Abstract
Introduction
Adenoidectomy, tonsillectomy, and adenotonsillectomy (AT), the most commonly performed surgical procedures in pediatric ear, nose, and throat (ENT), are usually the first significant challenge to their hemostatic system. Despite the best efforts to avoid it, bleeding remains the most concerning complication of AT. Early post-adenotonsillectomy (PAT) bleeding, although rare (incidence of 0.99–1.31%),1–4 requires immediate treatment. Most PAT hemorrhages (65%) in children occur within the first 24 h after surgery (early hemorrhages),4,5 their importance ranging from minor (self-limiting) to severe (life-threatening). Every surgeon would like to have predictive markers for this complication of one of the most common procedures performed in pediatric ENT.
In the last 20 years, many authors have studied the relationship between PAT hemorrhage and different variables: age of the patient, sex,1,3,4 indication of surgery (hypertrophy or recurrent infection of adenoids/tonsils 6 ), air temperature and pressure, surgical method used (cold dissection with cold hemostasis, cold dissection with hot hemostasis, hot dissection and hot hemostasis), 5 tonsil size or adenoid mass, 4 and different blood markers (fibrinogen, CRP).7,8
Lately, the lymphocyte count (absolute, relative, neutrophile to lymphocyte ratio (NLR), and platelet to lymphocyte ratio (PLR)) has been studied in relation to different types of bleeding (GI tract bleeding, intracranial, cardiothoracic hemorrhage, post-tonsillectomy hemorrhage)7,8; fibrinogen levels 9 and C-reactive protein levels10,11 have been associated with the risk of post-tonsillar or upper or lower GI tract bleeding.
The purpose of our study is to establish whether blood markers measured routinely preoperatively in children with adenoidectomy, AT, or tonsillectomy (AT procedures) (i.e. blood cell count, International Normalized Ratio (INR), prothrombin time (PT), activated partial thromboplastin time (aPTT), fibrinogen, CRP) play a predictive role for early postoperative bleeding (i.e. PAT bleeding, PAT bleeding). Focus has been placed on the presence of relative lymphocytosis (a value of the lymphocyte percentage above 55%) in the blood cell count of the patients and its possible connection to postoperative hemorrhage.
Patients and method
We conducted an observational retrospective study on 801 patients who underwent AT over a 6-month period; following the recommendations of 6 months; following the STROBE statement, two groups of patients were identified: the bleeding group (group B) (19 patients in which early postoperative bleeding occurred) and the no bleeding group (group A) (782 patients). The obvious difference between the number of patients in the two groups is addressed in the “Discussion” section. Patient records were anonymized and retrospectively analyzed.
Our Hospital's Ethics Committee has approved this clinical study (21832/04.06.2021). Parental written consent is routinely obtained for preoperative blood tests, surgery, and general anesthesia.
Inclusion criteria: All 801 consecutive patients undergoing three types of surgical procedures—adenoidectomy (A), AT, or tonsillectomy alone (T)—in our Department (ENT Department of M.Curie Children's Emergency Hospital) for a period of 6 months (June through November 2021) were included in the study. Indications for these three types of procedures were obstructive sleep apnea (simple adenotonsillar hypertrophy) or a history of recurrent local or regional infections (tonsilitis, adenoiditis, acute otitis media or effusion otitis media) (quinsy tonsillectomy was excluded from the study); indications for surgery were not analyzed separately in connection with postoperative bleeding.
The 801 patients (with similar ethnicity and ages between 1 and 18 years) were included in two groups. The bleeding group (B)—consisted of 19 patients in which immediate PAT hemorrhage occurred (i.e. hemorrhage in the first 24 postoperative hours). The control group (A) consisted of the rest of 782 patients, in which no bleeding occurred. During the 6 months study period, we encountered 19 immediate postoperative bleedings (within the first 24 h postoperatively) and 2 delayed ones (after up to 7 to 10 days after AT). Only early hemorrhages were taken into account in our study.
All procedures included in the study have been performed by six ENT consultants (with similar surgical experience) using the same surgical techniques under general anesthesia with orotracheal intubation (sevoflurane induction and maintenance, intravenous propofol used as needed). Acetaminophen and metamizole solutions were used for intra- and postoperative analgesia, by IV infusion (no opioids, no ibuprofen). Classical curettage adenoidectomy (with Beckmann's curette) was performed, aided by endoscopic control. Hyperextension of the child's neck and Nelaton's probe inserted through the child's nose were used to retract the soft palate cranially for enhanced direct visualization. Tonsillectomy was performed using classical cold knife (steel) dissection and a steel sectioning loop. Cold hemostasis was a routine in AT procedures, and bipolar electrocoagulation was minimally used only when needed.
