Abstract
In sub-Saharan Africa, the invasive plant Parthenium hysterophorus (Parthenium) is threatening ecosystem integrity, biodiversity, and smallholder livelihoods. But, there is no single effective method of controlling it. Desmodium intortum, Lablab purpureus, and Medicago sativa were tested for their capacity to suppress Parthenium, as well as the allelopathic potential of Desmodium uncinatum leaf crude (DuLc) extract. While the study investigated the effect of DuLc extract concentrations on seed germination and seedling growth in laboratory, pot, field plot, it also assessed the effect of selected suppressive plants on Parthenium growth. It was found that high levels of DuLc concentrations and suppressive plants inhibited Parthenium germination and growth. When Parthenium was grown with suppressive plants, its growth was inhibited compared to when it was grown alone. When grown with all three test plants, the stem height and total fresh biomass of Parthenium seedlings were lowered by more than 60% and 59% in pots, and 40% and 45% in plots, respectively. Parthenium seed germination was decreased by 57% in plots, 60% in pots, and 73% in petri dishes at higher DuLc concentrations (i.e. 75% and 100%). Parthenium seedling stem heights were 36% (in plots) and 30% (in pots) shorter when sprayed with higher concentrations of DuLc. Overall, the findings of this study suggest that suppressive plants and those containing allelochemicals can be employed as a management tool to combat Parthenium invasion in sub-Saharan Africa, notably in Tanzania.
Introduction
Alien invasive plants (AIPs) such as Parthenium hysterophorus L. (Asteraceae), often known as Parthenium, represent serious challenges to biodiversity, agriculture, and the delivery of ecosystem services in recipient ecosystems.1–6 It degrades the quality of rangelands in invaded ecosystems.7,8 Parthenium alters the vegetation structure of recipient habitats by releasing allelochemicals that hinder the growth of coexisting plants, resulting in monospecific stands of Parthenium.9–11 Its presence in pastures lowers the quality, quantity, and production of feed.3,12 If unchecked, Parthenium has the potential to hinder sustainable development and smallholder livelihoods13,14 as well as negatively impact biodiversity and ecosystem services. 11
Earlier research has found that maintaining diverse assemblages and/ or density of native or non-invasive forage plants, as well as using bio–herbicides made from native plants is the best way to reduce AIPs abundance in pastures or natural habitats.15–18 If native plant density, such as competitive diversified feed species, is preserved in invaded areas, IAP invasion may be reduced.17,18 In natural environments where pesticides are not recommended, using suppressive plants as a Parthenium management approach could be a viable option. Also, some research found that certain native plants had the allelopathic potential to decrease AIP germination and growth.19–21 Native plants are favoured over exotic plants because they have co–evolved with the allelochemicals of the wider biological community, 19 and their non–target effects are expected to be less severe than AIP allelopathic features. Thus, such plants with bio–herbicidal potential can be used as a management tool for suppressing Parthenium. This is due to the fact that synthetic herbicides are usually broad-spectrum, causing harm to native and ecologically important plant species.4,20,22,23 Synthetic herbicides can also kill or injure beneficial invertebrates, such as natural enemies, pollinators, and decomposers. 24
Therefore, the main goal was to assess whether increasing the density of suppressive plant species and using crude extracts as bio–herbicides from native/naturalized plants could reduce the growth vigour of Parthenium. Experiments were carried out to assess (i) the suppressive ability of Desmodium intortum (Fabaceae), Lablab purpureus (Fabaceae), and Medicago sativa (Fabaceae) forage legume plant species, and (ii) the inhibitory effect of Desmodium uncinatum (Fabaceae) leaf crude (DuLc) extract against Parthenium germination and growth. Test plant species were chosen for their non-invasive, drought-resistant, fast-growing, and importance as livestock fodder.25,26 To assess the effects of selected suppressive plants and different DuLc concentrations on seed germination and seedling growth, petri dish, pot, and field plot experiments were used. It was hypothesized that selected plants and DuLc crude extract will negatively affect Parthenium germination, seedling height, root length, diameter, and biomass.
