Abstract
Objective
This study aimed to investigate the effects of low-dose radiation on the abdominal aorta of mice and vascular endothelial cells.
Methods
Wild-type and tumor-bearing mice were exposed to 15 sessions of low-dose irradiation, resulting in cumulative radiation doses of 187.5, 375, and 750 mGy. The effect on the cardiovascular system was assessed. Immunohistochemistry analyzed protein expressions of PAPP-A, CD62, P65, and COX-2 in the abdominal aorta. Microarray technology, Gene Ontology analysis, and pathway enrichment analysis evaluated gene expression changes in endothelial cells exposed to 375 mGy X-ray. Cell viability was assessed using the Cell Counting Kit 8 assay. Immunofluorescence staining measured γ-H2AX levels, and real-time polymerase chain reaction quantified mRNA levels of interleukin-6 (IL-6), ICAM-1, and Cx43.
Results
Hematoxylin and eosin staining revealed thickening of the inner membranes and irregular arrangement of smooth muscle cells in the media membrane at 375 and 750 mGy. Inflammation was observed in the inner membranes at 750 mGy, with a clear inflammatory response in the hearts of tumor-bearing mice. Immunohistochemistry indicated increased levels of PAPP-A, P65, and COX-2 post-irradiation. Microarray analysis showed 425 up-regulated and 235 down-regulated genes, associated with processes like endothelial cell-cell adhesion, IL-6, and NF-κB signaling. Cell Counting Kit 8 assay results indicated inhibited viability at 750 mGy in EA.hy926 cells. Immunofluorescence staining demonstrated a dose-dependent increase in γ-H2AX foci. Reverse transcription quantitative PCR results showed increased expression of IL6, ICAM-1, and Cx43 in EA.hy926 cells post 750 mGy X-ray exposure.
Conclusion
Repeated low-dose ionizing radiation exposures triggered the development of pro-atherosclerotic phenotypes in mice and damage to vascular endothelial cells.
Introduction
Low-dose radiation primarily originates from medical radiation, radiation incidents, and regions with high background radiation. It bears significant implications for public health, particularly concerning its potential link to the cardiovascular system. Although a comprehensive investigation into the direct correlation between low-dose irradiation and blood circulation system incidence is lacking, epidemiological data suggest an association between cardiovascular diseases (CVDs) and exposure to low-to-moderate radiation levels.1–4 The International Commission on Radiological Protection (ICRP) has also emphasized the need to scrutinize the effects of low-dose irradiation on the cardiovascular system. 5
Vascular endothelial cells, situated as a single layer between the bloodstream and vessel walls, wield critical influence over the circulatory system. They modulate arterial wall functions and regulate contractile tension, thus playing a pivotal role.6,7 It is well-established that endothelial cells are sensitive to ionizing radiation. 8 The consequences of radiotherapy-induced changes in endothelial cell function may contribute to atherosclerosis, possibly due to DNA damage triggered by ionizing radiation. This, in turn, prompts endothelial cell activation, transitioning from a quiescent state to a pro-inflammatory phenotype.9–11 Repeated irradiation can compromise the protective capacity of endothelial cells, culminating in endothelial dysfunction, 12 ultimately fostering vascular damage and atherosclerosis development. 13
The emergence of an atherosclerotic phenotype in vivo coincides with increased expression of relevant markers. Notably, the levels of PAPP-A and CD62P are closely associated with the inflammatory response and platelet activation in vivo.14,15 The abundance of PAPP-A in the abdominal aorta's media and inner membranes exhibits a positive correlation with atherosclerosis. 16 CD62P, a 140 kDa cell adhesion molecule, can be induced on endothelial cells and platelets. Its interaction with the P-selectin glycoprotein ligand (PSGL-1) activates platelets, promoting thrombosis and exacerbating inflammation, thereby accelerating atherosclerotic plaque formation. 17 Studies have revealed that the binding of CD62P to PSGL-1 on dendritic cells contributes to atherosclerosis progression via DC activation through the TLR4 signaling pathway. 18 PAPP-A regulates the expression and secretion of monocyte chemoattractant protein-1, IL-6, tumor necrosis factor-α, and interleukin-1β via the NF-κB pathway. 19 Additionally, COX-2, as a downstream gene, is associated with inflammation, fibrosis, and changes linked to carcinogenesis.
This study employs mice to investigate the effect of multiple low-dose irradiations on in vivo atherosclerotic vascular development. Consequently, the levels of PAPP-A and CD62P were assessed to gauge abdominal aorta atherosclerosis progression, while the activation of P65 and COX-2 pathways was also examined. Subsequently, microarray analysis was employed to screen for gene alterations in endothelial cells following low-dose irradiation, further validating the in vivo findings. Finally, certain genes identified through the microarray analysis, encompassing those related to DNA damage, inflammatory factors, and the adhesion molecule ICAM-1, were corroborated in endothelial cells.
