Abstract
Pb(II) leakage from batteries, dyes, construction materials, and gasoline threaten human health and environmental safety, and suitable adsorption materials are vitally important for Pb(II) removal. Bone char is an outstanding adsorbent material for water treatment, and the effectiveness in Pb(II) removing need to be verified. In this paper, the transport characteristics of Pb(II) in columns filled with a sand and bone char mixture were studied at the laboratory scale, and the influences of the initial concentration, column height, inlet flow rate, and competing ion Cu(II) on Pb(II) adsorption and transport were analyzed. The Thomas and Dose-Response models were used to predict the test results, and the mechanisms of Pb(II) adsorption on bone char were investigated. The results showed that the adsorption capacity of the bone char increased with increasing column height and decreased with increasing initial Pb(II) concentration, flow rate, and Cu(II) concentration. The maximum adsorption capacity reached 38.466 mg/g and the saturation rate was 95.8% at an initial Pb(II) concentration of 200 mg/L, inlet flow rate of 4 mL/min, and column height of 30 cm. In the competitive binary system, the higher the Cu(II) concentration was, the greater the decreases in the breakthrough and termination times, and the faster the decrease in the Pb(II) adsorption capacity of the bone char. The predicted results of the Dose-Response model agreed well with the experimental results and were significantly better than those of the Thomas model. The main mechanisms of Pb(II) adsorption on bone char include a surface complexation reaction and the decomposition-replacement-precipitation of calcium hydroxyapatite (CaHA). Based on selectivity, sensitivity, and cost analyses, it can be concluded that bone char is a potential adsorbent for Pb(II)-containing wastewater treatment.
Introduction
With the rapid development of the social economy, industrialization and urbanization has accelerated. As a result, resource shortages, environmental pollution, and ecological deterioration have become more intense. In particular, heavy metal pollution has attracted a substantial amount of attention because it is highly hazardous. Lead (Pb), one of three heavy metal endocrine disruptors, has long been used in industrial products such as batteries, dyes, construction materials, and gasoline, and it is widely distributed in soil, water, and atmospheric environments. It can enter animal and human bodies through the food chain and drinking water, thereby affecting human health and environmental safety.1–3
Many techniques (chemical precipitation, membrane filtration, liquid-liquid extraction, ion-exchange and adsorption) have been used for heavy metal ion removal, and their numerous drawbacks include high toxicity, detection limits, operating complexity, and costs. However, adsorption is recognized as one of the most promising key methods due to its advantages, such as cost-effectiveness, simple operation, environmental friendliness, facile handling, and the availability of various adsorbent materials. 4 Awual et al.5–10 found that ligand-functionalized composite materials are effective, low cost, and recyclable adsorbents, and their potential in Pb(II), Co(II), P(V), Cu(II), and Cd(II) removal application was confirmed. Jiang et al. 11 found that the maximum adsorption capacities of amine-modified, biopolymer-derived core adsorbents for Cr(VI), Pb(II), and Cu(II) reached 497.1, 535.6, and 163.7 mg/g, suggesting that the adsorbents were effective for both anionic and cationic metal ions. Jangde et al. 12 noted that a xanthate-modified apple pomace (XMAP) adsorbent had a high capacity and was easy to recovery, eco-friendly, and effective for Pb(II) removal. Xu et al. 13 used tetraethylenepentamine modified bagasse (AM-BS) and phosphoric acid-modified bagasse (PA-BS) as adsorbents, and found that the modified biosorbents exhibited great potential for Cu(II) and Pb(II) multi-metal recovery. Xiong et al. 14 determined that phosphoric acid-modified sugarcane bagasse (PA-SCB) could adsorb 73.7 mg/g Pb(II), 4.4 mg/g Cu(II), 0.4 mg/g Cd(II), 0.3 mg/g Zn(II), 0.2 mg/g Ca(II), and 0.03 mg/g Mg(II), suggesting that Pb(II) could be efficiently removed from the multi-metal system. Amiri et al. 15 used ostrich bone ash (OBA) modified with nanoscale zerovalent iron (nZVI) particles as a composite adsorbent (OBA/nZVI) and concluded that it effectively removed Hg(II) and Pb(II) ions in the pH range of 5–9. Through fixed-bed adsorption tests, Tsai et al. 16 found that the maximum Pb(II) adsorption capacity of chitosan-coated bentonite (CCB) reached 13.49 mg/g when the initial concentration was 200 mg/L, the bed height was 2.0 cm, and the flow rate was 0.4 mL/min. Khan et al. 17 found that the Pb(II) adsorption capacity of multiwalled carbon nanotube-polyurethane (MWCNT/PU) reached 270.27 mg/g, and the breakthrough capacity was 239.05 mg/g, suggesting that the composite adsorbent was effective for Pb(II) removal. Mitra et al. 18 discovered that the maximum adsorption capacity of hyacinth roots increased with increasing flow rate and initial Pb(II) concentration, whereas decreased with increasing bed height. Gong et al. 19 noted that graphite oxide-coated sand (GO-sand) is an effective adsorbent for Pb(II) and methylene blue (MB), and the exhaustion time for MB adsorption increased, whereas that for Pb(II) adsorption decreased significantly in Pb(II)-MB blend solutions.