Materials and methods
Competition experiment and seedling growth parameters
Seedlings of Parthenium were obtained from invaded habitats, and seeds of D. intortum, L. purpureus, and M. sativa from Kibo Seed LTD in Arusha, Tanzania. The study tested the suppressive effects of D. intortum, M. sativa, and L. purpureus on the growth vigour of Parthenium seedlings under competitive conditions. From the 10th to the 27th of February 2019, the experiments were conducted at the Nelson Mandela African Institution of Science and Technology (NM-AIST, 3° 24.149′ S and 36° 47.790′ E, 1197 m a.s.l) in plots (1 m2) and plastic pots (763.82 cm2 surface area). Black clay dirt was obtained from field plots and used to fill pots to the brim. Twenty-five seeds of Parthenium and three test plant species (D. intortum, M. sativa, and L. purpureus) were seeded in five pots (which were kept in a naturally lighted screen-house) and five plots in monoculture (control) and mixture. The 11 different planting combinations were replicated five times, giving 55 planting plots and 55 pots (Table 1).
Pot and plot experimental planting design diagram with P. hysterophorus and suppressive plant species.

Respectively, P, L, D, and B stands for Parthenium, M. sativa (Lucerne), D. intortum, and L. purpureus. S0, S1, S2, and S3 refer to levels of suppressive species diversity.
While pots received 0.5 litres of water each day, plots received 4 litres per day in the morning. 49–day–old seedlings of Parthenium were plucked from each pot/plot without injuring the roots to investigate the test plant's suppressive effect on invasive growth. Growth parameters included stem height, shoot diameter, root length, fresh biomass aboveground (FBAG), dry biomass aboveground (DBAG), fresh biomass belowground (FBBG), and dry biomass belowground (DBBG), as well as total biomass. Seedlings were washed to remove dirt before being separated into below- and above-ground biomass components. Each component was dried for 12 h in a separate paper bag in a 70°C oven. The stem height (from the soil level to the tip of the highest plant component) and root length were measured with a metre ruler. The shoot diameter (above the first two seedling leaves) and biomass were measured using a digital calliper and an analytical balance, respectively.
Preparation of D. uncinatum leaf crude (DuLc) extract
Fresh D. uncinatum leaves were obtained in the Arusha region from Nkwaranga, Ngiresi, Sokoni, Sura, and Urisho. The leaves were collected early in the morning before sunrise, then air dried for 30 days at room temperature in the shade. It was done so in order to avoid UV light to degraded non-photostable allelochemicals present in the leaves. Non-photostable allelochemicals are substances that are unstable and readily evaporate when exposed to sunlight. The dried leaves were crushed finely and placed in paper envelopes to be preserved. DuLc extract concentrations were prepared based on techniques used by Ngondya et al., 20 i.e., 100 g of powder was soaked in 1 l of distilled water to form a crude extract. In a 4 litre plastic container, DuLc extract was stored in a dark room for 72 h. The extract was filtered using Muslin cloth and the filtrates were diluted with distilled water to create varying DuLc concentrations (100 ml each) relative to the original extract, namely 0%, 25%, 50%, 75%, and 100%.
Parthenium seed germination and seedling growth experiments
Experiments were undertaken in the lab (for petri dishes), pots, and in the field plots to determine the allelopathic effect of DuLc extract on Parthenium seed germination. Each treatment consisted of five glass petri dishes, five plots (1 m2), and five plastic pots (763.82 cm2 surface area) and, which were then replicated five times. After rinsing the petri dishes with distilled water, 25 seeds of Parthenium were sown in each petri dish. In both pots and plots, the same number of seeds were sown. The seeds were kept moist or irrigated with five different levels of DuLc (i.e. 0%, 25%, 50%, 75%, and 100%). Plots were evenly distributed at 0.5 m intervals. Every day for 20 days, the number of seeds that germinated was counted, and the proportion of seeds that germinated was calculated. In the growth experiment, 40 seeds were put in each of 25 pots and 1 m2 field plots. Plots/pots were carefully watered twice a week (0.5 l and 4 l per pot and plot, respectively). To avoid overcrowding, seedlings were trimmed to three per pot after germination. For 25 days, 20–day–old seedlings in each plot and pot were sprayed with 150 ml and 200 ml, respectively, of the different DuLc concentrations (i.e. 0%, 25%, 50%, 75%, and 100%) using a hand sprayer. To evaluate the effects of DuLc concentrations on seedling growth at different concentrations, ten 64–day old Parthenium seedlings per treatment were picked from each plot and three from each pot without harming the roots at the end of the trials. Stem heights and root lengths in centimeters (cm), diameters in millimetres (mm), and biomass in grams (g) were measured as described in section 2.1 above. The positions of petri dishes and pots in all trials were rotated weekly during the experiment to guarantee an even distribution of sunshine and consistent coverage of water.