Materials and methods
Cell culture
The Human artery endothelial cell line (HAEC) was procured from iCell Bioscience Inc. (iCell-0015a) and maintained in endothelial cell media (ScienCell). The culture medium was supplemented with 5% fetal bovine serum (FBS), 1% growth factors, and 1% penicillin-streptomycin solution. Culturing was carried out at 37°C in an environment with 5% humidity. The EA.hy926 cell line, acquired from the Cell Bank of Type Culture Collection of the Chinese Academy of Sciences, was cultured in Dulbecco's modified Eagle's medium (DMEM, Hyclone, USA) supplemented with 1% penicillin-streptomycin solution (Hyclone, USA) and 10% FBS (Gibco, USA). This cell line was also maintained at 37°C in an atmosphere with 5% humidity. Both cell lines were utilized for experimentation within passages 5–15. Irradiation of the cells was carried out using a KUBTEC XCELL 225 X-ray cabinet. According to the multiple low-dose irradiation to animals, the radiation dose for the injury effect of abdominal aortic endothelial cells was determined to be 187.5, 375, and 750 mGy, delivered at a dose rate of 108 mGy/min.
Animals and treatment
C57BL/6 (B6) mice were obtained from Beijing Vital River Laboratory Animal Technology Co. Ltd. These mice were maintained with unrestricted access to food and water. To establish the murine transplantation tumor model of Lewis cells, a tumor block was generated through the subcutaneous injection of Lewis cells into eight-week-old male C57BL/6 (B6) mice, with the transplantation being performed in the armpit region under anesthesia. This model aimed to simulate a low-immunity scenario, serving as a stress condition for comparison with normal mice to investigate the potential exacerbation of histopathological changes in the abdominal aorta and heart.
A total of 40 mice, comprising twenty wild-type mice and twenty tumor-bearing mice in good physical condition, were designated for irradiation. These mice were randomly divided into four groups, labeled as a, b, c, and d groups, with five mice in each group. Subsequently, both the wild-type and tumor-bearing mice received whole-body X-ray irradiation every other day for a total of 15 sessions. The administered doses were 0, 12.5, 25, and 50 mGy, leading to cumulative doses of 0, 187.5, 375, and 750 mGy, respectively. The X-ray parameters included a voltage of 150 kV, a current of 5 mA, a 2.5 mm-thick aluminum plate, an irradiation distance of 3 m, and an irradiation dose rate of 7 mGy/min. The wild-type mice were euthanized one month after the conclusion of irradiation, while the tumor-bearing mice were euthanized upon reaching a tumor volume of 1000 mm3.
It's noteworthy that our animal experimental protocols received approval from the Experimental Animal Welfare Committee of the National Institute for Radiological Protection (NIRP, act no. 004, 15 March 2023) under the jurisdiction of the Chinese Center for Disease Control and Prevention (China CDC). The study was conducted in accordance with Chinese regulations governing animal experimentation (Ministry of Agriculture, act no. 2001-464, 29 May 2001).
HE staining
The mice were euthanized using humane methods in strict compliance with the Guidelines for Humane End Points for Animals Used in Biomedical Research. 20 Subsequently, the entire length of the abdominal aorta, measuring approximately 3–4 cm, was harvested. In each group, four slices from the aorta were selected for data analysis. These tissue samples were rigorously preserved by fixation in 10% buffered formalin, followed by embedding in paraffin wax. Subsequently, they were sectioned at a thickness of 4 μm and subjected to H&E staining for further examination and analysis.
Immunohistochemistry
Immunohistochemistry was employed to determine the expression levels of PAPP-A, CD62P, P65, and COX-2. In brief, following the standard procedures of dewaxing, dehydration, rehydration, and antigen retrieval using microwave treatment, paraffin sections underwent blocking with 3% H2O2 in deionized water and 5% normal sheep serum in PBS (0.01 M).
Subsequently, the paraffin sections were subjected to incubation with specific primary antibodies targeting PAPP-A (1:400), CD62P (1:400), P65 (1:200), and COX-2 (1:200), all in the presence of 5% normal sheep serum in PBS (0.01 M). This incubation occurred at 4°C overnight. Following the primary antibody incubation, paraffin sections were treated with horseradish peroxidase-conjugated secondary antibodies and incubated at 37°C for 20 min. After thorough rinsing with PBS, a DAB staining solution was applied, and the sections were examined under a microscope. Hematoxylin was employed for counterstaining to visualize the nuclei.