Postoperative bleedings, either from the adenoid area (rhinopharynx) or the tonsillar fossa were treated conservatively (aspiration of blood clots, local compression with cotton swabs, local vasoconstriction agents such as ephedrine, Tranexamic acid intravenously), or by hemostasis under general anesthesia (electrocoagulation directed to the source of bleeding). All patients who suffered from postprocedure bleeding had good results, without death. All surgical procedures that take place in the operating theater were mentioned in the surgical protocols saved in the hospital database.
According to hospital protocols, blood tests are routinely measured before surgery in all patients—complete blood count, INR, aPTT, PT, CRP, and fibrinogen. These variables were registered in our database for all enrolled patients, using separate files for the two groups. All blood tests were performed by the same laboratory; patients with blood tests performed in other laboratories were excluded from the study.
The normal range values for the variables measured in the children in our laboratory were as follows: 5.5–15.5 × 103 /ml for white blood cells (WBCs), 2–8 × 103 /ml for lymphocyte count, 25–55% for lymphocyte percentage, 0–5 mg/l for CRP, 140–360 mg/dl for fibrinogen, 0.84–1.20 for INR and 24–37 s for aPTT.
Comparison between the two groups and statistical analysis of the mean values for the registered variables (absolute and relative lymphocyte count, CRP, INR, aPTT, fibrinogen) was made to identify the factors associated with postoperative bleeding. A two-tailed unpaired t-test with Welch's correction and two-sided Fisher's exact test were used to compare mean values of variables between groups and to estimate predictive values of variables. Calculation of area under ROC curve was used to appreciate whether the chosen 55% cut-off point for the relative lymphocytosis percentage is accurate. One and two—sided p-values and odds ratios were calculated; p˂0.05 was considered statistically significant.
Results
There was a difference in the number of patients in the two groups (782 vs. 19 patients). The difference between the mean age of each group (0.39 ± 0.79 years) was not statistically significant (p = 0.615)—the mean age in the non-bleeding group of patients (group A) was 5.34 years (1–18 years) and 4.95 years (2–14 years) in the bleeding group (group B) (Figure 1).

Age distribution of patients in study groups.
We found no difference in the frequency of the male/female sex distribution between the two groups (p = 0.641); thus the two groups are comparable. Group A (the no-bleeding group) consisted of 782 patients, 43.86% girls (343 patients), and 56.14% boys (439 patients). Group B (the bleeding group) consisted of 19 patients, 36.84% girls (7 patients) and 63.16% boys (12 patients).
The types of interventions performed were the following: for group A, 79.15% adenoidectomies (619 surgeries), 16.75% adenotonsillectomies (131 surgeries), and 4.09% tonsillectomies (32 surgeries); for group B, 63.16% adenoidectomies (12 surgeries), 31.58% adenotonsillectomies (six surgeries), and 5.26% tonsillectomies (one surgery).
AT procedures were more frequent in the bleeding group compared to other types of intervention (adenoidectomy, tonsillectomy), although the difference was not statistically significant (p = 0.068).
The mean value of leukocytes was similar between the two groups (8.62 × 103 /ml for the no-bleeding group and 8.48 × 103 /ml for the bleeding group), and the difference was statistically non-significant (p = 0.796).
When comparing the lymphocyte count between the two groups, there was no statistically significant difference (p = 0.981) (Figure 2). The mean lymphocyte count was 3.87 ×103 /ml in the no-bleeding group and 3.88 ×103 /ml in the bleeding group.

Comparison between mean values for absolute lymphocyte count in the no-bleeding vs. bleeding groups: p = 0.981 (p < 0.05) non-significant (unpaired t-test with Welch's correction, two-tailed value; Welch corrected t = 0.02368, df = 18.87).
When comparing the percentages of lymphocytes between the two groups, there was no statistically significant difference (p = 0.726) (median value of 45.49% in the control group, mean value of 46.44% in the bleeding group) (Figure 3).

Comparison between lymphocyte percentage between bleeding vs. non-bleeding patients: p = 0.726(P < 0.05), which was not significant (two-tailed unpaired t-test with Welch's correction; Welch corrected t = 0.3559, df = 18.92).
