Abstract
Background:
Epilepsy is the most common childhood neurological disorder in Nigeria. Treatment of epilepsy is long-term and sometimes lifelong with anti-seizure medications. There are conflicting reports on the effect of anti-seizure medications on serum folate. There is therefore a need to determine the effect of a commonly used anti-seizure medication's on serum folate levels of children. This would provide an evidence-based consideration for folic acid supplementation in children on anti-seizure medication as has been suggested by some studies.
Study objectives:
To determine whether serum folate levels were lower in children taking long-term carbamazepine or sodium valproate, compared to a control group.
Methods:
Serum folic acid levels were measured from well-nourished children between the ages of 1–17 years on carbamazepine and sodium valproate monotherapy and their age/sex-matched controls, using spectrophotometry.
Results:
The mean serum folate levels of patients on carbamazepine (43) and sodium valproate (22) were 0.032 mg/l ± 0.009 and 0.028 mg/l ± 0.008, respectively. The mean folate levels of the controls were 0.046 mg/l ± 0.03 (p = 0 001). No statistically significant difference was observed between the serum folate levels of children on the two anti-seizure medications, that is, carbamazepine and valproate.
Conclusion:
The children on treatment with carbamazepine and sodium valproate for more than 6 months had statistically significantly lower serum levels of folic acid compared to the standard reference range and controls. The serum folate levels of children on carbamazepine were not statistically different from those on sodium valproate.
Background of the study
Folic acid (Vitamin B9) is a water-soluble vitamin that is crucial for the production of DNA, RNA, and normal cell growth. In association with Vitamin B12, folate is required for the synthesis of red blood cells. Folate deficiency is associated with megaloblastic anemia. Emerging literature has implicated folic acid deficiency as a risk factor for childhood leukemia. 1
In the methionine cycle, folic acid deficiency results in the accumulation of homocysteine, a precursor of methionine. Hyperhomocysteinaemia is associated with cerebrovascular and neurodegenerative diseases.2–5
Epilepsy has a prevalence of 45.3–75.4% among neurological disorders of childhood in Nigeria.6–9 Neurological disorders constitute about 20% of all pediatric consultations in Nigeria.10,11 It is often a chronic disorder that affects the quality of life of not only the patients but also of their caregivers and families.12–14
Treatment of epilepsy is long-term and sometimes lifelong with anti-seizure medications (ASMs).
Antiepileptic medications have been reported to reduce serum folate levels15–18 resulting in recommendations for folic acid supplementation for children on ASMs. Some researchers have even implicated this reduction in serum folate in the etiology of drug-resistant epilepsy. 19 Some reports however have not documented a similar reduction in serum folate levels while others even suggested that the in folate reduction is necessary for seizure control.15,20,21 Any reduction in serum folate will put a significant pediatric population at risk of any possible effect of folate deficiency because of the long-term duration of the management of epilepsy.
Consequent to the conflicting findings from previous studies, there is, therefore, a need to establish the effect of these ASMs on serum folate status considering the high prevalence of childhood epilepsy in our environment in a population reported to have a high prevalence of micronutrient deficiency. 22 It remains to be seen whether determining routine folate levels and rectifying any deficiency might contribute to the prevention of other adverse effects. For example, might this decrease the possibility of a cerebrovascular accident resulting from the accumulation of homocysteine with the subsequent endothelial injury. 23 and also determine if routine folic acid supplementation may be necessary for the management of children with epilepsy.
Methods
Study design: This was a cross-sectional analytical study conducted in the Paediatric neurology clinic of the University of Nigeria Teaching Hospital Ituku-Ozalla. Ethical approval was granted from the Research and Ethics Review Board of the College of the Medicine University of Nigeria Ituku-Ozalla and written informed consent was obtained from parents of study participants. Similarly, assent was obtained from study participants 7 years and older. There are more than 2000 children registered for epilepsy treatment in the pediatric neurology clinic of University Nigeria Teaching Hospital.
Study participants: These were children 1–17 years of age who have attended the neurology (epilepsy) clinic and are on monotherapy with carbamazepine (CBZ) or sodium valproate for at least 6 months. Age and sex-matched controls were enrolled from the children's outpatient clinic of the University of Nigeria Teaching Hospital Ituku-Ozalla.
