthreshold insomnia (i.e. < 8, no clinically significant insomnia; 8–14, subthreshold insomnia; > 14, moderate to severe insomnia) (Table 2). 25 In line with this, we also found a statistically significant decrease in PSQI scores from a baseline of 7.8 to 4.4. The change in average PSQI score from one that was greater than 5 to below 5 indicated that on average, the sample population no longer met the PSQI threshold for poor quality sleep27,35 (Table 2). Although there were also statistically significant reductions in scores on ESS, PSS, CES-D, and GAD-7, the baseline scores were within the range of normal for each measure, and the magnitude of change in scores was unlikely of clinical significance.
Within our sampled population of HCWs, we found a significant reduction in perceived SOL compared to baseline but no significant changes in objective SOL in our population as a whole. The initial discrepancy between change in perceived SOL without change in objective SOL was partially reconciled by stratifying the sample based on their baseline objective SOL. Although our sample reported significant difficulty with sleep through self-report measures, we specifically excluded individuals with a formal diagnosis of sleep disorders such as insomnia. Thus, it was unsurprising that the average baseline SOL was within the “normal range,” 36 limiting room for potential improvement. However, in individuals who demonstrated a baseline SOL > 20 minutes, the intervention was associated with a significant decrease in objective SOL (Table 6). While we cannot rule out regression to the mean independent of the intervention, the results suggest that the study intervention is promising for improving objective SOL in individuals with more clinically significant objective deficits. Furthermore, in individuals with baseline objective SOL of less than 20 minutes, the intervention was associated with an alteration in perceived SOL that was closer to the objectively measured SOL (Table 3), suggesting that it helped correct sleep-state misperception. Sleep perception is inherently important for perceived sleep quality and overall quality of life, such that individuals who accurately estimate their total sleep time score higher on quality of life measures compared to individuals with poor sleep estimation. 37 The discrepancy at baseline between objective and subjective sleep SOL in individuals with an objective SOL < 20 minutes could be explained through sleep-state misperception, otherwise known as paradoxical insomnia, where individuals overestimate their SOL compared to objective measurements.38–41 Furthermore, previous research has shown that individuals with insomnia need a greater duration of prolonged undisturbed sleep to perceive sleep onset compared to healthy controls. 40 The findings of the current study suggest that noise-masking earbuds may assist with correcting both perceived and objective sleep disturbances in SOL. These findings highlight the importance of using both subjective and objective sleep measures when evaluating sleep interventions.
There were several limitations in the present study. The number of individuals who were enrolled (and subsequently completed the study, n = 77) was determined by the number of available earbud devices that had been gifted to the study team. A slightly smaller cohort of participants from those who completed the study had sufficient high-quality, objective data suitable for analysis (n = 61). Future studies would ideally rely on power analysis during study design to determine the number of participants required. In addition, the lack of a control condition limits our ability to definitively conclude that change outcomes were due to the intervention and not a regression to the mean. Future studies could randomize individuals to a sham condition (earbuds without noise masking function) to confirm our outcomes. Finally, although we confirmed adherence to use of the earbuds by self-report, utilizing other methods to confirm adherence (such as having participants submit photos) would strengthen confidence in future results. Despite these limitations, our findings highlight the potential value of a nonpharmacological sleep intervention in a population that is highly vulnerable to sleep problems.
The fact that our overall sample population lacked objectively measured sleep pathology at baseline despite reports of poor sleep highlights the role of perception in sleep disturbance. It suggests that there is still value in correcting this by using low-risk, nonpharmacological interventions. The high volume of HCWs who expressed interest in the study, noting sleep disturbance on prescreen, emphasizes both the prevalence of sleep problems in HCWs and the need for intervention. The high level of reported adherence to use of the earbuds (93.5% of participants wearing the earbuds for seven out of seven nights) suggests that electronic noise-masking earbuds represent a feasible treatment strategy for further study.
Conclusions
In a sample population of frontline HCWs during the COVID-19, use of electronic noise-masking earbuds improved sleep quality, in line with our original hypothesis, as demonstrated by a significant reduction in perceived SOL and ISI scores. Contrary to our hypothesis, participants did not experience improvements in objectively measured sleep parameters, which may have been due to lack of objective sleep pathology at baseline. For the subsample with clinically significant objectively measured SOL disturbance, the use of the earbuds was associated with objective improvements. Given the consequences of HCW sleep disturbance for both individual health and patient care delivery, easily implementable nonpharmacological interventions, like noise-masking earbuds, are an essential area for ongoing study.
