Abstract
Brain arteriovenous malformation (BAVM) is an abnormality in the cerebral vascular system. Although the upregulation of the Notch signalling pathway is a deterministic factor in BAVM, the mechanism by which this pathway is upregulated in patients with BAVM is uncertain. The effects of serum starvation and vascular endothelial growth factor (VEGF) stimulation on the Notch signalling pathway in brain microvascular endothelial cells (MECs) and mouse embryonic stem (mES)/embryoid body (EB)-derived endothelial cells were investigated in this study. The duration of serum starvation and VEGF concentration were changed, cell viability was measured, and reasonable time and concentration gradients were selected for subsequent studies. Protein and mRNA expression levels of Notch signalling pathway components in both MECs and mES/EB-derived endothelial cells were detected using western blotting and real-time PCR, respectively. Expression levels of the Notch1, Notch4, Jagged1, delta-like ligand 4 (Dll4) and Hes1 proteins and mRNAs were upregulated by lower VEGF concentrations and shorter-term serum starvation but inhibited by higher VEGF concentrations and longer-term serum starvation. This study revealed effects of changes in the duration of serum starvation and VEGF concentration on the expression of Notch signalling pathway components in both MECs and mES/EB-derived endothelial cells, potentially contributing to BAVM formation.
Keywords
Introduction
Brain arteriovenous malformations (BAVMs) are fast-flow vascular malformations that were first described more than 200 years ago. 1 BAVMs are lesions of the cerebral vasculature that shunt arterial blood flow directly into the venous system without passage through a capillary system, resulting in a series of cerebral haemodynamic disorders.2,3 The clinical manifestations of BAVMs include haemorrhages, seizures, headaches and non-haemorrhagic neurological deficits. 1
Patients with BAVMs are at risk of life-threatening vascular rupture and intracranial haemorrhage (ICH). The malformed blood vessels are fragile and prone to rupture, causing cerebral haemorrhage. 4 ICH is the first clinical symptom in approximately 50% of patients with BAVM. 5 Multiple factors might increase the risk of rupture and haemorrhage, such as age, a single draining vein, haemorrhage at initial presentation and diffuse arteriovenous malformation (AVM) morphology.6,7 However, the molecular mechanisms underlying BAVM remain unclear.8,9 Studies have found that upregulation of the Notch signalling pathway is associated with BAVM.3,10,11
The Notch signalling pathway, which was originally discovered in Drosophila, has emerged as a critical mediator of vascular angiogenesis and the development of human AVMs. 12 In mammals, the Notch signalling pathway consists of four structurally related receptor proteins (Notch1-4) and five ligands (Jagged 1 and 2 and delta-like ligand (Dll) 1, 3 and 4).1,13 The Notch pathway regulates cell fate and differentiation through cell-cell communication. 13 Notch receptors and their ligands are transmembrane proteins. 12 Cell-cell interactions mediate the activation of Notch receptors through ligand binding to the receptor extracellular domain, resulting in sequential cleavage events and the release of an active Notch intracellular domain (NICD) that subsequently translocates to the nucleus and initiates the transcription of downstream genes.3,10,14 Hes1 is a downstream Notch target gene.11,12
Although the activation of Notch signalling in BAVMs has been described, 3 the mechanism by which the Notch signalling pathway is upregulated in BAVMs remains obscure. Vascular endothelial growth factor (VEGF) is overexpressed in human BAVM tissues, and inhibition of VEGF signalling in experimental models prevents BAVM formation. 15 The presence of structural defects and immature sections of the vessel wall in BAVMs indicate that vessels are embryogenically maldeveloped. Myometrial hypertrophy, expansion of the outburst diameter and an abnormal increase and transparency of elastic fibres may result from vascular remodelling induced by cerebral haemodynamic changes due to an arteriovenous shunt. 16 Therefore, we speculated that elevated VEGF expression and serum starvation would upregulate the Notch signalling pathway.
Currently, many cases of BAVM regeneration after complete surgery or embolization have been reported, reflecting the mysterious cause of BAVM.17,18 Dysplasia during the embryonic period has been shown to cause cerebral AVMs, and even in adulthood, Notch1 expression in endothelial cells is sufficient to cause vascular malformations. 3 Although BAVM may be congenital, it still may be dynamic and continue to grow, regress or even reappear.1,19 Based on these results, the Notch signalling pathway is evolutionarily conserved and responsible for determining cell fate in both developing embryos and mature tissues. Thus, mouse embryonic stem (mES)/embryoid body (EB)-derived endothelial cells were utilized in this study.
In this study, we investigated the effects of serum starvation and VEGF stimulation on the Notch signalling pathway in brain microvascular endothelial cells (MECs) and mES/EB-derived endothelial cells. We changed the duration of serum deprivation and VEGF concentration and evaluated the proliferation of the treated cells. We examined the protein and mRNA expression levels of Notch signalling pathway components in both MECs and mES/EB-derived endothelial cells. Our studies enrich our understanding of the pathogenesis of BAVM and provide an experimental basis for guiding the clinical treatment of BAVM.
