Abstract
Piezoelectric signals were obtained from samples based on hydroxyapatite (HAp) and collagen (C) containing different glucose (G) concentrations. HAp was obtained by coprecipitation using Ca2+ and HPO42− as precursor ions in solution. C and G were added at the beginning of the coprecipitation method during the HAp growth. The presence of glucose in HAp and collagen samples drastically reduces the voltage amplitudes and considerably increases the relaxation times of the piezoelectric signals. HAp and collagen are the main constituents of bone, muscle, etc., then, it is possible to use piezoelectric technology for local and early detection of high glucose concentrations: small pressures applied by electrodes or by actuators placed in appropriate places on the body to establish a background concentration and, from this, to determine regions of the body with high local glucose concentrations: weak signals and large relaxation times are associated with a diminishing in the sensitivity, and are indicative of the presence of regions of abnormally high glucose concentrations.
Introduction
Glucose is the primary energy source of living organisms; it is carried through the blood system to reach all cells in the body. The ingestion of glucose in humans is high because it is the body’s principal energy source; it is absorbed in the small intestine, passing to blood and finally to cells, mediated by insulin. 1 It participates in several metabolic processes: the control of some hormones, the glycosylation of many enzymes,2,3 the synthesis of cellulose for the cell wall, the activation of cellular growth, the storage of reserve compounds, the formation and remodeling of bone, etc. Glucose possesses a high capacity to react with other molecules like hydroxyapatite (Hap) and collagen (C). However, high glucose levels in the blood produce undesired effects: diabetic foot, neuropathies, blindness, bone atrophy, etc.4,5
Glucose has five hydroxyls, the primary (–CH2–OH) being the most reactive because it has more mobility and less steric hindrance. Because glucose is a small molecule (≈1 nm), it can diffuse to react with the OH groups on the inner and outer surfaces of the HAp, interrupting the crystal growth by blocking the exposed functional groups and preventing Ca2+ and HPO42− ions from being deposited on the HAp to continue its growth; this reduces the crystal size, modifies the piezoelectric response, the ionic transport, and the electric double layer (EDL) formation. The HAp-G reaction can be schematically represented by:
Bone, made of HAp and C, requires considerable amounts of glucose for its growth, however, when glucose concentration exceeds certain limits, diabetic complications occur, including a loss of sensibility characterized by low voltage amplitude and long relaxation time. Glucose does not accumulate homogeneously in body tissues but preferentially affects regions such as legs, arms, hand and feet; an analysis of piezoelectric signals allows the determination of high glucose concentrations in the different regions of the body, proving to be an appropriate technique for early, rapid and non-invasive detection of diabetes. It can be established, as a hypothesis, that a significant reduction in the piezoelectric amplitude is indicative of high local glucose concentrations; because the amplitude of piezoelectric signals can be determined in a precise way, this may be an effective technique for early detection of diabetes; however, it is necessary to measure the piezoelectric voltages in different parts of the body to establish the background concentration (which corresponds to the average levels of glucose naturally found in the body) and determine those regions that show changes in voltage amplitude; a device, similar to that used to monitor vital signs, can be redesigned for simultaneous measurements of piezoelectric signals on different and appropriate parts of the body, to obtain a map of piezoelectric voltages showing the distribution of glucose on the body. Another possibility is the use of piezo actuators so that, on the one hand, they exert an appropriate pressure and that simultaneously measure the intensity of the piezoelectric signal. This work reports the dependence of the piezoelectric signals on glucose content.
