Abstract
The tensile strength and corrosion behavior of dissimilar welded joints are currently a subject of concern. In this work, gas metal arc welding (GMAW) and distinct welding parameters (welding current, arc voltage, and welding speed) were used to join 304 stainless steel (SUS304) and SS400 low carbon steel, and the ultimate tensile strength (UTS) of the dissimilar welded joints was investigated. A corrosion test was conducted by immersion in 3.5 wt.% sodium chloride (NaCl) solution for 7, 14, and 21 days. Based on tensile strength and Tafel testing, the welding parameters “Item 4” (welding current: 170 A, arc voltage: 20 V, welding speed: 40 cm/min) yielded good mechanical strength and low corrosion characteristics. The microstructure characterization showed that the area around the welded joints and SUS304 had more granular corrosion and corrosion tubercles with increasing immersion time. The chromium content gradually decreased. When exposed to the chloride environment, these welded joints easily underwent corrosion due to the loss of passivity. However, high-velocity oxygen-fuel (HVOF) spray used on the welded joints reduced the corrosion current density. Compared with the non-thermal spray sample (corrosion current density:7.49e − 05 A/cm2) while the corrosion current density (7.89e − 10 A/cm2) is five orders of magnitude lower. This spray effectively slowed down the corrosion rate of the welded joints and gave the structural objects good protection in the sodium chloride solution.
Keywords
Introduction
Welded joints of dissimilar metals are widely employed for different applications requiring certain special combinations of properties as well as for cost savings.1–3 Due to the differences in the chemical, physical, and mechanical properties of the base metals, welding dissimilar materials is usually more challenging than welding similar materials.4–6 Dissimilar welded joints between stainless steel and low carbon steel are widely used in the steam pipes of power plants, nuclear reactors, and petrochemical plants. Because stainless steel and low carbon steel have different mechanical properties and corrosion properties, they meet the requirements of the daily environment and reduce costs. Stainless steels, such as the austenitic type (e.g. SUS304), have better corrosion resistance and high tensile strength but are relatively expensive, 7 while low carbon steel is cheaper, readily available, and provides high impact strength.
Past research has shown that failures often occur at dissimilar welded joints.8, 9 The common reasons for failure include lack of tensile strength and corrosion. The mechanical properties of dissimilar welded joints have been extensively studied. However, most of the dissimilar welded joints suffer from the effects of acidic and salty (NaCl) environments, resulting in cracks and pore formation. Failure of the gas metal arc welding (GMAW) joints between carbon steel pipe and 304 stainless steel elbows has been studied by Ul-Hamid et al. 10 Chuaiphan et al. 11 have researched dissimilar welding between American Iron and Steel Industry (AISI) 304 stainless steel and AISI 1020 carbon steel plates using shielded metal arc welding (SMAW) and gas tungsten arc welding (GTAW). Welded joints produced using GTAW exhibited higher corrosion potential and toughness than those produced using SMAW. Wenyong Wu et al. 12 have suggested that the joint had the greatest corrosion current density due to galvanic corrosion between the base metals, ferritic stainless steel and carbon steel. Chen et al. 13 have proposed that differences in the microstructures and chemical composition led to varying degrees of the galvanic effect between the different regions of two A710 steel welded joints.
P. Poonnayoml et al. 14 have studied the butt joints of SS400 carbon steel and SUS430 stainless steel welded by the GMAW method and discussed the effect of welding current on the performance of the welded joint. D. Purnama1 et al. 15 have proposed for dissimilar metal welding to use 100% CO2 as the shielding gas which, compared with 90%Ar + 10%CO2 mixed gas, resulted in more inclusions. Y. K. Khdir et al. 16 have studied the effect of different heat inputs on the mechanical properties of dissimilar metal arc welding joints of austenitic stainless steel (AISI 304) and low carbon steel (CS). K. V. et al. 17 have proposed to analyze the corrosion rate of stainless steel 304, stainless steel 316, and 304 and 316 welded joints using an electrochemical Tafel polarization experiment. The results showed that the corrosion rate of the welded joints is the highest among the candidate materials, while the corrosion rate of stainless steel 316 is the lowest B. Seo et al. 18 have studied the corrosion properties of dissimilar FSW joints of 6061 aluminum alloy and HT590 low-carbon alloy steel, and found that galvanic corrosion affects the corrosion properties of the joints. Mahto et al. 19 have studied the corrosion properties of dissimilar metal FSW joints of AA6061-T6 aluminum alloy and AISI304 stainless steel. They proposed that the corrosion rate increased with decreasing grain size. C. Wang et al. 20 have studied the microstructure evolution and corrosion behavior of dissimilar 304/430 stainless steel welded joints. The results showed that during the former stage, the anti-corrosive deterioration of the weld is due to the formation of larger portions of martensite and phase boundaries. During the latter stage, increased Cr content and decreased fraction of the phase boundary are responsible for the improvement of the anti-corrosive property of the welded joint. V. Bhanu et al. 21 have used an unconventional welding process—laser beam welding. The results from tests conducted on the dissimilar welded joint of P91 and Incoloy 800HT can be used to support data collection for the behavior of dissimilar joining of martensitic steel and austenitic materials. D. Bokov et al. 22 have studied the effects of FSW tool pin angle during FSW of AA1100 aluminum alloy and St-14 steel. Y. Sun et al. 23 have studied the mechanical properties evaluation crack and propagation behavior in dissimilar metal welded joints of 304L austenitic stainless steel and SA508 low-alloy steel.
Based on the state of the welded joint corrosion, the galvanic effect changes the material strength of the welded joints. Consequently, the investigation of corrosion damage and potential precautions can be beneficial to the improvement of the structural strength of dissimilar welded joints.
In this work, the precipitation of microstructural corrosion products was tested using the thermal spray method to improve the corrosion resistance of welded joints. High-velocity oxygen-fuel (HVOF) spray technology was used on the surface of the welded joints to reduce the porosity and obtain a lower corrosion current density. Compared with the corrosion current density of the original welded joints, as the thickness of the HVOF spray layer increases, the corrosion current density becomes smaller. The corrosion rate then becomes slower, which can effectively slow down the corrosion of the welded joints.
Materials and experimental methods
Materials
The experimental procedure is shown in Figure 1. SS400 low carbon steel and SUS304 stainless steel were used as base materials. The dimensions of the sample were 300 mm