Materials and methods
Cell culture
mES cells (ATCC, CRL-1934, Rockville, MD, USA) were co-cultured with gamma-irradiation-treated mouse embryonic fibroblast (MEF) cells in high-glucose DMEM (HyClone, Beijing, China) containing 15% foetal bovine serum (FBS) (Gibco, Grand Island, USA), 10 ng/mL leukaemia inhibitory factor (LIF; Sigma, St. Louis, MO, USA), 1% penicillin, 1% streptomycin, 1% non-essential amino acids (Sigma, St. Louis, MO, USA) and 0.1 mM β-mercaptoethanol (Sigma, St. Louis, MO, USA). EBs were cultured in DMEM containing 10% FBS, 1% penicillin, 1% streptomycin (Invitrogen, Carlsbad, CA, USA), 1% non-essential amino acids and 0.1 mM β-mercaptoethanol to promote endothelial cell differentiation. Suspension culture was used for EB formation in 60 mm petri dishes. After MEF removal, suspension culture in a 60 mm culture dish can induce EB formation. After an incubation for 5 days, EBs were transferred to six-well plates for cell differentiation. 20 mES/EB-derived endothelial cells were identified by the expression of CD34, a representative endothelial biomarker.
Rat brain MECs were purchased from BeNa Culture Collection (Kunshan, China) and cultured in DMEM supplemented with 10% FBS, 100 IU/mL penicillin and 100 μg/mL streptomycin in a humidified incubator with 5% CO2 at 37°C.
Cell viability
MECs (2.5 × 103 cells/well) were seeded into 96-well plates with DMEM supplemented with 10% FBS and cultured for 24 h to allow cell attachment and to assess cell viability. The culture medium was then removed, and the cells were washed with PBS three times and then cultured with serum-free medium for 24 h. The cells were then cultured with serum-deprived medium (DMEM with 2% FBS) for 1, 3, 5 and 7 days or in serum-free medium containing various concentrations of VEGF (5, 10, 15, 20, 25, 30, 40, 50, 75, 100, 125, 150, 175 or 200 ng/mL) (Sigma, St. Louis, MO, USA) at 37°C for 24, 48, or 72 h. After the incubation, cell viability was examined using a Cell Counting Kit-8 (CCK-8; Dojindo, Kumamoto, Japan) assay. The absorbance was measured at 450 nm using a TECAN Infinite M200 microplate reader (Tecan Group Ltd., Männedorf, Switzerland).
Western blotting
MECs were cultured in serum-deprived medium (DMEM with 2% FBS) for 1, 3, 5 or 7 days or in serum-free medium with various concentrations of VEGF for 5 days. mES/EB-derived endothelial cells were cultured in serum-deprived medium (DMEM with 2% FBS) for 14 days or in serum-free medium with various concentrations of VEGF for 30 days. MECs or mES/EB-derived endothelial cells were lysed with RIPA buffer (Beyotime, China) containing 1% protease inhibitor (PMSF). Protein concentrations were measured using a bicinchoninic acid protein assay kit (Beyotime, China). The total proteins were separated via sodium dodecyl sulphate–polyacrylamide gel electrophoresis and transferred to polyvinylidene fluoride (PVDF) membranes (GE Healthcare Life Sciences, UK). The membranes were blocked with 5% milk for 1 h and incubated overnight at 4°C with antibodies against Notch1 (1:1000, ABclonal), Jagged1 (1:1000, ABclonal), Dll4 (1:1000, ABclonal) and Hes1 (1:1000, ABclonal). After washes with PBST, the membranes were incubated with horseradish peroxidase (HRP)-conjugated anti-rabbit (1:2000; ABclonal) or anti-mouse (1:2000; ABclonal) secondary antibodies for 1–2 h, followed by three washes with PBST. The target protein signal was detected and visualized using an enhanced chemiluminescence (ECL) kit (Beyotime, China).
Quantitative real-time polymerase chain reaction (qRT-PCR)
MECs were cultured in serum-deprived medium (DMEM with 2% FBS) for 1, 3, 5 or 7 days or in serum-free medium with various concentrations of VEGF for 5 days. mES/EB-derived endothelial cells were cultured in serum-deprived medium (DMEM with 2% FBS) for 14 days or in serum-free medium with various concentrations of VEGF for 30 days. Total RNA was extracted from MECs or mES/EB-derived endothelial cells using TRIzol Reagent (Invitrogen, Carlsbad, USA), reverse transcription (RT) was performed using a PrimeScript RT Reagent Kit with gDNA Eraser (Takara Biotechnology Co., Ltd., Dalian, China), and qRT-PCR was performed using SYBR Premix Ex Taq II (Takara Biotechnology Co., Ltd., Dalian, China). All procedures were performed according to the manufacturers’ protocols. The qRT-PCR oligonucleotide primers used in this study are shown in Table 1. Amplification and analysis were performed using a real-time PCR system (ABI PRISM 7500, Applied Biosystems, Foster City, USA). The PCR cycling conditions were as follows: 95°C for 30 s; 40 cycles of 95°C for 5 s and 60°C for 34 s; and one cycle of 95°C for 15 s, 60°C for 1 min, and 95°C for 15 s. qRT-PCR data were analysed with the 2−ΔΔCT method and normalized using the GAPDH cDNA as the internal control.
Nucleotide sequences of primers used for qRT-PCR.
Statistical analysis
All experiments were performed in triplicate and repeated at least three times. Data were analysed using SPSS software (Chicago, Illinois, USA). Data are presented as the means ± standard deviations (SDs) (n = 3) and were statistically analysed. The statistical significance of the results was determined using one-way analysis of variance followed by Tukey’s post hoc multiple comparison test. A p-value <0.05 was considered statistically significant. * indicates p < 0.05; ** indicates p < 0.01; and *** indicates p < 0.001.
