Abstract
Objective
This study aimed to assess osteoarthritic changes in the ankle joint in a surgical model of chronic ankle instability (CAI) in mice.
Methods
This experimental study was conducted this study using 14-week-old Institute of Cancer Research (ICR) male mice (n = 19), divided into the sham control ([SH], n = 5), anterior talofibular ligament resection ([ATFL]; mild sprain, n = 7), and ATFL + calcaneofibular ligament resection ([ATFL + CFL]; severe ankle injury, n = 7) groups. A combination of behavioral analysis, micro-computed tomography, immunohistochemical staining, and serological analysis through enzyme-linked immunosorbent assay was performed to evaluate the outcomes.
Results
A decrease in hind-leg standing was observed in the injured mouse groups, indicating signs of discomfort. Significant deterioration in cartilage integrity and joint structure was quantitatively confirmed in the ATFL + CFL group. Increased cartilage erosion and subchondral bone damage were observed. Serological analysis revealed elevated levels of inflammatory cytokines (TNF-α, IL-6, and IL-1β) in the ATFL + CFL group, suggesting a heightened inflammatory response.
Conclusions
This study demonstrated that severe CAI, particularly that involving both the ATFL and CFL, directly contributes to the development of ankle osteoarthritis. This highlights the critical need for targeted interventions in preventing or mitigating this progression.
Introduction
Lateral sprains are one of the most common sports-related injuries. 1 In addition to the initial discomfort of the ankle sprain, 20%–30% of patients could progress to chronic ankle instability (CAI). 2 A combination of mechanical and functional instability is the probable primary causes of CAI.3–9 In addition, if CAI is not treated adequately or is neglected over a long period of time, ankle osteoarthritis (OA) can develop. 10
Since a long-term prospective follow-up is required, and as confounding bias can easily occur during the study period, few cohort studies have investigated whether OA occurs in cases of CAI. Therefore, whether the ankle sprain or CAI is the cause of ankle OA remains unclear. An animal model that mimics the pathologies observed in humans with CAI could reduce the challenges associated with prospective research into human CAI. Several studies have introduced an ankle sprain model in rodents to evaluate the relationship between sprain and CAI or pain control in sprains.11,12 However, despite its high incidence and clinical importance, proper animal models to evaluate the impact of CAI on OA development and prevention are lacking.
Therefore, this study aimed to assess osteoarthritic changes in the ankle joint in a surgical model of CAI in mice. It was hypothesized that several pieces of evidence associated with an ankle OA, from clinical, radiological, histological, and serological analyses, would be identified in a surgical CAI mouse model.
Materials and methods
Ethics approval
All efforts were made to minimize the number of animals used and to reduce their pain and distress during the experimental procedures. The study was approved by the Institutional Animal Care and Use Committee of Wonkwang University (WKU19-63), Republic of Korea, and was conducted in accordance with the Guide for the Care and Use of Laboratory Animals, 8th Edition. 11 Animals were monitored daily for general health and signs of distress, including reduced activity, weight loss, abnormal posture, and poor grooming. Humane endpoints were predefined, and mice showing severe or persistent distress were humanely euthanized according to institutional animal care guidelines.
Animal group and experimental design
The reporting of this study conforms to ARRIVE 2.0 guidelines. 12 Fourteen-week-old male ICR mice (weighing 33–36 g, n = 19) were purchased from Samtako Co Ltd (Osan, South Korea). All mice were fed a standard chow diet, given free access to water, and maintained in a pathogen-free environment at an ambient temperature of 22–24 °C and relative humidity of 55–60% under a 12 h:12 h light/dark cycle. Environmental enrichment, including nesting material and shelters, was provided to promote the animals’ well-being throughout the study period. Mice were randomly assigned to one of the following three groups using a computerised random number generator: sham group ([SH]; control, n = 5), resected anterior talofibular ligament group ([ATFL]; mild ankle instability, n = 7), and resected ATFL/calcaneofibular ligament group ([ATFL + CFL]; severe ankle instablity, n = 7), and the mice were housed individually. A formal priori sample size calculation was not performed. The number of animals used in each group was determined based on previous studies employing similar surgical models and experimental designs, with consideration given to minimizing animal use while maintaining adequate statistical power to detect biologically relevant differences.