Inclusion criteria for the subjects were; children between the ages of 1–17 years, children on ASM monotherapy with CBZ or sodium valproate for periods above 6 months while the controls were well-nourished children in the same age range, not on treatment for any chronic illness including epilepsy.
Exclusion criteria comprise the presence of chronic illnesses other than epilepsy, children on medications for other chronic illnesses in addition to ASM, and the presence of malnutrition.
A total of 65 children on ASMs and 65 age and sex-matched controls were enrolled consecutively into the study. Eligible participants who gave consent were recruited consecutively till the total sample size was reached, then the control participants who met the inclusion and matching criteria were also consecutively recruited enrolled every clinic day till the study size was complete.
Blood samples from study participants were collected once a week during the pediatric neurology clinic and analyzed immediately. The serum was subsequently analyzed for folic acid levels using spectrophotometry. Data collected were analyzed using SPSS version 22 IBM Armonk NY USA.
Means and standard deviations were used to summarize the level of serum folate. The comparison of mean serum folate between study and control was done using the student t-test. The relationship between serum folate level and particular ASM (sodium valproate, CBZ, and control) was done using ANOVA. P-value < 0.05 level of significance was regarded as significant.
Results
Of the children on ASMs, 22 children were on sodium valproate while 43 were on CBZ. The gender distribution was 44 males and 21 females giving a male: female ratio of approximately 2:1.
The mean ages of the children on CBZ and valproate were 10.22 ± 4.99 and 7.13 ± 4.11 years respectively (p = 0.01). The mean age for the study participants (N = 130) was 9.41 ± 4.94 years. The control was age-matched with children on medication and has a mean age of 9.17 ± 4.09.
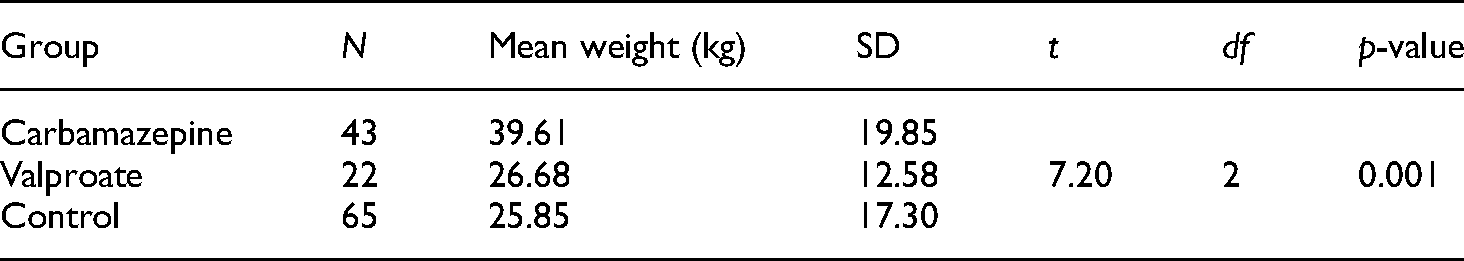
The mean body weight of children on CBZ and valproate was 39.61 ± 19.85 and 26.68 ± 12.58 kg, respectively (p = 0.001) (Table 1). All children on ASMs (CBZ and valproate) had a mean folate concentration of 0.031 ± 0.009 mg/l, while the mean bodyweight of the controls was 25.85 ± 17.30 kg.
Comparison of the body weights of the study subjects according to access to anti-seizure medications (ASMs).
The mean folate levels of patients on CBZ and sodium valproate were 0.032 ± 0.009 mg/l and 0.028 ± 0.008 mg/l, respectively (p = 0.001). The mean folate levels of age and gender-matched controls for CBZ and sodium valproate were 0.042 ± 0.005 mg/l and 0.05 ± 0.032 mg/l, respectively (Tables 2 and 3). The mean folate levels of all the controls were 0.046 ± 0.03 mg/l.
Distribution of mean serum folate levels of the test versus the control group.

Distribution of mean serum folate levels of the test versus the control group.