Theoretical considerations
Piezoelectric effect, ionic transport and electric double layer
The piezoelectric effect is a charge separation caused by mechanical compression; when stress is applied to a piezoelectric material, separate charges move towards the surfaces of the material creating an electric field in its interior Eo which gives rise to a voltage V; the electric field moves the charges present (mainly ions) towards charges of opposite sign; ionic mobility controls the output voltage of the piezoelectric profiles. Many biomaterials in the human body are piezoelectric: DNA, HAp, proteins (collagen, keratin, elastin), skin, hair, many amino acids, etc.9–13; the piezoelectric effect is a fundamental property of all biological systems since it is the main form of communication of all parts of the body with the brain14–16; piezo signals are commonly used to sense and control many processes in the body because the energy consumed by these processes is very low.17,18 As sensors, they detect mechanical stresses, deformations, fractures, injuries, etc., which can put in danger the integrity of the body. Because the presence of ions affects the piezoelectric voltage profiles, it is important to know the size, charge, and concentration of ions present in the system; there are several types of ions in the electrolyte in the body: Ca2+, HPO42−, OH−, etc.19,20; the field Eo transport these ions with different velocities near the charged surface modifying the voltage. The ionic transport is modeled as a series of jumps to neighbor sites; these sites must be of similar energy and size and percolated for long-distance conduction. However, when the ions concentration surrounding the jumping ion is high, the probability of jumping is reduced along with ionic transport; this effect is called “obstruction effect” and significantly controls ionic transport; for this reason ionic transport depends on the concentration. The electric field Eo moves the ions toward the charged surface of opposite sign and because they cannot cross the interface, they accumulate nearby forming the EDL.21,22 The EDL produces an electric field Ei in opposite direction respect to Eo, resulting in a reduction in the net field ET = Eo - Ei and consequently the voltage; as a consequence of the ion field Ei, the voltage profiles are decaying exponentials; then, V(t) was modeled as a double decaying exponential function:
Experimental methods
Materials
All HAp-based materials were obtained by the coprecipitation method reported elsewhere
23
; the reactants were calcium nitrate tetra-hydrated (Ca(NO3)2:4H2O), di-ammonium phosphate ((NH4)2HPO4), and ammonium hydroxide (NH4OH) used to adjust the pH, all from Aldrich Co. Collagen, in an aqueous solution, was obtained at 20% concentration (Eclat Laboratory) and used as received, while glucose was obtained from Dextrosa Anidar (
Samples preparation and characterization
For the piezoelectric determinations, the powders were mixed with 20% polyurethane (PU) used as a binder to stabilize the samples. 0.23 g of this mixture was placed in a stainless-steel mold to produce small disks of 12.7 mm diameter and 1.2 mm thickness; a pressure of 100 bars was applied to each disk for 1 minute; the application of this pressure compacts the sample while the chemical reaction that produces the PU begins; the samples were removed from the mold and left to rest for 1 day. All samples were hydrated by placing them for 15 seconds in a water fog produced by a commercial ultrasonic humidifier (Homedics model UHE-WB01) filled with distilled water; the moistened samples were quickly introduced between two stainless steel electrodes for electrical determinations and to avoid evaporation. The samples are required to be humidified for ionic transport.
Wide-angle X-ray diffraction (WAXRD) apparatus was used to determine glucose's effect on the HAp crystallite size. This analysis was accomplished on a Rigaku diffractometer model Ultima IV operating at 30 mA and 40 kV using a Cu Kα radiation wavelength of λ = 1.5406 Å; the angle 2Θ was varied from 5° to 80° at a scan of 2°/min. The crystallite size determination from the diffractograms, was carried out using JADE v8.1. The mechanical tests were performed in an Adamel Lhomargy (Evry, France) machine model DY22 used in compression mode with a velocity of 0.5 mm/min and a load cell of 500 daN; the Young moduli were obtained from the stress–strain slopes. The piezoelectric voltage profiles were obtained in two different modes: hold (H) and release (R); in H mode, the piezoelectric voltages were obtained while the maximum pressure (26.5 kPa) was applied for 3 minutes, while in R mode, the applied load was suddenly removed, and the voltage measured for 3 minutes without load. The piezoelectric analysis was obtained by measuring the voltages as a function of time using a digital multimeter USB Steren model MUL605 equipped with data communication software to the PC. The multimeter has a resolution of 0.01 mV in the 0 to 60 mV range with an accuracy of ±(0.5%) and an input impedance of 10 MΩ. The reproducibility was verified by repeating the measurements three times.