A higher incidence of relative lymphocytosis (lymphocyte percentage greater than 55%) was observed in the bleeding group (in 6 out of 19 patients, 31.58%) compared to the non-bleeding group (in 18.92% of the patients, i.e. in 148 out of 782 patients); but without statistical significance (Fisher's exact test p = 0.237, OR = 1.91) (Figure 4(a)).

(a) Presence of relative lymphocytosis in the bleeding vs. non-bleeding group: p = 0.237 (p < 0.05) non-significant (two-sided Fisher's exact test; odds ratio1.915; reciprocal of odds ratio 0.5221). (b) Relative lymphocytosis between the studied groups, with a cut-off point of 55%; ROC curve—area0.5300, std. error 0.07221; p = 0.655 (95% confidence interval) no correlation.
A percentage of lymphocytes above 55% seemed to have a weak predictive value for early postoperative bleeding in AT patients (with a sensitivity of only 31.58% but a specificity of above 80%); in other words, unless the percentage of lymphocytes was above 55% in a patient, he did not bleed with a probability of 80% in the first 24 h postoperatively.
We tried to find out whether this 55% limit was an adequate cut-off point for relative lymphocytosis in our patients. Analyzing the ROC curve for the relationship between the presence/ absence of bleeding and the percent of lymphocytosis, we found that this relationship was hazardous (area under the ROC curve AUC = 0.53) (Figure 4(b)) and no other cut-off point for the percentage value of lymphocytes could be linked to postoperative bleeding.
The mean CRP value was 1.95 mg/l in the non-bleeding group and, respectively, 1.24 mg/l in the bleeding group, with no statistical difference between the groups (p = 0.697) (Figure 5(a)). Not even the ROC analysis could reach statistical significance for the CRP values (Figure 5(b)).

(a) Comparison of CRP values in patients with postoperative bleeding vs. non-bleeding—p = 0.697 (p < 0.05)—non-significant (unpaired two-tailed test, t = 0.3900, df = 643). (b) Comparison of CRP levels in a patient with bleeding versus non-bleeding postoperative: area under the ROC0.5209, Std. Error 0.06938. p = 0.768, 95% confidence interval, no correlation.
The mean fibrinogenemia values were 280.0 mg/dl in the non-bleeding group and 257.4 mg/dl in the bleeding group. Patients with PAT bleeding showed lower fibrinogen levels compared to patients without PAT bleeding; this difference was statistically insignificant (p = 0.054) (Figure 6).

Comparison of blood fibrinogen levels in studied groups: p = 0.202(p < 0.05) non-significant (unpaired two-tailed t-test, t = 1.277, df = 621); area below the ROC 0.6011, Std. Error 0.06504, 95% confidence interval, p = 0.195, both without correlation.
There were no statistically significant differences between INR values in the group without bleeding (mean value 1.04) compared to the bleeding group (mean value 1.06) (p = 0.423).
There was no statistically significant difference between the aPTT values in the two groups: the mean value of aPTT was 31.47 in the non-bleeding group and, respectively, 31.83 in the bleeding group (p = 0.575).
Discussion
Comparing the values of different coagulation or inflammatory biomarkers measured preoperatively in the two groups of patients with and without bleeding (leucocyte count, lymphocyte count and percentage, CRP, fibrinogen, aPTT, INR), sex of the patient or type of intervention, we found no statistically significant difference (p > 0.05). No blood test variable could be associated with the PAT risk of bleeding (summary Table 1).
Blood markers in patients with and without PAT bleeding and their statistical significance (ns—non-significant).