There was a significant statistical difference between the mean folate levels of children on CBZ compared to the controls (t = 2.88, df = 106, p = 0.005). Similarly, the difference in the mean folate levels of children on valproate therapy was statistically significantly lower when compared to the value in controls (t = 2.66, df = 85, p = 0.009).
There was no statistically significant difference observed between the serum folate levels of children on the two ASMs, that is, CBZ and valproate (t = 1.57, df = 63, p = 0.12).
There was no statistically significant relationship between the duration of intake of any of the ASMs and the serum folate levels (Tables 4 and 5).
Duration of intake of carbamazepine and serum folate levels.

Duration of intake of sodium valproate and serum folate levels.
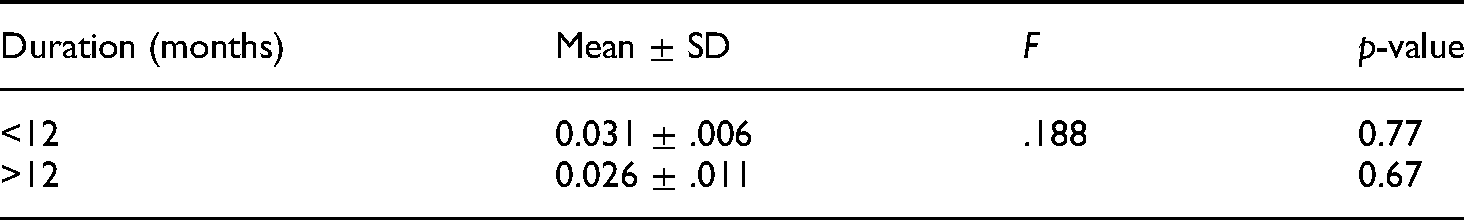
There was also no statistically significant difference in the levels of folate across the different age groups among all the participants on ASM treatment. Similarly, within the two groups of the cases, no statistical difference in the folate levels was noted also across the different age groups (p > 0.05) (Table 6). This was also the same with the controls as there was no statistically significant difference in the levels of serum folate as the different age groups (Table 7).
Correlation of mean serum folate levels with age groups of participants.

Correlation of mean serum folate levels with age groups of control participants.

It was also noted that sex did not influence the serum levels of the cases on antiepileptic medications (p > 0.05) (Table 8).
Distribution of mean serum folate levels by gender of participants.

Discussion
There was no statistically significant difference between the folate levels of children on CBZ and those on valproate. The mean levels of serum folate of the study group were lower than that of the controls and this was statistically significant, with the level of folate being lower with children on valproate compared to those on CBZ. By the definition of folate deficiency (serum folate < 0.03 mg/l), the participants on valproate were folic acid deficient while the children on CBZ had mean folate levels of 0.032 mg/l. The value 0.032 mg/l was a mean, it signifies that some children on CBZ are folate deficient also. A study by Akinsete and Boyo 24 documented a mean serum folate level of 0.082 ± 1.3 mg/l in healthy infants and pre-schoolers. This further confirms that the study population had reduced levels of folate in their serum. The higher level of folate reported by Akinsete and Boyo 24 may be because the participants were infants and young children all less than 36 months. In a similar study of 4478 children by Kreusler et al., 25 they noted that children 1 year and below had the highest folate level compared to other age groups. These children are still fed a milk-based diet and cereals that are commonly fortified with micronutrients when compared to the staples eaten by older children. This may explain the higher levels of serum folate observed in the younger children in these studies. This study did not include infants and hence cannot compare this finding among its study participants but noted that the youngest age group (1–6 years) of the control arm of this study had the highest level of folate (Table 7). This however was not statistically significant.
Children on ASM should be screened for common signs of folate deficiency which include glossitis, angular stomatitis, nausea and vomiting, cognitive impairment, and depression.
Folate metabolism is closely linked with vitamin B12 metabolism and its deficiency is associated with megaloblastic anemia, a clinical condition resulting from vitamin B12 deficiency.