Results
Figure 1(a)–(c) shows the diffractograms of HAp-based samples with different glucose concentrations. In Figure 1(a), corresponding to pure HAp, the reflections are narrow and well-defined; however, when glucose was added between 1% and 9% (Figure 1(b) and (c)), these become wider, less intense and not well defined as compared with pure HAp, indicating that a change in the structure of the crystal has occurred due to the presence of glucose: due to the small size of glucose and its high reactivity, they can diffuse on the inner and outer surfaces of HAp reacting with its functional groups, interrupting the crystal growth by blocking the exposed groups and avoiding that Ca2+ and HPO42− ions are deposited in the HAp so that it continues with its growth. Figure 2 was included due to the chemical reaction between HAp and PU; PU is an amorphous material (no reflections) produced by a reaction between polyisocyanate (R–N–CO–) and all OHs present in the system, including those of HAp; this reaction may produce conformational changes that affects the HAp crystalline structure; when 20% of PU is added to HAp, the reaction that produces PU has not been completed and can compromise the structure of the HAp. Figure 3(a) and (b) shows complete voltage profiles for (a) HAp + 9%G and (b) collagen + 9%G. The first part of the profile corresponds to H mode while the second to R mode.

X-ray diffractions of HAp with (a) 0%, (b) 1%, and (c) 9% of glucose.

X-ray diffraction of HAp containing 20% PU.

Complete piezoelectric profiles showing the H mode followed by the R mode for (a) HAp + 9%glucose and (b) collagen + 9%glucose.
Some piezoelectric characteristics of the HAp samples were included in Figure 4(a) and (b). The voltage–stress (V–σ) plot is reported in Figure 4(a) showing a linear dependence of V with σ at low stresses, meaning that HAp is a linear piezoelectric material. In isotropic piezoelectric materials, like HAp, the polarization is proportional to the stress σ: P = d33σ, where d33 characterizes the piezoelectric properties. Figure 4(b) shows the hysteresis curve in a current–voltage (I–V) plot; hysteresis curves of piezo materials are typically reported in a polarization-electric field (P–E) space; however, because P is proportional to I and E is proportional to V, both spaces, P–E and I–V, are equivalent.

Some piezoelectric characteristics of HAp: (a) voltage–stress and (b) current–voltage.
In Figure 5 is reported a plot of the crystallite size of the HAp as a function of the glucose concentration; two similar curves were included of the same sample where one was obtained averaging the crystallite size overall reflections of the diffractogram, while in the other, the average was done using the three most intense reflections; in both profiles was included the standard deviation. These profiles show a significant reduction in the HAp crystallite size from 22 to 10 nm due to reaction with glucose: glucose blocks the access to functional groups, preventing the reaction with the corresponding ions for crystal growth of HAp.

Size of crystallite as a function of the glucose content.
The mechanical results are reported in Figure 6(a) and (b). Figure 6(a) shows the stress–strain (σ–ε) curves for HAp with different collagen concentrations (10, 15, 20, and 5%); these profiles show a linear dependence of σ with ε with practically the same slopes; the Young moduli were obtained from the slope Y = σ/ε and are reported in Figure 6(b) as a function of collagen concentration; these Young moduli are small compared to those obtained from natural bone because these materials were obtained by a physical mixture of HAp and C agglutinated with PU instead of producing the HAp-C composite by the natural coprecipitation process as the human body does; Y is practically constant (Figure 6(b)) for collagen concentration less than 20% (a reduction from 16.64 to 16.45 MPa) meaning that it is HAp that supports almost the whole compression stress; however, when the collagen concentration exceeds 20%, corresponding to percolation concentration, the reduction was larger, from 16.45 to 16.02 MPa: at concentrations higher than 20%, the elastic property of collagen begins to play an important role to the mechanical properties of the mixture HAp-C.

(a) Stress–strain and (b) Young modulus of samples at different collagen concentrations.