The experimental flowchart.

Schematic of the dissimilar welded joints.
Chemical composition (wt.%) and mechanical properties of low carbon steel (SS400).

Chemical composition (wt.%) and mechanical properties of stainless steel (SUS304).

Chemical composition (wt.%) and mechanical properties of the filler wire electrode (GMX309L).
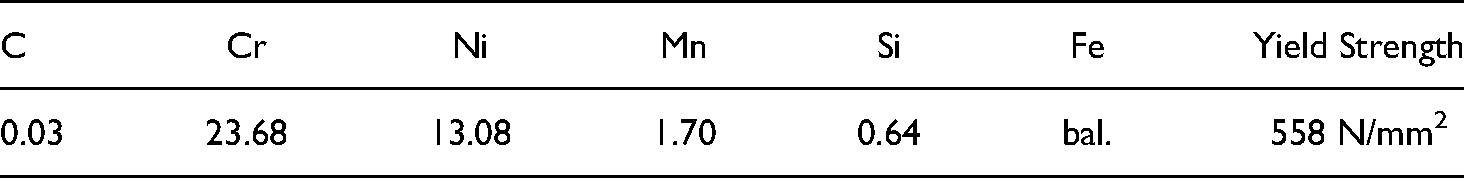
Welding Parameters and Input Heat Process
All welding experiments used argon (Ar) as the shielding gas, supplied at a constant rate of 10 l/m throughout the experiments. The welding of dissimilar joints was done by robotic arms (OTC DAIHEN, FD-V6), as shown in Figure 3. 2. To calculate the required heat input for arc welding procedures, the following formula can be used:

Welding equipment (OTC, FD-V6).
Ten samples were tested to explore the changes in the mechanical properties and corrosion characteristics after dissimilar joints were welded using different welding parameters (shown in Table 4).
Welding parameters and heat.

Tensile strength testing
Tensile strength testing is one of the most common and convenient material testing methods. When a material is subjected to an increasing axial load, the basic mechanical properties related to the relevant material can be obtained, including the ultimate tensile strength (UTS). The parameters of the tensile test pieces were based on the ASTM E8/E8M specification, as shown in Figure 4.

Tensile specimen dimensions (Unit: mm).
Tafel corrosion test
To evaluate the corrosion current density and corrosion potential of dissimilar welded joints, the sample test area was restricted to 0.237 cm2. The electrochemical corrosion test was performed in a three-electrode cell using a potentiostat (WonATech, ZIVE SP1). The working electrode (WE) was the welded joint sample, the reference electrode (RE) was a calomel electrode, and the counter electrode (CE) was platinum. The entire measuring system is shown in Figure 5. The Tafel corrosion tests were performed in 3.5 wt.% NaCl solution using the procedures stated in the ASTM G5-94 and G61-68 standards.

