Abstract
In this paper, a series of experiments were carried out to explore the effect of slurry pH on materials removal rate (MRR) and surface quality in lapping of Si3N4 ceramics wafers with fixed abrasive pad (FAP) using different slurries with different pH. Then, the scanning electron microscope was employed to detect the microtopography and abrade smooth of FAP for exploring the effect of slurries pH on self-conditioning of pad. Experimental results demonstrated that the roughness of wafers increase with the increasing of slurry pH value. The surface of wafers is the best when the pH = 1 and the worst When the pH = 13. The MRR decreases firstly and then decreases with the increasing of the pH. The MRR reaches the lowest when slurry pH = 7, and reaches the highest when slurry pH = 13. These results further suggest that the soften layer of SiO2 could be formed due to the reactions between water and materials on wafer surface, which facilitates increasing material remove rate and improving the surface quality. The hydrogen ion and triethanolamine in slurry could react with the copper in fixed abrasive pad, which also could enhance the materials remove rate and affect the surface quality.
Introduction
The Si3N4 ceramics is functional material with excellent properties, including high hardness, high strength, and good wear resistance and so forth. Thus, it is widely used in machinery, communication, aerospace, microelectronics and other fields to prepare high precise bear, precision steam nozzle and other high-performance devices.1–3 However, Si3N4 ceramics is easy to appear collapse and surface defects on processing due to its high hardness and brittleness, which can’t meet the requirements of devices for high precision and good surfaces quality. So, how to get smooth surface efficiently is still challenges in processing the Si3N4 ceramics work pieces.
The lapping and polishing with loose abrasive are traditional processing methods to get the smooth surface of work piece. The materials are removed by free abrasives with decreasing particle size step by step, and the better surface quality will be gotten at the end.4,5 So, it drew many researchers’ attentions6–8 to use the loose abrasive for processing the Si3N4 ceramics work pieces. Wan et al.
9
optimized the process parameters for chemical mechanical polishing of silicon nitride (
In order to get the good lapping process of Si3N4 ceramics lapping with FAP, this paper try to take the experiments with FAP, and get the materials remove rate and surfaces quality of slurries with different pH. The scanning electron microscope was also employed to detect the micro topography and abrade smooth of fixed abrasive pad to explore the effect of slurries pH on self-conditioning of pad. These works would provide the theoretical and experimental basis for the preparing the lapping slurry and obtaining optimized process of Si3N4 ceramics with FAP.
Materials and method
Lapping experiments
The lapping and polishing machine of ZYP300 (Maike Company, Shenyang) was employed to process square silicon nitride ceramic samples (Which mechanical properties are shown in Table 1) with side lengths of 50 mm and a thickness of 1.7 mm for lapping experiments, the processing principle is shown as Figure 1. The diamond particles with a size of W7 (The average diameter of abrasive particle =7 μm) was chose as the abrasive of FAP (which compositions and concentrations are shown as in Table 2). The detailed process of preparation FAP was introduced in Xiao-xiao et al. 19 Before experiments, samples were rough lapped with FAP of W14 (The average diameter of abrasive particle = 14 μm) diamond under the following lapping parameters: platen speed of (ω1 = ω2) 80 r/min, lapping pressure of 43.2 kPa, slurry of deionized water, the offset between the center of platen and the center of work-pieces is 40 mm, slurry feed speed of 60 ml/min and lapping time of 10 min. This work could ensure remove marks of roughing grinding on samples surface and keep the consistent surface condition of samples. After rough lapping, the surface roughness of samples is about Ra 100 nm, and the morphology and microstructures of their surface is shown in Figure 2. In order to maintain consistent surface properties of FAP, the surface of FAP was dressed under the same conditioning parameters for 20 min by using the same diamond dresser before each lapping experiment.
The mechanical properties of work pieces.


The processing principle of the lapping and polishing machine ZYP300.
The compositions and concentrations of FAPs used in lapping experiments.