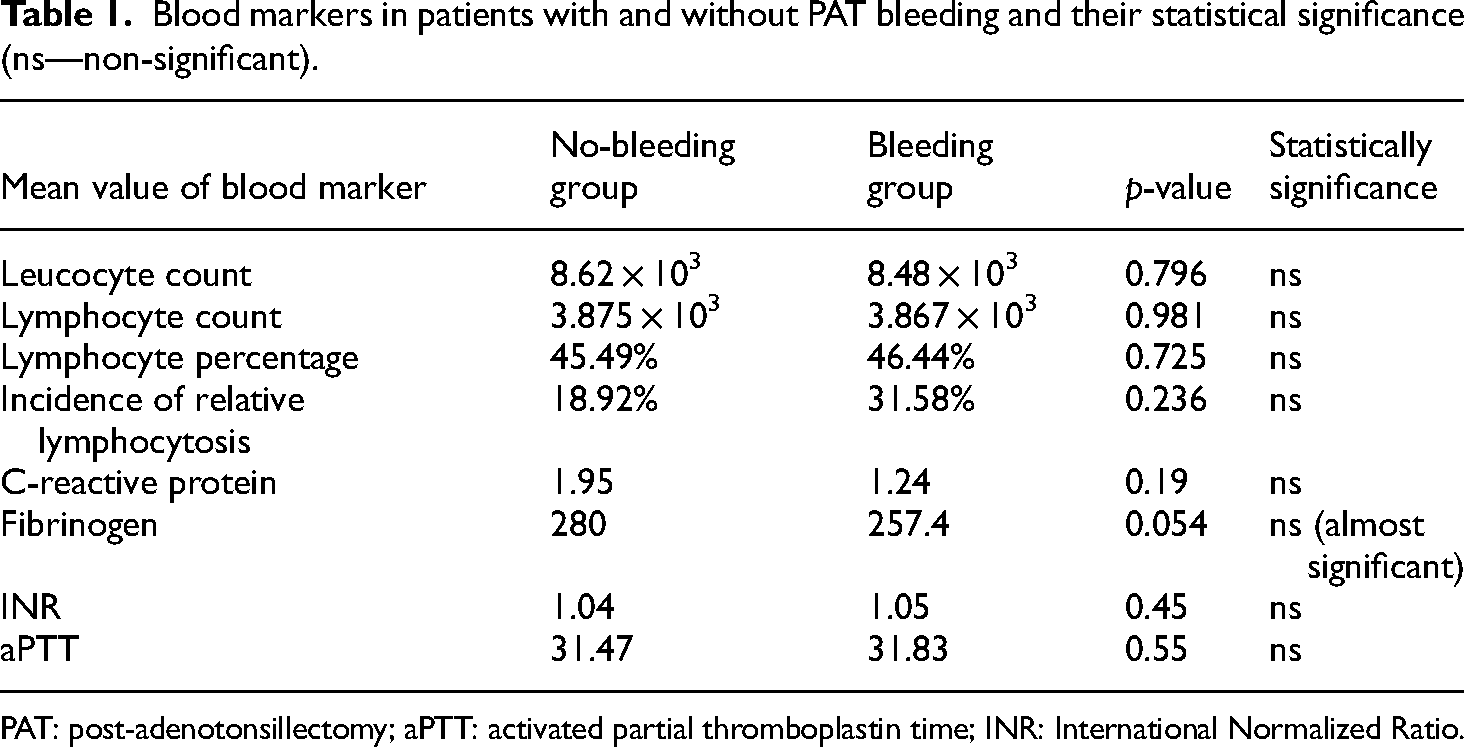
PAT: post-adenotonsillectomy; aPTT: activated partial thromboplastin time; INR: International Normalized Ratio.
First, there was a striking difference between the number of patients in the two groups (782 versus 19 patients), resulting from the fact that the incidence of postoperative bleeding is low after AT procedures (0.5–4.2%).1,2 The risk of PAT bleeding in our patients was 2.37%, including primary and secondary hemorrhages, consistent with data from the literature. Approximately one in 50 patients with AT bleeds postoperatively. Only 19 early bleedings after adenotonsilectomy (21 total bleeding during the study period, early and delayed bleeding) were recorded in our department over a period of 6 months. We excluded the delayed hemorrhages from the study, as the delayed hemorrhage risk cannot be correlated to blood markers measured on the day of the surgery. To include 800 patients in the bleeding group, we should have extended the study period to 20 years!
Procedures performed in our department were cold instrument dissection surgeries with minimal bipolar electrocautery hemostasis when necessary (to avoid negative side effects of electrocautery use: prolonged postoperative pain and symptoms of velopharyngeal insufficiency, malodeur). 12 This accounts for the fact that most postoperative hemorrhages were primary (89.48%), similar to data from the literature.4,5
In our study, the CRP values were compared for the group without bleeding versus the bleeding group. There was no statistical difference between the groups (p = 0.697). C-reactive protein is an acute phase response protein, and its serum level is elevated during inflammation.10,11,13 Few studies link acute inflammation to bleeding, and none of them correlates CRP value or relative lymphocytosis with PAT bleeding. In one study, CRP levels were higher in patients with lower GI tract bleeding than in patients without bleeding.10,11 In the same study, relative lymphocytosis was associated with postoperative bleeding after colonoscopy with a probability ratio of 6.71 (p = 0.014). 10
Although the timing of adenoidectomy and tonsillectomy is usually as far as possible from acute respiratory tract infection, the adenoid mass and the tonsils are considered sites of infection. Leucocyte count, CRP, and fibrinogen values measured before surgery could parallel the degree of local inflammation. However, we were unable to find that these inflammatory markers are significantly increased in patients with PAT bleeding. We can infer that PAT bleeding is not correlated with inflammatory markers!