Results
Effects of serum starvation on MEC cell proliferation
Cell viability was measured at 1, 3, 5 and 7 days of serum deprivation using the CCK-8 method to assess the effects of serum starvation of MECs. Cell viability gradually decreased with the extension of the serum starvation time. Serum starvation exerted a significant effect on cell viability (p < 0.05) (Figure 1).

Effects of serum starvation on MEC proliferation. Relative growth rates of MECs after culture in serum-free medium for 1, 3, 5 and 7 days. All experiments were performed in triplicate. Data are presented as the means ± SD (n = 3).
Effects of serum starvation on the Notch signalling pathway in MECs
The expression of Notch pathway-related proteins was assessed using western blotting to examine the relationship between serum deprivation and the Notch pathway in BAVM. Figure 2 displays the protein levels in MECs cultured with serum-free medium for 1, 3, 5 and 7 days. The expression levels of Notch1, Jagged1, Dll4 and Hes1 were all upregulated after serum deprivation. The expression levels of Notch1, Jagged1, Dll4 and Hes1 peaked after serum deprivation for 5 days (p < 0.05).

Effects of serum starvation on the expression of proteins related to the Notch signalling pathway in MECs: (a) western blots showing levels of the Notch1, Hes1, Jagged1 and Dll4 proteins, with β-actin serving as the internal control (n = 3) and (b) quantification of Notch1, Hes1, Jagged1 and Dll4 levels normalized to β-actin levels (n = 3). Data are presented as the means ± SD.
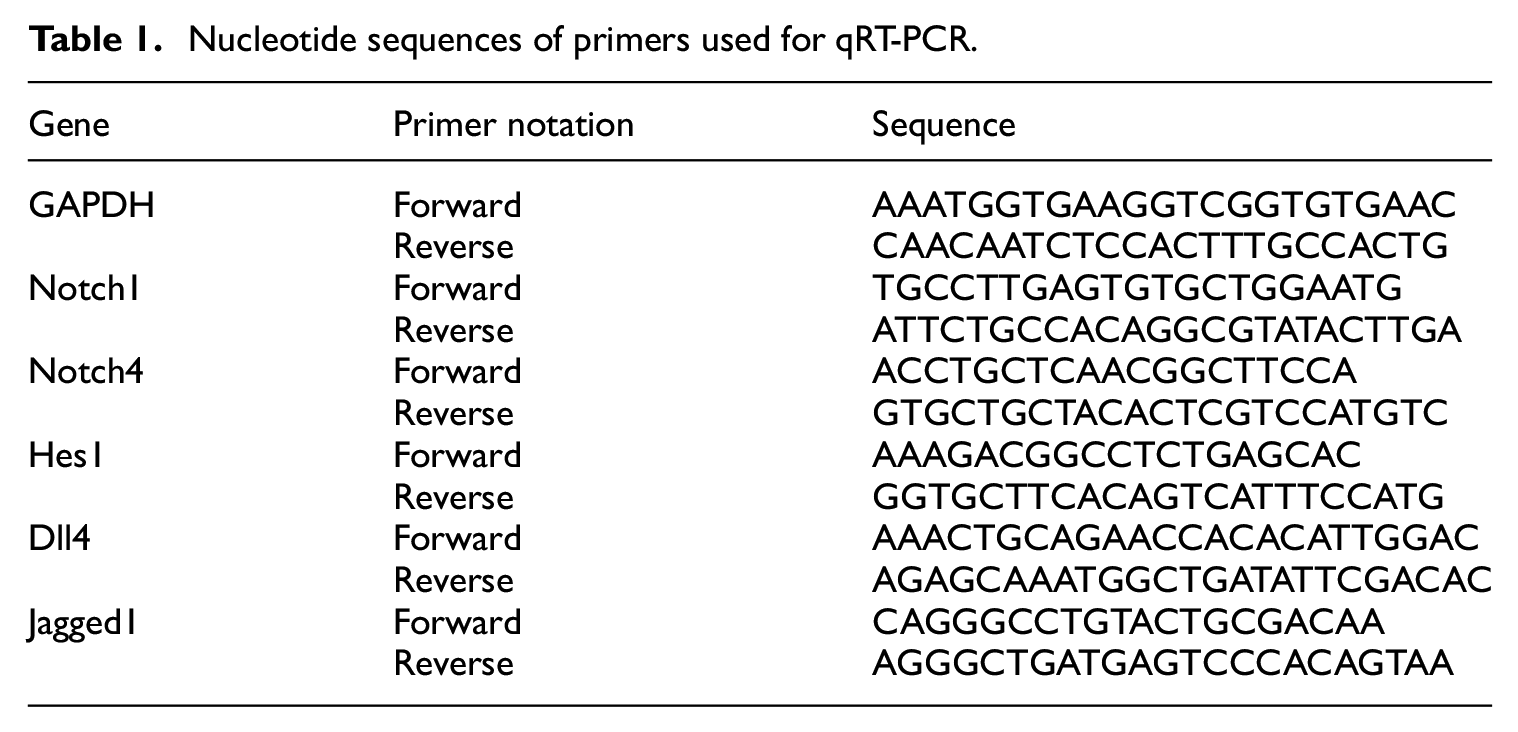
Expression levels of the Notch1, Notch4, Jagged1, Dll4 and Hes1 mRNAs in MECs cultured in serum-free medium for 1, 3, 5 and 7 days were detected using real-time PCR to further confirm the relationship between serum deprivation and the overexpression of components of the Notch signalling pathway. As expected, the highest expression levels of these mRNAs were also observed on day 5 (Figure 3). The results indicated that when the duration of serum starvation is relatively short, the expression of Notch signalling pathway components increases with time until reaching a plateau. Subsequently, the expression of Notch signalling pathway components decreases with prolonged serum starvation.