The morphology and microstructure of sample before experiment.
Ingredients of different lapping slurries are shown in Table 3, their pH values were adjusted by adding the phosphoric acid (H3PO4) and the triethanolamine (C6H15NO3, TEA). Samples were lapped by using the different slurry under the following conditions: platen speed was (ω1 = ω2) 80 r/min; Lapping pressure was 43.2 kPa; the offset between the center of platen and the center of work pieces was 40 mm; slurry feed speed was 60 ml/min and lapping time was 60 min.
pH and ingredients of slurries in experiment.

Every sample was cleaned in the ultrasonic cleaning machine for 5 min to remove off the debris and dust on surface before and after lapping experiment. After that, the weight of every sample was measured on the precision analytical balance of Sartorius CP225D (Precision: 0.01 mg), every sample was measured five times and averaged the results. After lapping experiment, the MRR (MRR, nm/min) was calculated according to the equation (1).

Where, Δm is the weight of sample after lapping (g), ρ is density of Si3N4 ceramics sample (g/mm3), a is the side length of the square sample (mm), t is the lapping time (min).
The surface morphology of samples and FAP detecting
The surface roughness and microstructure of ultrasonic cleaned samples were measured by using the optical contour testing system of CONTOURGT-K (Bruker Company, Precision: 0.1 nm) after lapping. Every samples were measured for five times on different positions (shown in Figure 3), and the average value was calculated as the roughness value. The 2D microstructure image and 3D morphology image of every sample were saved by using the optical contour testing system.

The different measuring positions on samples surface.
Before lapping experiment, the FAP was dressed by the diamond dresser under the same condition. After lapping, a square sample of size 2 mm×2 mm was got from the FAP surface by using the cutting tool. Because the microstructures and morphology of FAP could be estimated by using the scanning electron microscope (SEM). 20 Then, the microstructures of every FAP samples was measured by using SEM, and SEM work parameters is shown in Table 4. Their microstructures image were dealt by using the software of Photoshop for calculating their worn ratio, and the processing flow is shown as Figure 4.
The work parameters of SEM.