Unlike other reports in the literature,9,11 in our study, postoperative bleeding was not related to high fibrinogenemia. Blood fibrinogen levels measured preoperatively were compared between patients in the two groups: patients with PAT bleeding showed lower fibrinogen levels compared to patients without PAT bleeding, and this difference was almost statistically significant (p = 0.054). Some authors associate low fibrinogen levels in the blood with the risk of postoperative bleeding in cardio-surgery and neurosurgery patients.14,15 Other authors consider elevated preoperative levels of plasma fibrinogen to be a significantly worse prognostic factor for post-tonsillectomy hemorrhage. 9
Interestingly, we found that a value of lymphocyte percentage value above 55% had a weak predictive value for postoperative bleeding in AT patients (with a sensitivity of only 31.58%, but an acceptable specificity of above 80%). In other words, 80% of AT patients with a lymphocyte percentage that does not exceed 55% will not bleed postoperatively. Children with relative lymphocytosis may require stricter supervision (hospital stay) in the first 24 h after AT, especially if they live far from the hospital.
Relative lymphocytosis (defined as an increase in lymphocytes by more than 40% in the presence of a normal absolute WBC count in adults) can be a normal finding in children,8,10,11 especially under the age of 2, but can also be a feature of infection16–19 or a sign of inflammation: acute viral or bacterial infections, connective tissue disorders (Marfan's syndrome, LES, rheumatoid arthritis), thyrotoxicosis, splenomegaly with splenic sequestration of granulocytes. Signs and symptoms may range from mild/none to severe: fatigue, unexplained weight loss, fever, night sweats, enlarged lymph nodes, and easy bleeding or bruising due to a poorly functioning spleen.
The lymphocyte count is age-dependent. Its WBC count is raised in infants and decreases slightly with age. It is quite difficult to define relative lymphocytosis in children due to age variations.18,19 In our study, we define relative lymphocytosis as a percentage of lymphocytes above 55% (this was the upper limit given by our laboratory and also the upper limit for the percentage of lymphocytes adjusted to the mean age in both studied groups, approximately 5 years).18,19
Normal values of coagulation markers (INR, aPTT, thrombocytes) are usually reported in patients with PAT hemorrhage.14,20,21 Our results of this study were consistent with those of the previous authors.
Acetaminophen and Metamizole solutions were used for intra- and postoperative analgesia in our study patients, by IV infusion (no opioids, no ibuprofen). This was in order to eliminate bias due to the influence of ibuprofen on postoperative hemorrhage in AT. 22
AT procedures were encountered more frequently in the bleeding group in our study compared to other types of intervention (adenoidectomy, tonsillectomy) (a value of p less than 0.05 could have meant that AT increased the risk of postoperative bleeding 2.5 times compared to adenoidectomy), although the difference was not statistically significant (p = 0.068). The type of intervention could also not be related to the risk of postoperative bleeding.
The present study might have some limitations: indications for surgery were not analyzed separately in connection with postoperative bleeding, the lack of calculation of the sample size, and the lack of analysis of narrower age groups of patients 23 —as it is possible that the postoperative hemorrhage rate is different for different age groups, Despite our large sample size (801 patients), the fact that the study is based on a single-center surgical experience, with uniform surgical technique and anesthesiology protocols, can skew the results in unpredictable ways. The correlation between relative lymphocytosis and fibrinogenemia with the risk of bleeding after AT needs further investigation.
Conclusions
PAT bleeding remains unpredictable despite blood tests performed routinely prior to surgery.
Relative lymphocytosis (a value of the lymphocyte percentage above 55%) has a weak predictive value for postoperative bleeding in AT—80% of patients without relative lymphocytosis will not bleed postoperatively. Children with relative lymphocytosis may need longer supervision in the first 24 h after AT.
Footnotes
Acknowledgements
Publication of this paper was supported by the University of Medicine and Pharmacy Carol Davila, through the institutional program Publish not Perish.
Author contributions
V.E. involved in conceptualization, methodology, data curation, validation, formal analysis, and writing—original draft, reviewing; D.C.G. involved in methodology, formal analysis, software, resources, project supervision, and writing—review and editing. Both authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Institutional review board statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics’ Committee of M.S.Curie Hospital (decision 21832/04.06.2021).
Informed consent statement
Written informed consent was obtained from all the patients’ parents involved in the study.