Effects of serum starvation on the mRNA expression of Notch signalling pathway components in MECs. The mRNA levels of the Notch signalling pathway-related genes Notch1, Notch4, Jagged1, Dll4 and Hes1 were detected using real-time PCR (n = 3). Data are presented as the means ± SD.
Effects of VEGF on MEC proliferation
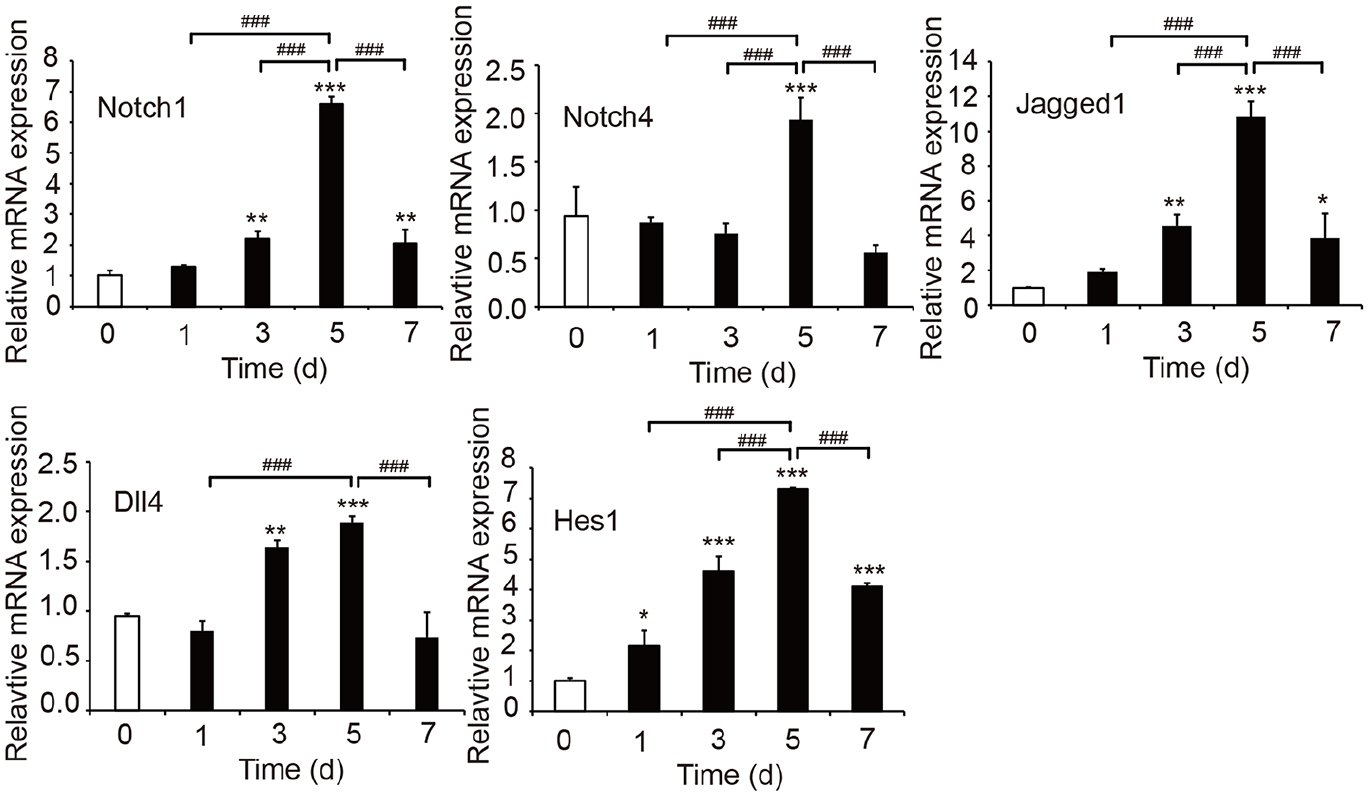
Cell viability was measured after cells were cultured with various concentrations of VEGF for 24, 48, or 72 h to investigate the effects of VEGF on MECs. When the VEGF concentration was <20 ng/mL, the viability of MECs increased with increasing VEGF concentrations. The highest viability of MECs was observed in the presence of 20 ng/mL VEGF, and then, cell viability gradually decreased with increasing VEGF concentrations (p < 0.05) (Figure 4). Thus, the effects of VEGF on the viability of MECs showed an inverted u-shaped trend. We selected VEGF concentrations of 5, 10, 20, 50, 100 and 150 ng/mL for subsequent experiments to optimize the experimental conditions.
Effects of VEGF on MEC viability (n = 3). Data are presented as the means ± SD.