The processing flow of figure by using Photoshop.
Results and discussion
The surface quality
Figure 5(a) shows that the roughness value of sample increases with the pH value of lapping slurry when lapping the Si3N4 ceramics with W7 FAP. When the lapping slurry pH = 1, the surface quality of lapping sample is the best and its roughness is only Ra = 37.8 nm. When the lapping slurry pH = 13, the surface quality of lapping sample is the worst and its roughness reaches to Ra = 58.3 nm. This is because of the soft layer formation on samples resulted by the chemical actions of slurry, and the soft layer could enhance the critical cutting depth of sample and improve the surface quality. Additionally, the chemical effect of lapping slurry on the matrix of FAP could promoting the self-conditioning of FAP, enhancing the extruding height of abrasive and increasing the average cutting depth, which would increase the roughness of sample surface. The surface quality of sample after lapping depends on the combination of the above two roles.
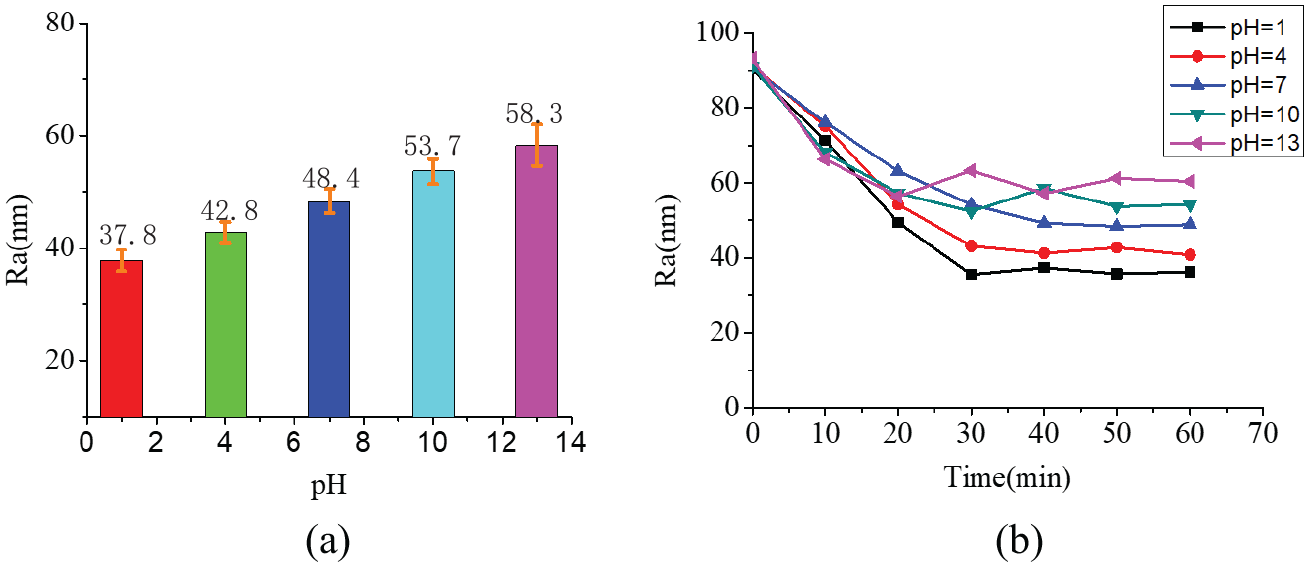
The surface roughness of work-pieces and changing over time lapping with different slurries: (a) the surface roughness of work-pieces and (b) the surface roughness of work pieces changing over time.
Figure 5(b) shows that the roughness of samples surface is about Ra 90 nm at lapping beginning, the bump on the surface of samples are removed gradually in lapping and the roughness value of samples surface are gradually decreased. At the beginning of lapping, the surface roughness decline velocity of the sample processed with slurry pH = 1 is the maximum, followed by pH = 4, pH = 13, and pH = 10. The surface roughness decline velocity of the sample processed with slurry pH = 7 is the minimum. After lapping for 30 min, the surface roughness value of samples processed with slurries besides slurry pH = 7 stops going down and reaches a relatively stable value, which indicates that the surface layer formatted resulting by rough lapping has been completely removed. The surface roughness value of samples processed with slurry pH = 7 reaches a relatively stable value after 50 min lapping. This is because the MRR of sample processed with slurry pH = 7 is the smallest, the surface roughness value reaches stable need the longest time.
Figures 6 and 7 shows the 2D and 3D surface topography of samples after lapping 60 min with different slurries. Among them, the surface of the sample processed with the slurry pH = 1 is the smoothest, and followed by pH =4, pH = 7, pH = 10, and pH = 13. There are only a few tiny scratches on the sample surface processed with slurry pH = 1, but many tiny scratches and a few of rough scratches on the sample surface processed with slurries of pH = 4, pH = 7, and pH = 10. The surface quality of that with slurry pH = 13 is the worst, who was covered by the rough scratches and big pits resulted by brittle removing of material. The results present that the extruding height of abrasive on FAP surface when lapping with slurry pH = 13 is taller than other slurries, and this would result the bigger cutting depth and formation of rough scratches and big pits.

