Abstract
Neuroprosthetic strategies seek to immediately alleviate deficits and reinstate voluntary control of movement. To facilitate recovery, it is crucial to gain a comprehensive understanding of the mechanisms involved in the return of intentional movement. Nevertheless, the precise relationship between the resurgence of cortical commands and the recovery of locomotion remains somewhat elusive. In the study conducted by Duguay, Bonizzato, Delivet-Mongrain, Fortier-Lebel and Martinez, we introduced a neuroprosthesis designed to deliver precise bi-cortical stimulation in a clinically relevant contusive spinal cord injury model. We conducted experiments in both healthy and spinal cord injured cats, where we fine-tuned the timing, duration, amplitude, and site of stimulation to modulate hindlimb locomotor output. In healthy cats, we observed a wide range of motor programs. However, after spinal cord injury, the induced hindlimb movements became highly stereotyped but were effective in modulating gait and reducing bilateral foot dragging. These results suggest that the neural basis for motor recovery traded off selectivity for effectiveness. Through a series of longitudinal assessments, we found that the restoration of locomotion following spinal cord injury was closely linked to the recovery of the descending neural drive. This underscores the importance of directing rehabilitation interventions toward the cortical target. The study results are discussed in terms of their impact and limitations.
Keywords
Spinal cord injuries (SCIs) produce persistent walking impairments that have a profound impact on both independence and the overall quality of life. Most SCIs are anatomically incomplete, signifying that certain neural pathways responsible for transmitting information to and from the brain remain intact. Even in instances of severe SCIs, which are clinically categorized as ‘motor complete’, these residual connections may play a role in the return of motor function. More specifically, the direct and indirect pathways originating from the motor cortex play a pivotal role in the planning, coordination, and execution of voluntary movements. Research conducted by my team in rodent models1–3 and by other researchers in humans4–6 has revealed that the recovery of walking abilities following incomplete SCIs is heavily contingent upon the activation, reinforcement, and adaptability of these neural pathways. Additionally, the direct stimulation of these preserved connections via targeted neurostimulation of the cortical region has resulted in previously unobserved immediate alleviation of locomotor deficits and sustained enhancements in the voluntary control of movements in rodent models of unilateral hindlimb paralysis. 2 Nevertheless, it remained uncertain whether cortical stimulation could be employed for reinstating bilateral locomotion control following severe SCIs that result in incomplete paraplegia.
To address this knowledge gap, a neuroprosthetic platform was developed to monitor and modulate bilateral hindlimb movements in cats, both in healthy condition and following severe SCIs. The system employed real-time processing of electromyographic signals from hindlimb muscles and utilized intracortical microstimulation (ICMS) to produce and modulate hindlimb movements. The goal was to investigate the efficacy of this platform in controlling hindlimb kinematics and alleviating locomotor deficits in cats with SCIs. 7
Phase-coherent ICMS in healthy cats
We first investigated the effects of phase-coherent ICMS on hindlimb kinematics in healthy cats (Figure 1(A)). ICMS was delivered to either the left or right motor cortex during treadmill walking. The timing of ICMS delivery was critical for its effectiveness, with the best results obtained when ICMS was delivered during the contralateral hindlimb's swing phase (Figure 1(B)). This phase-coherent ICMS significantly enhanced contralateral hindlimb flexion, resulting in increased step height and flexion velocity.

Phase-coherent uni-cortical stimulation modulated contralateral hindlimb movements in intact and SCI cats. Schematic representation of uni-cortical neurostimulation: Right cortex stimulation modulated left hindlimb flexion through descending projections in the intact state (A) and following a thoracic spinal cord contusion (E). Changes in step height in relation to ICMS delivery timing are presented in (B) and (F), while (C) and (G) show variations based on train duration. (D) and (H) display changes in step height associated with different amplitudes of stimulation. The dashed lines in (B) and (F) indicate the percentage of the step cycle during which the measures of ICMS-evoked movements were assessed in panels C-D and G-H. p: ** < 0.01; *** < 0.001; **** < 0.0001. Modified from Duguay, Bonizzato, Delivet-Mongrain, Fortier-Lebel and Martinez,7 under the CC BY-NC-ND license (http://creativecommons.org/licenses/bync-nd/4.0/). SCI: spinal cord injury.
