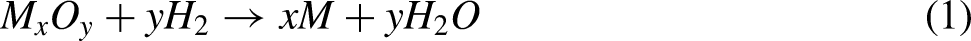Abstract
The press and sinter method remains the standard among powder metallurgy processes for powdered stainless-steel materials. It delivers low cost, low oxidation rate, and adequate corrosion resistance. Furthermore, 17-4PH is a martensitic stainless-steel that is commonly used for high-strength and medium-ductility stainless steel parts. However, a few studies have investigated the press and sinter method for producing 17-4PH parts. This shortage is due to the high hardness (low compressibility) of 17-4PH powder. Thus, the main objective of this study is to evaluate the press and sinter method in terms of the manufacturing process, the influencing factors, and the theoretical basis of press and sinter methods in conjunction with metal injection molding technology for the production of 17-4PH parts. First, the literature and monographs are examined and summarized to cover the previous results, research progress, development trends, and applications of press and sinter method 17-4PH parts. Following the theoretical analysis, the practical investigation was conducted by producing parts with cold pressing from 800 to 1600 MPa, followed by sintering: the sintering temperature was 1200 °C for one hour under a protective vacuum atmosphere. ImageJ analysis was performed to measure the sinter density. The results showed an increase in relative sinter density from 84.43% to 96.43% for 800 and 1600 MPa, respectively, while the earlier results reached 93.47%. Overall, the press and sinter method enables the production of high-hardness 17-4PH parts with a high density, without using additives like lubricants, wax, or alloying elements.
Keywords
Introduction
There are many different powder metallurgical manufacturing processes. For example, metal powder injection molding (MIM), additive manufacturing (AM), and hot isostatic pressing. In addition, the press and sinter method (PSM) remains the leader in manufacturing ferrous parts among all other powder metallurgical operations worldwide. According to Figure 1(a), the ferrous products obtained through PSM accounted for 63% of the total. A report on the metal powder market size in the United States from 2016 to 2027, which is shown in Figure 1(b), explains the high demand for PSM products compared to others.
Ferrous powder, such as 17-4PH (precipitation-hardened martensitic stainless steel), has high hardness, 3–4 times stronger than austenitic stainless steel such as 316. Precipitation-hardened stainless steel is usually chosen to produce stainless materials with high strength and medium ductility.3,4 17-4 PH parts are typically used in the aerospace, chemical, petrochemical, food, and general metal processing industries.5–8 Designers and engineers prefer 17-4PH stainless steel because of its beneficial properties such as high strength, hardness, easy modification by heat treatment, and corrosion resistance at temperatures below 300 °C.6,9,10 This is attributed to the alloy containing more chromium and nickel.4,11
However, PSM could not previously be used to produce high-density parts from 17-4PH powder due to the poor compressibility of the powder. Therefore, the second-best sintering method has been MIM, according to Figure 1(b). MIM includes the preparation of the starting material (polymer blend with metal-like properties), injection molding, debinding, and sintering.12–15 Similarly, PSM involves the compaction of the powder (metal or metal alloy, with or without polymers), usually in molds, followed by sintering of the molded bodies.16,17 The thermal debinding in a high-temperature burnout furnace should be done before sintering in the MIM process, which takes up to 50 h. 7 In contrast, there is no debinding step in PSM. Another advantage of PSM is that the particles bond together directly (without organic materials such as wax), resulting in less oxidation compared with MIM. 18 Therefore, PSM does not cause high pollution. 19 MIM is similar to PSM because both produce near-to-net shapes and complicated geometries, but MIM is more expensive than PSM. 7 Figure 2 shows the previous studies that performed the densification of sintered 17-4PH parts by PSM and MIM. Fewer PSM studies were published than MIM studies. The low compressibility of the 17-4PH powder, which leads to low densification, was the main reason for fewer PSM studies.20–22 PSM can produce stainless-steel parts with a sintered density of 6.8 to 7.3 g/cm3. 23 In contrast, the theoretical density of 17-4PH materials reported in the literature ranges from 7.75 to 7.85 g/cm3. 24

PSM stainless steel parts are cheaper than MIM stainless-steel parts, especially for 103 to 106 pieces with weights ranging from 0.1 to 9 kg per piece.19,34,35 PSM has several advantages for the mass production of small, intricately shaped components. 36 For example, money can be saved for gear pressing and sintering due to the reduced number of production operations. 25 PSM stainless steels exhibit higher quality than other types of stainless steel in terms of cost, control of size, and resistance to wear and corrosion. 23
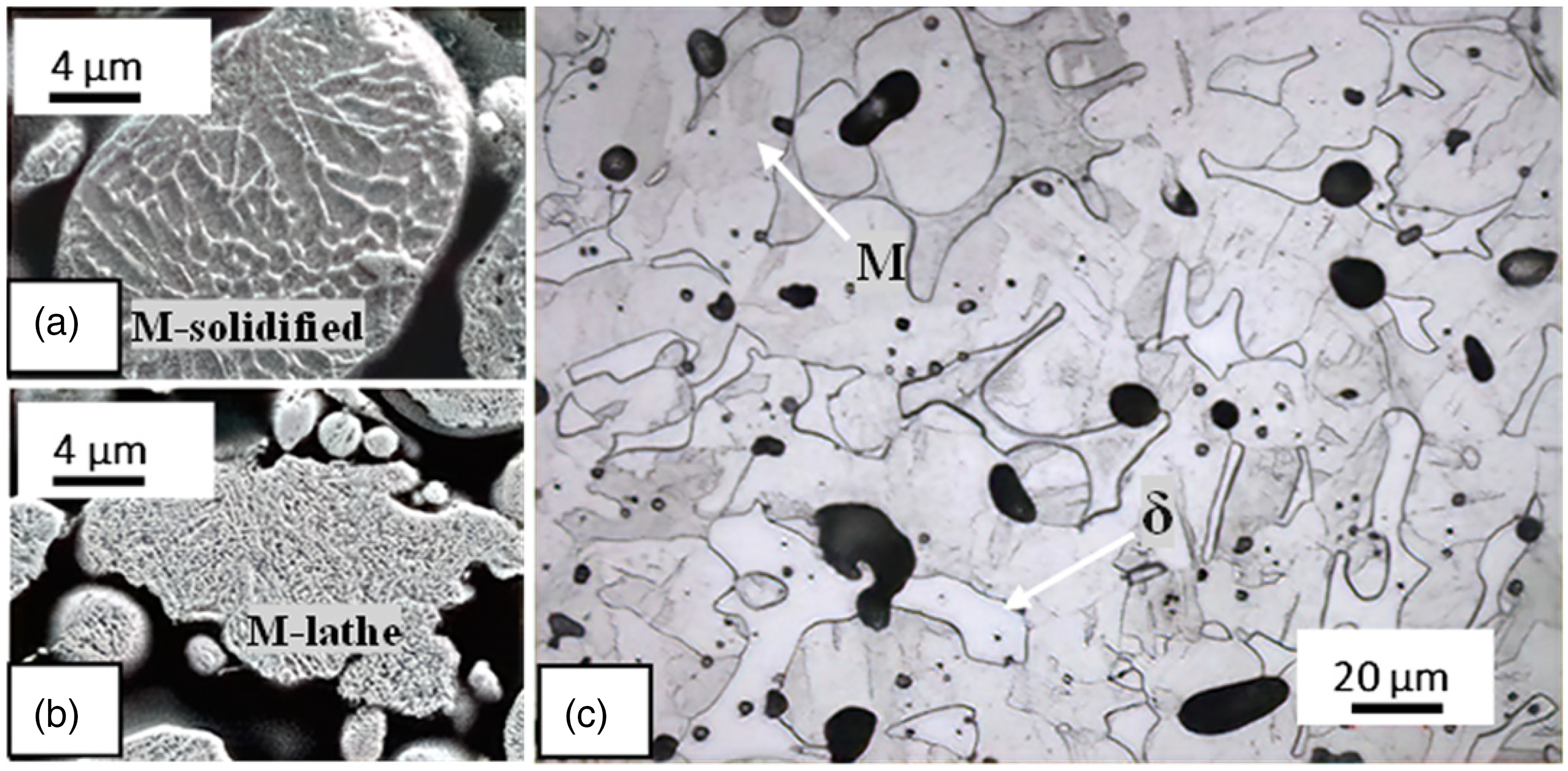
However, the high porosity of PSM parts made of 17-4PH is still the main issue. Steels with higher density typically have greater mechanical properties. 37 Thus, earlier studies attempted to increase the sinter density of 17-4PH parts produced by PSM. In 1994, Reinshagen and Witsberger demonstrated that aged, sintered PSM 17-4PH parts could achieve a relative sinter density of 93.47%. 11 Moreover, Schade et al. discovered that cutting the carbon content of conventional water-atomized 17-4PH powder (0.023 wt.% carbon) in half with a high-performance atomization (HPAT) technique increased the relative sinter density to 94.75%. 20 Additionally, in 2008, water-atomized PSM 17-4PH parts that were sintered and heat-treated reached the maximum relative sinter density of 92.45%. 11 In 2013, it was found that increasing the sintering temperature improved the sinter density by 92.19%. 3 Moreover, adding 0.6 wt.% of boron increased the relative sinter density of PSM 17-4PH parts from 84.54% to 96.4%. 9 Figure 3 shows the results of previous studies regarding how to increase the density of sintered PSM 17-4PH parts by increasing the cold pressing or sintering temperature (without alloying additions, postprocessing, or lowering the carbon content).

