Abstract
The current article was designed to assess the role of chitosan nanoparticles (CNPs) in the management of hepatic injury induced by the hepatocarcinogen 2-nitropropane (2-NP). Rats were divided into three groups. The first group served as a control, the second group was injected with 2-NP, while the third group was treated with CNPs 1 h before 2-NP injection every other day for 4 weeks. The 2-NP injection upregulated serum AST and ALT activities, as well as hepatic TNF- α, IL-6, and MDA levels and the expression of vascular endothelial growth factor (VEGF) and caspase-3, whereas GSH contents and SOD activity were decreased. Immunohistochemistry investigations revealed that the hepatic protein expression of collagen I, inducible nitric oxide synthetase, proliferating cell nuclear antigen, cluster of differentiation, and p53 were upregulated. hematoxylin and eosin (H&E) and Masson’s trichrome stains supported the previous parameters, and CNPs ameliorated most of the previous biochemical parameters. CNPs achieved promising results in the limitation of 2-NP hepatotoxicity.
Introduction
Increasing evidence suggests that multiple imperative functions of the liver, include food metabolism; clearing the body from poisonous substances, and regulating blood clotting factors. 1 The statistics of patients suffering from liver diseases is rapidly growing, presenting a major problem to healthcare infrastructures from both medical and economic perspectives. Liver diseases may convert into liver cancer, which is the fifth most common cause of death worldwide. 2 Exposure to chemicals is one of the imperative hazardous factors that lead to the development of liver injury and hence hepatocellular carcinoma HCC. 3 Wide evidence suggests that liver regeneration is a complex, tightly controlled process involving many inflammatory cells, growth factors, and hormones. Muted responses to oxidative stress, reduced expression of growth regulatory genes, diminished rates of DNA repair, and telomere shortening may contribute to a reduction of the hepatic regeneration capacity. 4
2-Nitropropane (2-NP) is utilized worldwide in animal models as a liver-injury inducer in addition to its use for different industrial purposes, such as the production of phentermine, chlorphentermine, and teclozan. 2-NP (an extraction solvent) is considered by WHO as food additives and fractionating solvent in the production of fats and oils besides being an intermediate in the synthesis of some pharmaceuticals, dyes, insecticides, and textile chemicals. 5 Moreover, it is an ingredient of tobacco smoke. 6 The hepatotoxicity of 2-NP is attributed to the propagation of reactive oxygen species (ROS), reactive nitrogen species (RNS), lipid peroxidation, disturbance in liver function ALT, AST, and GGT, decrease in superoxide dismutase and catalase.7,8 The oxidative stress leads to chronic inflammation, cellular damage, and necrosis, and hence, hepatocellular carcinoma. 9
Neutraceuticals are widely used nowadays due to their reduced toxicity. Among these is chitosan, which is a chitin extract from shrimps. Chitosan is valuable in diverse fields such as biomedicine, 10 pharmaceuticals, 11 and metal chelation.12,13 It can augment immunity, and it is applied as an anticancer medication. 14 Chitosan nanoparticles (10 mg/kg, p.o.) showed powerful pharmacological properties in the amelioration of an insulin-resistant status enhanced by a high-fat, high-fructose diet in rats. 15
The higher affinity of CNPs for negatively charged biological membranes is attributed to the CNPs positive charge. 16 Nanotechnology is urgent for the treatment of different diseases and tumors without any harmful effect on the normal cells 17 to avoid the adverse properties of chemotherapies. Nanoparticles with size 1–100 nm are so attractive in biomedicine due to their minute size and steadiness.16,18
During normal physiological conditions, angiogenesis is regulated by the balance between angiogenic enhancers and suppressors. VEGF is a vital key controller of tumor angiogenesis. 19 Proliferating cell nuclear antigen (PCNA) is a cell cycle indicator protein; it acts as a downhill secure throughout the DNA replication20,21 and in post-replication processes. A cluster of differentiation (CD)68 is an indicator of macrophages that adjust inflammatory responses and adaptive Th1 immunity to encourage angiogenesis as well as tissue pair. 22 There is an evidence that CNPs-mediated inhibition of angiogenesis was linked to impaired level of VEGF receptor 2. 23
The present study aims to assess the potential effect of CNPs to antagonize liver injury induced via 2-NP. This was achieved by monitoring liver function, aspartate aminotransferase, alanine aminotransferase activities, and inflammatory markers (TNF- α and IL-6) as well as oxidant/antioxidant biomarkers as hepatic malondialdehyde (MDA), reduced glutathione (GSH) contents, superoxide dismutase (SOD) activity. The protein expression of VEGF, collagen I, inducible nitric oxide synthetase (iNOS), PCNA, CD68, p53, and caspase-3 were assessed. Also, using hematoxylin and eosin (H&E) stain as well as Masson’s trichrome stains confirmed these results.