Reduced serum folate in children on CBZ and valproate has been documented in previous studies15–18 with several mechanisms having been proposed. These include interference in the metabolism of the coenzyme forms of folate, reduced intestinal folate absorption, alteration in enzyme activity.4,26 None of these reports noted clinical features suggestive of folate deficiency, neither are these features listed in the literature as adverse effects of these ASMs. A possible reason might be the plateauing of the reduction effect of the ASMs on folate, thus limiting the effects of the decreased folate levels, so that they do not manifest clinically further studies are required to ascertain this.
It was also observed that there was no statistically significant relationship between duration of intake of ASM and the serum folate levels which was similar to the finding from a report by Kishi et al. 27 This finding further gives credit to the hypothesis that the effect of ASMs plateau with time and the duration of intake then becomes insignificant with reference to the level of serum folate.
Findings from this study are different from some authors5,20,28 who reported no difference in folate levels between the controls and the study group of patients on CBZ and valproate. The underlying dietary intake of the study participants may have contributed to the outcomes of these studies as they were done in developed countries where the prevalence of micronutrient deficiency is minimal.
Also, it was observed in this study that the mean folate level of the control group was lower than the standard reference range for children. This probably implies an underlying nutritional efficiency among children in the study locale. This may probably be from poor dietary intake among children in this environment. The common causes of folic acid deficiency include inadequate diet, and or poor absorption. Opportunities for health education should be engaged to teach caregivers the best sources of folic acid and best methods of preparation as folic acid is both heat and light labile.
Food fortification with vitamins and minerals is a public health measure that is prevalent in developed economies that reduce and prevent micronutrient deficiency.
The lower folate level was not particular to any of the two ASMs as there was no significant difference between the mean folate of the two groups (CBZ and valproate). This shows that the effect of the ASMs on folate may be independent of each other, have a possible summative, lessening, or even synergistic effect. Further studies on this are required to enable physicians to determine appropriate ASM combinations for patients on polytherapy, which is common practice in the management of epilepsy. Paknahad et al. 29 studied patients on both monotherapy and polytherapy and also documented that patients who were on polytherapy had lower levels of folate compared to those on both monotherapies. The reason for this is uncertain.
It was observed that children on ASMs, CBZ, and valproate in this study weighed as much as 119% and 115%, respectively, of expected for age, and more than their age-matched controls whose weights were within the normal range (86%) for age. Weight gain is a recognized side effect of treatment with sodium valproate. Weight gain in as much 43–58% of patients (children and adults inclusive) and up to14% change was observed on treatment with valproate.30–32 Different mechanisms have been proposed to explain the weight gain associated with valproic acid. These include stimulation of the pancreatic beta cells, reduced peripheral absorption of glucose resulting in insulin resistance, rise in the levels of postprandial insulin and low density lipoproteins.33–34
The reason for the increased weight of the participants on CBZ is not clear. Although most literature has also not documented weight gain as a side effect of therapy with CBZ. Easter 35 reported that weight gain was associated with some patients on CBZ.
Conclusion
The findings from this study showed that children on treatment with CBZ and sodium valproate for more than 6 months had lower serum levels of folic acid compared to the standard reference range. The serum folate levels of these children were statistically significantly lower than that of their age and sex-matched controls that were not on an antiepileptic drug. It was also noted that the serum folate levels of the control group of the study were lower than the standard reference. The low serum folate levels of children on carbamazepine were not statistically different from those on sodium valproate.
This study would recommend further studies with a view to determine if the incorporation of low-dose folic acid into the treatment of patients on long-term intake of carbamazepine and sodium valproate would be required. Intermittent monitoring of the serum folate levels of children in our environment who are on long-term anti-epileptic medications especially carbamazepine and sodium valproate would have been ideal but is limited by cost and sometimes availability. There should be monitoring of patients on ASMs for any signs of folate deficiency, and if observed, folate supplementation commenced. Also, there should be a strengthening of policies and programs on food fortification with micronutrients (folic acid inclusive) to prevent an underlying deficiency in the general population.
Limitation of the study
The study did not have the opportunity to determine the baseline levels of the participant's folate level prior to the commencement of ASMs. We could not correlate the dose of the antiepileptic drug to the mean folate levels to determine if there was a dose-effect influencing the outcome. Also, the study would have preferred to use BMI rather than weight but does not have access to the data required for it. Similarly larger population numbers may be required for further evaluation of outcomes.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