Statistical analysis
Using one-way ANOVA, all growth parameters (stem height, shoot diameter, root length, FBAG, DBAG, FBBG, DBBG, and total biomass of Parthenium) were compared across suppressive species planting combinations and at varied DuLc extract concentrations. The Shapiro-Wilk test and Levene's test were used to confirm normality and homogeneity of variance, respectively. The post-hoc tests were performed using the Tukey-Kramer HSD test where the main effect terms were significant in ANOVA. Origin (2013) version 9.0 SR1 statistical software was used. Statistical significance was set at p values < 0.05.
Results
Effect of suppressive plants on Parthenium growth vigour
When grown in combinations containing suppressive plants, Parthenium seedlings had lower stem height, root length, stem diameter, and biomass than when grown in monoculture. When grown with L. purpureus, however, these parameters were shown to be significantly reduced (Tables 2 and 3). Overall, the stem height (Pot: F(7, 32) = 9.41, p < 0.0001, Plot: F(7, 32) = 3.26, p = 0.01), shoot diameter (Pot: F(7, 32) = 20.01, p < 0.0001, Plot: F(7, 32) = 1.67, p = 0.0151), and root length (Pot: F(7, 32) = 2.78, p = 0.0224, Plot: F(7, 32) = 11.77, p < 0.0001) of Parthenium seedlings grown with suppressive plants) differed significantly between planting density (Table 3). In pot experiment, the stem height of Parthenium seedlings was 77% (4.5 ± 0.4, p = 0.007) shorter when grown with three suppressive plant species (i.e. PLDB) than when grown in monoculture (Table 2 and 3). Also, it was > 60% shorter when grown with one (L. purpureus: 5 ± 0.6, p = 0.046) or two (L. purpureus and D. intortum: 5 ± 0.2, p = 0.022) suppressive species compared when grown alone (Table 2 and 3). When Parthenium seedlings were grown with suppressive plant species, the stem diameter decreased by > 30% compared to when grown alone (Table 2 and 3). When it was grown with two or three suppressive species, the invasive stem diameter was 79% (PBD: 0.70.2, p < 0.0001), 43% (PLD: 1.8 ± 0.4, p = 0.0019), and 70% (PLBD: 1.2 ± 0.1, p < 0.0001) smaller than when it was grown alone (Table 2 and 3). Furthermore, the invasive seedlings had a shorter root length when they were grown with suppressive plants than when they were grown in monoculture. For instance, when it was grown with three suppressive species (PLDB), Parthenium root length was 64% (4.6 ± 0.6, p < 0.0001) shorter compared to when it was grown alone (Table 2 and 3).
Percentage change (%) of Parthenium seedlings growth parameters when grown with suppressive plant species at different diversity levels in planting mixtures.

FBAG, DBAG, FBBG, DBBG stands for fresh biomass above-ground, dry biomass above-ground, fresh biomass below-ground, and dry biomass below-ground, respectively.
Growth parameters (Mean ± S.E) of Parthenium seedlings grown in monoculture and with suppressive plant species.