Surgical procedures
Each mouse was anesthetized by inhalation of 4% isoflurane with supplemental oxygen for 2 min in an induction chamber, followed by maintenance with 1.0–1.5% isoflurane during the surgical procedure. Under these conditions, mice recovered within approximately 50–80 s after removal from anesthesia. After confirming adequate anesthesia, the right ankle was shaved and disinfected with chlorhexidine. Using a microscope and sterile instruments, a longitudinal incision was made in the anterolateral skin of the ankle to expose the ankle joint. Then, the skin was gently retracted to expose the lateral structures of the ankle joint (Figure 1(a)). To expose the ATFL, the peroneal tendon is identified and retracted as it lies directly along the ATFL in mice and is superficially visible.13,14 For the ATFL group, the ATFL was resected (Figure 1(b)). For the ATFL + CFL group, the ATFL and CFL were resected (Figure 1(c) and (d)). In the SH group, no ligament was damaged. The incision was closed with simple sutures using Nylon 4-0. After surgery, all mice were allowed to recover under a warming lamp until they were able to move freely. All mice were housed individually in cages postoperatively. No specific exclusion criteria were applied in this study. All animals and data points were included in the final analysis, as no significant postoperative complications, abnormal weight changes, or behavioral issues were observed.

Anatomical structure and location of the ankle ligament in mice. (a) The ATFL is identified between the EDL and EDQ in the anterolateral view after longitudinal skin incision and retraction. (b) The ATFL was resected in the ATFL group and ATFL + CFL group. (c) The CFL was identified under the distal part of the fibula. (d) The CFL was resected in the ATFL + CFL group. ATFL: anterior talofibular ligament; CFL: calcaneofibular ligament; EDQ: extensor digiti quinti; EDL: extensor digitorum longus.
Physical activity and clinical assessment
According to a previous study that analyzed pain-like behaviors in animal models, hypersensitivity to mechanical stimulation of the hind legs is considered to reflect joint pain. 15 Therefore, behavior was assessed by evaluating the number of times to stand on the hind leg for 60 min at the same time every 4 weeks, from 4 weeks after surgery, using video recording analysis. 15 In addition, to evaluate the clinical severity of arthritis, bodyweight, paw thickness, and ankle thickness were assessed immediately before sacrifice. Paw thickness and ankle thickness were measured independently by three researchers using digital calipers (Mitutoyo, Andover, UK), and the average thickness of both hind limbs was used. Paw thickness was determined by dorsoventral measurement, and ankle thickness was measured at the widest part of the ankle joint.
Micro-computed tomography (micro-CT)
High-resolution micro-CT scanner (SkyScan1173; Bruker-CT, Kontich, Belgium) was used to evaluate radiological changes (800 images were acquired using a tube voltage of 130 kV, a current of 66 μA, and a 1.0-mm aluminum filter).
Histopathological assessment
At the end of the experiment, mice were euthanized by cervical dislocation. Death was confirmed by the absence of reflexes and respiratory movement before tissue collection. Ankle joint bone tissue was fixed in 10% neutral buffered formalin for 24 h, decalcified in Calci-Clear Rapid® (National Diagnostics, Atlanta, GA, USA) for 1 week and then embedded in paraffin. Sections (4 μm thick) were cut on a Leica RM2255 microtome (Leica Microsystems, Bannockburn, IL, USA) and stained with haematoxylin (BBC Biochemical; Mount vernon, WA, USA) and eosin (BBC Biochemical) (H&E), safranin O (MilliporeSigma; St Louis, MO, USA) or toluidine blue (MilliporeSigma). Images were taken using a PannoramicTM P250 Flash digital slide scanner (3DHISTECH Ltd, Budapest, Hungary), and a dedicated viewing program (CaseViewer; 3DHISTECH Ltd) was used for observation. Histological assessment of cartilage degradation was performed using the Osteoarthritis Research Society International (OARSI) scoring system, a validated method for evaluating OA severity in murine models. Safranin O-stained sagittal sections of the ankle joint were scored by two blinded observers on a scale of 0 to 6, as described in the OARSI guidelines. 16 The average score was used for statistical analysis.