Materials and methods
Chemicals
Chitosan was purchased from iHerb (product code: NTB-21260), whereas 2-NP was purchased from Sigma-Aldrich and Merck Co. (St. Louis, MO, USA).
Preparation of chitosan nanoparticles
The chitosan nanoparticles (CNPs) were prepared by the ionic gelation method of Calvo et al. 24
Characterization studies
The mean hydrodynamic diameter potential of CNPs was 43.58 ± 1.5 nm with +9 to +12 mV. zeta potential. According to TEM, it had an average size of 40 ± 12 nm, with no agglomeration of other particles. The sample code is NS0115. The particle size is 40 nm, with a fine nanostructure of a few nanometers’ particles. The concentration was 30 mg/ml equivalent to 3% w/v. Water was used as the dispersion medium. CNPs possess a narrow particle size distribution without agglomeration. Figure 1 shows CNPs imaged by TEM.

TEM image of CNPs.
Animals and doses
Eighteen Wistar albino rats (n = 6, male, 190–210 g) were held at standard conditions for 1 week before the beginning of the experiment. The animals were supplied with a chew diet and water ad libitum. Rats were left about 1 week before the experiment for accumulation. The experimental protocol was permitted by the Animal Care and Use Committee of the College of Pharmacy, King Saud University (no.: KSU-SE-20-16). The rats were divided into three groups of six rats each. Group, I was administered olive oil and designated as a control. Group II rats have injected IP with 1 ml of 200 mg/kg 2-NP 9 dissolved in olive oil every other day for 4 weeks. Group III rats were treated daily with CNPs orally at 500 mg/kg every other day 1 h before 2-NP injection for 4 weeks.16,25 Four weeks later, the rats were subjected to CO2, then sacrificed via decapitation; blood samples were collected for serum separation. The livers were removed, and a 20% w/v homogenate was set in phosphate-buffered saline. Centrifugation of the homogenate took place, followed by a supernatant collection for measuring the biochemical parameters. The liver sections were fixed in 10% formalin for histological and immunohistochemistry examinations. Some parts of liver were saved under liquid nitrogen to carry out western blot analysis.
Evaluation of markers of liver function
Serum alanine aminotransferase (ALT) and aspartate aminotransferase (AST) were assessed using reagent kits (Randox, County Antrim, United Kingdom) according to the manufacturer’s instructions.
Assessment of oxidants /antioxidants parameters
The degree of lipid peroxidation (malondialdehyde) in the liver tissues was determined according to the method of Uchiyama and Midori. 26 The content of GSH was determined by the method of Ellman. 27 The activity of superoxide dismutase (SOD) was measured according to the method of Marklund and Marklund. 28
Estimation of IL-6 and TNF-α
Interleukin-6 (IL-6) was measured using a highly sensitive rat enzyme-linked immunosorbent assay (ELISA) kit (IBL International GmbH, Germany) following the manufacturer’s instructions. Tumor necrosis factor- α (TNF- α) was measured using the ELISA assay kit following the instructions supplied by the manufacturer (DuoSet Kits, R&D Systems, USA).
Western blot analysis of VEGF and caspase-3 protein expression
The liver sections were homogenated in lysis buffer (20 mM HEPES, 2 mM MgCl2, 1 mM EDTA, 1 mM DTT, 0.1% SDS, 1 mM PMSF, pH 7.4) on ice. The supernatants were harvested by centrifugation at 12,000g at 4°C for 10 min. The protein concentrations were determined using a Bradford assay. The proteins (20 µg) were separated using 12% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS–PAGE) and transferred to PVDF membranes. The membranes were blocked for 1 h at room temperature with 5% nonfat milk in Tris-buffered saline Tween-20 (0.1% TBST) and incubated with the corresponding primary antibodies for VEGF and caspase-3. For protein quantification, the bands were scanned and quantified with b-actin as an internal control. The membranes were incubated with ECL reagent for 2–10 min and exposed to X-ray film. 29
Histological study
Dehydration and embedding of the liver samples in paraffin wax were carried out after their fixation in 10% formalin. Then, sections were stained with either H&E or Masson’s trichrome (MT) stain. All sections were inspected using a light microscope.