As a result of the above literature, PSM is still the most demanded technology worldwide in the powder metallurgy market for producing stainless-steel parts. According to Figure 1(b), PSM will also be demanded in the future. However, PSM is still unable to provide high-density 17-4PH parts. The densification is considerably affected by many forming factors such as cold pressing, powder compressibility, powder particle shape, particle size distribution, lubricant addition, sintering temperature and time, and sintering atmosphere. Therefore, this review article focuses on the 17-4PH materials produced by PSM and compares them with those produced by MIM, summarizes the latest research on the forming factors, investigates the causes of low densification results, and finds the theoretical solutions to approve the densification property. This report also includes practical experiments that validate the academic gaps in a parallel manner.
Cold pressing
The densification of green compacts (i.e. powders before sintering) affects the strength and shrinkage ratio of the sintered parts. Lower green density means lower sinter density 20 and increased sinter shrinkage. 38 Consequently, low sinter density leads to poor mechanical properties. 39
Cold pressing is the process of repackaging metal powders. Pressing begins with the reduction of the pore space and increasing the powder's efficiency, eliminating the bridges created during filling, and forming new surfaces free of oxide. 40 However, the metal powder's compressibility (hardness) is the main factor that directly affects green densification. The high compressibility of metal powder (low hardness) leads to an elevated green density. The effect of cold pressing on metal powders with different hardness (pure iron 104 HV, titanium 144 HV, and 304L 205 HV) was investigated. 41 The findings indicated that the powder hardness and densification behavior had an inverse relationship.
Moreover, different powders of stainless steel with different compressibility (434L, 304L, and 17-4PH) were cold pressed up to 690 MPa. 20 The results showed that the parts made from 434L and 304L (high ferrite content and high compressibility) had a higher density than those made from 17-4PH (containing martensite and low compressibility). Since ferrite is less hard than martensite, it is possible to achieve higher green and sinter densities. 42
Previously, the effect of cold pressing on the green densification of PSM 17-4PH parts was investigated by different studies, which is shown in Figure 4(a). The highest cold press applied by previous studies was 760 MPa. As a result, the maximum relative green density was approximately 80.67%. Additionally, the effect of particle size distribution has been investigated, which is also presented in Figure 4(a). The lowest average particle size was around 45 µm. However, the increase in cold pressing has had a more significant impact than reducing particle size on improving green densification. 26

In the present work, Oerlikon water-atomized powder consisting of martensitic stainless steel (17-4PH) with a particle size of − 15/ + 45 µm was sieved using a 32-µm mesh and cold-pressed under vacuum up to 1600 MPa. The results shown in Figure 4(b) indicate that cold pressing increased the green density, and at 1600 MPa, the relative green density was around 90%, which was much higher than in previous studies.
Sintering
The energy stored in the cold-pressed metal part is released during sintering. Sintering is a complicated process in which the development of the microstructure occurs through the action of several different transport mechanisms. 43 In the solid state, the sintering of crystalline solids can occur by vapor transport, and surface, lattice, or grain boundary diffusion, in addition to plastic deformation by dislocation migration. 44 In the sintering of powdered steel, the green parts are solidified to the desired composition using temperatures below the melting point (but above half the melting temperature) for the appropriate amount of time. 40 During the sintering process, there are three kinetic stages: bonding after the reduction of oxides from the surfaces of powder particles (bonding between adjacent particles with the formation necks); densification (adhesion between the solid and porous phases); and grain coarsening (significant grain growth and interaction between pores and grain boundaries). 45 Thus, a higher green density, which means there are more points of contact between the particles, speeds up adhesion and densification during sintering. 40
Most metal (M) oxides in steel powdered materials are reduced by reduction reactions to start the necking stage during sintering. The reduction media can be (a) hydrogen, (b) carbon, or (c) carbon monoxide.
46
Moreover, the reduction process can take place at temperatures of less than 500 °C. Hydrogen comes from moisture in the powder and an atmosphere that contains hydrogen. Carbon comes from graphite lubricants or steel powder mixtures. Between 725 and 750 °C, the peak of thermal expansion shows that martensite BCC is transforming into austenite FCC. The initial sintering shrinkage peak appeared between 964 and 1050 °C. The isothermal sintering peak can be seen at temperatures ranging from 112 to 1143 °C. This peak started with heating shrinkage and ended with delayed shrinkage. The hydrogen peak shows that high sinterability can be reached by reducing the silicon oxides that cover the 17-4PH powder between 1250 and 1308 °C. This peak is reached when hydrogen is used as a protective atmosphere.