Blood biochemical indicators
Blood samples were collected from mice by drawing 300–500 μL of blood from the orbital sinus after inhalation anesthesia with isoflurane. The orbital sinus was selected as the site of blood collection because it allows for the efficient collection of a relatively large volume of blood in mice with minimal distress when performed properly under anesthesia. Blood samples were centrifuged at 1300 × g for 15 min at 4 °C. Levels of serum tumor necrosis factor (TNF)-α, interleukin (IL)-6, and IL-1β (R&D Systems, Minneapolis, MN, USA), and mouse cross-linked C-terminal telopeptide of type I (CTX-1), a bone resorption marker (MyBioSource, San Diego, CA, USA), were measured using enzyme-linked immunosorbent assay (ELISA) kits according to the manufacturers’ guidelines.
Statistical analysis
All experiments were performed at least three times, and statistical analyses were performed using GraphPad Prism software (Version 8.0; GraphPad Software, La Jolla, CA, USA) with a significance level set at p < 0.05. Normality was assessed using the Shapiro–Wilk test for one-way analysis of variance (ANOVA). The preference and acceptance tests were analyzed using one-way ANOVAs with post-hoc pairwise comparisons applying Tukey's HSD test. Residual diagnostics were conducted to check the normality and linearity assumptions.
Results
CAI reduces physical activity and increases paw thickness
To confirm the behavioral changes and clinical findings of OA in mice following the induction of CAI, a comparative analysis was performed among the SH, ATFL, and ATFL + CFL groups. No significant differences in body weight changes were observed among the groups (p = 0.340) (Figure 2(a)). Physical activity was measured by the number of times mice stood on their hind legs. The ATFL and ATFL + CFL groups showed significantly reduced rearing at 18 weeks (SH vs. ATFL, p = 0.006; SH vs. ATFL + CFL, p = 0.004), 22 weeks (SH vs. ATFL, p = 0.003; SH vs. ATFL + CFL, p < 0.000), and 26 weeks (SH vs. ATFL, p = 0.000; SH vs. ATFL + CFL, p = 0.000) as compared to the SH group (Figure 2(b)). The ATFL + CFL group developed significant swelling and increased paw thickness, accompanied by reduced activity, as compared to the SH and ATFL groups (Figure 2(c) and (d)). While the ATFL group showed a tendency toward increased paw thickness compared to the SH group, this difference was not statistically significant. In contrast, the ATFL + CFL group showed a significant increase in paw thickness relative to both the SH and ATFL groups. These findings indicate that additional injury to the CFL exacerbates osteoarthritis severity compared to ATFL injury alone. However, ankle thickness did not change significantly, even in the ATFL + CFL group (p = 0.615) (Figure 2(e)).

Analysis of physical activity and clinical changes related to osteoarthritis in a chronic ankle instability mouse model. (a) Bodyweight change in each group was determined on the indicated days. (b) Physical activity was measured as the number of times a mouse reared onto the hindlimbs during a 60-min period at 4, 8, and 12 weeks after surgery. (c) Representative photographs of SH, ATFL, and ATFL + CFL group mice. (d) Paw thickness and (e) ankle thickness were measured at 12 weeks after surgery. Statistically significant differences are indicated by *p < 0.05, **p < 0.01 or ***p < 0.001 versus the SH group. SH group: sham, control; ATFL group: resected anterior talofibular ligament, mild ankle sprain; ATFL + CFL group: resected ATFL/calcaneofibular ligament, severe ankle injury; W: weeks.