Figure 7(a)–(f) shows the effect of increasing glucose concentration from 0% to 9% on voltage amplitude and relaxation time for H and R modes. Figure 7(a) shows typical piezoelectric voltage profiles of HAp and collagen samples for H and R modes; these were shifted vertically for comparison purposes; all voltage profiles were fitted using a double exponential function (Eq. (2)) to obtain the voltage amplitudes and the relaxation times. Samples without glucose have large voltage amplitudes and short relaxation times in H mode; however, when glucose was increased up to 9%, the amplitudes were severely reduced from 88.8 to 4.8 mV, and the relaxation times enlarged from 1.3 to 9.9 s; these changes in voltages and times are reported, for all samples, in Table 1. Figure 7(b)–(f) shows, as histograms, effects of glucose on the amplitudes and times of the profiles of HAp and collagen for H and R modes: Figure 7(b) changes in Vo1; Figure 7(c) changes in Vo2; Figure 7(d) changes in τ1; Figure 7(e) changes in Vo1 and Vo2; Figure 7(f) changes in τ1. Histograms provide better visualization of these changes: blue boxes correspond to 0% G and red ones to 9% G, while the red arrows indicate reduction and blue ones increment. In all cases, glucose produces a reduction in the voltage amplitude and an increment in the relaxation times. Because the H mode is directly related to the intensity of the applied stress and the R mode to the healing process, the intensity for H is significantly larger with respect to R. The reduction in amplitude in H mode is smaller for collagen (from 22 to 10 mV) than for HAp (from 89 to 5 mV) which means that collagen is less sensitive to the reaction with glucose because the change in morphology has a limited affectation. Figure 8 provides a general view of the dependence of the amplitudes Vo1 and Vo2 of HAp and C samples as a function of glucose concentrations for H mode.

Plots of HAp and C with 0 and 9% G for H and R modes: (a) voltage profiles; (b) Vo1; (c) Vo2; (d) τ1; (e) Vo1 and Vo2; (f) τ1; blue is for 0%G and red for 9%G.

Voltage amplitude Vo1 and Vo2 as a function of glucose content for HAp and collagen.
Vo1, Vo2, τ1, τ2 for HAp and collagen containing 0 and 9% glucose.

Discussion
Figure 1(a)–(c) shows that when glucose concentration was increased, the XRD reflections become wider, less intense, and not well-defined with respect to pure HAp; this is produced by the reaction HAp-G which interrupts the crystal growth reducing the crystallite size and increasing the polydispersity. The diffractogram of HAp-20%PU reported in Figure 2 shows that the chemical reaction with PU did not modify the HAp crystalline structure. The voltage profiles reported in Figure 3(a) and (b) for (a) HAp + 9% G and (b) collagen + 9% G show two stages: the first one, corresponding to H mode, was obtained under applied pressure conditions, in the second stage, for R mode, the pressure was released; in both cases the profiles were decaying exponentials.
The voltage–stress (V–σ) plot reported in Figure 4(a) shows that, at low stresses, there is a linear relationship of V with σ, meaning that HAp is a linear piezoelectric material; the slope in the linear regime is directly related to d33 (= P/σ), which is a constant that characterizes linear, homogeneous and isotropic piezoelectric materials; however, at higher stresses, the V–σ relationship is no longer linear and, once V has reached its maximum value, it begins to decay due to the presence of defects like fractures or possible catastrophic failure. Figure 4(b) shows a hysteresis curve in an I–V space which is equivalent to P–E space where typically are reported hysteresis curves: P is proportional to I,
Figure 5 shows the effect of glucose on the crystallite size of HAp; there is a significant reduction in the crystallite size from 22 to 10 nm produced by the reaction with glucose: this can react with the functional groups of internal and external HAp surface, preventing the deposition of ions in solution to continue the HAp growth; this reaction reduces the crystallite size (Figure 1(b) and (c)) and the mechanical properties of HAp, causing complications such as brittle bones, osteoporosis, etc. Due to the small size of glucose and its high reactivity, small amounts of glucose are required to produce a severe reduction in the crystallite size; the reduction in crystallite size reported in Figure 5 supports the assumption of blocking of functional groups of the HAp by glucose molecules. Glucose can react with Ca2+, HPO42−, and HAp-OH, consuming them and interrupting the crystal growth: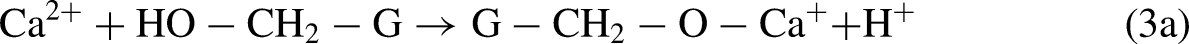



Respect to mechanical properties, there is a linear relationship stress–strain for HAp at different collagen concentrations (10, 15, 20, and 25%) (Figure 6(a)). The Young moduli, obtained from the slope Y = σ/ε, are reported in Figure 6(b) as a function of collagen concentration; this figure shows that Y is practically constant for collagen concentration less than 20%, (from 16.64 to 16.45 MPa) meaning that meaning that HAp is the one who supports the compression stress; however, when the collagen concentration exceeds 20% (percolation concentration), the reduction of Y was larger (from 16.45 to 16.02 MPa); in this case the elastic property of collagen contributes in an incipient but important way to the mechanical properties of HAp-C. In this figure the discrete points correspond to experimental data and the continuous line to a fitting using an exponential function: Y = 16.67 - 0.005 e0.2[C] where [C] is the collagen concentration. Figure 7(a) shows typical piezoelectric voltage profiles of HAp and collagen, while Figure 7(b)–(f) shows, as histograms, the effect of glucose on voltage amplitudes and characteristic times for HAp and C in H and R modes.