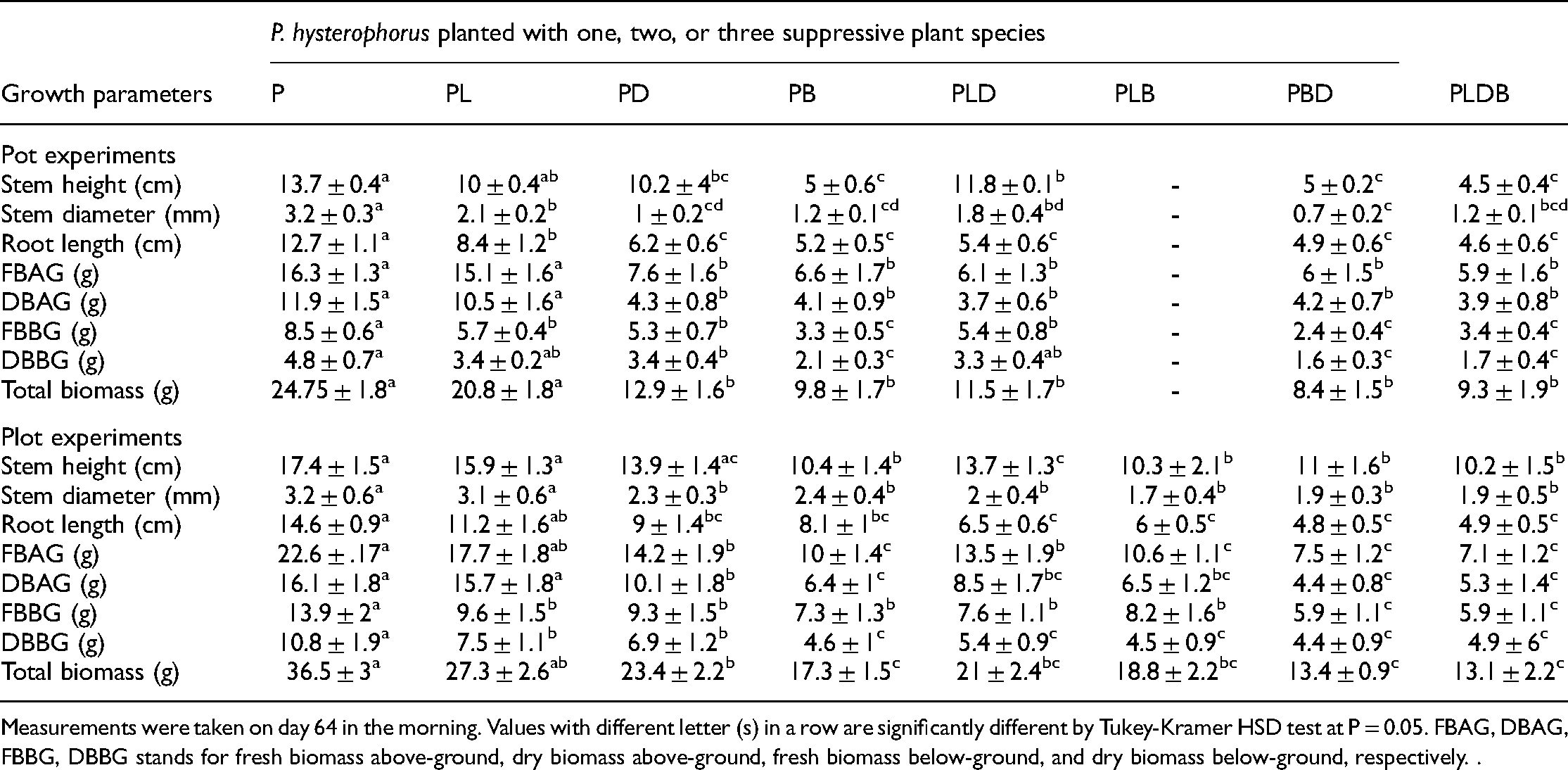
Measurements were taken on day 64 in the morning. Values with different letter (s) in a row are significantly different by Tukey-Kramer HSD test at P = 0.05. FBAG, DBAG, FBBG, DBBG stands for fresh biomass above-ground, dry biomass above-ground, fresh biomass below-ground, and dry biomass below-ground, respectively. .