Tafel measuring equipment.
Microstructure corrosion products
An environmental scanning electron microscope (ESEM, FEI Quanta 200, FELMI-ZFE) was used to investigate the surface morphology of the dissimilar welded joints. The ESEM analyzes the X-rays emitted by a material when it is probed using electromagnetic radiation and is commonly used to analyze chemical compositions.
Thermal spray technology
High-velocity oxygen-fuel (HVOF) spray was used on the welded joints. Our method used a combination of oxygen and various fuel gases, including hydrogen, propane, propylene, and kerosene. The 2.3 mm diameter thermal spray wires were pure aluminum (Sulzer Metco Inc.). The thicknesses of the sprayed layers were 150 and 450 μm.
Results and discussion
Ultimate tensile strength and input heat
The UTS test and input heat results of the dissimilar welded joints were produced by the different welding parameters given in Table 5. As can be seen from the table, the welding parameter sets with 8.4 and 8.5 for the current/voltage ratio showed the highest average UTS values. The increasing heat input of sets 1–4 resulted in increasing tensile strength, but for set 5 the tensile strength decreased when the input heat was too high. Five of the experiments (BW-1–BW-5) were performed under constant welding current and welding speed; only the arc voltage changed during welding. The fourth set of welding parameters (BW-4) had the best average UTS (331.294 MPa). Following a similar pattern to the first 5 sets, the increasing heat input of sets 6–8 led to increased UTS, but the UTS of sets 9 and 10 decreased when the input heat was too high. In addition, in experiments BW-6–BW-10, arc voltage and welding speed were constant, with only the current changing while welding. The eighth set of welding parameters (BW-8) had the best average UTS (331.541 MPa) in this group.
Ultimate tensile strength and input heat.

These test results showed that the efficiency of the joints and tensile strength increased with increasing heat input up to a threshold, beyond which excess heat input led to reduced efficiency. 16
Corrosion analysis
The same ten sets of welding parameters were applied to the electrochemical corrosion experiments on the dissimilar welded joints. The samples were kept in 3.5 wt.% NaCl solution for 7, 14, and 21 days to observe the corrosion current characteristics.
Within 7 days and among samples BW-1–BW-10, BW-1 had the largest corrosion current (1.11e − 5 A/cm2). It also had the fastest corrosion rate. The Tafel test results of BW-1–BW-5 after 7 days are shown in Table 6 and Figure 6. Within samples BW-6–BW-10, BW-6 had the largest corrosion current (7.87e − 5 A/cm2). It also had the fastest corrosion rate. The Tafel test results of BW-6–BW-10 after 7 days are shown in Table 7 and Figure 7.
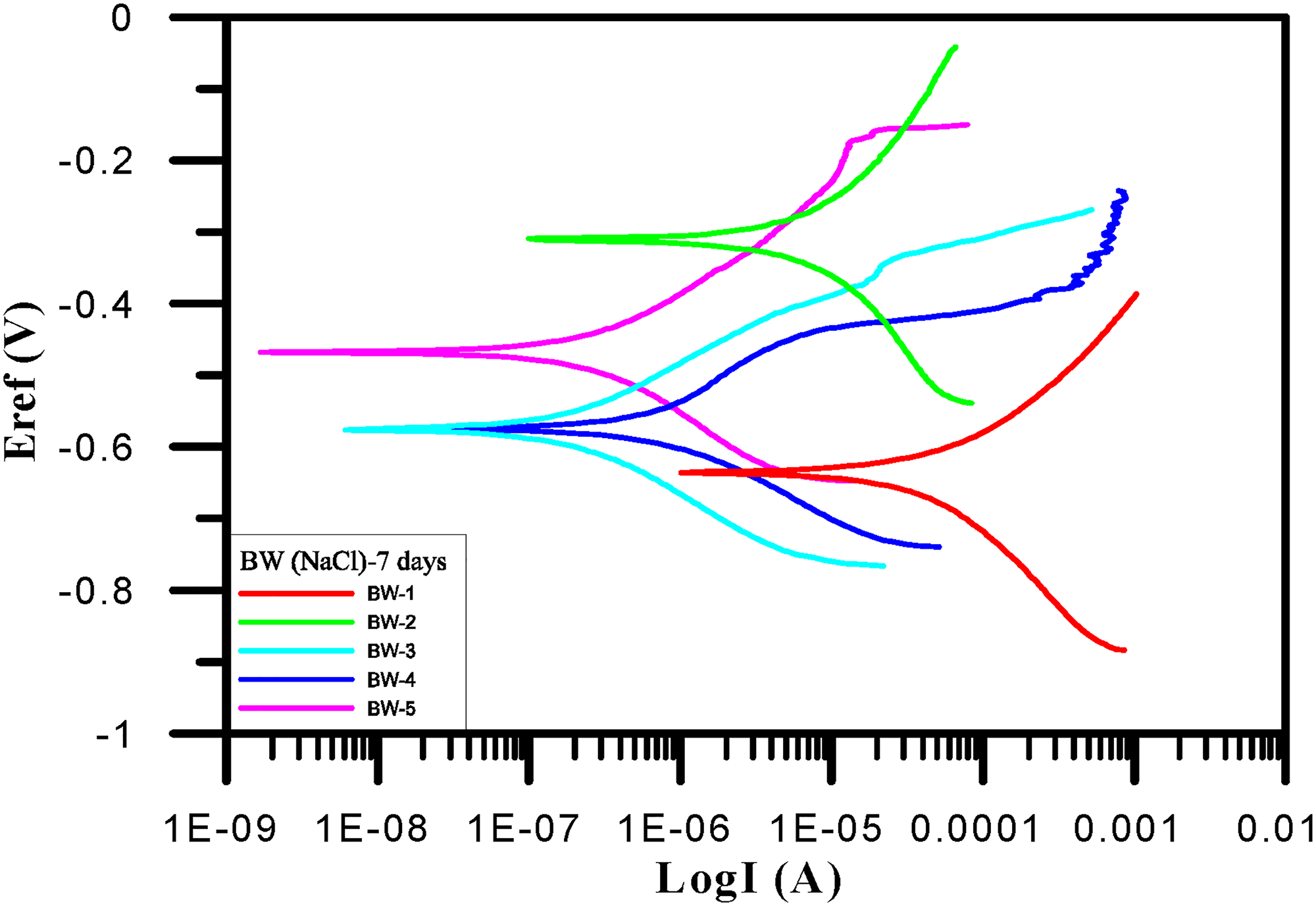
Tafel polarization curves (BW-1∼BW-5) for 7 days.