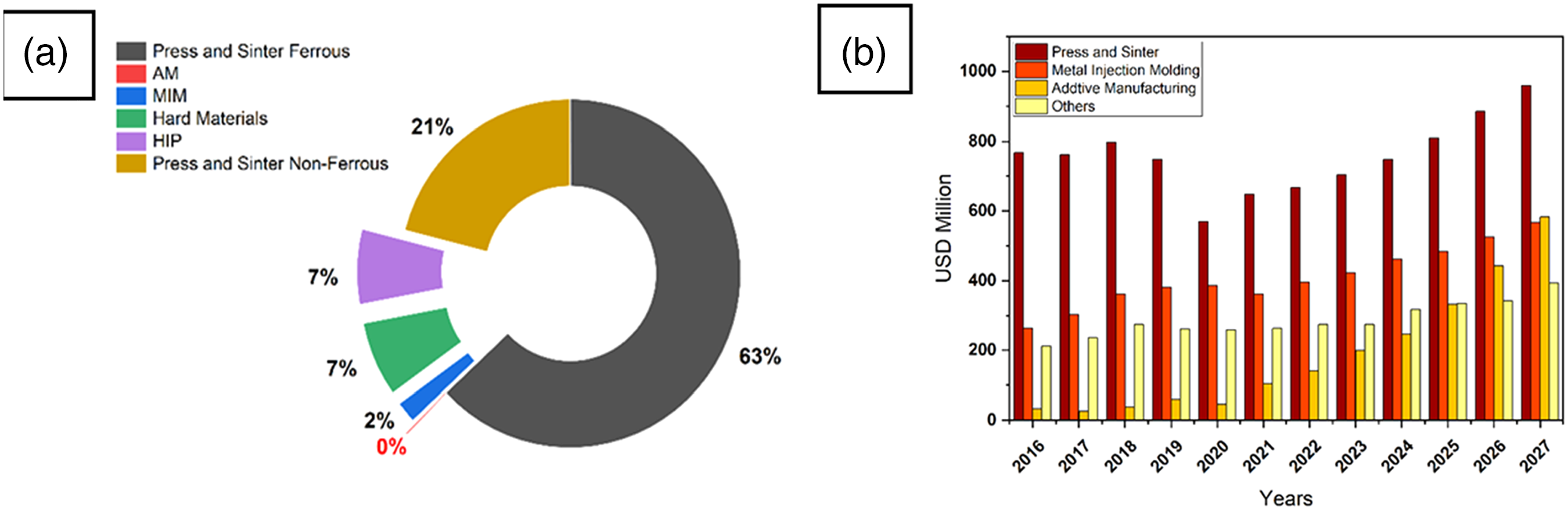
To confirm the first peak (thermal expansion), water-atomized 17-4PH powder was examined before sintering, and the microstructure showed a solidified martensite, as shown in Figure 6(a). After sintering at 780 °C and subsequent quenching, the microstructure showed lathe martensite, emphasizing austenitization. Another example of the microstructure of a PSM 17-4PH part is shown in Figure 6(b). At the end of one hour of sintering at 1360 °C in pure hydrogen, the microstructure showed martensite, delta-ferrite (δ-ferrite), and pores. A detailed explanation will be given in the following sections of this review.
In addition to increasing cold pressing, PSM stainless-steel parts can be improved by changing the sintering conditions (temperature, time, and atmosphere), and thus controlling the grain growth, oxidation, and the ratio and shape of pores. High sintering temperatures can eliminate tiny pores, make the remaining pores spherical, and homogenize the stainless-steel structure. Sometimes, higher temperatures can distort the PSM steel's final dimensions, but spherical pores increase the mechanical properties of a sample compared to irregular pores. The pores are the most critical factors when it comes to predicting and modifying the mechanical properties.
Factors related to the properties of sintered stainless-steel materials
Sintering time and temperature
Densification
Increasing the sintering temperature and time affects the ratio and shape of the pores. The effect of sintering temperature and time on the densification of 17-4PH parts produced by PSM has been studied. 27 The results are shown in Figure 7. They show higher relative sinter densities with increasing sintering temperature and time. The highest and lowest relative densities were 92.19% (using the highest temperature of 1300 °C and a time of 120 min) and 85% (using a low sintering temperature of 1200 °C), respectively.

Sintered density of 17-4PH materials as a function of the sintering time and temperature and (reproduced from Ref. 27 ).
The previous experiments performed to improve the sintering densification of PSM 17-4PH parts are summarized in Figure 8(a). Increasing the sintering temperature from 1300 to 1340 °C for the same sintering time and cold pressing (60 min, 600 MPa) resulted in a 2.3% increase in the relative sinter density. In addition, increasing the sintering time from 20 to 60 min at a fixed cold pressing and sintering temperature (690 MPa, 1260 °C) was investigated. 20 The results showed an increase in the relative sintering density from 89.6% to 93.47% for 20 and 60 min, respectively.
For the practical experiments conducted herein, the sintering conditions were established as follows: one hour and up to seven hours as holding times; 1200 °C as a sintering temperature; and vacuum as a protective atmosphere. The relative sinter density was measured by ImageJ analysis as the average value of 12 readings for 12 optical photos taken from different positions for each cross-sectionally cut and polished sample of PSM 17-4PH. These densification results are shown in Figure 8(b). The sintered densities increase with the green densities, Figure 4(b) for comparison. The relative sinter density of the green samples cold-pressed at 800 and 1600 MPa increased from 84.43% to 96.43%, respectively. In earlier research, the maximum relative sinter density was 93.47%. In addition, an extension of the sintering time, up to seven hours, improved the densification properties. The optical photos and the results of ImageJ analysis of the practical part of this review are shown in Figure 9. The fraction of pores decreases with increasing cold pressing, and the shapes of pores change from irregular to more spherical shapes. According to a previous study, increasing the cold pressing from 200 to 600 MPa transformed the shapes of the pores of 316L into regular shapes, and the volume fraction of regular pores increased from 10% to 22%. 50

The optical microscopic photos of press and sinter 17-4PH samples with indicated different cold pressings and calculated porosity (%) by ImageJ; sintered at 1200 °C for one hour.
In a previous study, the relative density of 17-4PH samples prepared by MIM was controlled from 61% to 99% by increasing the sintering temperature from 1200 to 1350 °C. 28 The results showed that not only did the pore fraction decrease but also the shapes of the pores became more regular as the sintering temperature increased. Additionally, the relative sinter density of MIM 17-4PH parts increased up to 98% when the sintering temperature was increased from 1150 to 1300 °C under the same production conditions. 48 In addition, the effect of sintering time and temperature on the densification of a MIM 17-4PH part containing 1% FeB was investigated. 51 The results showed that changes in sintering temperature, from 1265 to 1285 °C, and time, from 10 to 45 min, led to a nearly complete density (around 99%) due to the formation of the eutectic liquid phase, which exhibited high diffusivity and rapid densification. However, Figure 10 depicts the appearance of the δ-ferrite phase during sintering at 1300 °C, which may be responsible for the rise in the density of 17-4PH sintered parts. The light gray is the martensitic phase, and the δ-ferrite is the dark gray phase. The microstructure of the parts sintered at 1150 °C included both austenitic grain boundaries and martensitic layers.

Microstructures of 17-4PH produced by metal injection molding after sintering at (a) 1150 °C and (b) 1300 °C (reproduced from Ref. 48 ). M, γ, and δ refer to martensite, austenite, and delta-ferrite, respectively.
However, increasing the sintering time at elevated temperatures can lead to overheating of the steel structure. For example, the effect of using different sintering times on the properties of HK30, an austenitic stainless steel, was studied. 49 Sintering at 1270, 1280, and 1300 °C revealed that, for the same holding time, density increases with increasing temperature. At 1280 °C, the highest density of 7.61 g/cm3 was reached after seven hours of holding time, and further holding led to a slightly lower density, indicating the possibility of overheating. A higher sintering temperature of 1300 °C led to the overheating of the microstructure, reducing the bond strength between grain boundaries. As a result, the density and mechanical properties decreased further.
Mechanical properties
The porosity ratio affects the mechanical and physical properties of powdered steel parts. The balanced effects of reduced porosity and grain coarsening enhanced tensile strength and elongation, while yield strength remained unchanged. 52 Usually, a decrease in porosity raises the yield strength, but it was terminated by the grain-coarsening phenomenon. Moreover, by increasing the sintering time from 20 to 60 min, the sintered density of conventional water-atomized 17-4PH green compacts has also increased from 6.8 to 7.3 g/cm3. 26 The high density resulted in high mechanical properties. Conversely, pores reduce mechanical properties by acting as stress concentrations and promoting fracture growth. 53 In another study, when the sintering time was increased from 20 to 40 min, the sinter density increased, and subsequently, the hardness increased from 59 to 65 HRA. 20 In addition, the effect of different sintering temperatures on MIM 17-4PH parts was investigated. 14 The results showed that the 17-4PH parts had a higher relative density at the high sintering temperature of 1360 °C than at 1200 °C, but there was no significant change in hardness. Table 1 shows the mechanical properties of previously studied PSM 17-4PH parts made using different conditions.
The mechanical properties of 17-4PH parts manufactured by press and sinter and compared to wrought processing.