The 2D surface topography of work piece after lapping 60 min with different pH slurries: (a) pH = 1, (b) pH = 4, (c) pH = 7, (d) pH = 10, and (e) pH = 13.
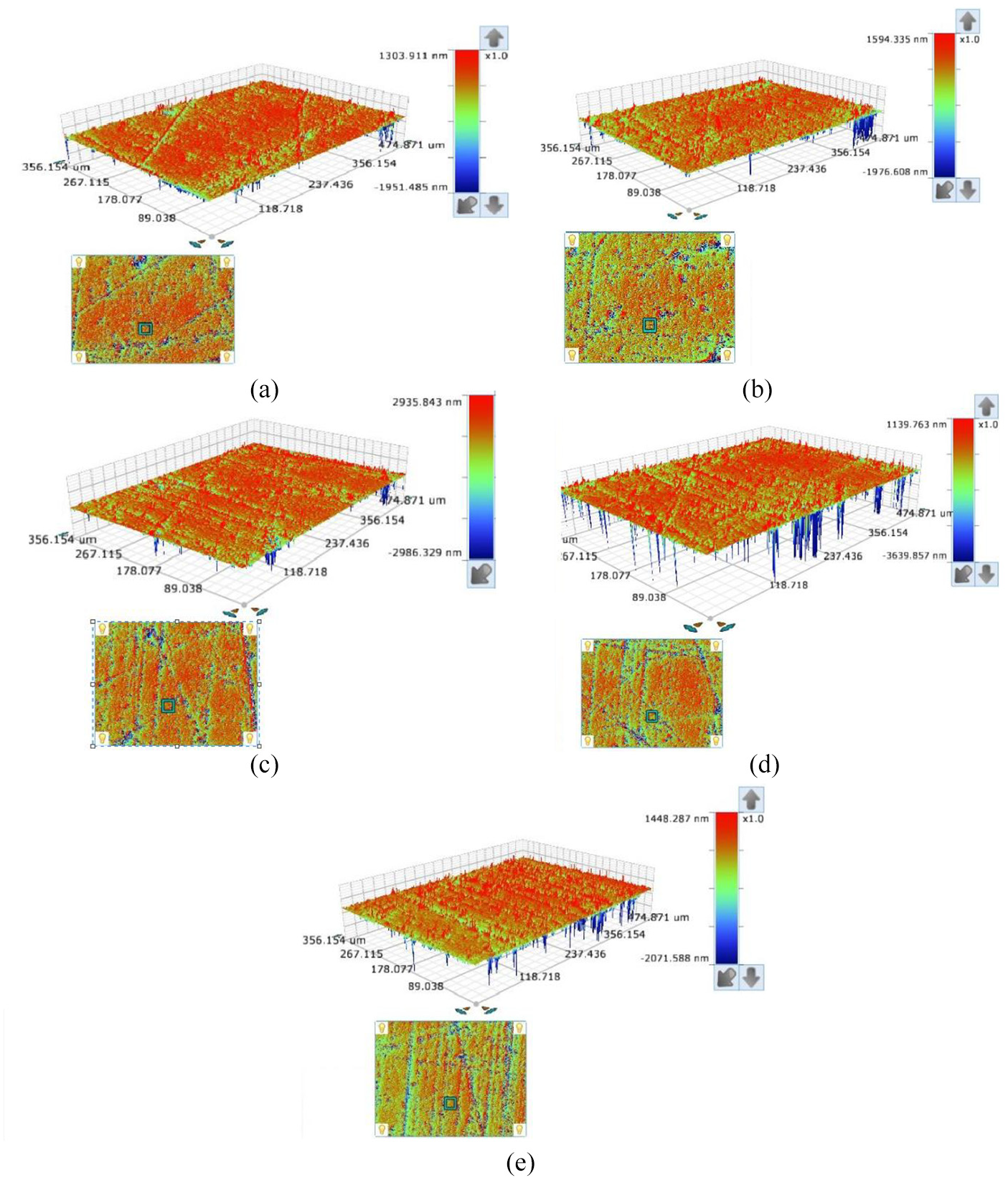
The 3D surface topography of work piece after lapping 60 min with different pH slurries: (a) pH = 1, (b) pH = 4, (c) pH = 7, (d) pH = 10, and (e) pH = 13.
The instant high temperature would be formed owing to the extrusion between the FAP and work piece when lapping,21,22 the chemical reaction expressed as the equation (2) 23 would be occurred between the materials on work piece surface and the soften layer formed on work piece surface. When the lapping slurry is acidic, the phosphoric acid added in slurry could speed up the chemical reaction as the catalyst, it also facilitates the forming of the soft layer SiO2 and helps removal the materials on work piece surface.