The paraffin sections were observed under a standard light microscope, equipped with a digital camera, with the targeted markers identified by their characteristic brown coloration. The integrated optical density (IOD) value was quantified utilizing IPP (Image Pro Plus) software, from which the mean density (IOD/AREA) was computed. The IOD/AREA values served as indicators of PAPP-A, CD62P, P65, and COX-2 levels in the analyzed samples.
Microarrays analysis
Total RNA extraction from HAEC cells was carried out using Trizol (Life Technologies, Carlsbad, CA, USA). Subsequently, the Ambion® WT Expression Kit was employed to perform reverse transcription of RNA into cDNA. The resulting cDNA fragments were subjected to hybridization on the GeneChip Affymetrix Mouse Clariom® S Array (Affymetrix). Following hybridization, the GeneChips underwent a series of washing and staining steps within the Affymetrix Fluidics Station 450. Data analysis was conducted utilizing the Robust Multichip Analysis algorithm, applying Affymetrix default analysis settings and employing global scaling as the normalization method.
GO enrichment analysis
Gene Ontology (GO) analysis was employed to investigate gene functionality by leveraging differentially expressed genes and arranging them into hierarchical categories according to molecular biological processes. For statistical analysis, both the chi-square test and the two-sided Fisher's exact test were utilized.
Pathway enrichment analysis
Pathway analysis was employed to identify significant pathways among differentially expressed genes, utilizing the Kyoto Encyclopedia of Genes and Genomes (KEGG) database as a reference. The comprehensive KEGG database was parsed to extract all the genes and pathways relevant to the study. Subsequently, the study pathway network was constructed, making use of the pathway topology information available within the KEGG database.
Cell counting kit 8 assay
To evaluate the effect of X-ray irradiation on cell viability, the Cell Counting Kit 8 (CCK8) assay was conducted. In a nutshell, EA.hy926 cells were seeded into 96-well plates at a density of 6000 cells per well, with three replicate wells established for each experimental group. Subsequently, the cells were cultured in the incubator for 24 h, 48 h, and 72 h following irradiation with X-ray doses of 187.5, 375, and 750 mGy, respectively. At the specified time points, 10 µL of CCK8 solution (Dojindo, Tokyo, Japan) was added to each well, followed by a 3 h incubation in a cell culture incubator. The optical density of each well was then determined at 450 nm using a Thermo Scientific microplate reader.
Immunofluorescence staining
EA.hy926 cells were initially seeded onto glass slides and allowed to incubate for a duration of 12 h. Following 1 h irradiation, the cells underwent fixation using 3% paraformaldehyde, permeabilization with 0.25% Triton X-100 in PBS and blocking with 5% BSA. Subsequently, the cells were subjected to probing with the primary antibody against γH2AX (1/400; Cell Signaling #9718 s) overnight at 4 °C.
After thorough washing, the cells were incubated with a secondary antibody, Alexa Fluor 568 (1/600; Cell Signaling #8889 s), for 1 h. Coverslips were then applied, with 4’,6-diamidino-2-phenylindole incorporated for nuclear staining, and subsequently sealed. The quantification of γH2AX foci was carried out using a fluorescence microscope (LSM 700, Carl Zeiss, Oberkochen, Germany). Image analysis was conducted using ImageJ software.
Reverse transcription quantitative PCR
Total RNA extraction from the cells was carried out utilizing Trizol (Invitrogen). Subsequently, cDNA was synthesized from 1 μg of total RNA employing the PrimeScript RT Master Mix (TOYOBO, Japan). Reverse transcription quantitative PCR (RT-qPCR) was conducted using the Fast SYBR Green Master Mix (TOYOBO, Japan) on a 7500 Fast Real-Time PCR system. Quantification of gene expression was performed using the delta-delta CT method, with target gene expression levels being normalized to β-actin expression levels. The primer sequences used are provided below:
ICAM-1, ATCTGTGTCCCCCCTCAAAAGTC (forward), CCATCAGGGCAGTTTGAATAGC (reverse); IL-6, 5’-ACTCACCTCTTCAGAACGAATTG-3’ (forward), 5′-CCATCTTTGGAAGGTTCAGGTTG-3’ (reverse); Cx43, 5′-TCTGAGTGCCTGAACTTGC-3’ (forward), 5′-ACTGACAGCCACACCTTCC-3’ (reverse); β-actin, 5’-ATCACCATTGGCAATGAGCG-3’ (forward), 5’-TTGAAGGTAGTTTCGTGGAT-3’ (reverse).
Statistical analysis
The GraphPad Prism 7.0 software was used to conduct graphs and statistical analyses. One-way ANOVA statistical method was used for comparisons for multi-group. Differences were considered statistically significant at P < 0.05.