332(10): 332 per Vickers with 10 kgf as test load; HPA 17-4PH: high performance atomized 17-4PH mixed with 0.75 wt.% Acrawax C; HQ: 1040 °C for 1 h and oil quenching; P: cold-pressing in MPa; PLT: powder layering technique; RD: 60% reduction at 500 °C; S: sintered; UTS: ultimate tensile strength; WA17-4PH: water-atomized 17-4 PH with 1.0 wt.% Li stearate; YS: yield strength.
* UTS and YS according to correlation model of Yrieix and Guttman; - means not included.
Pores and grain size
Multiple factors influence the shape of the pores, including the powder particle morphology, sintering temperature and time, and the atmosphere. Using gas-atomized and water-atomized powders for producing MIM 17-4PH parts has been investigated. 31 The sintering results at 1250 °C showed that the pores between particles were quite different for the MIM 17-4PH made of gas-atomized and water-atomized powders (see Figure 11). Spherical powder particles produce more rounded pores compared to irregular powder particles. The shape of the particles in gas-atomized samples is spherical, while it is irregular in water-atomized samples. 31 In another study, the MIM 17-4PH parts sintered at 1300 °C had round pores compared to those sintered at 1150 °C. 62 Moreover, adding alloying elements can affect the pore morphology. Increasing the boron content in MIM 17-4PH parts reduced the volume of the pores and changed their shapes to be more spherical. 65 Consequently, the mechanical properties of the sintered 17-4PH materials may be related to the pore shapes, which are related to the form of the powder particles, as mentioned earlier. The tensile properties of MIM 17-4PH parts made from gas-atomized powder over a range of sintering temperatures (1250−1350 °C) were around 10% higher than those of these parts made from water-atomized powder. 66 More rounded pores also led to higher ductility. 11 Regular pore geometries and low porosity at elevated sintering temperatures contributed to the increase in the tensile strength of parts made of chromium alloy steel that were produced by PSM. 38

Pores in (a) gas and (b) water atomized 17-4PH parts sintered at 1250 °C, manufactured by metal injection molding (reproduced from Ref. 31 ).
Furthermore, high porosity causes the hardness values to vary within the same manufactured part. For wrought steel, the porosity reduces heat transfer, favoring bainite formation rather than martensite, which may explain the tremendous difference in hardness with decreasing density. 64 Additionally, the martensite phase is harder than the bainite phase.
The effect of sintering temperature or time on the shapes of the pores has also been observed in PSM 316L parts. Both the 17-4PH and 316L parts showed similar sintering behavior. 27 Increasing the sintering temperature from 1200 to 1300 °C for PSM 316L parts decreased the pore fraction from 20% to 12%, and the shapes of the pores changed from irregular to more regular. 61 However, sintering at a temperature of 1200 °C was considered insufficient and resulted in low mechanical properties. 63
Increasing the sintering time and temperature increases grain growth and decreases the porosity ratio and the diameter of the pores. 52 For example, the average grain sizes of the samples were 45, 64, and 76 μm after sintering at 1280 °C for 5, 7, and 9 h, respectively. 67 The higher the sintering temperature, the larger the average grain size. 68 However, tensile strength and elongation depend more on porosity than on grain size, because the fracture is mostly caused by the growth of micropores and coalescence. 52
In the case of PSM 17-4PH parts, there is no study directly showing the influence of sintering temperature or time on the size and form of porosity. There is also a lack of suitable sintering conditions to solidify the green compacts of 17-4PH with enhanced mechanical properties. As mentioned above, a sintering temperature of 1200 °C leads to insufficient sintering, even for 316L, which has greater compressibility than 17-4PH. Therefore, it is necessary to investigate the optimal sintering conditions for PSM 17-4PH parts.
Particle size and carbon content
Densification
Fine powder particles and low-carbon content improve the densification properties of 17-4PH parts. The effect of gas-atomized 17-4PH powders in the range of 10 to 32 µm was investigated by MIM. 66 The result showed that the particle size distribution at higher sintering temperatures (above 1300 °C) had little effect on the tensile strength. On the contrary, the highest mechanical properties (tensile strength, yield strength, and elongation) were obtained at the lowest sintering temperature (1149 °C) with the lowest particle size (10 µm).
There is an inverse connection between the carbon content and the densification property of powdered steel materials. The effect of using water-atomized 17-4PH powder was studied, but it was prepared using two different methods: conventional atomization (CAT) and HPAT. 20 HPAT 17-4PH powder has lower carbon and nitrogen content and finer particles than the CAT powder. The results showed that the sintered parts made of HPAT powder had a higher density (7.4 g/cm3) than those made of CAT powder (7.3 g/cm3), regardless of the sintering conditions. Moreover, parts with low-carbon content in the range of 0.02 to 0.04 wt.% gave optimum combinations of strength and ductility, although the wide specification range for carbon is 0.00 to 0.070 wt.% in the MIM 17-4PH parts. 11 The percentage of carbon in low-carbon stainless steel must be kept low to prevent carbide precipitation from chromium carbides. For example, in austenitic stainless steels that are cooled slowly, the carbon content must be in the range of 0.02–0.03 wt.%. The results showed that, as the amount of carbon in the material increased, the tensile strength, yield strength, and hardness all increased, but elongation decreased. 40
Nitrogen has a comparable effect to carbon on martensite formation. Control of nitrogen content is equally essential for maximizing ductility. 11 Nitrogen acts as an austenite stabilizer in 17-4PH parts. The denser austenite slows the rate of shrinkage and densification. It also prevents the formation of the favorable ferrite phase, which hinders densification. 47 During sintering, nitrogen penetrates the steel and forms nitrides. This impedes the diffusion of iron or chromium and restricts the densification rate. 67
The phase diagram of 17-4PH with a carbon content of less than 0.04 wt.% indicates that the liquid phase occurs slightly above 1360 °C (see Figure 12). 7 The formation of δ-ferrite during high sintering temperatures and the development of the microstructure during sintering are summarized in the following structures: ferrite (α); austenite (γ); and γ + δ-ferrite. The phase diagram shows that the formation of δ-ferrite strongly depends on the carbon content, and δ-ferrite drops with increasing carbon content. 32 However, an investigation of the microstructural evolution of MIM 17-4 PH stainless steel at room temperature revealed that a martensitic phase occurs. 69 The martensite transforms into γ at approximately 700 °C, which is consistent with the corresponding phase diagram in Figure 13. The second-phase transformation is the transformation of γ to δ-ferrite, which starts at nearly 1200 °C and is related to the dilatometric peaks explained in the Sintering section.