The study also examined the impact of ICMS duration and amplitude on hindlimb movements. A 100 ms stimulation duration was found to be optimal, leading to the most significant increases in step height (Figure 1(C)) and flexion velocity. Additionally, varying ICMS amplitude demonstrated precise control over contralateral step height (Figure 1(D)) and toe trajectories. These findings established that phase-coherent ICMS could effectively modulate hindlimb kinematics in healthy cats. In essence, this implies the feasibility of accurately regulating limb movement trajectory and intensity through cortical stimulation.
Phase-coherent ICMS in SCI cats
Next, we tested the effects of phase-coherent ICMS in cats with contusive SCIs (Figure 1(E)). Cats with thoracic spinal cord contusions initially exhibited complete bilateral hindlimb paralysis. However, two to three weeks after SCI, all cats displayed weight-supported bilateral hindlimb locomotion, albeit with dragging deficits.
Similar to our observations in healthy cats, phase-coherent ICMS delivered during contralateral hindlimb swing execution significantly enhanced contralateral hindlimb flexion, thereby reducing dragging deficits in cats with SCIs (Figure 1(F)). ICMS delivered during other phases of the locomotor cycle had minimal effects or even disrupted locomotion.
The study also confirmed that the duration and amplitude of ICMS remained critical factors in modulating hindlimb flexion after SCI. A 100 ms stimulation duration was optimal, leading to increased step height and reduced dragging (Figure 1(G)). Varying ICMS amplitude continued to provide precise control over contralateral step height and toe trajectories and was efficient in reducing dragging (Figure 1(H)), with no significant impact on the ipsilateral hindlimb. This finding is particularly promising, suggesting that cortical neuroprosthetic interventions can help restore some degree of functional limb movement even after a severe injury.
Diverse movement synergies in healthy cats vs. SCI Cats
One notable observation was the diversity of motor outputs produced by ICMS in intact cats. Different electrode sites within the hindlimb motor cortex representation led to qualitative changes in hindlimb trajectories during swing (Figure 2(A)). Six distinct motor programs were identified in intact animals, resulting in various hindlimb movement patterns (Figure 2(B)). These movements remained consistent within the same electrode but varied in the extent of trajectory modulation.

Phase-coherent uni-cortical stimulation produced a variety of motor synergies in an intact cat, while producing a single stereotyped lift movement after SCI. (A) The location of implanted arrays within layer V of the hindlimb motor cortices is indicated on the left. A parasagittal section of a model brain (i, ii) illustrates the specific location targeted by the multi-shank arrays with respect to layer V (indicated by a dotted trace) of the hindlimb motor cortex (4γHL). Movements induced by phase-coherent stimulation of each electrode site during locomotion in the intact state and after SCI are depicted on the right. (B) Four distinct movements were elicited by stimulating various electrode sites in an intact cat, with the peak modulations of each tested electrode overlaid. (C) Conversely, in the same cat after SCI, there was a lack of movement diversity. The peak modulations of each tested electrode were superimposed. Modified from Duguay, Bonizzato, Delivet-Mongrain, Fortier-Lebel and Martinez,7 under the CC BY-NC-ND license (http://creativecommons.org/licenses/bync-nd/4.0/).
However, this diversity in motor outputs was lost in cats with SCIs. Following SCI, all cats exhibited upward-directed modulation of hindlimb flexion, regardless of the stimulation site or amplitude (Figure 2(C)). The loss of movement diversity indicated a more limited range of motor responses following SCI. This loss underscores the idea that there might be restrictions on the spectrum of motor responses achievable through cortical neuroprosthetic interventions. It emphasizes the need for personalized approaches in such scenarios, which could potentially combine the effectiveness of cortical stimulation to modulate hindlimb flexion with the stimulation of spinal
8
or reticular circuits9,10 shown to recruit various spinal synergies.