Tafel polarization curves (BW-6∼BW-10) for 7 days.
BW-1∼BW-5 corrosion potential and corrosion current density for 7 days.

BW-6∼BW-10 corrosion potential and corrosion current density for 7 days.

Within 14 days and among samples BW-1∼BW-10, BW-2 had the largest corrosion current (8.68e − 5 A/cm2). It also had the fastest corrosion rate. The Tafel test results of BW-1–BW-5 after 14 days are shown in Table 8 and Figure 8. Within samples BW-6–BW-10, BW-6 had the largest corrosion current (1.65e − 6 A/cm2). It also had the fastest corrosion rate. The Tafel test results of BW-6–BW-10 after 14 days are shown in Table 9 and Figure 9.

Tafel polarization curves (BW-1∼BW-5) for 14 days.

Tafel polarization curves (BW-6∼BW-10) for 14 days.
BW-1∼BW-5 corrosion potential and corrosion current density for 14 days.
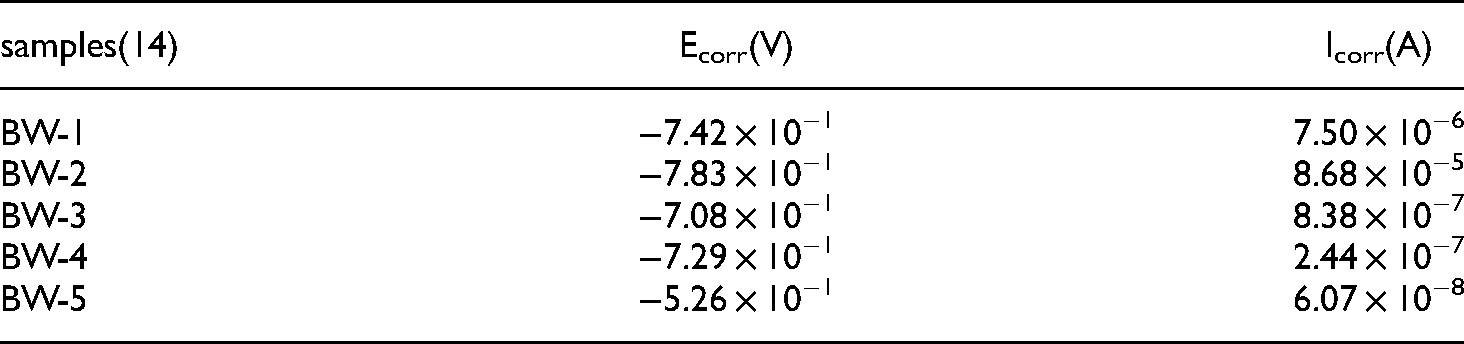
BW-6∼BW-10 corrosion potential and corrosion current density for 14 days.