Effects of VEGF stimulation on the Notch signalling pathway in MECs
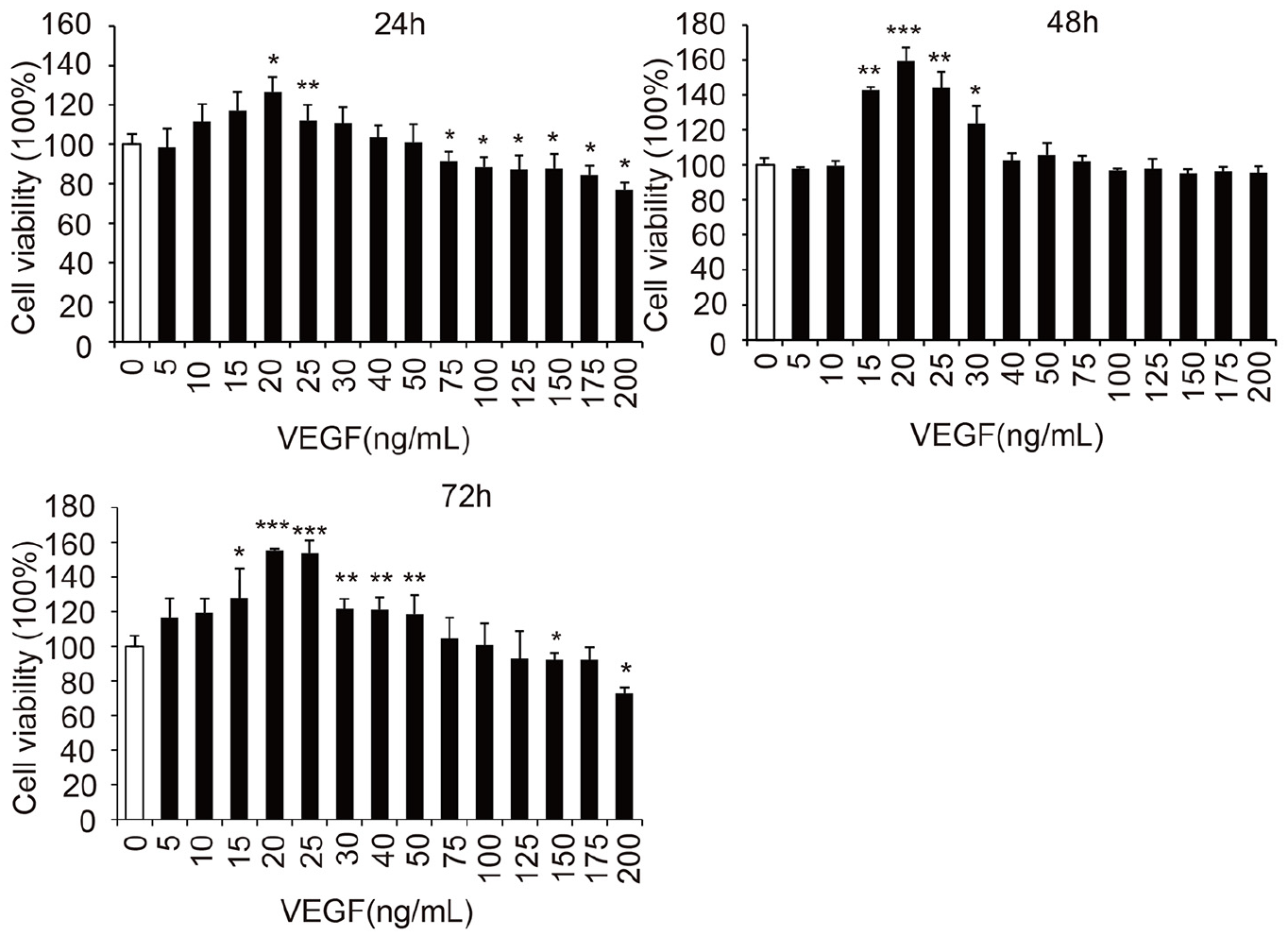
Expression levels of Notch pathway-related proteins and mRNA were measured using western blotting and real-time PCR to explore the relationship between the Notch signalling pathway and VEGF in BAVM. As presented in Figure 5, the Notch1, Jagged1, Dll4 and Hes1 protein expression levels increased with increasing VEGF concentrations between 0 and 20 ng/mL. The highest protein expression levels were observed in cells treated with 20 ng/mL VEGF. However, when the VEGF concentration exceeded 20 ng/mL, the protein expression levels began to decrease with increasing VEGF concentrations. As illustrated in Figure 6, the Notch1, Notch4, Jagged1, Dll4 and Hes1 mRNA expression levels showed the same trends as the protein expression levels. These results suggest that the expression of Notch signalling pathway components was gradually upregulated when the VEGF concentration was relatively low. After the Notch1, Notch4, Jagged1, Dll4 and Hes1 protein and mRNA expression levels (Notch signalling pathway) peaked, the expression of components of the Notch signalling pathway was downregulated by VEGF at concentrations >20 ng/mL.

Effects of VEGF on the protein expression of Notch signalling pathway components in MECs: (a) Western blot analysis of Notch1, Hes1, Jagged1 and Dll4 protein levels, with β-actin serving as the internal control (n = 3) and (b) quantification of Notch1, Hes1, Jagged1 and Dll4 levels normalized to β-actin levels (n = 3). Data are presented as the means ± SD.