Phase diagram of 17-4PH alloy (reproduced from Ref. 7 ).
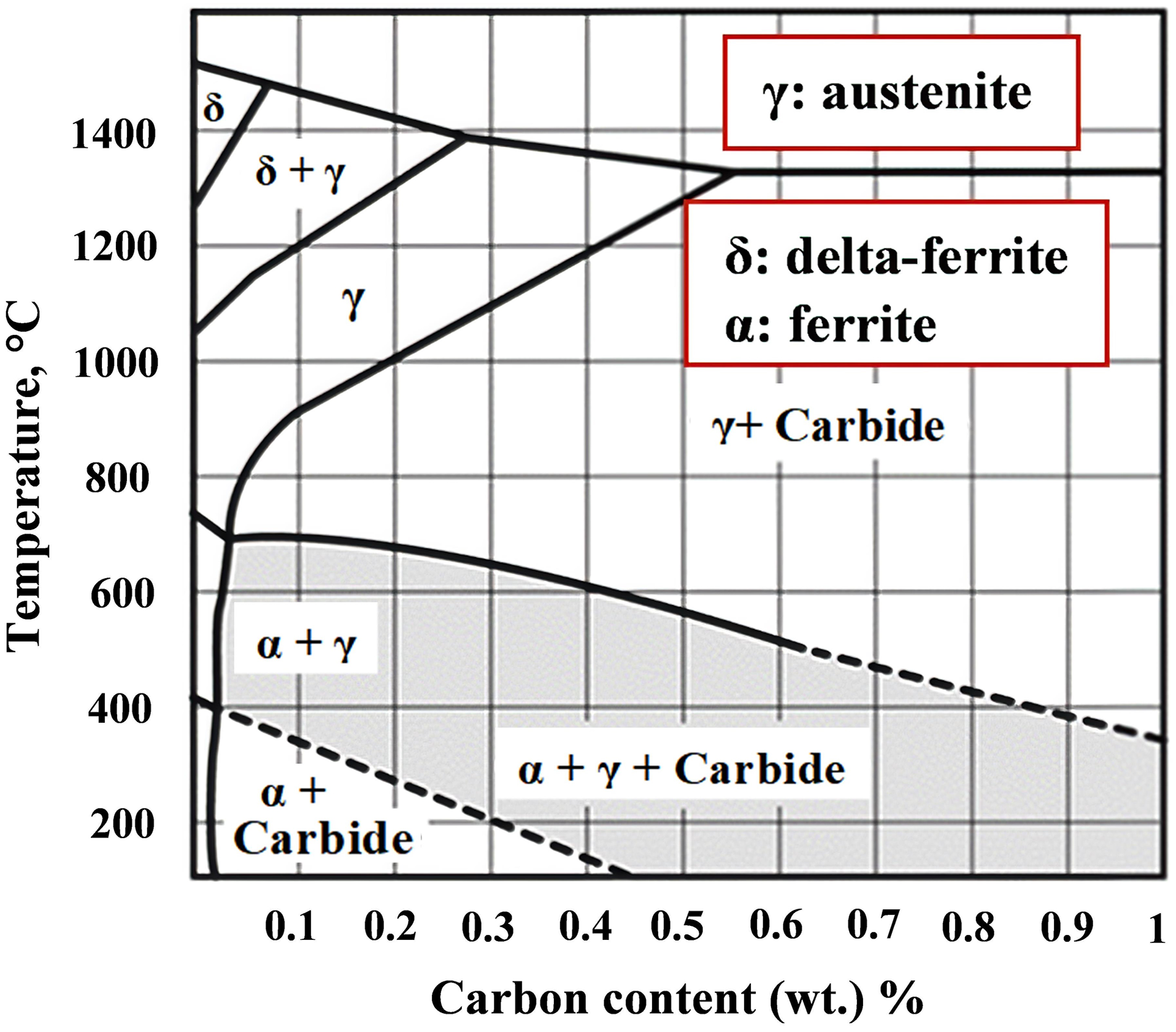
Phage diagram of 0.1 C%-steel, 18% chromium, and 4% nickel (reproduced from Ref. 69 ).
The above studies discussed how carbon content affects density, mechanical properties, and ferrite volume fraction. However, the effect of carbon content on carbide formation and oxidation (reduction) remains unclear in PSM 17-4PH parts. In addition, the conducive relationship between carbon content and sintering conditions (temperature and time) for the formation of carbides and oxides has not been clarified.
Residual carbon effect
Furthermore, the formation of δ-ferrite decreased the pores and improved the densification of sintered 17-4PH materials. 47 This phenomenon is observed because the diffusion volume of the ferrite phase structure (BCC) is faster than in the austenite phase structure (FCC) of low-carbon steels. 7 According to Baba and Kyogoku, an abrupt decrease in tensile strength occurred at high residual carbon content, which they attributed to the effect of retained austenite. 6 The consequence of retained austenite on the mechanical behavior of 17-4PH parts produced by the selective laser melting method (an AM process) was investigated. The results showed that the parts with large amounts of retained austenite exhibited stress-induced transformation to martensite during tensile testing. This behavior was associated with much lower yield strengths, faster strain hardening over a wider range of strains, and a later start to local plastic deformation. 70
A study was conducted to investigate the relationship between carbon content and densification property and retained austenite formation in the area of press and sinter of 17-4PH parts, as mentioned in the literature. However, the influence of carbon content on the volume fraction of δ-ferrite, the retained austenite, and phase transformation during sintering requires further research. Additionally, more research is necessary regarding the effects of carbon content on the mechanical and physical properties of PSM 17-4PH parts.
Sintering atmosphere and oxidation
Atmosphere
By controlling the phases in the microstructures of 17-4PH materials, it is possible to determine a connection between the sintering atmosphere and the densification property. δ-ferrite formation in PSM 17-4PH parts produced under different sintering atmospheres has been studied. 47 The results illustrated that the appearance of δ-ferrite began in a pure hydrogen (H2) environment with a sintering temperature above 1220 °C, which can be seen in Figure 14(a) along the grain boundaries (whitest color). Other results obtained using H2 and hydrogen/nitrogen (H2/N2) atmospheres at an elevated sintering temperature (1360 °C) revealed that no δ-ferrite phase formed and that the microstructure contained significant amounts of porosity, as shown in Figure 14(b). Unfortunately, these results cannot be compared with other studies (e.g. Figure 12) due to the absence of carbon content. Additionally, it was mentioned that δ-ferrite formation can occur at a temperature lower than 1220 °C, as shown in Figure 15, according to the two-phase master sintering curve model. 69 Moreover, Ar + 20% H2 was used as an atmosphere to sinter PSM 17-4PH green compacts. 33 A range of sintering temperatures from 1240 to 1290 °C for 90 min was used. A chromium–nickel–martensite matrix with a few small islands of δ-ferrite at the grain boundaries comprised the final microstructure. Thus, the formation temperature of the δ-ferrite depends on the chemical composition of the powder, the sintering atmosphere, and the residual carbon. 31

Microstructures of press and sinter 17-4PH parts (a) quenched from indicated temperatures in pure H2, and (b) quenched after sintering for 1 h at 1365 °C under the specified atmospheres (reproduced from Ref. 47 ).