Bone char is an outstanding adsorption material for water treatment, and its effectiveness in removing toxic materials has been confirmed by researchers. Wang and Luo 20 found that the Pb(II) adsorption capacities of bone char and cellulose-modified bone char (MCC-BC) reached 89.9 and 115.7 mg/g, showing that modified bone char is an efficient adsorbent for Pb(II) removal. Begum et al. 21 found that the maximum As(V) adsorption capacity of cattle bone char was 0.13 mg/g, and the highest removal rate of 62% was achieved after 3 days of treatment. Villela-Martinez et al. 22 studied the adsorption of As(V) and fluoride on bone char and found that both pollutants could be effectively removed by bone char. Furthermore, the presence of As(V) did not affect the fluoride adsorption capacity of bone char. Ferreira et al. 23 measured Cu(II) removal rates of 36.2% and 97.3% in single and binary systems, respectively, suggesting that Cu(II) has a higher affinity to bovine bone char. Liu et al. 24 investigated the adsorption characteristics of Cu(II) on modified pig bone char (PBC), and found that the Cu(II) adsorption capacity of PBC@SiO2-NH2 was 30 mg/g, suggesting that PBC could be a promising adsorbent for heavy metals after specific functionalization. Sellaoui et al. 25 indicated that in both single and ternary systems, bone char more effective removed Cu(II) than Cd(II) and Zn(II), which were adsorbed to a lower extent. Through fixed-bed column tests, da Silva et al. 26 investigated the adsorption equilibrium of Zn(II) and Cd(II) on bovine bone char and found that the optimal flow rate for minimizing the diffusional resistances of the bed was 4 mL/min for different concentrations of Zn(II) and Cd(II). Rojas-Mayorga et al. 27 revealed that proper pyrolysis conditions could improve the metal uptake of bone chars by 143% and found that the adsorption capacities ranged from 68.3 to 119.4 mg/g, with a removal rate trend of Cd(II)>Zn(II)>Ni(II).
As the main heavy metal pollutant, Pb(II) is widely distributed in soil, water, and atmospheric environments, thereby threatening human health and environmental safety. Bone char is an outstanding adsorbent with the advantages of a large specific surface area, porous characteristics, good regeneration, wide distribution, and stable physical and chemical properties. However, it has the disadvantage of a strong odor, and it is difficult to degrade under natural conditions. 28 To date, few studies have been conducted to investigate the adsorption characteristics of Pb(II) on bone char, especially in relation to its transport characteristics in fixed-bed column tests. In this study, the adsorption and transport characteristics of Pb(II) in fixed-bed column filled with a mixture of sand and bone char were studied, and the effects of the initial concentration, column height, inlet flow rate, and competing ion Cu(II) on the adsorption and transport performance of Pb(II) were investigated. Furthermore, the Thomas and Dose-Response models were used to predict the test results, and the mechanism of Pb(II) adsorption on bone char was revealed. Based on selectivity, sensitivity, and cost analyses, it can be concluded that bone char is a potential adsorbent for Pb(II)-containing wastewater treatment.
Materials and methods
Test materials
The test materials were fine sand (Fujian ISO standard sand, China) and commercial bone char. After being ground by a high-speed grinder, the bone char consisted of very fine particles and met the test requirements. The main chemical components of the bone char were determined using an X-ray fluorescence spectrometer, as shown in Table 1. The Fujian sand (ISO standard) was passed through a 0.5-mm standard sieve, rinsed with clean water until no impurities were left, dried for 12 h at 105°C, and cooled for use. Based on a gradation test, the uniformity (Cu) and curvature (Cc) coefficients of the sand were calculated to be 1.912 and 0.803, respectively, indicating poorly graded sand. Moreover, the specific weight (Gs) was 2.627, and the maximum (ρdmax) and minimum (ρdmin) dry densities were 1.662 and 1.351 g/cm3, respectively.
Chemical composition of bone char.