Effects of VEGF on the mRNA expression of Notch signalling pathway components in MECs. The mRNA expression of Notch signalling pathway-related genes Notch1, Notch4, Jagged1, Dll4 and Hes1 was detected with real-time PCR (n = 3). Data are presented as the means ± SD.
Effects of serum starvation on the Notch signalling pathway in mES/EB-derived endothelial cells
Experiments were performed to investigate the relationship between serum starvation and cerebral AVM during embryonic development, and Figure 7 displays protein levels in mES/EB-derived endothelial cells cultured with serum-free medium for 5, 7, 10 and 14 days. Notch1, Jagged1, Dll4 and Hes1 expression increased with prolonged serum starvation over relatively short times, and the highest protein expression was observed after serum starvation for 10 days. The protein expression levels began to decrease when serum starvation lasted for more than 10 days (Figure 7). The results of real-time PCR further revealed that the changes in mRNA expression were consistent with the trends in protein expression (Figure 8). Based on these results, serum starvation mediates the expression of Notch signalling pathway components in mES/EB-derived endothelial cells and that the induced signal increase decreases upon long-term serum starvation. This finding is consistent with the effect of serum starvation on the expression of Notch signalling pathway components in MECs.

Effects of serum starvation on protein expression in mES/EB-derived endothelial cells: (a) western blots showing levels of the Notch1, Hes1, Jagged1 and Dll4 proteins, with β-actin serving as the internal control (n = 3) and (b) quantification of Notch1, Hes1, Jagged1 and Dll4 levels normalized to β-actin levels (n = 3). Data are presented as the means ± SD.
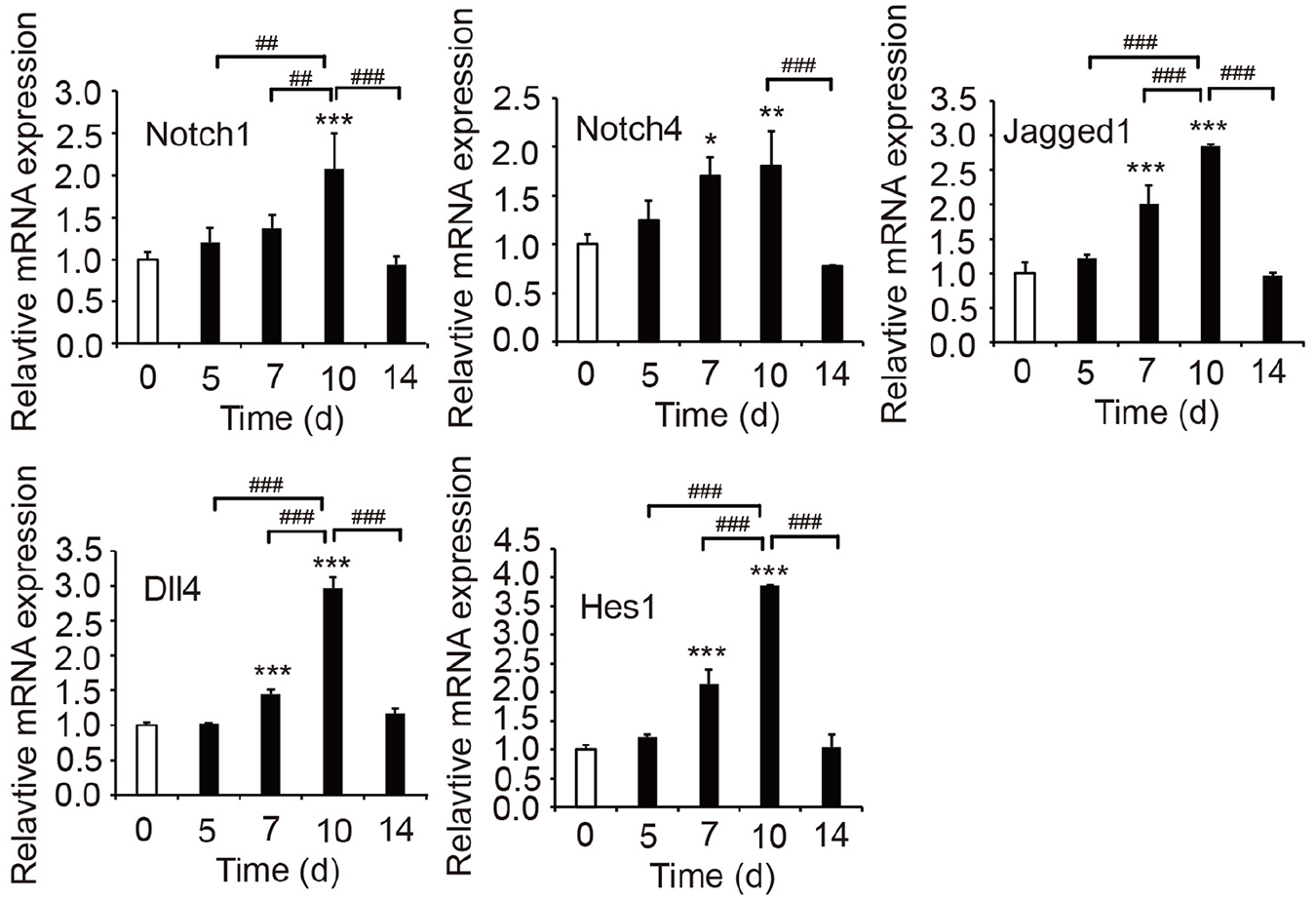
Effects of serum starvation on mRNA expression in mES/EB-derived endothelial cells. The mRNA expression of the Notch signalling pathway-related genes Notch1, Notch4, Jagged1, Dll4 and Hes1 was detected with real-time PCR (n = 3). Data are presented as the means ± SD.