Delta-ferrite percentage with sintering temperature of 7-4PH parts manufactured by metal injection molding (reproduced from Ref. 69 ).
The exact role of the N2 atmosphere was found in another type of steel. The influence of the sintering atmosphere on the densification properties of HK30 materials was studied. 67 The results showed that the density was lower when sintered with N2 than with Ar. However, HK30 parts sintered in N2 had a higher hardness and tensile strength than those sintered in Ar. This was because N2 was dissolved in the austenitic microstructure and formed precipitates at the grain boundaries.
Overall, sintering in a vacuum atmosphere lowers the oxygen content. For example, the results of sintering water-atomized iron powder pre-alloyed with 1.8 wt.% Cr showed that the oxygen content in the green compacts before sintering was 0.15 wt.%, while the highest oxygen content after sintering in a vacuum was 0.02 wt.%. 38
To achieve a higher final density, oxidation during sintering should be avoided entirely. Sintering in an Ar atmosphere or vacuum is the preferred choice for 17-4PH materials. Depending on the sintering atmosphere, Cr2O3 may be formed. The effect of using different sintering atmospheres (vacuum, Ar, and N2/H2) to produce MIM parts from gas-atomized 17-4PH stainless-steel powder was investigated by X-ray analysis. 18 The results are illustrated in Figure 16. No oxygen was found in the case of vacuum or Ar, whereas oxygen was observed when N2 + H2 was used as a protective atmosphere. Cr2O3 was created after sintering in the N2 + H2 atmosphere, but no Cr2O3 was formed when sintering in the Ar and vacuum atmospheres. In addition, it was found that the increase in Cr2O3 was related to the higher sintering temperature Cr2O3 formed on the surface of the sintered particles, which stopped diffusion and made the material less dense.

Spectra of line EDS analysis of metal injected molding 17-4PH parts sintered at 1050 °C under (a) vacuum or Ar, (b) mixture of N2 and H2 atmosphere (reproduced from Ref. 18 ).
Oxides source
Steel powder atomized with water contains more oxide than powder atomized with gas. Therefore, water-atomized sintered powdered materials will always contain small amounts of fine oxides. 11 In addition, these oxides might exist either on the surface or within the particles. As mentioned in Section 3, surface oxides need to be reduced for sinter necks to form between powder particles.
The transformation of the surface oxides and their entrapment inside the sinter necks can occur during the heating phase. 38 Oxidized particles enriched with Si and Mn were found on the surface of H13 steel powder atomized with water. 68 After sintering, these oxide particles of about 1–3 μm were randomly distributed in the grain boundaries and matrix. Additionally, the gas-atomized powder has a more regular shape than the water-atomized 17-4PH powder. Therefore, the gas-atomized powder results in more regular pores (see Figure 11). These regular pores reduce the oxidation rate during the sintering process because they reduce the amount of exposed surface area. 36
Furthermore, different oxides can form in water-atomized steel powders, such as Cr2O3, NiO, and SiO2. Si is considered critical because most of the oxidation that occurs during water atomization is the oxidation of Si to SiO2, causing reduction. This reduction depends on the sintering conditions and results in residual oxides in the sintered component. Mn, however, increases oxidation during water atomization. 40 Several critical oxides in austenitic stainless steels were residues from the water atomization process: Cr2O3, MoO3, and SiO2. 16 Cr2O3 has high stability and is challenging to reduce, while Mo and Si must be removed before or during the sintering process. Moreover, the mechanical properties of stainless-steel parts can be improved by reducing oxides. For example, it was found that the mechanical properties of MIM 17-4PH parts made from gas-atomized powder were improved compared with MIM 17-4PH parts made from water-atomized powder, mainly due to the lower Si, O, and SiO2 contents in the case of gas-atomized 17-4PH powder. 24
Even though certain oxides, such as Cr2O3, MoO3, and SiO2, were found in the pressed and sintered austenitic stainless-steel parts, those analyses did not adequately investigate the oxides that may be present in the PSM 17-4PH parts. The phases with different sintering temperatures were well explained, 47 but the study did not address the oxides.
Heat treatment
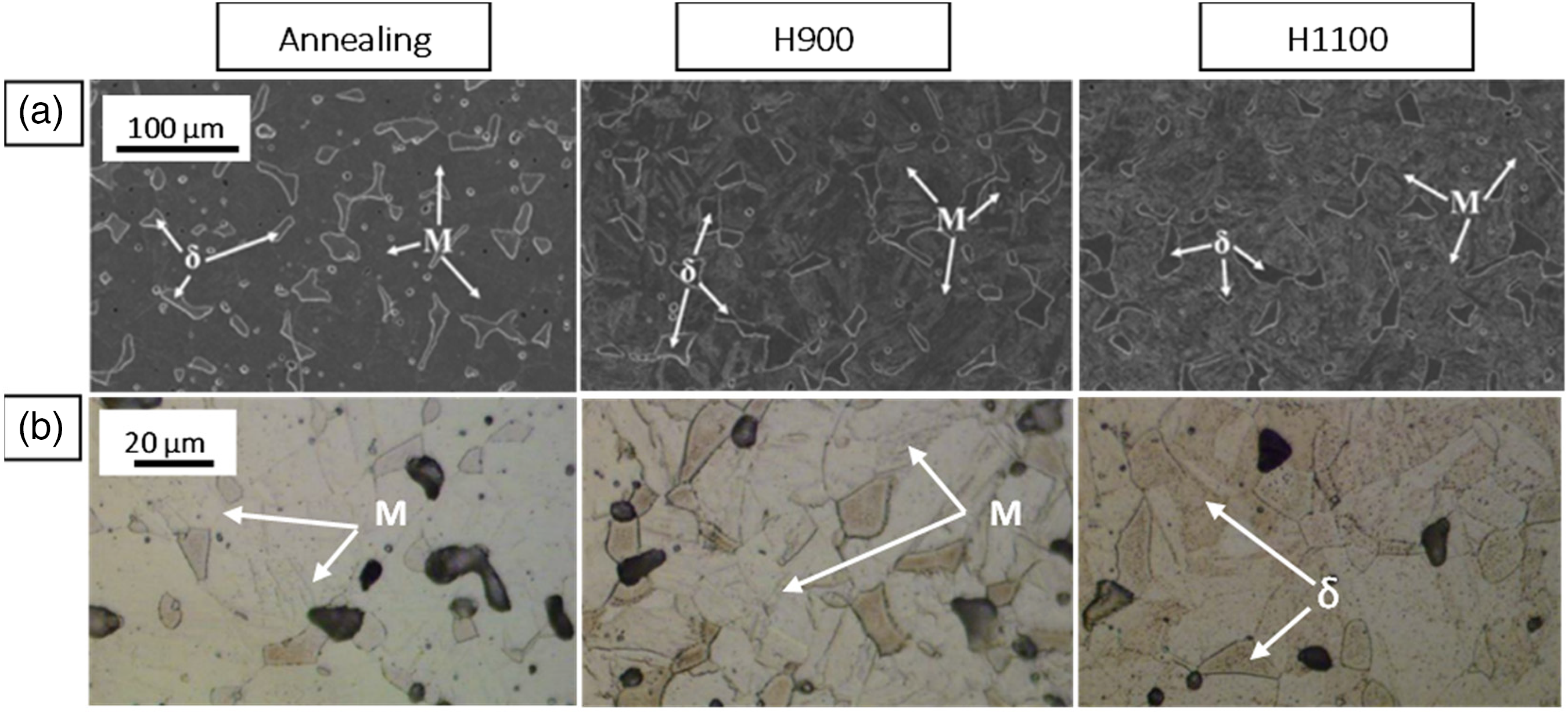
Heat treatment is the postprocessing of a manufactured metal part with the intention to modify properties, such as strength, ductility, and hardness, by controlling the microstructure and achieving the desired phases, grain growth, and precipitation. For instance, after annealing a CoCrFeMnNi alloy, the strain increased by 5%. 71 In addition, ferrite dissolution reduced the hardness of 316 stainless steel by 25% when solution-treated at 1050 °C. 72 Thus, many studies investigated different conditions for sintered parts produced from water- and gas-atomized 17-4PH using PSM, MIM, and fused deposition modeling.11,24,73 However, each experiment used a different atmosphere, hold time, heating rate, and cooling rate. Solution annealing, H900, and H1100 made up most of these conditions, as presented in Figure 17. The volume fraction of δ-ferrite increased after the solution heat treatment. Therefore, this condition is appropriate for a reasonable ductility requirement. An increase in strength, hardness, and a decrease in ductility is noted after applying H900 (generally 482 °C for 1 h), which formed Cu precipitates (observed by optical microscope) and less δ-ferrite compared to the as-sintered parts. Thus, δ-ferrite impacts tensile strength and ductility. 7 Increasing the volume fraction of ferrite in low-carbon steel leads to a further rise in ductility and a loss in tensile strength due to the elasticity. 74 However, an accumulation of δ-ferrite and larger Cu precipitates occurred during H1100 (552 °C for 1 to 2 h). Consequently, an elemental map was used to examine the black spots (not the big pores), which were distributed in the microstructure of the water-atomized MIM 17-4PH parts. 24 The results revealed the presence of SiO2 due to the presence of Si near oxygen sites, in addition to Cu precipitates (see Figure 18).