Within 21 days and among samples BW-1–BW-10, BW-2 had the largest corrosion current (7.49e − 5 A/cm2). It also had the fastest corrosion rate. The Tafel test results of BW-1–BW-5 after 21 days are shown in Table 10 and Figure 10. Within samples BW-6–BW-10, BW-6 had the largest corrosion current (4.00e − 7 A/cm2). It also had the fastest corrosion rate. The Tafel test results of BW-6–BW-10 after 21 days are shown in Table 11 and Figure 11.
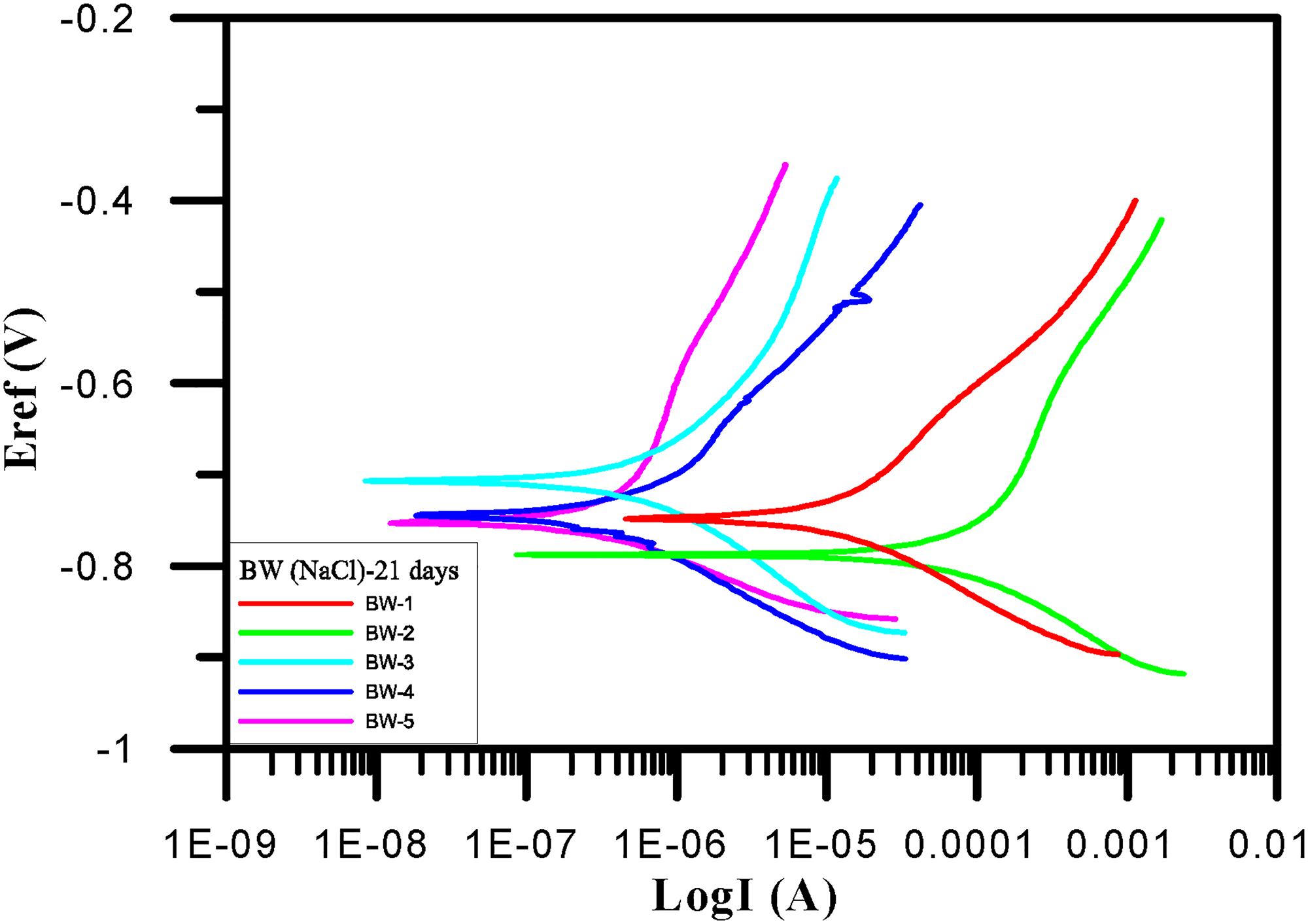
Tafel polarization curves (BW-1∼BW-5) for 21 days.
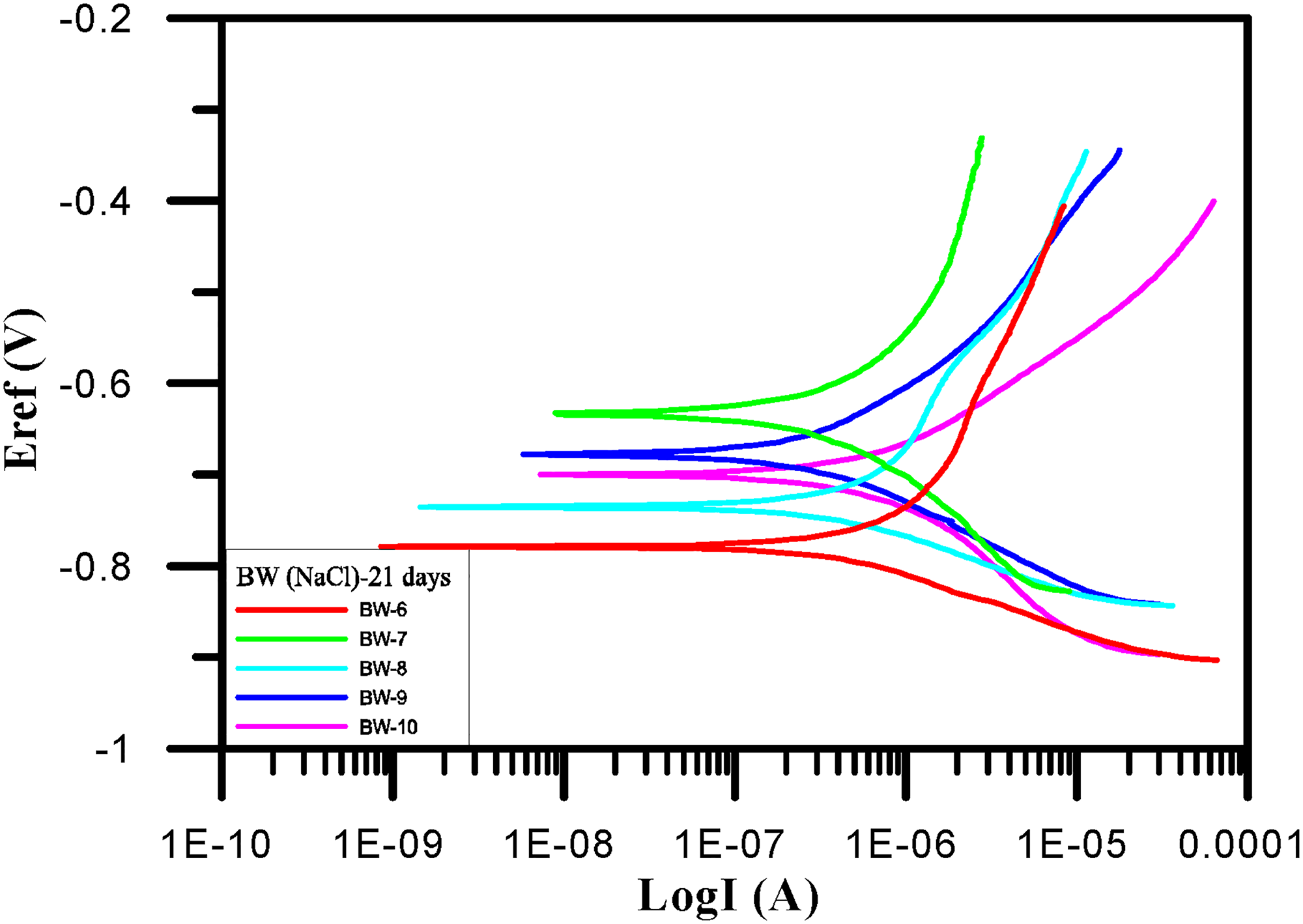
Tafel polarization curves (BW-6∼BW-10) for 21 days.
BW-1∼BW-5 corrosion potential and corrosion current density for 21 days.