Results
Low-dose irradiation induced the abdominal aorta tissue injury
The extent of tissue damage in the abdominal aorta was assessed through HE staining. The results revealed a notable thickening of the inner membranes of the abdominal aorta, with an increase of at least twofold observed after irradiation with doses of 375 mGy and 750 mGy (Figure 1A, c1 and d1) in comparison to the 0 mGy group (Figure 1A, a1). Furthermore, in the 750 mGy irradiation group (Figure 1A, d1), there was not only an abundance of oval foam cells but also evidence of inflammatory cell infiltration, along with a small number of neutrophils. Additionally, in the cumulative doses of 375 mGy and 750 mGy groups (Figure 1A, c1 and d1), the smooth muscle cells in the media membrane exhibited a more irregular arrangement than in the 0 mGy group (Figure 1A, a1).

He staining results of the abdominal aorta and hearts in mice irradiated with different doses of X-ray. I-inner membrane, M-media membrane, O-outer membrane, FC-foam cell, IC-inflammatory cell, IS-intercellular membrane, BBC-boundary between myocardial cells. (A) HE staining results of the abdominal aorta in wild-type mice; (B) HE staining results of the abdominal aorta in tumor-bearing mice; (C) HE staining results of hearts in wild-type mice; (D) HE staining results of hearts in tumor-bearing mice. Bar = 50 µm or 10 µm.
HE staining of the abdominal aorta demonstrated that in all tumor-bearing mice irradiation groups (Figure 1B, b1, c1, and d1), the contour of the outer membrane became indistinct, the outer membrane thickened with inflammatory cell infiltration, smooth muscle cells exhibited significant disarray, and there were substantial numbers of oval foam cells, contrasting with the 0 mGy group (Figure 1B, a1).
Furthermore, in the cumulative doses of the 375 mGy group (Figure 1C, c1), the intercellular septum appeared enlarged compared to the 0 mGy group (Figure 1C, a1). Meanwhile, in the cumulative doses of the 750 mGy group (Figure 1C, d1), the boundary between cardiomyocytes became less distinct. In all tumor-bearing mice (Figure 1D), particularly evident after 750 mGy irradiation (Figure 1D, d1), the arrangement and compactness of cardiomyocytes declined. Cardiomyocytes exhibited deformation, and their arrangement became distorted following 375 mGy irradiation in tumor-bearing mice (Figure 1D, c1). Notably, HE staining of the hearts in tumor-bearing mice highlighted a clear inflammatory response.
Low-dose irradiation induced the initiation and progression of abdominal aortic atherosclerosis in mice
The levels of PAPP-A and CD62P following radiation exposure serve as indicators for estimating the progression of abdominal aorta atherosclerosis. The findings revealed a significant increase in PAPP-A levels within the inner membranes of wild-type mice in the cumulative dose groups of 187.5 mGy, 375 mGy, and 750 mGy compared to the control group (Figure 2A). This suggests a progression of atherosclerosis following irradiation. In the 0 mGy group, there was no detectable expression of PAPP-A, but a positive correlation was observed between the expression of PAPP-A and the radiation dose in the other groups. Additionally, the expression of PAPP-A was higher in the outer membrane of tumor-bearing mice in the cumulative dose groups of 375 mGy and 750 mGy (Figure 2B).

The level of PAPP-A and CD62P was examined by immunohistochemical staining in the abdominal aorta (indicated by arrows), followed by semi-quantitative analysis. (A) The PAPP-A level in the abdominal aorta of wild-type mice irradiated with different doses of X-ray; (B) The PAPP-A level in the abdominal aorta of tumor-bearing mice irradiated with different doses of X-ray; (C) The CD62P level in the abdominal aorta of wild-type mice irradiated with different doses of X-ray; (D) The CD62P level in the abdominal aorta of tumor-bearing mice irradiated with different doses of X-ray. (*means P < 0.05, compared with control).
Furthermore, the results indicated an increase in the expression of CD62P in the inner membrane of wild-type mice in the irradiation groups when compared to the control group, with a notable increase observed in the 187.5 mGy group (Figure 2C).
PAPP-A activates the Nf-κb pathway in the mice of radiation-induced abdominal aorta atherosclerosis phenotype
The assessment of P65 levels within the inner membranes of abdominal aorta atherosclerosis revealed an elevation in P65 expression in wild-type mice exposed to cumulative doses of 375 mGy and 750 mGy (Figure 3A). Additionally, in the cumulative dose groups of 187.5 mGy and 375 mGy, there was a significant increase in P65 levels within the inner membranes of tumor-bearing mice (Figure 3B). These findings indicate that PAPP-A activation is associated with the NF-κB pathway in mice with radiation-induced abdominal aorta atherosclerosis.