Immunohistochemical investigation
Some paraffin liver sections were immunostained to detect collagen I, iNOS, p53, PCNA, and CD-68 antibodies. Immunostaining was applied following the streptavidin-biotinylated horseradish peroxidase protocol (Novalink Max Polymer detection system, Novocastra). The method involved inhibition of endogenous peroxidase activity with 3% H2O2 in distilled water for 5 min and washing the sections in TBS (Sigma; T 5030-100 TAB; pH 7.6) twice for 5 min for each. The blocking of non-specific binding of the antibodies was carried by incubation with protein for 5 min (Novocastra). The sections were incubated with rabbit polyclonal anti-collagen-I, iNOS, PCNA, p53, and CD-68 (dilution 1:2000) for 60 min at room temperature. The sections were then washed in Tris buffer three times for 3 min for each and incubated with the secondary antibody for 30 min (Novocastra) and then washed in Tris buffer three times for 3 min each. Then peroxidase was distinguished with the working solution of diaminobenzidine (DAB) substrate (Novocastra) for 10 min. Finally, the sections were washed in distilled water for 10 min. The sections were counterstained with Mayer’s hematoxylin and then mounted in DPX (a mixture of styrene, a plasticizer, and xylene). For the negative control sections, the same procedure was followed but with the omission of the incubation with the primary antibodies. All of the sections were imaged using an Olympus DP72 light microscope. 30
Calculation and justification of the sample sizeselected for this study
The sample size was calculated by the resource equation method; E = Total number of animals - Total number of groups. In this method, a value E is calculated based on the decided sample size. The value of E should lie within 10 to 20 for optimum sample size. 31 In the present study E = 15; the number of animals = 6 which is permitted by the ethics committee of King Saud University.
After dissection livers were separated and divided into three portions; the first portion was homogenized in phosphate-buffered saline to form 20% homogenate, the second portion was kept in 10% formalin, whereas the third portion was kept under nitrogen for molecular analysis
Statistical analysis
The results were statistically analyzed by comparing the different groups’ values with those of controls. The data are presented as mean ± standard deviation (SD). Significant differences between the values were studied using a post hoc 12 ANOVA test.
Results
2-NP injection for 4 weeks induced a significant elevation in serum AST and ALT activities, as well as hepatic TNF- α, IL-6, and MDA levels (p ≤ 0.001); GSH level and SOD activity significantly decreased compared to control values. The concurrent administration of CNPs with 2-NP significantly decreased AST and ALT activities and MDA levels (p ≤ 0.001). It also significantly modulated TNF-α, IL-6, GSH levels, and SOD activity (p ≤ 0.001), matched with 2-NP injected group (Figures 2–4).

ALT and AST activities of control as well as 2-NP and CNP-administered groups.

GSH level, SOD activity, hepatic MDA, IL-6, and TNF-α levels in control as well as in 2-NP intoxicated and CNPs-administered groups.

IL-6 and TNF-α levels in control as well as in 2-NP intoxicated and CNP-administered groups.
Protein expression of VEGF and caspase-3 were significantly upregulated upon 2-NP injection, while CNPs significantly downregulated their levels compared to the 2-NP injected group (p ≤ 0.05) and (p ≤ 0.01), respectively (Figure 5).

Protein expression of VEGF and caspase-3 in control as well as in 2-NP intoxicated and CNP-administered groups.
Histopathologically, the liver sections stained with H&E from rats that had received 2-NP revealed a mild inflammation in the lobule, while the section exposed to CNPs showed a marked amelioration of hepatic inflammation and restored normal architecture of the liver. Liver sections from the rats that received 2-NP showed a slight increase of collagen fiber in the portal tract, while those exposed to CNPs showed a normal amount of collagen (Figure 6). The immunohistochemistry study revealed that collagen I, iNOS, PCNA, P53, and CD-68 showed strong immune reactivity in the 2-NP injected group, as compared to the control group. However, the rats treated with CNPs presented with moderate immune reactivity (Figures 7 and 8).