CAI increases synovial inflammation and joint destruction
Micro-CT findings demonstrated articular destruction of the ankle joint of the hind paw. While no significant change was seen in the ATFL group, the ATFL + CFL group demonstrated significantly increased inflammation and destruction of the tarsal metatarsal joint, calcaneus, or tarsal bone as compared to the SH group (Figure 3(a)). Examination of microstructural characteristics revealed a decrease in total porosity in both the ATFL and ATFL + CFL groups. By micro-CT analysis of the ankle joint, bone mineral density (BMD) and bone volume fraction (BV/TV) were shown to be significantly increased in the ATFL and ATFL + CFL groups (Figure 3(b)–(d)). Histological evaluation revealed inflammatory cell infiltration, cartilage degradation, and pannus formation in the ankle joints of the ATFL + CFL group, with more pronounced changes compared to the SH and ATFL groups (Figure 4(a)). The ATFL + CFL group exhibited marked synovial proliferation and joint space narrowing, indicating progressive joint destruction. To quantitatively assess osteoarthritic severity, OARSI scoring was applied to Safranin O–stained sections. The OARSI scores were significantly higher in the ATFL + CFL group compared to the SH group (p < 0.001), and moderately elevated compared to the ATFL group, reflecting a higher degree of cartilage degeneration and OA progression (Figure 4(b)). These findings support the conclusion that combined ligament injury accelerates osteoarthritic changes in the ankle joint.
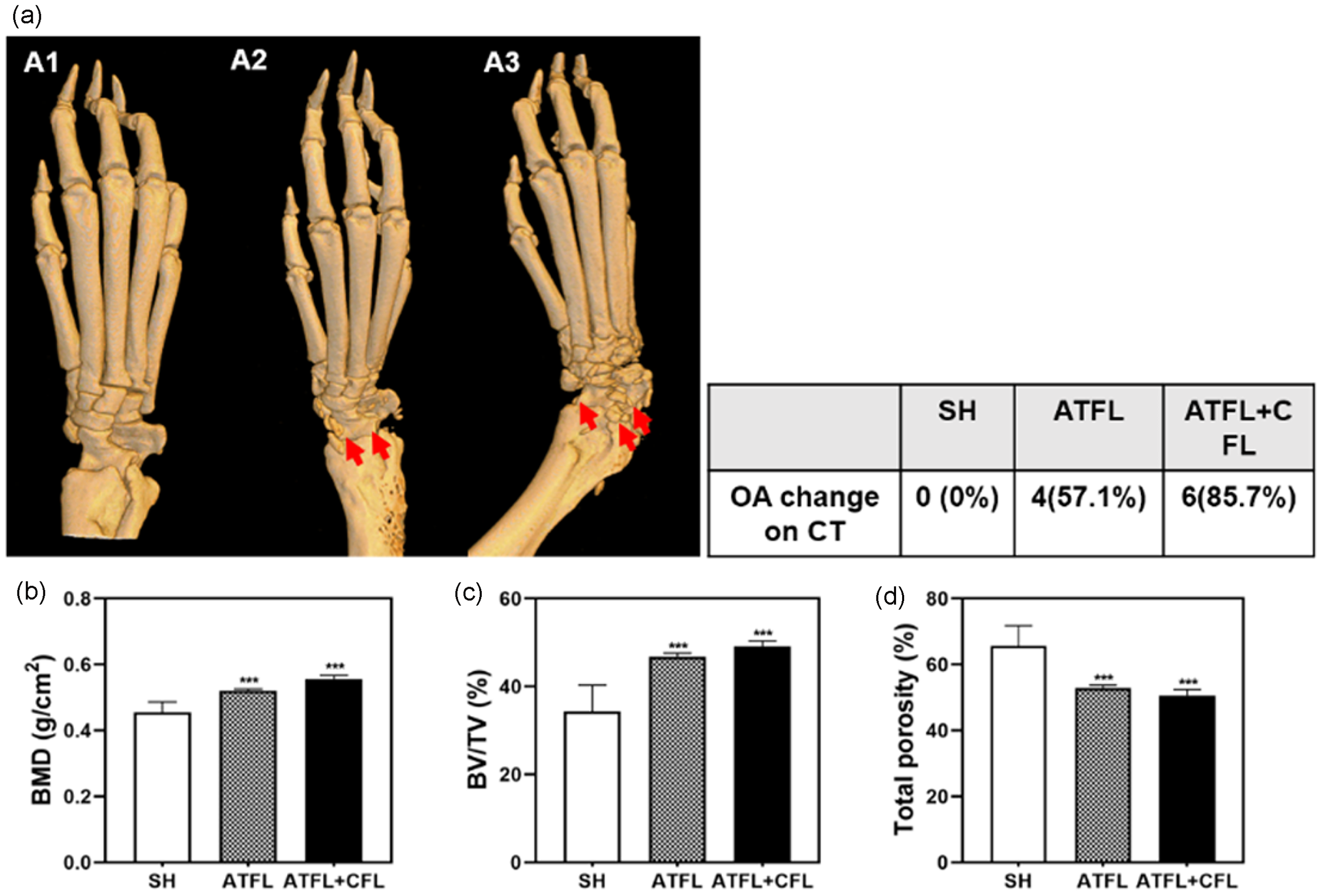
Micro-computed tomography (CT) analysis in a chronic ankle instability mouse model. (a) Micro-CT images of the ankle joints of the SH group (A1), ATFL group (A2) and ATFL + CFL group (1C) mice. Osteophyte and loose bodies were identified in the ATFL group and ATFL + CFL group (A, right). The red arrow indicates an osteophyte and a loose body in the ankle joint. (b–d) Quantitative analysis of bone mineral