In plot experiments, Parthenium seedlings' stem height decreased by 51% (PB: 10.4 ± 1.4, p < 0.0001) and 57% (PBD: 10.3 ± 2.1, p < 0.0001), and 41% (PLDB: 10.2 ± 1.5, p < 0.0001) when grown with two or three suppressive plant species than when grown in monoculture (Table 2 and 3). While the root length of Parthenium seedlings decreased by > 50% when grown with two or three suppressive species, it decreased by < 35% when it was grown with one suppressive plant species or alone (Table 2 and 3). Furthermore, Parthenium stem diameter was < 47% smaller when grown with two or three suppressive plant species than when grown in monoculture. In addition, when Parthenium was grown with just one suppressive species (e.g. PB or PD), the diameter was around 25% less than when it was grown alone (Table 2 and 3). Moreover, the average FBAG and DBAG of Parthenium seedlings differed significantly between planting density in pot (FBAG: F(7, 32) = 13.99, p < 0.0001, DBAG: F(7, 32) = 10.33, p < 0.0001) and plot (FBAG: F(7, 32) = 11.68, p < 0.0001, DBAG: F(7, 32) = 9.42, p < 0.0001) experiments (Table 3 and 2). The average fresh biomass below-ground and dry biomass below-ground was significantly different between planting density in both pot (FBBG: F(7, 32) = 22.78, p < 0.0001, DBBG: F(7, 32) = 15.94, p < 0.0001) and plot (FBBG: F(7, 32) = 3.23, p = 0.0105, DBBG: F(7, 32) = 3.85, p = 0.0038) experiments (Table 2 and 3). In general, when planted with two or three suppressive plant species, as well as when grown with L. purpureus alone, the invasive biomass was negatively influenced.
In pots, P. hysterophorus FBAG and DBAG were decreased by > 50% when grown with two or three suppressive plant species compared to when grown alone (Table 2 and 3). Similarly, P. hysterophorus FBBG and DBBG were > 40% lower when grown with two or three suppressive species than when grown in monoculture (Table 2 and 2). In plot experiments, P. hysterophorus FBAG and DBAG were also reduced by > 40% when grown with two or three suppressive species compared to when grown in monoculture (Table 2 and 3). However, when grown with L. purpureus alone, the invasive biomass was reduced by > 50% (Table 2 and 3). When grown with three suppressive plant species, FBAG and DBAG were decreased by > 65% compared to when grown alone (Table 2). Parthenium FBBG and DBBG levels were reduced by > 40% when grown with two suppressive species compared to when grown alone (Table 2). The FBBG and DBBG of Parthenium were > 50% lower when grown with three suppressive species than when grown in monoculture (Table 2). Further, when Parthenium seedlings were grown with a single suppressive species, D. intortum or L. purpureus, FBAG and DBAG were > 35% lower than when grown alone or with M. sativa (PL: 17.7 ± 1.8, p = 0.3615 and 15.7 ± 1.8, p = 1.000, FBAG and DBAG, respectively, Table 2 and 3).
Furthermore, the study found that the total fresh biomass (FBAG + FBBG) of Parthenium seedlings was significantly different between suppressive plants density in pot and plot experiments (F
(7, 32)
= 23.00, p < 0.0001; and F(7, 32) = 12.38, p < 0.0001, respectively, Table 3). Parthenium total biomass was > 50% lower when it was grown with two or three suppressive plat species than when it was grown alone in pots (Table 2). Also, it was > 45% lower when it was grown with one suppressive species (PD: 12.9 ± 1.6, p < 0.0001, and PB: 9.8 ± 1.7, p < 0.0001) than when grown in monoculture or with M. sativa (PL: 20.8 ± 1.8, p = 0.6618) (Table 2 and 3). In plots, total biomass was reduced by > 40% when grown with two or three suppressive species compared to when grown in monoculture (Table 2). Additionally, when the invasive was grown with a single suppressive species (PD: 23.4 ± 2.2, p = 0.0048, and PB: 17.3 ± 1.5, p < 0.0001) the total biomass was reduced by > 35% compared to when it was grown alone or with M. sativa species (PL: 27.3 ± 2.6, p = 0.1016). In each planting combination
Parthenium seed germination
The germination of Parthenium seeds was delayed at higher concentrations (i.e. 75% and 100%) of DuLc extract compared to lower concentrations (Table 4). Under 25% DuLc concentrations and in the control treatment, seedlings had emerged on days 1–4 (Table 4). Under 100% DuL concentrations, Parthenium seed germination was suppressed by 73% in petri dishes (F (4, 20) = 13.88, p < 0.0001), 60% in pots (F (4, 20) = 17.82, p < 0.0001), and 57% in plots (F (4, 20) = 18.73, P < 0.0001) (Table 5). In general, the seed germination inhibition increased with increasing DuLc extract concentration treatment (Table 5).