The levels of P65 and COX-2 were examined by immunohistochemical staining in the abdominal aorta (indicated by arrows), followed by semi-quantitative analysis. (A) The P65 level in the abdominal aorta of wild-type mice irradiated with different doses of X-ray; (B) The P65 level in the abdominal aorta of tumor-bearing mice irradiated with different doses of X-ray; (C) The COX-2 level in the abdominal aorta of wild-type mice irradiated with different doses of X-ray; (D) The COX-2 level in the abdominal aorta of tumor-bearing mice irradiated with different doses of X-ray. (*means P < 0.05, compared with control).
Activation of Nf-κb promoted the COX-2 level in the mice of radiation-induced abdominal aorta atherosclerosis
A notable positive correlation was observed between the expressions of PAPP-A, P65, and COX-2 within the inner membranes, displaying a similar pattern. Specifically, there was a significant increase in COX-2 levels within the inner membranes in the cumulative dose group of 750 mGy (Figure 3C). In tumor-bearing mice, not only did the level of COX-2 increase in the media membrane but there was also a significant elevation of COX-2 within the inner membranes in the cumulative dose groups of 375 mGy and 750 mGy (Figure 3D).
The altered genes were studied by microarray analysis after 375 mGy X-ray irradiation in the endothelial cell
Gene expression changes were assessed in HAECs harvested 24 h following exposure to 375 mGy of X-ray irradiation. Microarray analysis revealed 660 differentially expressed mRNAs between the irradiation and control groups (fold change > 1.5; P < 0.05), with 425 genes exhibiting upregulation and 235 genes showing downregulation.
Further enrichment analysis was conducted, including GO and Pathway analysis. The GO analysis results indicated that the upregulated genes were associated with biological processes such as type I interferon signaling, endothelial cell-cell adhesion, IL-6 signaling, and NF-κB signaling (Figure 4A). In contrast, the downregulated genes were linked to processes related to smooth muscle cell migration and cell viability (Figure 4B). Based on the analysis of the KEGG database, 29 significantly upregulated pathways were identified, encompassing crucial pathways like the chemokine signaling pathway, TNF signaling pathway, and NF-κB signaling pathway(Figure 4C). Additionally, 20 significantly downregulated pathways were identified, including pathways such as oxidative phosphorylation, RNA polymerase activity, tyrosinase metabolism, and others (Figure 4D).