density (BMD) (B), bone volume fraction (BV/TV (c), and total porosity (d) of the mouse ankle joints. ***p < 0.001 versus the SH group. SH group: sham, control; ATFL group: resected anterior talofibular ligament, mild ankle sprain; ATFL + CFL group: resected ATFL/calcaneofibular ligament, severe ankle injury.

Histological assessment and OARSI scoring in a chronic ankle instability (CAI) mouse model. (a) The ankle joint was sectioned and stained with hematoxylin and eosin (H&E), safranin O, or toluidine blue. (b) OARSI scores were obtained from Safranin O–stained sections to evaluate osteoarthritic severity. Data are presented as mean ± SD. p < 0.05 was considered statistically significant.
CAI stimulates proinflammatory cytokine production
The mechanisms underlying CAI-induced OA were examined. Because various cytokines are involved in joint inflammation and OA progression, we measured cytokine levels using ELISA. Significant increases in TNF-α, IL-6 and IL-1β were observed in the serum of the ATFL + CFL group compared to the SH group (Figure 5(a)–(c)). Additionally, the effect of CAI on blood markers related to bone resorption was assessed. Serum CTX-1 levels tended to increase in the ATFL + CFL group compared to the SH group, but the difference was not statistically significant (Figure 5(d)).
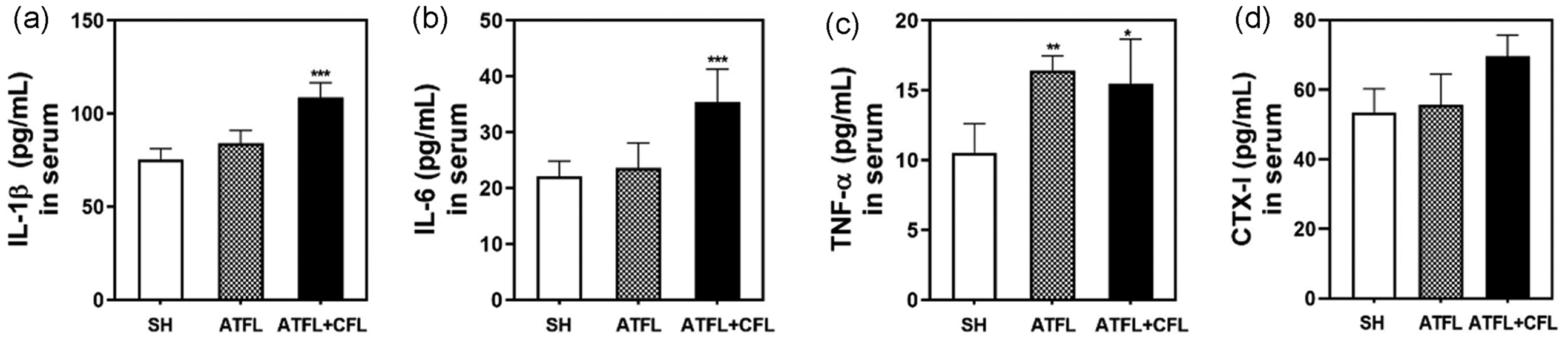
Analysis of proinflammatory mediators and a bone resorption marker in the serum of a chronic ankle instability model mice. The expression levels of (a) interleukin (IL)-1β, (b) IL-6, (c) tumor necrosis factor-α (TNF-α), and (d) C-terminal telopeptide of type I (CTX-1) were determined using murine enzyme-linked immunosorbent assays. Statistically significant differences are indicated by **p < 0.05, **p < 0.01, or ***p < 0.001 versus the SH group. SH group: sham, control; ATFL group: resected anterior talofibular ligament, mild ankle instability; ATFL + CFL group: resected ATFL/calcaneofibular ligament, severe ankle instability.