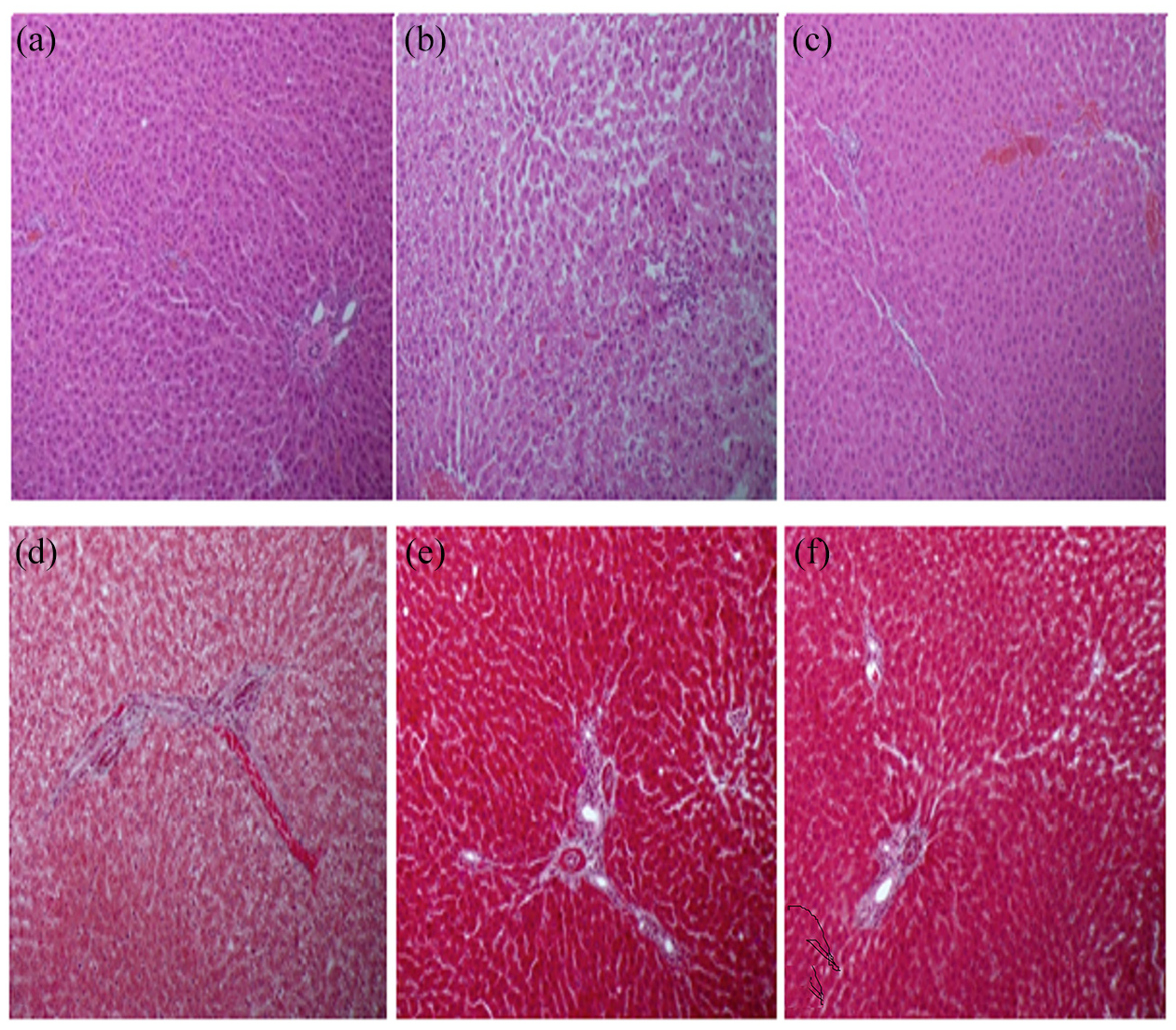
(a–c) Light photomicrograph of rat liver stained with hematoxylin and eosin (H&E): (a) section from normal control rat showing normal hepatic cells and sinusoids, (b) section from rat that received 2-NP showing mild inflammation in the lobule, (c) section from rat exposed to CNPs showing marked improvement of hepatic inflammation and normal histology, (d–f) light photomicrograph of Masson’s trichrome (d) section from normal control rat showing unremarkable increase of collagen fibers in the portal tract, (e) section from rat that received 2-NP showing slight increase of collagen fiber in the portal tract, and (f) section from rat exposed to CNPs showing a normal amount of collagen.