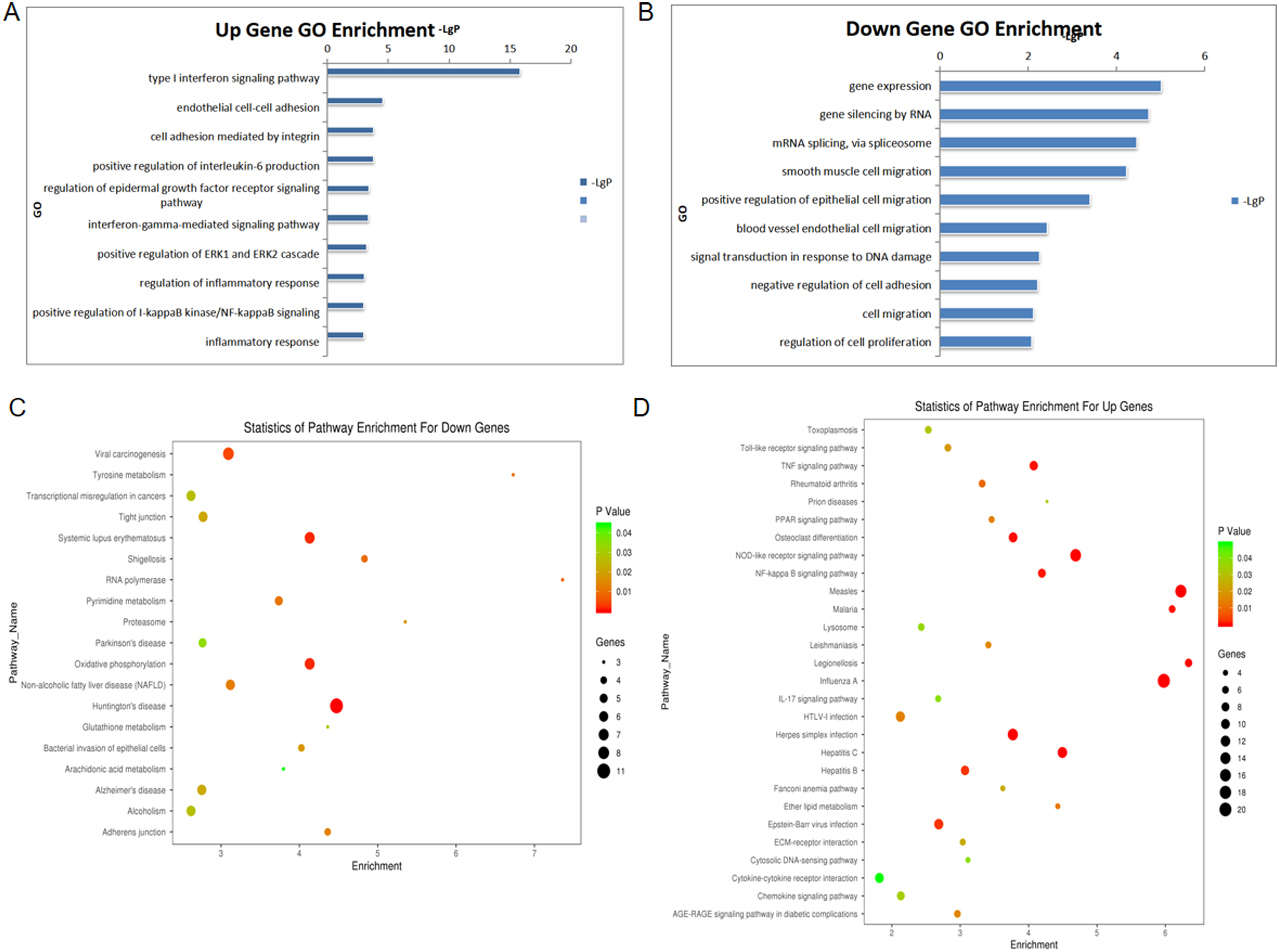
Comparison between differential expression of mRNA in HAEC cells following 375 mGy X-ray irradiation. (A, B) GO term cluster expressed genes in HAEC cells at 24 h after 375 mGy X-ray irradiation; (C, D) KEGG pathway enrichment analyses of differentially expressed genes in HAEC cells at 24 h after 375 mGy X-ray irradiation.
Low-dose irradiation induced the endothelial cell damage
To validate the effects of low-dose radiation, both the CCK8 assay and immunofluorescence staining of γ-H2AX foci were conducted.
The CCK8 assay results revealed that a dose of 750 mGy resulted in the inhibition of viability in the EA.hy926 cell line, particularly evident at the 72 h post-irradiation time point (Figure 5A). The γ-H2AX assay, which involves the quantification of γ-H2AX foci within cell nuclei, is an effective method for detecting radiation-induced DNA double-strand breaks. Figure 5B displays the images depicting γ-H2AX foci in EA.hy926 cells. The results from immunofluorescence staining demonstrated a dose-dependent increase in the number of γ-H2AX foci (Figure 5C).

Endothelial cell injury effects by ionizing radiation (IR) in EA.hy926. (A) Cell viability was measured by the CCK8 assay in EA.hy926 after irradiation; (B) Representative images of γ-H2AX foci formation in EA.hy926 are shown; (C) Number of γ-H2AX foci in EA.hy926 1 h after 187.5, 375 and 750 mGy X-rays irradiation. (*means P < 0.05, **means P < 0.01, compared with control; n = 3).
Low-dose irradiation induced a pro-inflammatory state in the endothelial cell
The inflammatory status of endothelial cells plays a crucial and early role in atherosclerosis development. Key pro-inflammatory cytokines like IL6 and adhesion factors such as ICAM-1, which are implicated in atherosclerosis, were assessed. RT-qPCR results revealed a significant increase in IL6 and ICAM-1 secretion in EA.hy926 cells at the 72 h time point following exposure to 750 mGy X-ray irradiation (Figure 6A, B).

The expression changes of IL-6, ICAM-1, and Cx43 mRNA in EA.hy926 cells were analyzed by RT-qPCR at 72 h after irradiation of 187.5, 375, and 750 mGy. (*means P < 0.05, compared with control; n = 3).
Interestingly, several studies have indicated that Cx43 typically exhibits low expression levels in healthy endothelium, and its increased expression has been associated with enhanced atherosclerotic lesions in vivo.21,22 The results from RT-qPCR demonstrated elevated expression of Cx43 in EA.hy926 cells at 72 h after irradiation with doses of 187.5, 375, and 750 mGy X-rays (Figure 6C).