The number of Parthenium seeds that germinated under different concentration treatments (i.e. 0%, 25%, 50%, 75%, 100%) of Desmodium uncinatum leaf crude (DuLc) extract in pots, plots, and Petri experiments over the period of 20 days. Desmodium uncinatum leaf crude extract delayed P. hysterophorus seed germination at higher concentrations.

Parthenium seeds germination percentages (mean ± se) under different D. uncinatum leaf crude (DuLc) extract concentrations over a 20 days in plots, pots, and Petri dishes experiments.

Values with different letter(s) in a row are significantly different by Tukey-Kramer HSD test at p < 0.05.
Parthenium seedling growth parameters
Parthenium seedlings' stem height sprayed with DuLc differed significantly in both plot and pot experiments (F (4, 20) = 11.21, p = 0.0001, and F(4, 20) = 16.87, p < 0.0001, respectively, Table 6). In pot experiments, stem heights (mean ± se) of Parthenium seedlings under 75% (11.3 ± 0.4 cm) and 100% (12.1 ± 0.5 cm) treatments were approximately 30% shorter than those sprayed with lower DuLc concentrations (i.e. < 50%) in the pot experiments (Table 6). A similar trend was observed in plot experiments when Parthenium seedlings’ stem heights sprayed with 75% and 100% DuLc concentrations were 21 ± 0.2 cm and 20.3 ± 0.5 cm, respectively; this was about 36% shorter than those sprayed with lower DuLc concentrations (i.e. < 70%) and control (Table 6). Parthenium seedlings’ root length sprayed with DuLc extract concentrations differed significantly in both plot and pot experiments (F (4, 20) = 27.80, p < 0.0001, and F(4, 20) = 3.83, p = 0.0181, respectively, Table 6).
Different growth parameters of Parthenium seedlings treated with D. uncinatum leaf crude (DuLc) extract for 25 days in pots and plots experiments under different concentrations.

Values with different letter(s) in a row are significantly different by Tukey-Kramer HSD test at p = 0.05. FBAG, DBAG, FBBG, DBBG stands for fresh biomass above-ground, dry biomass above-ground, fresh biomass below-ground, and dry biomass below-ground, respectively.
In pot experiments, the root length was about 51% shorter when sprayed with a higher DuLc concentration, i.e., 75% (7.7 ± 0.6 cm) or 100% (8.8 ± 0.6 cm) than those sprayed with a lower concentration, i.e., 25% and 50% (Table 6). In plot experiments, the root length of Parthenium seedlings in 50%, 75%, and 100% DuLc concentrations was 45% shorter than those sprayed with 25% DuLc concentration and control (Table 6). The stem diameter of Parthenium seedlings differed significantly under different DuLc concentrations in both experiments (Pots: F(4, 20) = 3.19, p = 0.0351, and Plots: F(4, 20) = 12.26, p < 0.0001, Table 6). In pot experiments, the stem diameter was approximately 31% smaller at higher DuLc concentrations (i.e. 75%: 2.4 ± 0.1 mm and 100%: 2.2 ± 0.2 mm) than those sprayed with 25% and 50% concentrations (Table 6). Similarly, the diameter of seedlings sprayed with 50%, 75%, and 100% DuLc concentrations in plot experiments was slightly smaller than those sprayed with 25% concentration (Table 6).
The average FBAG of Parthenium seedlings differed significantly across different DuLc extract concentrations in both experiments (Plots: F(4, 20) = 3.31, p = 0.031, and Pots: F(4, 20) = 16.16, p < 0.0001) experiments (Table 6). Also, FBBG in both experiments differed significantly between treatments (Plots: F(4, 20) = 51.85, P = 0.031, and Pots: F(4, 20) = 15.95, P < 0.0001, Table 6). In both plot and pot experiments, the seedlings sprayed with a 100% concentration of DuLc extract had lower FBAG and FBBG. Respectively, the FBAG of Parthenium seedlings in pots (10.7 ± 0.9 g) and plots (110.1 ± 6.1 g) was 30% and 33% smaller when it was sprayed with higher DuLc concentrations (i.e. > 70%) than those at lower concentrations (Table 6). Similarly, the FBBG Parthenium seedlings in pots (2.1 ± 0.3 g) and plots (6.2 ± 0.3 g) were 78% and 60% smaller than FBBG at lower DuLc concentrations (Table 6).