Glucose does not accumulate homogeneously throughout the body; some specific parts (foot, legs, arms, etc.) are more affected, showing high local glucose concentrations, which leads to diabetic disorders characterized by a loss of sensibility, weakness, poor blood flow, etc.; in an acute case, there is a sudden and intense onset characterized by strong piezoelectric signals of high amplitude and short duration; in the chronic case, the symptoms grow slowly, characterized by weak signals of small amplitude and long duration. Intense actions produce high-intensity piezoelectric signals which alert the brain that some action of important intensity is happening; the amplitude for an acute case is around 18 times more intense with respect to chronic one and 16 times more intense for hold with respect to release. The R signals correspond to small and residual signals producing a minor alert; this is generally related to the healing process however, this is not always the case: the presence of glucose significantly attenuates the intensity of the piezoelectric signals that reach the brain and, consequently, it pays little attention to this fact producing, generally, a worsening of the situation.
The inhomogeneous distribution of glucose in the body means that some regions are more affected by a high concentration of glucose; then, in addition to having tests that measure the average glucose throughout the body, it is equally or more important to have information on local glucose concentrations throughout the body; this can be done using the piezoelectric technique because is sensitive to the glucose concentration; it is convenient and necessary to obtain piezoelectric voltages in different parts of the body to build a map of piezoelectric voltages and, from this, a map of glucose concentrations; this information allows the physician to determine potential problems in different regions of the body. Clip-like devices, similar to those worn on the finger to monitor vital signs, can be redesigned to measure the voltages in different body parts simultaneously; piezoelectric signals are a promising alternative for early and non-invasive detection of diabetes. The piezoelectric determination in different body parts involves different materials, some of which may negatively influence glucose determination in specific regions.
According to Figure 8, the voltage amplitudes Vo1 and Vo2 of HAp and collagen in H mode suffer a significant reduction due to glucose; this mode is directly related to a severe action: the more serious the injury, the more intense the piezoelectric signal. This figure provides a general vision of how the piezoelectric amplitudes are affected by the presence of glucose. The important reduction happens when glucose reaches 3%; at higher concentrations, the intensity remains constant. A small green arrow was included indicating the glucose concentration of 2.0 g/L (≈0.2%), corresponding to the lower limit concentration for diabetes diagnosis: blood glucose concentration (BGC); this is a clinical value to specify a threshold between a healthy person and a diabetic. A loss of sensibility (a reduction in intensity) is a good criterion for establishing the “diabetes local threshold concentration” (DLTC) which is 3%G in HAp, as reported in Figure 8; this value does not correspond to the one obtained from a glycosylated hemoglobin analysis which is an average taken from the whole body.