Discussion
Epidemiological studies have indeed provided evidence indicating an association between exposure to low-dose irradiation and an elevated risk of CVD.1–4 Nevertheless, direct investigations into the causative relationship between low-dose ionizing radiation and CVD have been relatively scarce. Notably, the underlying biological mechanisms responsible for the low-dose effect may diverge from those governing the high-dose counterpart. 23 Atherosclerosis, a prevailing etiological factor, frequently underpins the pathogenesis of CVDs. In this context, atherosclerosis acts as a comorbidity factor that intensifies and expedites structural cardiac degeneration in response to low-dose ionizing radiation. 24
Research findings have substantiated that low-dose irradiation constitutes a risk factor for carotid atherosclerotic disease. 25 Following exposure to a 100 mGy low-dose X-ray thoracic irradiation, atherosclerosis-prone ApoE-/- mice exhibit acute and early inflammatory reactions. 26 Similarly, LDLR-/- mice manifest an atherosclerotic phenotype after exposure to a 100 mGy low-dose irradiation. 27 Furthermore, endothelial cells subjected to a single X-ray dose of 500 mGy exhibit elevated levels of pro-atherosclerotic and pro-inflammatory cytokines, namely, IL6 and CCL2. 9 The conducted study has disclosed a pro-atherogenic phenotype subsequent to multiple low-dose irradiations in mice. PAPP-A, recognized as an early marker of atherosclerosis, concurrently signifies an increased local inflammatory state and has been documented in coronary atherosclerosis. 28 Significantly, a substantial positive correlation exists between PAPP-A and COX-2 expression in coronary arteries. 29 The current investigation has elucidated that PAPP-A possesses the capability to activate NF-κB through a sequence of cascading reactions, ultimately eliciting COX-2 production.30,31 Notably, our statistical analysis has confirmed a significant positive correlation between PAPP-A and COX-2 levels within the media and inner membranes (Figure 2, 3). Our data unequivocally illustrate that under the precondition of heightened NF-κB, PAPP-A, COX-2, and CD62P, positive elements are localized within endothelial cells of the inner membrane and the cytoplasm of smooth muscle cells in the media membrane.
Indeed, studies have illuminated that tumors exert not only a detrimental influence on the immune system at the primary tumor site but also extend their effect to the systemic immune milieu.32,33 Utilizing a tumor-bearing mouse model to simulate a low-immunity scenario, we have juxtaposed it with wild-type mice to explore whether abdominal aorta and heart histopathological alterations would be exacerbated within the context of a stressful environment. Histological examination via HE staining has unveiled that wild-type mice exhibit inner membrane thickening, inflammatory cell infiltration, and foam cell formation, while tumor-bearing mice subjected to irradiation display outer membrane thickening, inflammatory cell infiltration, and foam cell formation. Both groups, whether wild-type or tumor-bearing, within the irradiation cohort have exhibited heightened intercellular septation, reduced tissue compactness, and blurred intercellular demarcations. Notably, HE staining of the hearts of tumor-bearing mice has unequivocally indicated a pronounced inflammatory response. Immunohistochemistry has further substantiated increased expression of NF-κB, COX-2, and CD62P in both wild-type and tumor-bearing mice. Remarkably, PAPP-A expression predominantly registers higher in the adventitia of tumor-bearing mice.
To corroborate the results derived from our animal experiments, we employed endothelial cells for in vitro investigations. Endothelial cells include a multifaceted role encompassing essential secretory, metabolic, and immunological functions. When subjected to external stimuli, the endothelium's typical physiological functions become disrupted, setting in motion a cascade of molecular alterations that culminate in atherosclerosis and CVD.34,35 Radiation exposure induces various forms of cellular damage, with DNA double-strand breaks holding particular significance. In our investigation, we observed a dose-dependent escalation in the number of γ-H2AX foci in endothelial cells following low-dose irradiation, a finding congruent with previous findings. 36
Another critical aspect addressed is the propensity of low-dose irradiation to shift the endothelial quiescent phenotype towards a pro-inflammatory state. This pro-inflammatory phenotype is characterized by the upregulation of chemokines, cytokines, and adhesion molecules.37,38 Atherosclerotic plaques develop as inflammatory cells migrate from the bloodstream into the intima, where they assume a foam cell phenotype. 39 The expression of inflammatory and adhesion factors by endothelial cells assumes primary significance in this context. Pathway analysis results indicate the upregulation of the chemokine signaling pathway, TNF signaling pathway, and NF-κB signaling pathway, indicative of an inflammatory activation. Our research discerned heightened expression of IL-6 and the adhesion molecule ICAM-1 in endothelial cells subsequent to low-dose irradiation, signifying the activation of endothelial cells and their transition from a quiescent to a pro-inflammatory state. Existing studies have suggested that Cx43 exhibits low expression in healthy endothelium but undergoes a substantial upregulation in atherosclerotic lesions in vivo. 21 It has also been reported that the elevation of Cx43 expression enhances adhesion factors. 40 In our investigation, we observed an increase in connexins Cx43 levels in EA.hy926 cells at 72 h following exposure to 187.5, 375, and 750 mGy X-irradiation. In light of the aforementioned findings, it is reasonable to conclude that multiple exposures to low-dose ionizing radiation induce the initiation and progression of pro-atherosclerotic phenotypes in both mice and vascular endothelial cells.
However, it is essential to acknowledge certain limitations inherent in this paper. Regarding sample size determination, while the experimental sample size exceeds the minimum required for statistical analysis (≥3), an efficacy analysis was not undertaken.