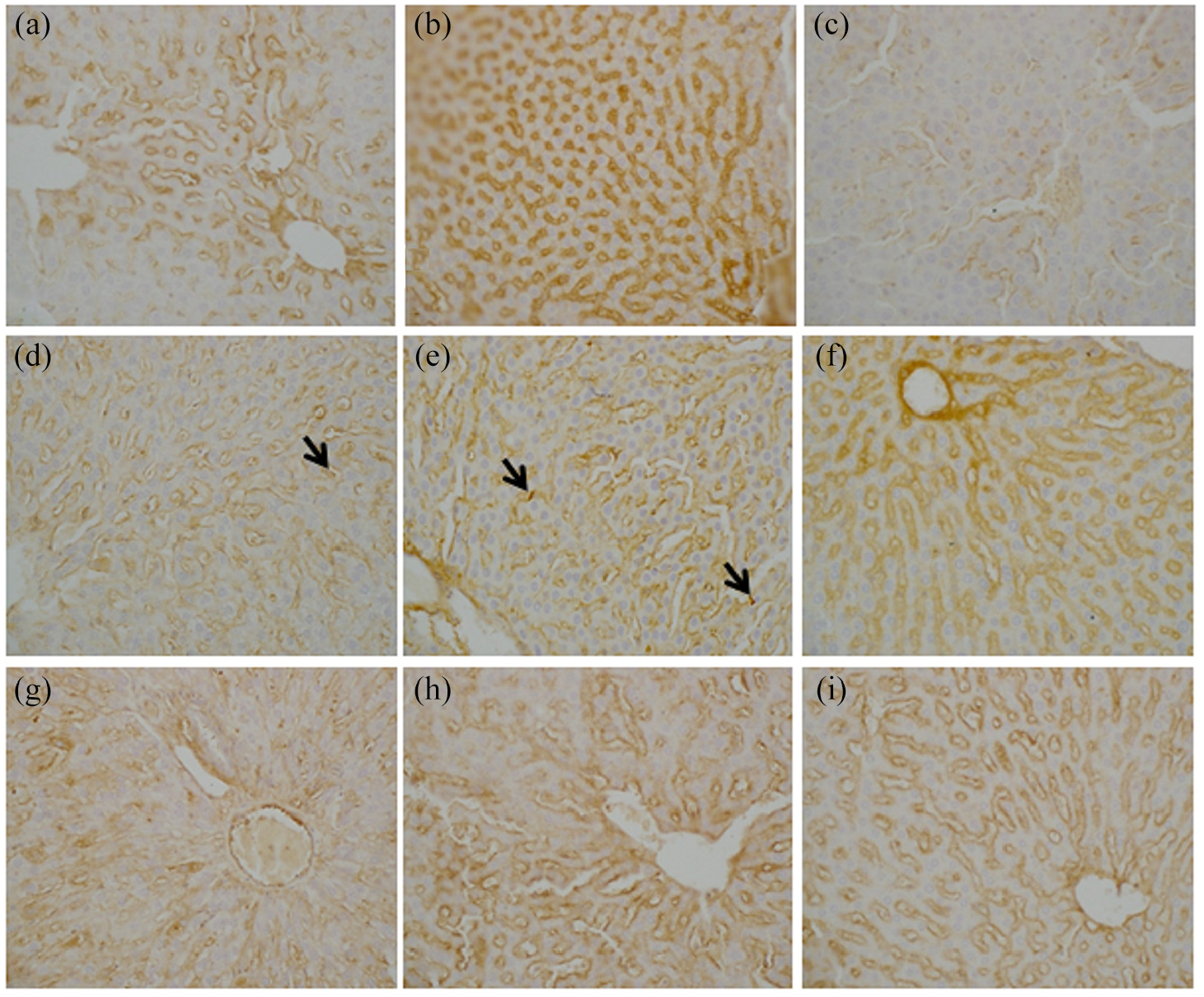
(a–c) Anti-iNOS immune stained sections of liver from rat: (a) section from control rat showing immune staining in the endothelium of the hepatic sinusoids and blood vessels while hepatocytes are negative in staining, (b) section from rat received 2-NP showing immuno-stained around hepatic sinusoids, and (c) section from rat exposed to CNPs showing weak staining on hepatic sinusoids, (d–f) anti-CD68 immune stained liver sections of rats (d) section from control rat showing minimal immune staining for CD68, characteristic for Kupffer cells (arrows) in the wall of the hepatic sinusoids, (e) section from rat received 2-NP showing immuno- stained (arrows) around hepatic sinusoids and stains normally Kupffer cells, (f) section from rat exposed to CNPs showing normal positive staining in some Kupffer cells, (g–i) anti-collagen I immune stained liver sections of rats (g) section from normal control rat showing minimal immune stain around the central vein and portal tracts, (h) section from rat exposed to 2-NP showing collagen stains with positivity around portal tracts and central vein, and (i) section from rat exposed to CNPs showing collagen stains with slight positivity around portal tracts and central vein.