Authorship
All authors made a substantial contribution to this work, helped draft and revise the manuscript, and approved the final submitted version, and participated sufficiently in the work to take public responsibility for appropriate portions of the content. HCH contributed to software and technological implementation; investigation; data curation; writing—original draft; writing—review and editing; visualization; and project administration. SLM was involved in conceptualization; methodology; resources; writing—review and editing; and funding acquisition. KKG was involved in conceptualization; writing—review and editing. RLJ was involved in formal analysis; data curation; visualization; and writing—review and editing. MDS was involved in formal analysis; data curation; visualization; and writing—review and editing. CNE was involved in conceptualization; methodology; supervision; resources; funding acquisition; and writing—review and editing. AMN was involved in conceptualization; methodology; supervision; writing—original draft; and writing—review and editing.
Supplemental Material
sj-docx-1-sci-10.1177_00368504241242276 - Supplemental material for Harnessing technology to improve sleep in frontline healthcare workers: A pilot study of electronic noise-masking earbuds on subjective and objective sleep measures
Supplemental material, sj-docx-1-sci-10.1177_00368504241242276 for Harnessing technology to improve sleep in frontline healthcare workers: A pilot study of electronic noise-masking earbuds on subjective and objective sleep measures by Heinrich C Haller, Susan L Moore, Katherine K Green, Rachel L Johnson, Mary D Sammel, C Neill Epperson and Andrew M Novick in Science Progress
Supplemental Material
sj-docx-2-sci-10.1177_00368504241242276 - Supplemental material for Harnessing technology to improve sleep in frontline healthcare workers: A pilot study of electronic noise-masking earbuds on subjective and objective sleep measures
Supplemental material, sj-docx-2-sci-10.1177_00368504241242276 for Harnessing technology to improve sleep in frontline healthcare workers: A pilot study of electronic noise-masking earbuds on subjective and objective sleep measures by Heinrich C Haller, Susan L Moore, Katherine K Green, Rachel L Johnson, Mary D Sammel, C Neill Epperson and Andrew M Novick in Science Progress
Footnotes
Data availability
The data that support the findings of this study as well as the protocol are available from the corresponding author, AMN, upon reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Review and approval for this study was obtained from the Colorado Multiple Institutional Review Board (protocol #21-2676). All participants provided consent to participation via an electronic consent form approved by the Institutional Review Board.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Eunice Kennedy Shriver National Institute of Child Health and Human Development (grant number K23HD110435), Ludeman Center for Women's Health Research, University of Colorado, and the National Institute on Aging (grant number U54AG062319).
Guarantor
AMN takes full responsibility for this article, including the accuracy and appropriateness of the reference list.
Supplemental material
Supplemental material for this article is available online.
Author biographies
Heinrich C Haller is a medical student at the University of Colorado Anschutz Medical Campus. His research is focused on women's mental health, the use of technology to improve psychiatric outcomes, and psychedelic drug interventions.
Susan L Moore is a Research Assistant Professor in the Colorado School of Public Health and holds a PhD in Health and Behavior Sciences as well as a Master's of Science in Public Health. She specializes in digital health and clinical informatics research and evaluation technology interventions.
Katherine K Green is an Assistant Professor at the University of Colorado Anschutz Medical Campus where she is Medical Director of the Sleep Center. She holds an MD, with specialty training in Otolaryngology and Sleep Medicine, as well as a Master's of Science in Anatomy and Cellular Biology. Her primary research is on sleep-wake disorders, and the use of surgical interventions for obstructive sleep apnea.
Rachel L Johnson is a Senior Research Instructor in the Colorado School of Public Health and holds a Master's of Science in Biostatistics. She has expertise in the use of generalized linear mixed models and survival analysis, with a focus on research in women's mental health.
Mary D Sammel is Professor of Biostatistics, Psychiatry, and Obstetrics and Gynecology at University of Colorado Anschutz Medical Campus and is Associate Director of the Center for Innovative Design and Analysis. She hold a ScD in Biostatistics. Her research interests include observational study design and longitudinal data methods, with specializations in women's health, sex differences, and aging.
C Neill Epperson is the Robert Freedman Professor and Chair of the Department of Psychiatry at the University of Colorado Anschutz Medical Campus. She holds an MD, with specialty training in Psychiatry. Her research focuses on women's mental health across the lifespan, the influence of early life stress and hormones on the brain, and the use of novel technologies to improve psychiatric outcomes.
Andrew M Novick is an Assistant Professor at the University of Colorado Anschutz Medical Campus. He holds an MD with specialty training in psychiatry, as well as a PhD in basic biomedical sciences. His research focuses include the neurobiology of sex steroids and reward processing, as well as the evaluation of novel drug therapies for psychiatric disorders.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