Bi-cortical stimulation in healthy and SCI cats
The study further explored the potential for bi-cortical stimulation to bilaterally modulate hindlimb trajectories in both healthy and SCI cats. By alternately stimulating both left and right motor cortices, we aimed to achieve bilateral control of foot trajectories during locomotion.
Bi-cortical stimulation indeed induced alternated increases in hindlimb flexion in healthy cats. The alternated stimulation enhanced bilateral locomotor patterns, improving hindlimb flexion and reducing dragging deficits. This effect was immediate but disappeared when the stimulation ceased.
To ensure that the stimulation of one cortex did not interfere with the other, the study combined stimulation from both cortices independently. This analysis confirmed that the specific cortical control of contralateral hindlimb movements was independent of interactions with the homologous cortex.
In SCI cats, phase-coherent bi-cortical stimulation also improved the bilateral locomotor pattern, enhancing hindlimb flexion and alleviating dragging deficits. The beneficial effects of ICMS were observed immediately but disappeared upon discontinuation of the stimulation. Again, the study confirmed that the specific effects of cortical stimulation on contralateral hindlimb movements were independent of homologous cortex stimulation. The ability to achieve bilateral control is of paramount importance for coordinated and balanced locomotion. The prospect of accomplishing this through cortical neuroprosthetic interventions holds great promise for enhancing mobility and overall quality of life in individuals with spinal cord injuries.
Correlation between locomotion and stimulation recovery
The study further explored the relationship between locomotor recovery and the re-expression of cortically induced gait modulation. Cats with SCIs displayed different profiles of locomotor recovery, with varying timelines for the return of weight-supported locomotion and obstacle avoidance capacity.