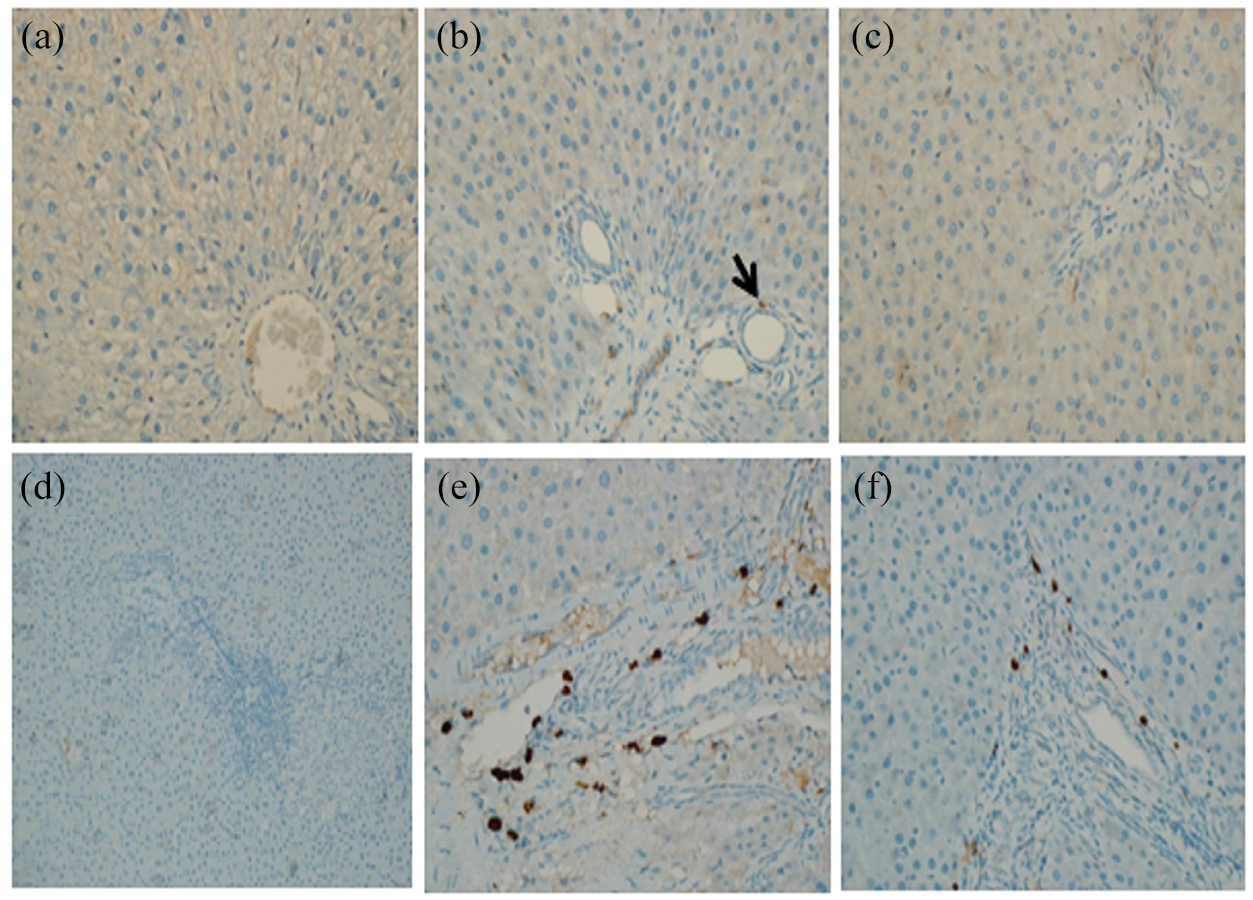
(a–c) Anti-PCNA immunostained liver sections of rats: (a) section from normal control rat showing a minimal positive nuclear staining, (b) section from rat received 2-NP showing the positivity of PCNA staining in the nucleus and indicating an increase in the proliferation rate, (c) section from rat exposed to CNPs showing mild positive nuclear staining, (d–f) anti-P53 immunostained liver sections of rats, (d) section from normal control rat showing weak immune staining in the inflammatory cells in the portal tract, (e) section received 2-NP showing positive staining in the inflammatory cells in the portal tracts, and (f) section from rat that received CNPs showing focal areas of moderate intense nuclear staining at the junction between portal tract and hepatocyte.