BW-6∼BW-10 corrosion potential and corrosion current density for 21 days.

After 21 days of the Tafel test, the BW-2 sample had the largest corrosion current (7.49e − 5 A/cm2) among samples BW-1–BW-5. Sample BW-4 had the smallest corrosion current (2.82e − 7 A/cm2). Among the BW-6∼BW-10 samples, BW-6 had the largest corrosion current (4.00e − 7 A/cm2). Sample BW-9 had the smallest corrosion current (1.72e − 7 A/cm2). The data are shown in Table 12 and Figure 12.

Corrosion current density of dissimilar welded joints for 7, 14, and 21 days.
Corrosion current density for 7, 14 and 21 days.

Microstructure and corrosion product compositions analysis
A scanning electron microscopy (SEM) image of the boundary of a dissimilar welded joint and SUS304 after 7 days immersed in 3.5 wt.% NaCl solution is shown in Figure 13(a). Figure 13(b) shows the joint after 14 days of immersion, and Figure 13(c) shows it following 21 days of immersion.
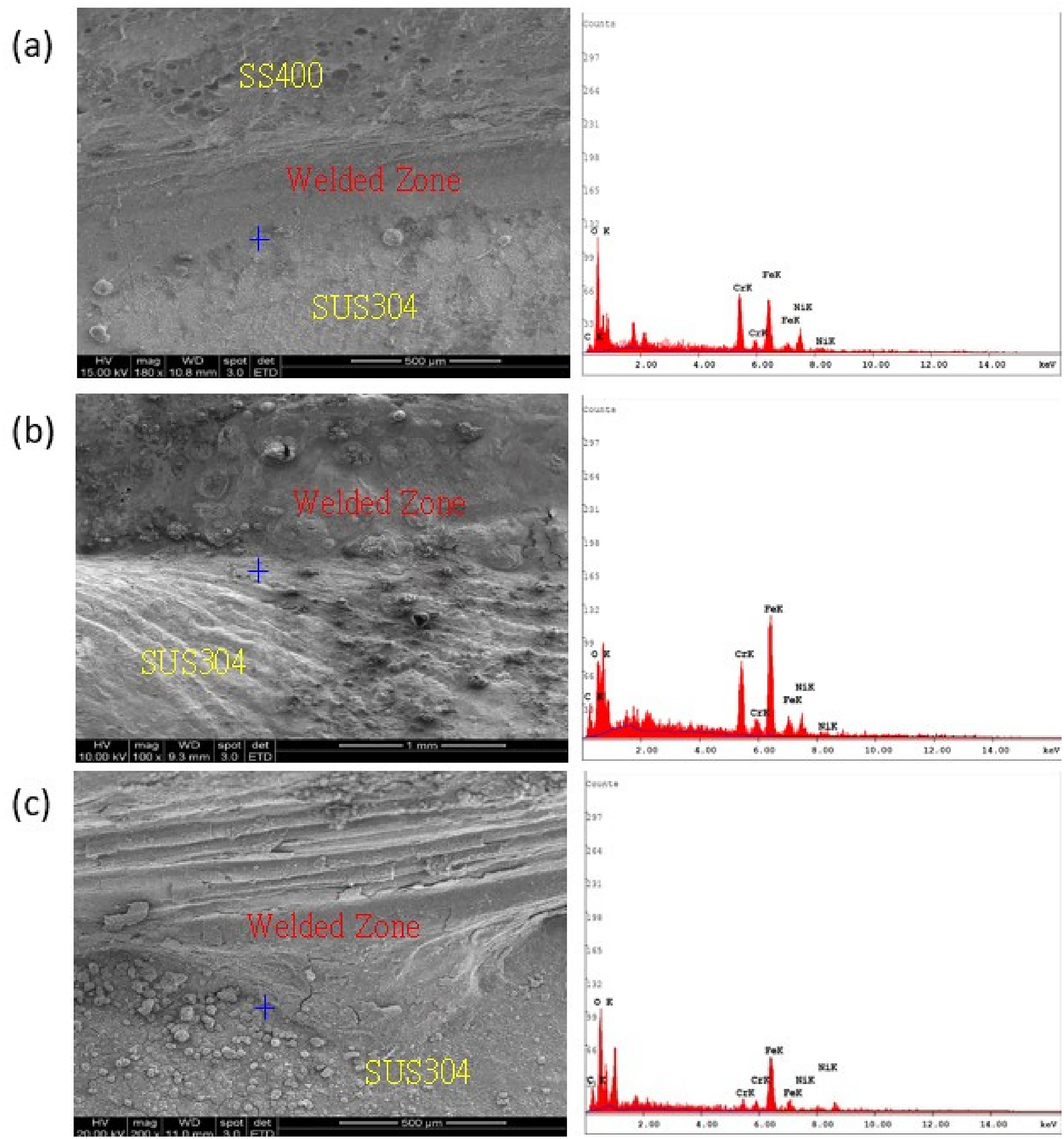
SEM images and Energy-dispersive X-ray spectroscopy (EDS) elemental analysis for (a) 7 days, (b) 14 days, and (c) 21 days.
In Figure 13, there were many deposits visible on the surface of the metals near the junction of the dissimilar welded joint and the SUS304. These corrosion products were in the form of granules and tubercles, as shown in Figure 14.

SEM image of the corrosion products.
Table 13 presents the elemental chemical composition of the boundary of the welded joints and SUS304 after 7, 14, and 21 days immersed 3.5 wt.% NaCl solution. The main components of the grain boundary layer of the welded joints and SUS304 were chromium (Cr), as well as chromium depletion and corrosion carbides.
Elemental composition of the grain layer of the welded joints and SUS304.