Interestingly, the timeline of locomotor recovery closely correlated with the timeline of expression of cortical stimulation capacity (Figure 3(A)). The cats that recovered locomotion earlier also exhibited earlier and stronger responses to cortical stimulation. This correlation suggests that the restoration of locomotion is intimately tied to the recovery of descending motor control pathways. Understanding this relationship could offer valuable insights into devising rehabilitation strategies for individuals with SCIs. It implies that interventions aimed at enhancing neural plasticity and motor recovery might also enhance the effectiveness of neuroprosthetic devices. 2
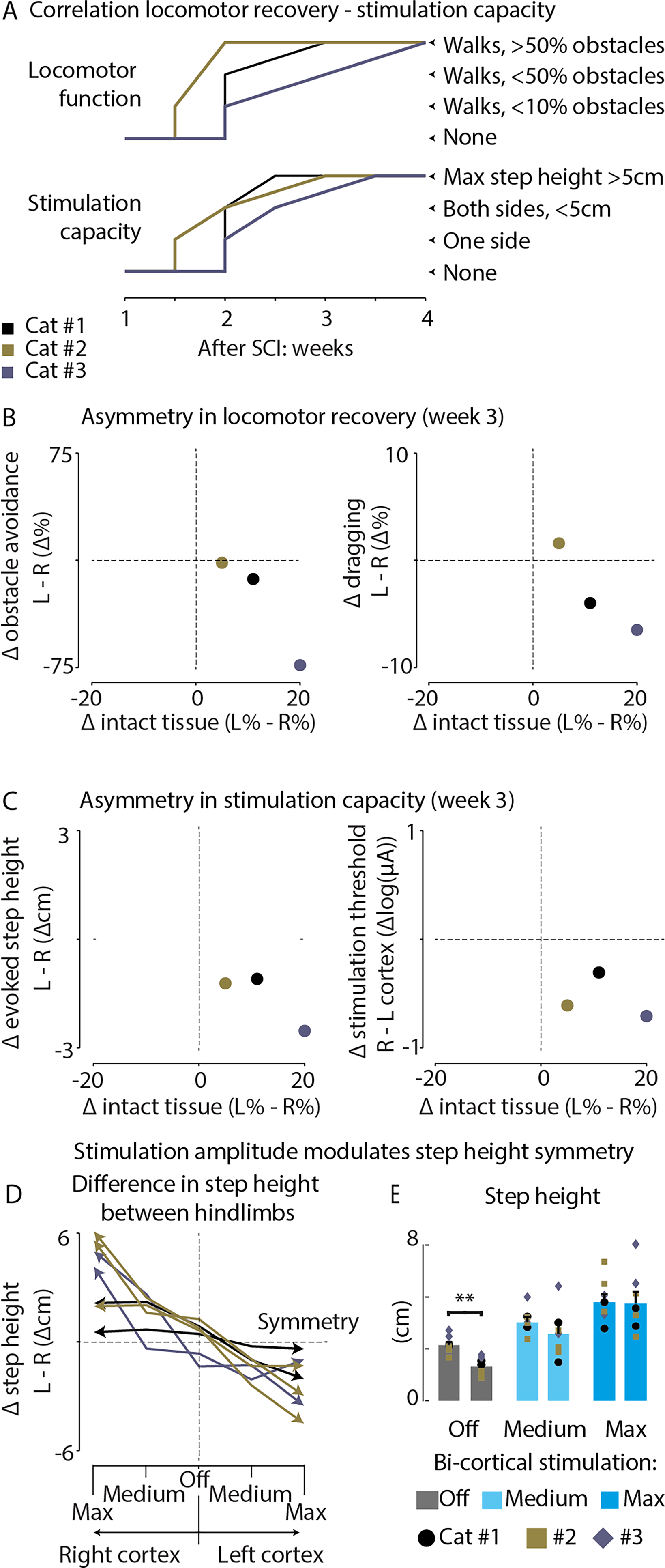
The return of locomotion correlated with the re-emergence of cortically induced gait modulation. (A) In the top section, it is shown that cats began to recover overground walking 1.5–2 weeks after SCI and could clear obstacles (>50%) during weeks 2–4 post-SCI. In the bottom section, the first responses to cortical stimulation started to appear 1.5–2 weeks after SCI and evolved into strong leg flexions (>5 cm) during weeks 2.5–3.5 post-SCI. The timeline of locomotor recovery in each cat closely aligns with the timeline of cortical stimulation capacity expression. (B) Asymmetries in the outcomes of contusive lesions (spared tissue) corresponded to asymmetries in locomotor capacity, specifically related to obstacle avoidance and dragging. (C) Asymmetries in contusive lesion outcomes were also associated with differences in stimulation capacities across the two cortical hemispheres, including stimulation thresholds and the ability to modulate step height. (D) By adjusting the stimulation amplitude to the left and right cortex, it was possible to reduce the asymmetry in step height between the left and right hindlimbs. Altering the stimulation amplitude to the contralateral cortex could mitigate asymmetry caused by the lack of elevation in a particular hindlimb. (E) Step height was compared between hindlimbs during spontaneous locomotion and during bi-cortical stimulation at medium and maximal amplitude. In the absence of stimulation (stimulation off), there were differences in step height between hindlimbs. However, bi-cortical stimulation corrected step height and restored symmetry between hindlimbs. Modified from Duguay, Bonizzato, Delivet-Mongrain, Fortier-Lebel and Martinez,7 under the CC BY-NC-ND license (http://creativecommons.org/licenses/bync-nd/4.0/).