Discussion
In this study, the effects of CAI were investigated using a surgical mouse model. The results demonstrated that the ATFL + CFL group, in which both the ATFL and CFL were resected, revealed significantly greater clinical severity, such as indicated by paw thickness, decreased rearing behavior, greater erosion of the cartilage on three-dimensional micro-CT, and aberrant histology. More specifically, quantitative analysis of arthritis-related inflammatory cytokines in serological analysis by means of ELISA supported the findings indicating arthritic changes of the ankle joint in the ATFL + CFL group with induced gross instability.
The recurrence rate of ankle sprains is high, and as a result, a considerable number of patients who have suffered from ankle sprains will go on to develop CAI.17,18 Persistent ankle instability leads to ongoing disability and sensorimotor control deficits, which are associated with reduced physical activity and quality of life.17,18 Not surprisingly, these residual impairments are thought to persist for the remainder of the patient's life, in part due to the link between CAI and posttraumatic ankle OA.17–20 However, a lack of lifelong prospective studies, due to logistical reasons, limits our understanding of whether CAI affects the development of ankle OA. The optimal prospective study design to enable the systematic determination of factors that contribute to the development of ankle OA would include an evaluation of individuals before and after development of CAI. Given the time and difficulty associated with performing such systematic investigations of arthritic changes after CAI using clinical and radiological evidence, such studies are currently not feasible in humans. However, a longitudinal, live-animal experimental design avoids these concerns and allows us to quantify the outcome of CAI-related ankle OA by applying systematic perturbations to the ankle joint.
Importantly, the lateral ligament complex of the mouse ankle have anatomic locations and functions similar to those in humans; thus, the mouse ankle is a useful model for studying neuromuscular adaptations after an ankle injury.13,14,16–21 While mouse models are well-established for ankle sprains, a surgically induced CAI mouse model has not been used.13,14,16,17 In a previous study, a rat model of posttraumatic OA was significantly different from the pathophysiology of CAI because the tendon and ligament tissues were excised. 22 In another study, a group with simple transection of the ATFL (n = 7), a group with ATFL + CFL transection (n = 7), and a SH operation group (n = 5) were analyzed using the same method as in the current study, but no behavioral, histological, or serological differences were found among the three groups. 13 In our study, unlike the previous surgically induced mouse model that transected both the ATFL and CFL, CAI was generated in a mouse model by resection with a few defects of the ATFL and CFL, to prevent ligament healing. Subsequently, the OA changes of the ankle joint in this CAI mouse model were verified through clinical, radiological, histological, and serological analyses. Therefore, the strength of this study is that it demonstrated the induction of posttraumatic OA by CAI in an animal model. Furthermore, this study could serve as a basis for subsequent studies to evaluate the drugs and genetic pathways that prevent or alleviate OA caused by CAI.