In addition, it was found that DBAG of Parthenium seedlings differed significantly under DuLc extract concentrations in both field plots (F (4, 20) = 6.30, p = 0.0019) and pots (F (4, 20) = 42.39, p < 0.0001) experiments (Table 6). Likewise, DBBG in pots (F (4, 20) = 13.19, p < 0.0001) and plots (F (4, 20) = 5.14, p = 0.0052) experiments was significantly different (Table 6). The seedlings sprayed with 100% DuLc concentration had lower DBAG and DBBG in both experiments (Table 6). The DBAG in pots (2.1 ± 0.1 g) and plots (10.1 ± 0.8 g) was 50% and 41% smaller than the DBAG under lower DuLc concentrations, respectively (Table 6). The DBBG of Parthenium seedlings in pots at high DuLc concentration (i.e. > 70%) treatments were 67% (0.7 ± 0.5 g) lower compared with that in lower concentration (i.e. > 70%) treatments (Table 6). With 75% and 100% DuLc concentrations, the DBBG in plots was considerably reduced by 75% (0.7 ± 0.0 g) compared to DBBG in lower concentrations (Table 6).
Discussion
The findings of this study show that suppressive forage species in planting combinations with the Parthenium, as well as DuLc concentrations, inhibited invasive growth and germination. Across a wide range of suppressive forage species and a high proportion of D. uncinatum, Parthenium growth metrics were consistently low. This reveals that D. uncinatum leaves contain active allelochemicals, and that suppressive plants can be utilized to control Parthenium invasions. When a high density of suppressing plants (D. intortum, L. purpureus, and M. sativa) was planted with Parthenium, the growth rate, biomass, stem height, and root length were all reduced. This suggests further that when competitive forage plants are planted alongside Parthenium at high density in an invaded habitat, the invasion may be countered. Previous research showed that when invasive plants (e.g. Parthenium) were planted in a combination with suppressive forage species, their growth vigour was reduced.2,10,18,27
The effectiveness of the test plants to restrict Parthenium seedling growth could be attributed to their ability to compete with the invasive for resources (light, space, moisture, and nutrients). As a result, the resources available to Parthenium were reduced due to their high density. Furthermore, the juvenile seedlings of Parthenium were shaded by a large ground cover and/or broad leaves of L. purpureus, inhibiting their growth. This means that, contrary to prior reports, Parthenium seedlings cannot thrive in the shade.27,28 Though D. intortum can be used to inhibit Parthenium, it should be combined with other legume fodder plants or grasses such as Urochloa mutica, Digitaria eriantha, and Pennisetum clandestinum, as these grass species grow well together.25,29 Medicago sativa, despite its rapid growth rate and high stem height, may not be able to effectively control Parthenium when grown alone. Medicago sativa, on the other hand, can help with Parthenium management when combined with ground cover forming forage grasses. The enhanced suppressive efficacy of L. purpureus may be owing to its high stem height, root system, and biomass, in addition to its ability to generate ground cover. The lack of suppressive ability of M. sativa, on the other hand, could be related to its tiny leaf area and inadequate ground cover.