In chemical mechanical processing, the surface quality depends on not only the average cutting depth of abrasives, but also the removal mode of materials. When the average cutting depth is deeper than the critical cutting depth of brittle-ductile transition, the most of material will be removed by brittle fracture mode, and the surface quality will become worse. In contrast, most material will be removed by ductile removal mode, and the surface quality will be better. The softened layer could be formed because of the chemical effect of slurry, so the critical cutting depth of brittle-ductile transition would be enhanced and the surface quality would become better. Under the acid condition, phosphoric as catalyst, the chemical reaction shown as equation (2) will be accelerated because of the catalyst of the phosphoric acid. Therefore, the surface quality of work piece processed with the acid slurry is better than that with the neutral and alkaline slurry. This is consistent with the experimental results shown in in Figures 5–7.
The MRR
Figure 8(a) presents that the MRR decreases with the increase of pH value of slurry when the pH is less than 7, and the MRR is the lowest and only 40.21 nm/min when pH = 7. On the contrary, the MRR increases with the increase of pH value of slurry when the pH is more than 7, and the MRR is the highest reaching 95.43 nm/min when pH = 13.This is because that the materials removal of work piece mainly depends on the mechanical action of abrasive in lapping and polishing with FAP. The soft layer on work piece surface would be formed owing to the chemical effect of slurries, its hardness is softer than that of work piece materials and could enhance the cutting depth of abrasive. 17 Additionally, the chemical effect of lapping slurries on the matrix of FAP could enhance the self-conditioning of FAP, it could increase the extruding height of abrasive 20 and MRR of work piece.

MRR and changing over time lapping with different slurries: (a) the MRR and (b) the MRR changing over time.
To explore the performance change of FAP over time, the MRR at different time was analyzed, and the change over time lapping with different slurries is calculated, which is shown as Figure 8(b). The results show that the cutting performance of FAP is well at lapping beginning, and the MRR are both higher than 100 nm/min. With the lapping continues, the cutting performance of FAP is sharply become worse, and the MRR is acutely declined. This is owing to the worn of heave on FAP surface and diamond abrasive, resulting in FAP and workpiece surface roughness decrease.
The MRR is not decline continually after lapping for 30 min when pH = 10 and pH = 13, and stabilize at about 50 nm/min. This phenomenon indicates that the worn and shedding of old abrasive and the exposure of new abrasive reach balance, and the FAP has a better self-conditioning performance under this condition. However, when pH = 7, the MRR is still continued to decline after lapping for 30 min. Until lapping for 50 min, the MRR fell to a stable value and less than 10 nm/min. This displays that the self-conditioning performance of FAP is very bad when lapping only using the deionized water as slurry, and the FAP appears a phenomenon of “glazing” after lapping for 50 min, the cutting performance of FAP is seriously become worse.



When lapping with the acid slurry, the chemical reaction will be slowly occurred between H+ ions, water, the oxygen in slurry and copper powder added in the FAP. The detailed equations are expressed as the equations (3)–(5).24,25 When there are many H+ ions in lapping slurry, the OH– ions will reduce, and it is helpful to occur the chemical reaction expressed by equation (6). This will accelerate the corrosion of copper powder in FAP surface and result the structure loosen of FAP matrix, and it is beneficial to enhance the self-conditioning of FAP matrix and the self-sharpening of abrasives.




When the lapping slurry is alkaline, the complex reaction would be occurred between the triethanolamine added in slurry and the Cu2+ ions,26,27 and the detailed equations are shown as the equations (6)–(9). If the concentration of triethanolamine in slurry is large, the chemical reaction of equation (6) will be easily occurred. So, the copper powder in FAP will be dissolved and corroded, and the self-conditioning and cutting property of FAP will also be enhanced.
Because of the complex reaction of the triethanolamine and the Cu2+ ions, the plenty of Cu2+ ions were consumed, so the corrosion of copper powder in the alkaline slurry was greater than that in the acidic slurry. Therefore, it is deduced that the self-conditioning performance of FAP lapping with the alkaline slurry is better than that with the acidic slurry according to the chemical roles; the bulging height of abrasives on FAP surface lapping with the alkaline slurry is higher than that with the acidic slurry; and the MRR of the alkaline slurry is also higher than that of the acidic slurry. Those also explains the experiment results which were shown in Figure 8.
The microstructures of FAP surface
To explore the change of FAP processing property under different condition, the worn area ratio λ is presented and defined as the following equation (10).
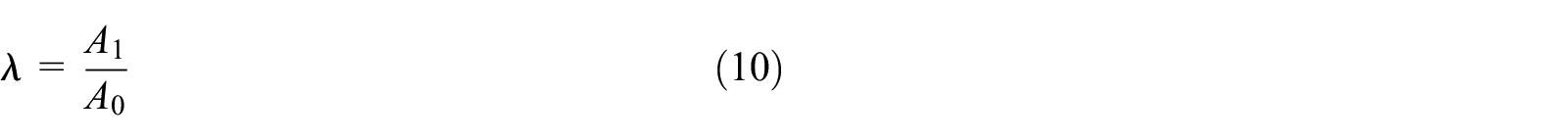
Where, λ is the ratio of worn area, A1 is the worn area of FAP, A0 is the whole area of FAP.
Figure 9 shows the surface microstructures of dressed FAP before lapping experiment. The surface of FAP is very rough, and the conditioning of FAP and the dissolution of pore-forming materials lead to many pits and projections of matrix and abrasive covering the whole surface of FAP. It displays that the FAP has well cutting performance and many big chip pockets at this moment.