Altered foot posture and ankle instability may influence proximal joint alignment and load transmission. Previous studies have demonstrated that changes in foot posture, such as hallux valgus or rearfoot pronation, are associated with varus knee alignment and medial knee osteoarthritis.23,24 This biomechanical coupling suggests that chronic ankle instability, by altering foot posture and gait mechanics, may contribute not only to local joint degeneration in the ankle, but also to compensatory changes affecting knee joint health over time.
CAI has been described as an isolated ATFL tear in 80% of cases, or an ATFL and CFL tear in 20% of cases. 25 The ATFL is the weakest and most frequently injured component of the lateral ankle ligament complex.25,26 Isolated CFL injuries are uncommon; instead, the CFL is typically torn secondary to an ATFL tear. 26 In this study, the isolated ATFL group demonstrated inferior outcomes as compared to the SH group in terms of histological analysis as well as TNF-α in serological analysis. However, OA changes were significantly greater in the ATFL + CFL group than in the ATFL group. These results might provide indirect evidence that, in humans, gross instability of the ATFL and CFL is more likely to induce posttraumatic OA than instability of the ATFL alone.
In comparison with prior rodent models of ankle instability, such as the one described by Wikstrom et al., 14 which primarily assessed neuromuscular function and balance following ligament transection, our model more effectively captures the structural and inflammatory changes associated with posttraumatic osteoarthritis. The model by Wikstrom et al. involved both CFL and ATFL + CFL groups, which does not fully reflect the typical injury pattern observed in humans, where CFL injuries rarely occur in isolation. 14 In contrast, our model, which includes isolated ATFL resection and combined ATFL + CFL resection, more closely mimics the clinical progression of lateral ankle sprains, where ATFL injury is often followed by CFL injury.25,26 By combining detailed anatomical surgery with micro-CT imaging, histological staining, and cytokine analysis, this study provides a more physiologically relevant and reproducible platform for investigating the pathophysiology of CAI-induced osteoarthritis and evaluating potential therapeutic interventions.
This study has some inherent limitations that should be addressed. A few functional differences between mice and humans should be considered. In mice, the tibiotalar joint is more flexed, and the calcaneus bears less weight compared to the human ankle. These differences impose limitations on the mouse models in terms of being true for human ankle OA. 22 Second, due to the limitations of the experimental schedule and the research scale, the life span of mice during the experiment did not correspond to adult age. In addition, although a comprehensive understanding of the disease pathology requires longitudinal studies, our analyses were based on a single time-point. 27 Prediction of OA changes in older mice is difficult, and estimating the lifelong outcome in the ATFL resection only group is not simple. Nevertheless, the fact that ankle OA occurred in the ATFL + CFL group within a period of 12 weeks is meaningful in that it confirmed that the insufficiency of the lateral ligament complex contributed to the development of ankle OA. Third, ICR mice were used instead of rats, as these mice are larger than conventional strains and more suitable for precise and delicate surgeries under a microscope. Finally, future studies are planned to investigate the molecular pathways underlying CAI-induced OA using transgenic models, such as knockout mice.
Conclusion
This study demonstrated that surgical induction of CAI by resection of the ATFL + CFL in a mouse model may result in the development of OA of the ankle joint, as evaluated through analysis of behavior, clinical severity, micro-CT images, histology, and serology analysis. This model aligns with current theoretical frameworks of posttraumatic OA due to CAI and may serve as a valuable tool for future investigations into therapeutic strategies.
Footnotes
List of abbreviations
Acknowledgements
An earlier version of this manuscript has been posted as a pre-print on Research Square (![]() ).
28
).
28
Ethical considerations
The study was approved by the Institutional Animal Care and Use Committee of Wonkwang University (WKU19-63), Republic of Korea, and the study was conducted according to standard guidelines.
Authors’ contributions
SH Lee contributed to the study conception and design, as well as drafting the manuscript. JY Kim and SY Eun were responsible for data acquisition, analysis, and interpretation. GH Lim and MH Kim assisted in manuscript preparation. All authors critically reviewed the manuscript for significant intellectual content and approved the final version for submission.
Funding
This study was supported by a grant from the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2020R1G1A1102304, NRF-2022R1C1C2010740).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