Also, previous research had found that greater native plant density reduced the abundance of AIPs in pastures.15–17,27 Setaria incrassata, Cenchrus ciliaris, Panicum maxicum, L. purpureus, and Eulalia aurea at high density, for example, reduced Parthenium growth. 27 When the AIP grass Megathyrsus maximus was planted with suppressive plants Myoporum sandwicense, Dodonaea viscosa, and Plumbago zeylanica at higher density, the reproductive tiller and biomass of Megathyrsus maximus were lowered. 17 As a result, increasing plant density and maintaining different forage plant groups in different environments may help to reduce AIP invasion and the number of seeds in the soil. 16
Furthermore, it was found that high concentrations of DuLc (> 70) inhibited the germination of Parthenium seeds and slowed seedling growth compared to lesser amounts. Only high concentration treatments inhibited Parthenium seedling growth, which is consistent with earlier findings.20,30,31 As previously reported in other studies, DuLc has the potential to be used as a bio-herbicide.20,22,32–34 This shows that DuLc may be useful in limiting the spread of Parthenium. Other research reports on the efficacy and bio-herbicide potential of D. uncinatum back up the findings of the current study. For example, Hooper et al. 33 and Pickett et al. 35 found that intercropping D. uncinatum with grains, such as sorghum, maize, and millet, inhibited the weed Striga hermonthica by allelopathy. Isoschaftoside, a C-glycosylflavonoid, 33 and three isoflavanones are among the allelochemicals found in D. uncinatum.32,33 These allelochemicals may be present in DuLc as well, and are responsible for Parthenium germination and growth inhibition. Nonetheless, understanding the efficiency of certain allelochemicals found in D. uncinatum leaf exudate requires identification of individual allelochemicals.
Allelochemicals found in DuLc extract have been shown to reduce the ability of Parthenium seedlings to compete with other plants. However, Parthenium, like other AIPs, may not be eradicated by a single treatment alone, necessitating coordinated management measures.18,36 Other management measures, like traditional weeding and chemical approaches, should be used with a broader strategy, according to this. In addition, biological control agents such as Zygogramma bicolorata, 18 metabolites of fungal species, 37 or bio-herbicides from other native plants could be added to Parthenium management utilizing the DuLc bio-herbicide.1,37 It is important to emphasize, however, that DuLc should be made from D. uncinatum young leaves obtained from several places that are free of pesticide contamination. 38
Overall, this research has shown that suppressive plants and DuLc can be utilized as a management tool to limit or eliminate Parthenium invasion. Both management approaches (using native plants' allelopathic and suppressive potential) are preferred for combating Parthenium because they are technically low-cost (because the plants used are readily available) and environmentally friendly (because they are expected to cause less harm to the environment than conventional herbicides). They can also be utilized as feed, ground cover to prevent erosion and biodiversity protection. However, local people must be included in these management approaches in order to achieve long-term sustainability in controlling Parthenium. They may be taught and aided in the planting of suppressive fodder species, as well as the preparation and application of plant crude extracts with bio-herbicide potential. Furthermore, to achieve effective Parthenium management, a coordinated national plan to combat AIPs is required.
Conclusions
Parthenium was found to be vulnerable to competition from suppressive plants and allelochemicals from DuLc, especially at increasing density and concentrations. Planting combinations with varying levels of planting density and DuLc concentrations had a negative impact on Parthenium germination and growth. Because of their detrimental influence on seed germination and seedling growth, suppressive fodder species and native or naturalized allelopathic plants can be efficiently employed as AIP management tools for Parthenium. However, the study suggests that appropriate management strategies be integrated for effective Parthenium control. The study further suggests that more research be done to find D. uncinatum allelochemical components that are ideal for large-scale, low-impact deployment in Parthenium management. As a result, the study indicates that D. uncinatum, which has bio-herbicidal capacity, can be used in sub-Saharan Africa to manage Parthenium, which poses a danger to natural and agricultural ecosystems, biodiversity, and local communities' livelihoods. In general, additional research is needed to uncover new native-wild plant species with suppressive capacity and bio-herbicide potential for the management of biological invasion.
Footnotes
Acknowledgements
The author acknowledges the support of African Centre of Excellence, “Centre for Research, Agricultural Advancement, Teaching Excellence and Sustainability in Food and Nutritional Security (CREATES)”, in the School of Life Sciences and Bioengineering at the Nelson Mandela Institution of Science and Technology (NM - AIST) in Arusha, Tanzania, for funding this work. The author also thanks Idea Wild for supporting us with research equipment.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the World Bank through its African Centre of Excellence, “Centre for Research, Agricultural Advancement, Teaching Excellence and Sustainability in Food and Nutritional Security (CREATES)”, in the School of Life Sciences and Bioengineering at the Nelson Mandela Institution of Science and Technology (NM - AIST) in Arusha, Tanzania, for funding this study, and Idea Wild for supporting us with research equipment.