Discussion
Drug delivery systems have attracted increasing attention worldwide in the last decades in the medicinal and pharmaceuticals industry. The current treatment modality for 2-NP-mediated liver injury is extensively investigated. The protective effect of resveratrol, 32 binaphthyl diselenide, 33 or moringa oleifera 34 as a treatment for 2-NP induced hepatotoxicity was documented. CNPs are well-recognized delivery systems broadly used as polymeric carriers in the nanomedicine field. CNPs are carbohydrates of natural origin that are biocompatible and biodegradable polycationic polymers and have penetration properties. Liver disease is considered a remarkable health problem because of the obstacle of current therapies, due to the high risk of recurrence and the lack of drug selectivity in addition to other serious side effects. Therefore, there is an urgent need for new liver-targeting therapeutic strategies. Nanomedicine uses nanoscale carriers as delivery systems to target and deliver drugs and/or diagnostic agents to specific organs. CNPs can be successfully used in the preparation of nanoparticles that, for their peculiar surface-properties, can specifically interact with a liver tumor by passive and active targeting. The previous article highlights the use of CNPs for the therapy and limitation of HCC induced by 2-NP. 35
The present study aims to elucidate the hepatoprotective effect of chitosan nanoparticles versus 2-NP-induced liver injury through measuring liver function, oxidative stress, angiogenic, apoptotic, and proliferating cell antigen biomarkers
2-NP induces abnormal DNA replication and is considered a hepatotoxicant in animals. 36 Nitrite and acetone produced by the action microsomal enzymes on 2-NP represent the main culprit behind its hepatotoxicity and hence the propagation of ROS and RNS. 8 The oxidative stress triggers the activation of inflammatory mediators (TNF-α, IL-6, COX-2, and iNOS), leading to chronic inflammation, cellular damage, and necrosis.37,38 In the present work, the mortality rate was 10%; moreover, injection with 2-NP induced liver injury and caused an elevation in serum ALT and AST together with the alteration in the oxidative status manifested by an increase in lipid peroxidation and the expression of iNOS with a contaminant decline in SOD activity and GSH level. This is in agreement with the study which reported that 2-NP induced oxidative stress and ROS affecting liver cell membrane integrity, leading to the leakage of liver enzymes and releasing oxidative stress and the nitrosative stress biomarkers. 8 Previous studies reported that CNPs is a promising candidate for drug delivery in liver diseases. The studies shed light on the therapeutic potential of CNPs and their power to mitigate hepatotoxicity caused by carbon tetrachloride, diethylnitrosamine, and acetaminophen. They concluded that CNPs could improve liver function parameters (ALT, AST & ALP) and oxidative stress biomarkers including glutathione peroxidase (GPx), SOD, MDA, catalase, alpha-fetoprotein, and carcinoembryonic antigen and histological picture of the liver in rats treated with carbon tetrachloride or diethylnitrosamine or acetaminophen.39,40
Herein, the level of inflammatory markers (TNF- α and IL-6) were overexpressed as well. While CNPs administration downregulates such levels. Mosa et al. 41 reported that the CNPs treatment improved the inflammatory responses (IL6 and TNF-α) in rats’ gastric tissue after hydroxyapatite nanoparticles’ oral uptake.
In the present study, histopathological investigations displayed a mild inflammation in the lobule and a slight increase in collagen fiber in the portal tract after 2-NP intoxication. It was documented that histological examination of liver sections of haloperidol- treated rats showed that CNPs treatment decreased that destructive progress. 42
Accordingly, Borges et al. 9 reported that AST, ALT, and lipid peroxides were elevated while superoxide dismutase, catalase, and ascorbic acid levels were decreased in rats that received a single dose of 2-NP. Herein, concurrent administration of CNPs limited 2-NP-induced hepatic injury and collagen deposition and also suppressed the levels of AST and ALT, as well as oxidative stress and TNF- α and IL-6 levels. CNPs administration resulted in a marked amelioration of hepatic inflammation and normalized collagen content. It was reported that CNPs have a direct antioxidant effect by lowering oxidative stress and inhibiting MDA elevation and glutathione depletion as well as significantly suppressing iNOS and NF-κB, the initiator of the inflammatory cascade.43,44