Test method
Figure 1 shows the schematic diagram of the column test. Wastewater was pumped from the bottom to the top, and a peristaltic pump was used to accurately control the flow rate. The columns were plexiglass tubes with an internal diameter of 2 cm, an external diameter of 3 cm, and heights (Z) of 10, 20, and 30 cm. The filling material for the columns was 95% sand and 5% bone char by mass. The columns were filled in layers using a dry packing method, and the filling density of the column was controlled at ρb = 1.475 g/cm3 during packing. The masses of the materials required to fill the columns with different heights were calculated and are listed in Table 2.

Schematic diagram of the lab-scale column test.
Filling materials for different bed depths.

The sand and bone char were weighed based on the calculated masses and fully mixed, and then the columns were filled with the mixture in layers. After each layer was added, it was pressed to the same height by a stainless steel compactor to ensure the uniformity of the particles. Two filtration membranes with a pore diameter of 0.22 μm were placed at each end of the column. A geotextile layer of the same diameter was placed between the filtration membranes to prevent the adsorbents from flowing out with the solution, thus ensuring the clarity of the effluent and the accuracy of the measurement results. The encapsulated lower end of the column was connected to a peristaltic pump. After the column was saturated with distilled water at a certain flow rate, a lead solution with a given concentration was introduced by the peristaltic pump at a certain flow rate and flowed through the soil from the bottom to the top. Samples of the effluent solution were collected at the outlet every 30 min, and the concentration of Pb(II) ions was measured using an atomic absorption spectrophotometer. The test was terminated when the ratio of the effluent concentration to the initial concentration reached 0.95. The test was repeated three times under the same conditions, and the average was determined to eliminate experimental error. In addition, a set of blank tests was conducted using sand as the sole filling material to eliminate the influence of the sand on the adsorption of the bone char.
The breakthrough and termination points were defined as the Pb(II) concentration of the solution at Ct/C0 = 0.1 and Ct/C0 = 0.95, respectively. The porosity of the columns was e = Vv/VL. Here, C0 and Ct are the initial and effluent Pb(II) concentrations (mg/L) at each time point, and VL and Vv are the volumes of the column and voids in the column (L), respectively. The porosity of the columns used in the experiment was e = 0.345.
Theory models
Adsorption and transfer theory
In the design of a column, the length of the column should be greater than the length of the mass transfer zone. Because the mass transfer zone occurs inside the column, it is not easy to measure directly. If sampling points are set at different locations within the column, the flow and concentration distribution of the fluid within the column would be affected. Here, the zone corresponds to the length of the column through which mass transfer occurs between the breakthrough and termination points, and tδ denotes the time it takes for the main adsorption mass transfer zone to move along the column. 29

where tb and tx are the breakthrough and termination times (min), respectively.

where D is the height of the adsorbent (cm) and tf is the time required for the initial formation of the main adsorption zone.

where f is the solute adsorption capacity coefficient of the adsorbent in the adsorption zone.

where Vx and Vb are the termination and breakthrough volumes (mL), respectively.

where Q is the maximum adsorption capacity per unit mass of the adsorbent (mg/g).

where S is the saturation rate of the adsorbent (%).
Thomas model
The Thomas model 30 assumes that the reaction driving rate follows a second-order reversible kinetic reaction mechanism and is expressed as

where kTh is the Thomas rate constant (mL/mg min); q0 is the maximum concentration of the solute in the solid phase, that is, the adsorption capacity of the adsorbent (mg/g); m is the mass of the adsorbent (g); F is the flow rate (mL/min); C0 is the initial concentration (mg/L); and t is the time.
Dose-response model
The Dose-Response model 31 can be expressed as follows:

When b = q0m/C0, the above formula can be transformed into a linear form:

where a and b are the model parameters and V is the solution volume.
Results and discussion
Influence of the initial concentration on the adsorption and transport characteristics
To investigate the influence of the initial concentration on the transport of Pb(II), tests were conducted in a column with a height of 20 cm at a flow rate of 4 mL/min, and the initial concentration of Pb(II) was 200, 300, and 400 mg/L. As shown in Figure 2, the Pb(II) breakthrough curve for bone char was consistent with a previously reported fluoride breakthrough curve. 32 When the initial concentration was high, the Pb(II) transport rate in the column was high, and Pb(II) flowed through the column in a short time. The main reason for the accelerated transport of Pb(II) was the high concentration gradient; at a given seepage rate, the transport rate was controlled by the dispersive mass transfer of Pb(II). Furthermore, the Dose-Response model fit the experimental data better than the Thomas model. With increasing the initial concentration of metal ions, both the breakthrough and termination times decreased, and the breakthrough curve became steeper. Tables 3 and 4 show that as the initial concentration increased, the Pb(II) adsorption capacity per unit mass of adsorbent decreased, and the length of the main adsorption mass transfer zone decreased. These results can be explained by the decrease in the available adsorption sites on the adsorbent with increasing initial concentration and time. Furthermore, as the concentration increased, the load rate and the driving force for the mass transfer process increased, leading to a decrease in the ability of the adsorbent to continue to adsorb ions from the solution. The solute permeation theory suggests that due to molecular diffusion, the solute continuously permeates from the surface to the interior, and the instantaneous diffusion rate at the interface is proportional to the concentration gradient at the interface. Therefore, changing the concentration gradient affects the saturation rate and breakthrough time. In other words, the concentration affects the diffusion of the solute, thereby the initial concentration of Pb(II) greatly influenced the adsorption process.
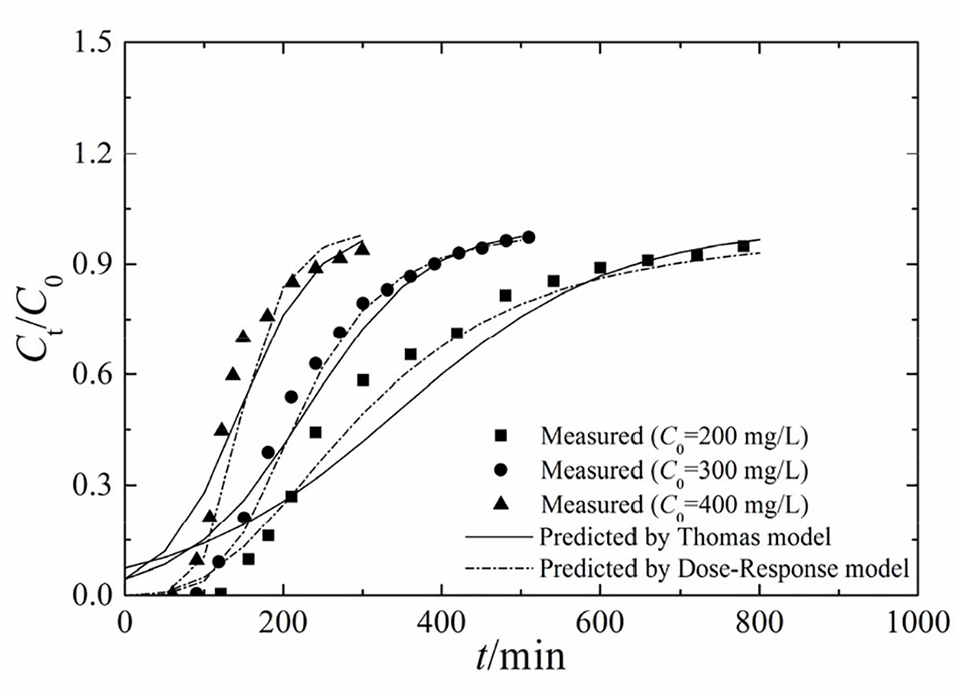
Comparison of the measured and predicted breakthrough curves at different initial concentrations.
Parameters of the Thomas and Dose-Response models at different initial concentrations.

Adsorption and transport parameters at different initial concentrations.