Effects of VEGF stimulation on the Notch signalling pathway in mES/EB-derived endothelial cells
Since AVM is congenital, we next stimulated mES/EB-derived endothelial cells with different concentrations of VEGF and extracted protein and mRNA from the treated cells after 30 days of culture to further investigate the pathogenesis of AVMs. We detected the expression of Notch pathway-related proteins with western blotting. Increases in VEGF concentration from 0 ng/mL to 10 ng/mL lead to increased Notch1, Jagged1, Dll4 and Hes1 expression. The highest protein expression was observed after cells were exposed to 10 ng/mL VEGF, and the levels began to decrease with increasing VEGF concentration when the concentration exceeded 10 ng/mL (Figure 9). Real-time PCR further confirmed that the trends at the mRNA level were consistent with those at the protein level (Figure 10). Thus, relatively low VEGF concentrations induce the expression of Notch signalling pathway components in mES/EB-derived endothelial cells, but when the VEGF concentration is too high, negative-feedback inhibition of the Notch signalling pathway occurs. This effect shares the same trend for the effects of VEGF on the Notch signalling pathway in MECs.

Effects of VEGF on protein expression in mES/EB-derived endothelial cells: (a) western blots showing Notch1, Hes1, Jagged1 and Dll4 protein levels, with β-actin serving as the internal control (n = 3) and (b) quantification of Notch1, Hes1, Jagged1 and Dll4 levels normalized to β-actin levels (n = 3). Data are presented as the means ± SD.

Effect of VEGF on mRNA expression in mES/EB-derived endothelial cells. The mRNA levels of the Notch signalling pathway-related genes Notch1, Notch4, Jagged1, Dll4 and Hes1 were detected using real-time PCR (n = 3). Data are presented as the means ± SD.
Discussion
BAVMs are the most common type of vascular malformation in the central nervous system. Previously, investigators estimated that the annual detection rate of BAVMs is 1.12–1.42 cases per 100,000 people. 5 BAVMs result in haemorrhagic stroke in young people, most commonly in people between 20 and 40 years of age; account for half of the haemorrhagic stroke cases in children; account for 1%–2% of all stroke cases; and can cause epilepsy in adolescents.2,3 BAVMs pose a serious threat to human health. According to statistics, the average annual bleeding rate is 0.5%, the rebleeding rate is 29%, 5%–25% of patients die within 1 year after haemorrhage, and 25%–40% have permanent neurological deficits. In recent years, with the rapid development of microsurgical neurosurgery, neurointerventional surgery and neuroradiological surgery, BAVMs are currently being treated using various methods, including medical management, endovascular embolization, neurosurgery and stereotactic radiotherapy. 21 Even among patients with BAVM and confirmed complete embolization who underwent surgical resection by cerebral angiography, reports of recanalization and recurrence in later stages persist. Regardless of the use of resection, embolization or radiotherapy, all affected patients have certain complications (incidence rate of 12%–41%), of which normal perfusion pressure breakthrough syndrome characterized by blood-brain barrier damage and acute cerebral oedema is the most serious. To date, no specific medications are available to treat BAVMs. 22 The current primary treatment plan for BAVM is surgical resection, but the efficacy is controversial. Current morbidity data suggest that for unruptured AVMs, invasive management poses a higher risk than non-invasive management. 23 Therefore, the lack of targeted therapy and the high mortality rate indicate that a better understanding of the mechanisms underlying AVM is needed.
The Notch pathway, which is likely the most widely used signalling pathway in intercellular communication, is involved in cell fate determination and tissue patterning in metazoans and is a key mediator of normal arteriovenous differentiation. Although the activation of Notch signalling may not be the primary cause of AVM development, its involvement at any stage of disease progression makes it a possible target for therapeutic development. Other than surgical excision, endovascular embolization and radiotherapy, no treatment has been developed to prevent bleeding from BAVMs, and each of these methods carries the risk of disability or death. Approximately 20% of patients with BAVM cannot be offered these methods due to excessive risk. 11
According to previous studies, the Notch signalling pathway is upregulated in BAVMs. One study identified Notch1 upregulation in human samples. The results of immunohistochemistry and western blotting of surgically resected BAVM samples showed that Notch1, the Notch1 ligands Jagged1 and Dll4 and the downstream Notch1 target Hes1 were activated in BAVM. 11 Another study found that Notch1 and Notch4 expression levels were increased in patients with BAVM. 1 Murphy et al. 10 observed that upregulation of Notch signalling mediates AVMs not only in the brain but also in other organs in the mouse, such as in the liver, skin, uterus and lungs, in studies of mice and embryos. The authors also suggested that endothelial overexpression of a constitutively active Notch4 intracellular domain (Notch4) results in BAVM in young mice and that activation of the Notch signalling pathway is potentially a crucial molecular candidate in BAVM pathogenesis. 10 Another study on a mouse model showed that Notch4 normalization reduces blood vessel size in AVMs. 3 These findings suggest that a treatment targeting the Notch signalling pathway may be a promising BAVM therapy and could be recommended as an alternative approach to surgery, embolization or radiotherapy. 1
In the present study, serum deprivation significantly inhibited MEC proliferation. The protein and mRNA expression levels of Notch1, Notch ligands (Jagged1 and Dll4) and the downstream Notch target Hes1 in MECs were all increased after serum deprivation, suggesting that the Notch signalling pathway is upregulated by serum deprivation. This observation is consistent with the previously reported decisive role of serum deprivation in gene expression in adipose tissue-derived stromal cells (ASCs). 24 Our data also showed the upregulation of the Notch signalling pathway in MECs during the early stage of serum starvation (<5 days). The expression of Notch signalling pathway components peaked at day 5 and then decreased thereafter.