VEGF is a strong enhancer of vascular permeability and angiogenesis, which is essential for tumor development and progression. 45 The expression of cytokines activate VEGF formation in the immune system and inflammatory cells, which has a vital role in the propagation of multi-organ system dysfunction in discriminating injury. 46 PCNA is involved in the post-replication repair. 47 Cancer is often associated with high levels of PCNA. 48 A positive correlation was observed between VEGF2 and PCNA immunoreaction concerning the duration of 2-NP injection. 49 CNPs play an important role in the reduction of tumor growth and represents a new class of anti-cancer drug. CNPs administration to the experimentally Eldrich carcinoma-bearing mice, reduced tumor size, decreased MDA level, and increased GSH level. Moreover, it significantly decreased inflammatory and angiogenic markers (TNF-α, PDGF, and VEGF). 4
In the current study, 2-NP induced overexpression of the apoptotic factors (caspase-3 &P53), angiogenic factor (VEGF), collagen I, cell cycle marker (PCNA), inflammatory marker (CD-68), Meanwhile, treatment with CNPs downregulated the previous protein expressions. It was revealed that CNPs have pro-apoptotic powers against metastatic melanoma RPMI7951 cells and cultured Schwann cells(hydrogen peroxide-induced apoptosis model) since it inhibited specific caspases.50,51
The angiogenic factor VEGF is one of the key regulators of tumor angiogenesis. 19
However, CD68 is an essential prototypic component of inflammation that drives neoplastic progression.52,53 Previous studies revealed that the apoptotic marker and cell cycle arrester p53 expression was upregulated upon 2-NP administration. 54
Proliferating cell nuclear antigen (PCNA), a cell cycle marker protein, functions as a sliding clamp during DNA replication. 21 PCNA is involved in protein-protein interactions 20 and the DNA damage tolerance pathway known as post-replication. Previous studies have indicated that clusters of differentiation of CD-68 are a marker for macrophage inflammatory responses and adaptive Th1 immunity promoting angiogenesis, as well as tissue remodeling and repair. 22 There is evidence that CNPs-mediated inhibition of angiogenesis is linked to impaired levels of vascular endothelial growth factor receptor 2. 23 Previous results documented that CNPs have specific cytotoxic effects on tumor cells, tumor growth inhibition, and necrosis blockage of VEGF-induced endothelial cell activation and apoptosis.23,31,55 CNPs nanoparticles revealed a moderate enhancement in glomerular damage and showed mild positive reactions to PCNA in the glomeruli and renal tubules in the cortical and medullary portions for the establishment of its antiproliferative effect. 56 Chitosan is valuable in diverse fields such as biomedicine, 10 pharmaceuticals, 11 , and metal chelation.12,13 It can augment immunity, and it is applied as an anticancer medication. 14
In the current work, CNPs successfully ameliorated 2-NP-induced hepatoxicity. It significantly down regulated ALT and AST activities (p ≤ 0.001). as well as MDA level, also it improved 2NP- induced histological changes in the liver. Comparable to the present study, Ibrahim et al. 33 reported that binaphthyl diselenide prevented 2-NP-induced alterations in ALT and AST activities (p ≤ 0.05). Moreover, binaphthyl diselenide prevented hepatic histology changes and lipid peroxidation. This demonstrated that CNPs is more effective against 2-NP intoxication. An important finding of this research is that CNPs decreased MDA levels indicating the antioxidant power of this compound. Probably, CNPs protective effect on 2-NP- induced hepatic injury appears to be correlated to its antioxidant potential. Likewise, Resveratrol ameliorated 2-NP- induced hepatic oxidation injury in rats, it decreased SOD level, reduced caspase- three activity, DNA damage by its antioxidant and scavenging properties. 32 Additionally, melatonin decreased VEGF expression in 2-NP- induced hepatocellular carcinoma in mice. 48 The extract of olive oil and fig attenuated 2-NP- induced alterations in mice submandibular gland by down-regulating IL-6 and TNF-α.57,58 Moreover, the extract of moringa leaf significantly lowered the increased MDA level that was induced by 2-NP in the liver of obese mice. 34 These effects may be linked to its antioxidant activity.
Chitosan is a novel strategy in treating liver disease and identifying its related signaling pathways via ameliorating Angiogenic, oxidative stress, and apoptotic markers.
Limitation mortality rate reached 1/10 of the total animal account so it is recommended to increase the number of animals in each group.
Moreover, 2-NP was observed to be irritant to animals so it is preferred to increase the percentage of olive oil used as a vehicle
Conclusion
CNPs are considered as promising candidates against 2-NP hepatotoxicity through the suppression of oxidative stress, inflammation, and the expressions of p53, VEGF, CD-68, and PCNA.
Footnotes
Author contributions
All authors participated in all parts of the work
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Authors extend their appreciation to the Deanship of Scientific Research, King Saud University for funding this work through the scientific group (Grant No. RG-1441-467).
Ethic
The experimental protocol was permitted by the Animal Care and Use Committee of the College of Pharmacy, King Saud University (no. KSU-SE-20-16).