The Pb(II) adsorption performance of different materials were analyzed, and Table 5 summarized the maximum adsorption capacities of the different adsorbents. It can be seen that the Pb(II) adsorption capacity of bone char is inferior to that of conjugate or modified adsorbents, whereas superior to that of natural adsorbents. Overall, it can be concluded that bone char has an excellent adsorption capacity for Pb(II), suggesting that it is suitable for the treatment of Pb(II)-containing wastewater.
Comparison of the Pb(II) adsorption capacities of different adsorbents.

Influence of the column height on the adsorption and transport characteristics
The characteristics of Pb(II) transport in columns with heights of 10, 20, and 30 cm were comparatively analyzed at an initial Pb(II) concentration of 200 mg/L and an inlet flow rate of 4 mL/min. As shown in Figure 3, the Pb(II) and crystal violet dye breakthrough curve trends for bone char are similar. 47 When the column height increased, the Pb(II) transfer time in the column was extended, leading to a longer contact time with the bone char, and the adsorption was closer to equilibrium. The corresponding increase in adsorption resulted in a decrease in the concentration of Pb(II) in the liquid phase, which further lengthened its transport time. On the other hand, the mass of bone char was greater in the longer column, which also increased Pb(II) adsorption and delayed its transport. 48 Furthermore, the Dose-Response model fit the experimental data better than the Thomas model. With increasing column height, the breakthrough curve tended to flatten, and the breakthrough and termination times both increased. When the column height was 30 cm, the curve predicted by the Thomas model deviated significantly from the experimental data points. Tables 6 and 7 indicate that as the column height increased, the adsorption mass transfer zone was expanded, and the adsorption capacity increased. These results can be explained by the fact that the adsorbent amount and the number of adsorption sites on the adsorbent increased with increasing column height.

Comparison of the measured and predicted breakthrough curves for different bed depths.
Parameters of the Thomas and Dose-Response models for different bed heights.
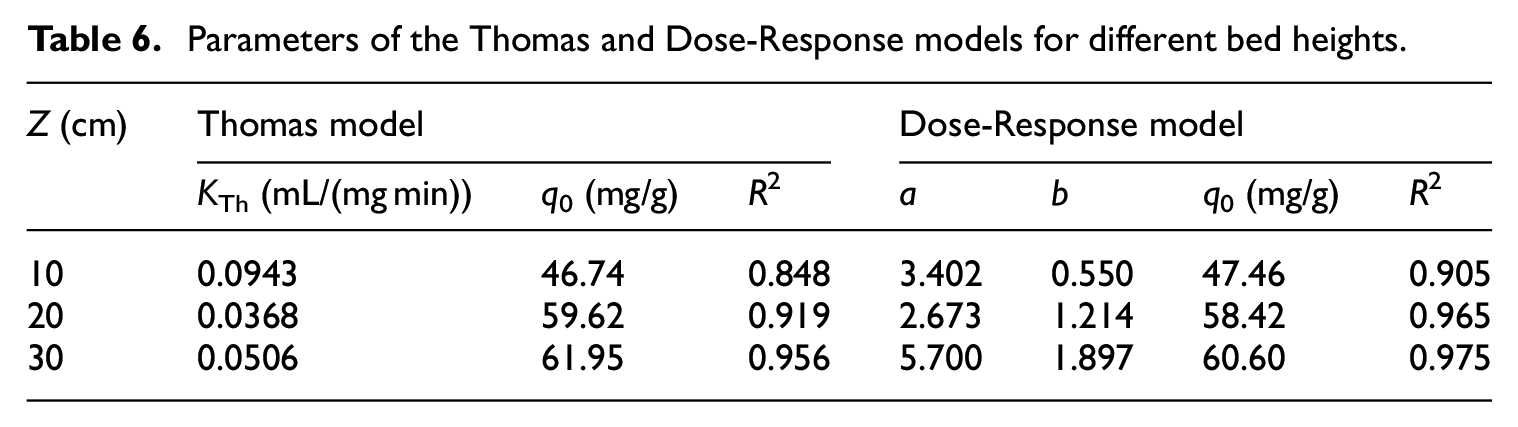
Adsorption and transport parameters for different bed heights.