Clinical evidence has provided two theories about the origin of BAVM. First, BAVMs are believed to be congenital and caused by the failed regression of embryonic AV connections that persist into postnatal life. BAVMs are also suggested to result from improper venule remodelling. 25 Angiogenesis is a complex multistep process that is regulated by growth factors, particularly VEGF, in endothelial cells and is very important for life. VEGF deficiency results in embryonic death and impaired tissue maintenance and regeneration. 26 As shown in our previous study, VEGF expression is upregulated in surgically excised human BAVM specimens. 27 Notch signalling is intertwined with VEGF in the process of angiogenesis.28–30 As illustrated in Figure 11, the Notch ligand Dll4 is upregulated in response to VEGF/VEGFR2 (VEGF receptor 2) signalling in tip cells during angiogenesis. An increase in Dll4 expression in tip cells activates Notch in the neighbouring stalk ECs.

Schematic diagram of Notch signalling. In sprouting angiogenesis, increased expression of Dll4 in tip cells is achieved by both endothelial and non-endothelial derived VEGF to activate VEGFR2. Tip cells express high levels of Dll4 that activate Notch in the stalk cells to promote Notch signalling.
In the current study, an effect of the VEGF concentration on MECs was observed. When the concentration was less than 20 ng/mL, MEC viability was increased with increasing VEGF concentrations, but when the concentration was >100 ng/mL, cell viability decreased with increasing VEGF concentrations. The expression levels of Notch signalling pathway mRNAs and proteins showed the same trend. When the VEGF concentration was >20 ng/mL, expression levels of the Notch1, Jagged1, Dll4 and Hes1 proteins and mRNAs were all downregulated with increasing VEGF concentrations compared with those in cells treated with 20 ng/mL VEGF.
An insufficient nutrient supply prompts the formation of new vessels from the walls of existing vessels in a process termed angiogenic sprouting. 31 BAVMs disrupt the normal vessel hierarchy and are developed by replacing capillary beds, which separate arteries from veins, with enlarged and tangled vessels. Preservation of embryonic cerebrovascular system characteristics in BAVMs has led to the speculation that BAVMs form during early brain development. 10 In the present study, mES/EB-derived endothelial cells were used to evaluate the effects of VEGF and serum starvation on the Notch signalling pathway and to further understand the effects of VEGF and serum starvation on the development of cerebral AVMs.
VEGF upregulated the Notch signalling pathway in mES/EB-derived endothelial cells. At a VEGF concentration less than 10 ng/mL, the Notch signalling pathway was upregulated in mES/EB-derived endothelial cells. However, compared with 10 ng/mL VEGF, concentrations >10 ng/mL suppressed the Notch signalling pathway. A similar trend was observed in MECs: compared with 20 ng/mL VEGF, concentrations >20 ng/mL suppressed the expression of Notch signalling pathway components. Based on these data, the induction of the Notch signalling pathway decreases the sensitivity of cells to a VEGF gradient, implying a negative-feedback loop between VEGF-Notch signalling, as previously reported.28,32
Serum deprivation also significantly stimulated the Notch signalling pathway in mES/EB-derived endothelial cells. The highest expression levels of Notch signalling pathway components were observed on day 10 and then decreased thereafter. The trends in mES/EB-derived endothelial cells were similar to those in MECs: in the early stage of serum starvation (<5 days), the Notch signalling pathway was upregulated in MECs, and the expression levels of Notch signalling pathway components began to decrease with prolonged serum starvation. VEGF transcription was reported to be significantly upregulated with serum deprivation, and VEGF expression is stimulated by serum starvation.24,33 Therefore, in the present study, with an extended serum deprivation time, the induced expression of Notch signalling pathway components decreased the sensitivity of cells to serum deprivation and the VEGF gradient, and thus serum deprivation significantly reduced cell proliferation. All these events may lead to a decrease in protein and mRNA expression levels under serum deprivation conditions.
Nevertheless, previous studies on BAVM suggest that multiple pathways rather than a single pathway are involved in BAVM pathogenesis.34,35 More comprehensive studies on gene and protein expression and further in vivo investigations are required to completely elucidate the underlying mechanisms.
Conclusions
Serum starvation and certain concentrations of VEGF upregulate components of the Notch signalling pathway in both MECs and mES/EB-derived endothelial cells, suggesting that serum starvation and VEGF stimulation may potentially affect cerebral AVM activity. The data in this study provide preliminary but useful knowledge to guide further investigations and the clinical treatment of cerebral AVM.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant number 81471213) and the Fundamental Research Funds for the Central Universities (grant numbers N152004001 and N172004007).