Results reported for the uptake of glucose in mice skeleton in vivo show that bone accumulates a significant fraction of the total glucose ingested increased by the fact that the tests subjects were young with a high number of osteoblasts to produce rapid growth of the skeleton: the bone cells require a considerable amount of energy to maintain the skeleton in good conditions. The glucose absorption by bone was reported as the percentage of glucose injected dose (GID) per gram of tissue weight, that is %GID g−1. The glucose uptake by the skeleton, in vivo, was 4.38 ± 0.56% GID g−1 for the tibia and 2.65 ± 0.20% GID g−1 for the femur, given an average of 3.5 ± 0.38% GID g−1. This result agrees with the one reported in Figure 8: when glucose added (GA) per gram of HAp (GA g−1) reaches 3%, GA g−1 there is a drastic reduction in the voltage amplitude of the piezoelectric signal with the corresponding loss of sensibility. The value of 3.0% GA g−1 is the threshold for high local glucose accumulation, that is for local diabetes. The profiles reported in Figure 8 can be used to determine the onset of diabetes.
In Figure 5, the crystallite size is reported as a function of the glucose content; in this figure, a small green arrow was included at 2.0 g/L (≈0.2%) corresponding to BGC; the reduction in crystallite size is a consequence of the interaction of glucose with HAp. Studies have been reported on the interaction of the HAp surface with hydroxyl groups, 24 leading to the conclusion that on the HAp surface, there are more OH groups 1.67 with respect to calcium 25 1.33. This means that a small number of glucose molecules are required to block the crystal growth and reduce the crystallite size. Astala and Stott 26 found that a surface rich in Ca2+ ions is more active to OH− molecules than a surface rich in HPO42−; the interaction energy OH-HAp was determined for different crystalline planes finding that there is a strong interaction OH−Ca indicating a thermodynamically favorable adsorption process. From Figure 5, less than 1% of glucose is required to reduce drastically the crystallite size from 22 to 10 nm. The crystallite size is directly related to the piezoelectric voltage, being this a general behavior for piezoelectric materials. 27 From Figure 5, it is possible to infer that a small glucose concentration located in a specific part of the body produces a substantial reduction in the growth of the HAp crystallites, negatively affecting bone development. This figure provides a clear explanation of how glucose affects bone, reducing the crystallite size, decreasing the intensity of the piezoelectric signals, and reducing the sensibility in regions of the body affected by a high local glucose accumulation.
Conclusions
Three types of samples were prepared: HAp, HAp-G, and C-G at different glucose concentrations and two compression modes, H and R. For HAp-G, the voltages were reduced when the glucose was increased, meaning that the chemical reaction between HAp and glucose produces a blocking in the growth of the crystal reducing the signal intensity and sensibility. For C-G, the situation is similar but on a smaller scale, producing small conformational changes in collagen because the collagen structure hinders some active groups. In all cases, the amplitudes are reduced, and the times increase when glucose is augmented. For HAp at 3% glucose, the intensities are drastically reduced; then, it is possible to use the piezoelectric technique to have an early and non-invasive diagnosis of diabetes.
Footnotes
Acknowledgments
The authors are in debt to Dra. Beatriz Millan Malo for her valuable contribution to the X-ray diffraction analysis and the determination of crystallite size. Additionally, they want to mention M. in S. Guillermo Vázquez for a partial analysis of the piezoelectric profiles.
Data availability
All data generated or analyzed during this study are included in this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author biographies
Susana Vargas got her PhD in chemistry in 2001 from Universidad Autonoma Metropolitana, Mexico. She works at the Universidad Nacional Autonoma de México as a technician. Her main research field is the synthesis and characterization of new ceramic and polymeric materials. She has more than 80 research papers published in international journals.
Rogelio G. Rodriguez-Vargas, Graduated in Biotechnology; he obtained his degree in 2023 from the Instituto Tecnologico de Estudios Superiores de Monterrey.
Francisco Quintanilla got his PhD in food in 2008 from Universidad Autonoma de Querétaro, México. He works at the Universidad del Valle de México. Her main research field is the synthesis and characterization of polymers foodstuffs and water processing. He is the author of some papers in international journals.
Rogelio Rodríguez got his PhD in physics in 1987 from Universidad Autonoma Metropolitana, Mexico. He is a senior research at the Universidad Nacional Autonoma de México. His main research fields are: synthesis and characterization of new ceramic and polymeric materials, coating, composites, catalytic materials polymers, composites and catalysts. He is the author of more than 190 research papers published in indexed international journals.