Conclusion
In vivo, these findings imply that PAPP-A potentially facilitates COX-2 production through the upregulation of NF-κB. Furthermore, multiple exposures to low-dose X-ray irradiation instigate the development of pro-atherosclerotic phenotypes in mice.
In vitro, low-dose X-ray irradiation triggers DNA damage and enhances the secretion of adhesion and inflammatory factors within endothelial cells. Subsequently, multiple exposures to low-dose ionizing radiation initiate and advance pro-atherosclerotic phenotypes in both mice and vascular endothelial cells while causing cellular damage.
Footnotes
Acknowledgment
We thank Xiaohe Dong, Renjun Peng, Sinian Wang, and Dongshu Liu (The People's Liberation Army of China (PLA) Rocket Force Characteristic Medical Center) for their generous advice, assistance, and facility. We thank Jie Chen of our group for the data analysis and processing, image editing, writing the discussion part, grammar revision, etc.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Beijing Municipal Natural Science Foundation (7202139 and 7162137) and the National Natural Science Foundation of China (82273551 and 31570852).
Author biographies
Meng-Meng Liu received her MS degree in Radiation Medicine from China CDC Key Laboratory of Radiological Protection and Nuclear Emergency, National Institute for Radiological Protection, Beijing, P.R. China in 2023. She is currently working at Heilongjiang Provincial Hospital. Her research interests include radiation damage and radiobiological effects.
Chun-Yan Ding received the BS degree in Applied Physics (specialized in medicine) from Shandong First Medical University, Taian, China, in 2012 and an MS degree in Radiology from China CDC Key Laboratory of Radiological Protection and Nuclear Emergency, National Institute for Radiological Protection, Beijing, P.R. China, in 2019.
Zhi-Hui Li received her MS degree from Shanxi Medical University, Taiyuan, China, in 2017. She is currently working at the Department of Thoracic and Cardiovascular Surgery in Yanhua Hospital, Beijing, China, where she was engaged in clinical work.
Ru-Han Yi received her BS degree in Preventive Medicine from Southern Medical University, Guangzhou, China, in 2022. She is currently working towards an MS degree in Radiology with the China CDC Key Laboratory of Radiological Protection and Nuclear Emergency, National Institute for Radiological Protection, Chinese Center for Disease Control and Prevention, Beijing, China. Her research interests include radiation damage and radiobiological effects.
Li-Ping Ma received her BS degree in Preventive Medicine from Shanxi Medical University in 2019. She received her M.S. degree in Radiological Medicine from Chinese Center for Disease Control and Prevention in 2022. She is currently working toward the PhD degree with the Institute of Military Medical Sciences, Academy of Military Sciences, majoring in Health Toxicology. Her research interests include Radiation Medicine, Radiobiology, Radiotoxicology, and Radiation Damage Effects.
Xiang-Ming Ou, bachelor’s degree, chief technician, is engaged in the study of radiation protection dosage for medical diagnostic and treatment equipment.
Hai-Xiang Liu received her BS degree in Biotechnology North China University of Science and Technology, Tangshan, China, in 2010, an MS degree in Genetics from Anhui Medical University, Hefei, China, in 2013, and a PhD degree in Radiation Medicine from National Institute for Radiological Protection, Chinese Center for Disease Control and Prevention, Beijing, China in 2022. Her research interests include radiation biomarkers and related molecular mechanisms.
Ling Gao, Research Fellow, Doctoral Supervisor. Engaged in research on the effects and mechanisms of radiation damage. the PhD degree in institute of Radiology and Medicine, Academy of Military Medical Sciences, Beijing, China in 2008. From 2008 to 2010, she conducted postdoctoral research in the School of Life Sciences/the State Key Laboratory of Bio-membrane and Membrane Bio-engineering, Tsinghua University, Beijing, China. Since 2010, she has been working at the radiation medicine from National Institute for Radiological Protection, Chinese Center for Disease Control and Prevention, Beijing, China.
Qing-Jie Liu, PhD, Researcher, Doctoral Supervisor, received his BS degree in Preventive Medicine from Hebei Medical College, China in 1993, MS degree in Labour Health and Occupational Diseases (Radiation Genetics) from Chinese Academy of Preventive Medical Sciences (CAPMS) in 1996, and PhD degree in Radiology from Academy of Military Medical Sciences (AAMS) in 2001. From 1999 to 2001, he was a visiting scholar at the Laboratory of Molecular Genetics, Cedars-Sinai Medical Centre, University of California, Los Angeles, U.S.A. From 2001 to 2004, he was a postdoctoral fellow at the Institute of Basic Medical Sciences, Peking Union Medical College, China. He is currently the deputy director of the Institute of Radiation Safety, CDC, and is mainly engaged in the screening of radiation biomarkers by multi-omics.