Influence of the inlet flow rate on the adsorption and transport characteristics
The flow rate is an important factor to consider when evaluating the efficiency of an adsorbent in large-scale industrial wastewater treatment. When C0 = 200 mg/L and Z = 20 cm, the flow rates at the ion inlet were 2.5, 4.0, and 5.0 mL/min; that is, the experiment was conducted under hydraulic loads of 0.795, 1.274, and 1.592 mL/min cm2. As shown in Figure 4, at flow rates of 2.5, 4.0, and 5.0 mL/min, the breakthrough occurred at 360, 150, and 120 min, respectively. Therefore, when the flow rate was low, both the breakthrough and termination times increased, and the breakthrough curve tended to be flat, which is consistent with the adsorption characteristics of Mn(II) on bone char. 49 Furthermore, the Dose-Response model fit the breakthrough curve of the experimental data well. As shown in Tables 8 and 9, the length of the adsorption mass transfer zone and the adsorption capacity of the adsorbent decreased as the flow rate increased. These results can be explained by the fact that when the flow rate was low, the retention time of the adsorbed substance in the column was longer, leading to a longer contact time between the adsorbent and heavy metal ions and, therefore, a higher adsorption efficiency of the adsorbent. The adsorption capacity of the adsorbent easily reached saturation at high flow rates because the adsorption mass transfer space quickly reached the upper part of the column. Ko et al. 50 explained the influence of the flow rate on the kinetic adsorption process from a microscopic perspective and concluded that the change in flow rate would affect ion diffusion but not the adsorbent. When the inlet flow rate was high, the resistance between the liquid and membrane was reduced, and the external mass transport coefficient increased. Additionally, Chen and Wang 51 conducted research on the adsorption kinetics of a column to confirm that both the external mass transport coefficient and the dispersion coefficient increased with increasing flow rate. Therefore, when the flow rate was high, solute diffusion in the pores of the adsorbent was restricted, and the solute retention time in the column was insufficient to achieve equilibrium adsorption. 52 As the flow rate increased, the utilization rate of the column decreased, leading to a decrease in the Pb(II) adsorption efficiency of the bone char.

Comparison of the measured and predicted breakthrough curves at different feed flow rates.
Parameters of the Thomas and Dose-Response models at different feed flow rates.

Adsorption and transport parameters at different feed flow rates.

Influence of competing ions on the adsorption and transport characteristics
To study the competitive adsorption characteristics of Cu(II) and Pb(II) at different initial concentrations, the parameters were set as follows: the flow rate was 4 mL/min, the column height was 20 cm, the Pb(II) concentration was 200 mg/L, and the initial concentration of Cu(II) was 0, 25, and 50 mg/L. The measured and predicted breakthrough curves of Pb(II) adsorption are shown in Figure 5. The coexisting Cu(II) and Pb(II) competed for the adsorption sites on the adsorbent, reducing the breakthrough and termination times. In the Pb(II)-Cu(II) bisolute system, the transport rate of Pb(II) increased as the Cu(II) concentration increased. This result was mainly due to the significant inhibition of Pb(II) adsorption on bone char by Cu(II). As shown in Tables 10 and 11, the influence of Cu(II) on Pb(II) adsorption increased with increasing initial Cu(II) concentration, and the adsorption capacity and main adsorption mass transfer zone decreased.
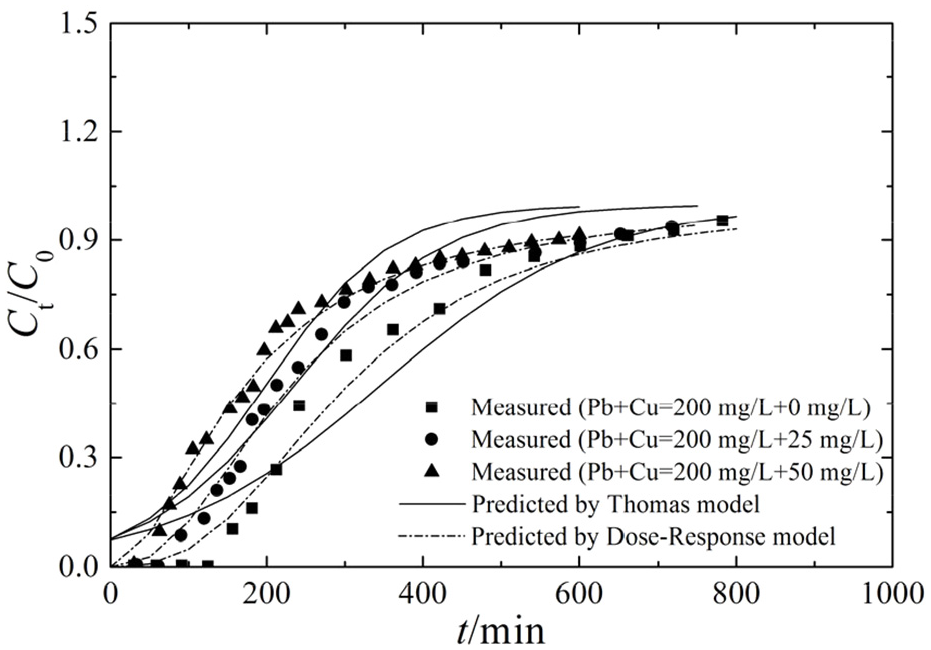
Comparison of the measured and predicted breakthrough curves for competitive adsorption.
Parameters of the Thomas and Dose-Response models for competitive adsorption.