The elemental map of Si, O, and Cu after heat treatment for 1-h under vacuum followed by quenching in N2 (reproduced from Ref. 24 ).
After annealing, H900, and H1100 heat treatments, it can be deduced that the gas-atomized MIM 17-4PH parts had more δ-ferrite and less SiO2 than the water-atomized parts. Table 2 summarizes the mechanical properties of PSM 17-4PH parts as a result of the heat treatment conditions.
Represents the changing in mechanical properties according to the heating treatment conditions of 17-4PH sintered parts.
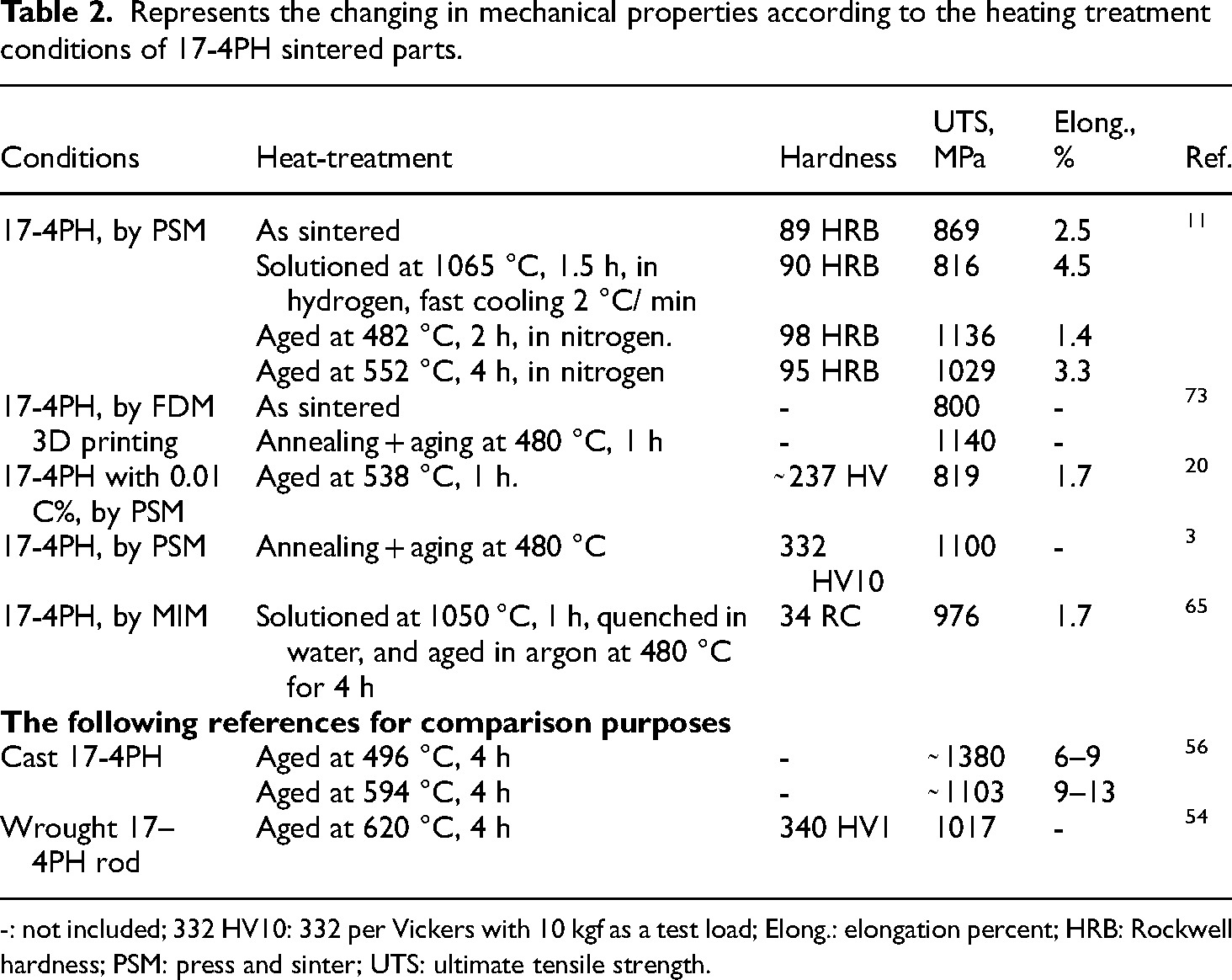
-: not included; 332 HV10: 332 per Vickers with 10 kgf as a test load; Elong.: elongation percent; HRB: Rockwell hardness; PSM: press and sinter; UTS: ultimate tensile strength.
Corrosion performance
The corrosion of stainless-steel materials in aqueous media is primarily caused by an electrochemical mechanism. This mechanism involves two types of reactions: anodic (oxidation of the metal) and cathodic (reduction in the aqueous medium).75,76 These processes produce a protective layer of Fe2O3. 76 In the case of 17-4PH materials, Fe2O3 and/or Cr2O3 might form, which function as a barrier between the metal and the service environment. 77 In addition, the following equations show an electrochemical reaction in a solution of sulfuric acid in water. 78
Oxidation reaction:
Iron oxides to form ferrous ions 