As the immersion time increases, the chromium content gradually decreased. When the chromium content is less than 12%, the original protection and passivation of the boundary layer of the welded joint and SUS304 were lost This area was thus prone to corrosion, and the corrosion rate was faster than in any other area.
Thermal spray analysis
After the thermal spray procedure, the electrochemical corrosion experiments were performed. 1. There are five test samples in the thermal spraying experiment. The original BW-2 sample has no spray coating, and the others are sprayed with 150 μm and 450 μm, respectively. For the welding parameters, the welding current is 170A, the arc voltage is 18V, and the welding speed is 40 cm/min. The thickness of the sprayed aluminum layer for Samples 1 and 2 is 150 μm. In contrast, the thickness of the sprayed aluminum layer for Samples 3 and 4 is 450 μm. After the Tafel electrochemical corrosion test, the corrosion potential and corrosion current density were measured, as shown in Table 14. The unsprayed BW-2 sample had the largest corrosion current (7.49e − 5 A/cm2). The corrosion current densities of the 150 μm thick samples are 2.91e − 8 A/cm2 and 4.75e − 8 A/cm2. The corrosion current densities of the 450 μm thick samples are 7.79e − 10 A/cm2 and 7.89e − 10 A/cm2.
Thermal spray corrosion potential and corrosion current density.

The anti-corrosion protection of the welded joints is strengthened by the thermal spray coating technology. With the increased coating thickness, the corrosion current density is minimized and the total resistance is maximized, both of which improve the anti-corrosion ability of the welded joints.
Conclusions
The main results of the study of GMAW dissimilar welded joints of SUS304 and SS400 steels support the following conclusions:
The BW-4 sample had a higher UTS and a lower corrosion current density than the other samples, signifying good mechanical strength and low corrosion characteristics with welding parameter 4 (current: 170 A, voltage: 20 V, speed: 40 cm/min). After 21 days, the original unsprayed BW-2 sample had the largest corrosion current (7.49e − 5 A/cm2). By applying an aluminum spray layer with a thickness of 450 μm, a lower corrosion current was obtained (7.79e-10 A); the corrosion current for the unsprayed BW-2 sample was up to five orders of magnitude higher. As the thickness of the sprayed layer increased, the corrosion current decreased, effectively slowing down the corrosion of the welded joints and ensuring good protection of the structural objects in the sodium chloride solution. The sample was immersed in sodium chloride solution for 21 days, and the EDS showed that the content of chromium was significantly reduced, which increased the probability of corrosion.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author biographies
Chung-Hsing Huang received an MS degree from the Nan Jeon University of Science and Technology, Institute of Engineering Science and Technology, Taiwan in 2014. He is currently pursuing a PhD in school at the Department of Mechanical Engineering at Kaohsiung University of Science and Technology, Taiwan.
Chang-Hsiang Hou received an MS degree from Taiwan National Cheng Kung University of System and Naval Mechatronic Engineering in 2005 and received his PhD from Taiwan National Kaohsiung University of Science and Technology in 2021.
Tso-Sheng Hsieh received his BS degree from the Department of Naval Architecture and Marine Engineering of Chung Cheng Institute of Technology, National Defense University, Taiwan, in 1991 and his MS degree from the Department of Mechanical Engineering of Kun Shan University, Tainan, Taiwan, in 2007. He received his PhD degree from the Department of Mechanical Engineering of Kaohsiung University of Applied Sciences, Taiwan, in 2016. He subsequently began lecturing in the Department of Industrial Engineering and Management, Fortune Institute of Technology, Kaohsiung, Taiwan and became an assistant professor in 2017. His research interests include optical fiber sensors and structure analysis.
Liren Tsai is currently an associate professor in the Mechanical Engineering Department at the National Kaohsiung University of Science and Technology (NKUST). His research interests include high speed impact experimental stress analysis and computational solid mechanics. Tsai received his PhD in mechanical and aerospace engineering from the Case Western Reserve University and his bachelor's degree in National Tsing Hua University, Taiwan.
Chia-Chin Chiang received his BS and MS degrees from the Department of Mechanical and Mechatronic Engineering at National Taiwan Ocean University, Taiwan, in 1998 and 2000, respectively, and received his PhD from National Taiwan University, Taiwan, in 2005. He subsequently began lecturing at the Department of Mechanical Engineering of Kaohsiung University of Science and Technology, Taiwan, and became a professor in 2015, and distinguished professor in 2021. His research interests include fiber Bragg gratings (FBGs), optical fiber sensors, smart materials and structures.