Furthermore, the study found that asymmetries in spinal lesion outcomes correlated with asymmetries in locomotor capacity and differential stimulation capacities across the two cortical hemispheres (Figure 3(B) and (C)). Cats with right-dominant spinal injuries displayed right-dominant locomotor deficits and exhibited lower excitability of the right hindlimb responses to cortical stimulation during the early recovery period. Remarkably, even in cats exhibiting an asymmetrical gait pattern, fine-tuning the stimulation amplitude individually on each cortex allowed for the control of each hindlimb's trajectory in a phase-dependent manner (Figure 3(D) and (E)). These findings underscore the importance of customizing neuroprosthetic interventions for individuals, acknowledging that the location and severity of spinal cord injuries can significantly differ among individuals. Cortical neuroprosthetic approaches may be effective for individuals exhibiting asymmetrical gait patterns.
In summary, Duguay, Bonizzato, Delivet-Mongrain, Fortier-Lebel and Martinez7 have devised an innovative stimulation strategy to alleviate walking deficits in rats with lateralized SCI and cats with bilateral SCI. Our approach combined the advantages of supraspinal approaches 11 and patterned stimulation techniques 8 that have been rapidly translated to ongoing clinical trials. The bi-cortical neuroprosthesis tested in this study shows promise in providing precise and personalized control over gait patterns, even in cases of asymmetrical locomotion. This suggests that individuals with varying degrees of motor impairments resulting from SCI or stroke could potentially benefit from cortical neuromodulation approaches.
The study provides valuable insights, but it is equally important to embrace its limitations as opportunities for further exploration and enhancement. The use of a cat model for contusive SCI serves as a useful tool for specific aspects of SCI research. However, it may not fully replicate the intricacies of human SCI, potentially leading to distinct recovery patterns and physiological responses. Acknowledging these differences emphasizes the need to consider the unique characteristics of human SCI when interpreting these findings. To bridge the gap between animal models and human applications, human subject research is essential to validate the effectiveness of cortical neuromodulation in clinical settings.
Although supported by rat studies aimed at clarifying the mechanisms of cortical stimulation,2,12,13 the study's focus on immediate cortical stimulation for locomotion modulation offers immediate insights, but there is a vital need to explore its long-term effects on locomotor recovery, which holds great significance for potential clinical applications.
The challenges encountered in modulating extensor activity through cortical stimulation after SCI highlight the necessity for refining approaches to restore natural gait patterns. Embracing multifaceted interventions, which could involve a combination of cortical with spinal 14 or peripheral 15 stimulation, may be pivotal for achieving full restoration of locomotor patterns in the context of severe SCI.
Considering less invasive methods, such as transcranial magnetic stimulation, could expand the applicability of these interventions, potentially making them more accessible and acceptable to a broader population. 6
While the study discusses the potential for clinical translation, the next crucial step lies in validating its safety and effectiveness in human patients through direct human data or clinical trials. These limitations, viewed through a constructive lens, guide future research towards comprehensive and highly effective motor recovery strategies.
Footnotes
Declaration of conflicting interests
MM filed a patent application (U.S. No. 62/880,364) covering a device that allows one to perform cortical stimulation during movement. She is also a shareholder of a start-up company focused on developing neurostimulation technologies.
Funding
The author discloses receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by the Natural Sciences and Engineering Research Council of Canada (RGPIN-2015-03860), the TransMedTech Institute and the Morton Cure Paralysis Fund. M.M. was supported by a salary award from the Fonds de Recherche Québec Santé.
Author biography
Marina Martinez received her Ph.D in Neuroscience from the Université d'Aix-Marseille, France, in 2009. Subsequently, she conducted postdoctoral research at the laboratory of Pr. Serge Rossignol, Université de Montréal, Canada. In 2014, she joined the Hotchkiss Brain Institute at the University of Calgary as an Assistant Professor in the Department of Cell Biology and Anatomy. Currently, she holds the position of Associate Professor in the Department of Neuroscience at Université de Montréal and Hôpital du Sacré-Coeur de Montréal. Her research focuses on unraveling the mechanisms of motor recovery after spinal cord injury and developing assistive neurotechnologies aimed at reversing paralysis.