Although few studies have reported the corrosion properties of 17-4PH powdered sintered materials, the porosity ratio and pore geometry are the main defects responsible for the lower corrosion performance of powdered stainless steels. Isolda Costa et. al. investigated the effect of pores of 17-4PH MIM parts with a porosity of 3% in naturally aerated phosphate buffer solution at 25 ± 2 °C. 79 The results showed that these parts were subjected to slightly more pitting corrosion than the parts fabricated by conventional metallurgy due to the built-in porosity. The pores increase the geometrical area of the alloy exposed to the corrosive medium and induce localized corrosion.78,79 Furthermore, the effect of an aerated medium of 3% NaCl electrolyte on the pitting resistance of 17-4PH parts manufactured by MIM and conventional technology was compared. 80 Pitting attack by the chloride ions toward the protective layer of both conventional and MIM 17-4PH parts was observed. This attack was slightly higher for MIM parts compared to conventional parts due to the higher porosity in MIM parts. Additionally, this undesired influence of pores was found in additive-manufactured 17-4PH parts. Two different powder particle size distributions were used to investigate the effect of pores size on the corrosion performance of 17-4PH parts manufactured by laser powder bed fusion (LPBF). 77 The smaller pores from finer powder with D50 = 17 µm resulted in improved corrosion performance compared to the larger pores from wide-range powder with D50 = 17 to 43 µm, at an energy density of 64 J/mm3. Moreover, the pores, especially the large pores, are responsible for the worse corrosion behavior of the LPBF parts compared with forged parts. 81
Surface roughness, nonmetallic inclusions, and phase microstructure all play a role in corrosion performance. Improving surface roughness by the shot peening process resulted in a decreased the corrosion rate of 17-4PH parts manufactured by direct metal laser sintering. 76 These surfaces work as a concentration of hydrogen, the same as porosity in an acid medium. 78 Additionally, the inclusions, such as MnS, stressed martensite, incoherent Cu precipitates, and M23C6 carbides, lead to an acceleration of the corrosion rate in the 17-4PH parts manufactured by conventional metallurgy. 75 Therefore, heat treatment is necessary to control the microstructure of 17-4PH parts. The effect of aging temperature on the corrosion performance of PSM 17-4PH parts sintered at 1340 °C and solutioned at 1040 °C in a diluted sulfuric acid medium was investigated. 78 The results revealed decreasing corrosion rates with decreasing aging temperatures from 500 to 480 °C. This can happen as a result of coherent Cu precipitates and stress relief in the martensite matrix. 75
More research into corrosion performance is required, particularly for 17-4PH parts manufactured by PSM. Since the porosity is the main factor that influences the passivity of the stainless-steel powdered materials, the corrosion performance should be investigated in parallel with the densification of the PSM 17-4PH parts and in different corrosive aqueous media, for example, NaCl and H2So4. Additionally, different heat treatment conditions need to be performed on these parts to investigate the effects of other important microstructural factors on the corrosion rate. These factors can be the coherent distribution of Cu precipitates, the structure of the martensitic matrix, and the volume fraction of δ-ferrite and austenite.
Conclusions and prospects
PSM remains the leader among the powder metallurgy processes used for manufacturing stainless-steel materials because it enables low cost, low oxidation rate, and adequate corrosion resistance. According to the data provided in the metal market powder size report from 2016 to 2027, the ferrous parts produced by PSM have the highest demand among other technologies. However, using PSM in the production of 17-4PH parts is still limited due to the low compressibility of this martensitic stainless-steel powder, which leads to low densification. Therefore, this review analyzed the previous investigations to examine the factors influencing the limitations of 17-4PH parts. These factors included the 17-4PH powder characteristics (particle size, atomization method, chemical compositions, and particle morphology) and PSM parameters (cold pressing, sintering temperature, sintering time, and sintering atmosphere). Additionally, this paper found a series of scientific gaps in the literature. Moreover, the previous work on PSM 17-4PH parts helped to summarize the optimal parameters that were applied and suggest theoretical solutions to improve the densification properties. The conclusions can be summarized as follows:
Previous PSM studies used a range of cold pressing up to 760 MPa, and the maximum relative green density was around 80%. Therefore, establishing a clear relationship between cold pressing (higher than 760 MPa) and green density is necessary. Additionally, most PSM studies used a particle size distribution of 30 to 100 µm. Thus, further studies are needed to investigate 17-4PH with a particle size smaller than 30 µm. Moreover, a sintering time of 60 min at a temperature of 1260 °C resulted in sufficient sintering of the PSM 17-4PH parts. The increase in density did not change much when the sintering temperature was raised above 1300 °C. The high diffusion rate of δ-ferrite demonstrated the densification property of PSM 17-4PH parts. δ-ferrite can be formed at a sintering temperature lower than 1220 °C. Carbon, nitrogen, and the sintering atmosphere are the main factors affecting the volume fraction of δ-ferrite. However, the influence of carbon content and residual carbon on the carbide formation, the volume fraction of retained austenite, oxidation, microstructural phases, and mechanical properties of parts made from PSM 17-4PH has yet to be carefully examined. Moreover, vacuum and argon sintering atmospheres were effective for 17-4PH parts to remove oxides and reduce porosity due to the high affinity of carbon to oxygen, but the nitrogen atmosphere caused a decrease in density. Cr2O3 can form in sintered 17-4PH parts at elevated sintering temperatures and is also related to the sintering atmosphere (hydrogen and nitrogen). Cr2O3 was not found when a vacuum was used as the protective atmosphere. Further studies need to investigate the dependence of oxide formation on the sintering temperatures of the PSM 17-4PH parts and the possibility of elimination of oxides by reducing the pore ratio. Gas-atomized 17-4PH powder has a more uniform shape than water-atomized powder, which improved the resulting strength of sintered parts. Gas-atomized 17-4PH powder also led to less oxidation during the sintering process. This was because the pores were more regular. For high strength and hardness with acceptable ductility, H900 was a preferred heat treatment condition. The solution and annealing heat treatments were suitable for increasing the ductility. Using the H1100 condition resulted in lower strength and hardness and higher ductility than H900. Additionally, as the amount of δ-ferrite in the microstructure of 17-4PH parts increased, their tensile properties decreased.
Correspondingly, applying a cold pressing of more than 760 MPa was one of the theoretical suggestions that was determined in this review. Herein, the effect of applying a range of cold pressing (800–1600 MPa) using water-atomized 17-4PH powder with a low particle size distribution ( + 15/−32 m) was investigated. The experimental results showed an improvement in green densification with increasing cold pressing from 78% at 800 MPa to 90% at 1600 MPa. In addition, the relative sinter density determined by ImageJ increased from 84.43% to 96.43% for the samples cold pressed at 800 and 1600 MPa, respectively.
Finally, the literature did not mention how an extended sintering time affects the properties of PSM 17-4PH parts. Most studies used a sintering time of 20 to 90 min. However, it was mentioned in the literature that increasing the sintering time to seven hours at a sintering temperature higher than 1280 °C led to overheating and negatively affected the densification of PSM HK30 steel parts. Therefore, the effects of increasing the sintering time (over two hours) on the final PSM 17-4PH properties are unknown. However, at a sintering temperature of 1200 °C, the experimental results showed that increasing the sintering time up to seven hours had a better impact on densification.
In summary, it was found that green densification was the most critical factor, which directly influenced the sintering density, and consequently, the mechanical properties. Increasing the sintering temperature was also beneficial, but it may cause oxidation and carbide precipitation. Moreover, increasing sintering time can result in improved density, but it must be controlled to keep grains from overgrowing. Furthermore, the initial 17-4PH powder characteristics affected the final product density. Finally, PSM can be used to achieve near full density, high mechanical properties, and economic 17-4PH products.
Footnotes
Acknowledgments
This work was financed by the UMA3 project, which has received funding from the European Union's Horizon 2020 research and innovation program under grant agreement no. 952463.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the UMA3 project (grant no. 952463).
Author biographies
Mohammed Qasim Kareem is a PhD student in materials engineering. His area of research is casting, powder metallurgy, AM of metal, heat treatment and microstructure analysis.
Tamás Mikó holds a PhD in Material Sciences and Technologies. His area of research is Material Sciences and Technologies.
Gréta Gergely is an associate professor in materials science. Her area of research is Al alloys, composite materials, bioceramics and image analysis.
Zoltán Gácsi is a professor in Physical Metallurgy. His area of research is computer model for physical metallurgy process, Solidification, Quantitative microscopy, Stereology, and Image analysis.