Adsorption and transport parameters for competitive adsorption.

Mechanisms of Pb(II) adsorption on bone char
As shown in Figure 6, the adsorption band at 3425–3570 cm−1 in the infrared spectrum of bone char represents the −OH stretching vibration. After the bone char was loaded with Pb(II), the characteristic peaks at 3425–3570 cm−1 became stronger, indicating that −OH participated in the process of Pb(II) adsorption on bone char. The adsorption bands at 1619, 1460, 1049, and 571 cm−1 were attributed to the C=O, C=C, PO43−, and Ca vibration, respectively. 53

FTIR spectra of (1) unloaded and (2) Pb(II)-loaded bone char.
As shown in Figure 7, the main component of the bone char was calcium hydroxyapatite (CaHA), and the X-ray diffraction (XRD) patterns of the bone char and the bone char loaded with Pb(II) were similar, indicating that no secondary crystalline phase appeared after the bone char was loaded with Pb(II). The mechanisms of Pb(II) adsorption on bone char mainly include a surface complexation reaction and the decomposition-replacement-precipitation of CaHA. 54 The surface complexation reaction can be expressed by the following formula:


X-ray diffraction patterns of (1) unloaded and (2) Pb(II)-loaded bone char.
The decomposition of Ca2+ from CaHA and its replacement by Pb2+ in CaHA precipitates can be expressed as

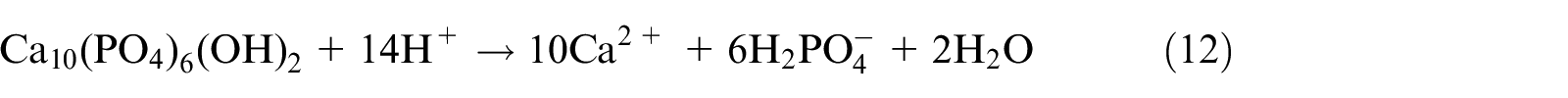
Conclusions
In this study, the effects of the initial concentration, column height, inlet flow rate, and competition ion Cu(II) on Pb(II) adsorption and transport were investigated through fixed-bed column tests. The test results indicated that the adsorption capacity of the bone char increased with increasing column height, whereas it decreased as the initial concentration of Pb(II), the flow rate, and the concentration of added Cu(II) increased. The maximum adsorption capacity reached 38.466 mg/g, and a saturation rate of 95.8% was achieved at an initial Pb(II) concentration of 200 mg/L, inlet flow rate of 4 mL/min, and column height of 30 cm. In the binary competitive system, increasing the Cu(II) concentration lead to decreases in the breakthrough and termination times, and a faster decrease in the Pb(II) adsorption capacity. In addition, the breakthrough curve predicted by the Dose-Response model fit the experimental results well and was significantly better than the predicted results of the Thomas model. Furthermore, the mechanisms of Pb(II) adsorption on bone char mainly included a surface complexation reaction and the decomposition-replacement-precipitation of CaHA. According to the test results with respect to the sensitivity, selectivity, and cost-effectiveness, bone char is a potential adsorbent for tackling the problem of water pollution by Pb(II) in a cheap, easy and ecological way to supply clean water to human society and safe-guarding the public health.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Basic Research Program of Shaanxi Province under Grant (No. 2021JM-535); and Special Fund for Scientific Research by Xijing University under Grant (No. XJ18T01).