The surface microstructures of FAP after conditioning.
Figures 10 and 11 shows the surface microstructures and worn area ratio of pad after lapping 60 min with different slurries. These SEM images dealt by using Photoshop clearly display the worn area on FAP surface (green areas on SEM images shown in Figure 7(f)–(j)). The worn area ratio of FAP under different conditions were calculated according to the SEM images dealt by using Photoshop, and the calculating results is shown in Figure 7(k). These SEM images show that all the FAP surface appear some degree of bluntness and abrasion after lapping for a long time, also the processing property of FAP becomes worse and chip pockets get smaller. According to the bluntness and abrasion of FAP surface after lapping with different slurries, the arrangement of slurries is as follow: pH = 7, pH = 4, pH = 1, pH = 10, and pH = 13. Among them, the bluntness and abrasion of the FAP after lapping with slurry of pH = 7, is the most serious. The most projections of matrix and abrasive are disappeared, the surface of FAP becomes smoother, most of pits on FAP surface are plugged up by debris, and the big worn area of FAP appears on surface. This indicates the cutting performance of FAP is seriously declined at this condition. The bluntness and abrasion of the FAP after lapping with slurry of pH = 10 is the slightest, the roughness of FAP is no significant different from that before lapping experiment, the pits and projections of matrix and abrasive are also covering the FAP surface, and the worn area on FAP surface are very small. It suggests that both the self-conditioning property and the cutting performance of FAP are well when lapping with slurry of pH = 13 at this moment. The roughness, bluntness and abrasion of the FAP lapping with slurry of pH = 1, pH = 4, and pH = 10 are both between pH = 7 and pH = 13.

The surface microstructures of pad after lapping with different pH slurries: (a) pH = 1, (b) pH = 4, (c) pH = 7, (d) pH = 10, (e) pH = 13, (f) pH = 1 after dealt by Photoshop, (g) pH = 4 after dealt by Photoshop, (h) pH = 7 after dealt by Photoshop, (i) pH = 10 after dealt by Photoshop, and (j) pH = 13 after dealt by Photoshop.

The worn area ratio of pad after lapping with different pH slurries.
The discussion on mechanism
In lapping, the effect of lapping slurry on MRR and surface quality could be due to two aspects. On one hand, the chemical interaction between the lapping slurries and the work piece results the soften layer forming on the surface of work piece, which could cause the microstructures of wafer surface changed and lead to the larger critical cutting depth of work piece. These results would improve the surface qualities of work piece in lapping. On the other hand, the chemical interaction between lapping slurries and the additives in FAP could lead to dissolution of additives, which could improve the self-conditioning of FAP and slow down the blunt of FAP. These results would change the morphology of FAP surface and raise the MRR.
In this study, the acid slurries not only could result the soften layer formation but also lead to the dissolution of adding copper in FAP. However, the alkaline lapping slurries hardly has obvious chemical interaction with work piece material, and it only could result the dissolution of adding copper in FAP. The effect mechanism of slurries on lapping process is shown as Figure 12. So, the surface quality of lapping with acid slurries are better than that of lapping with alkaline slurries, and the lower the pH, the better the surface quality, these well explain the experiment results in Section 2.1. Because the dissolution of copper resulted by the acid slurry is slower than that resulted by the alkaline, so role of FAP self-conditioning with acid slurry is weaker than that with alkaline slurry. So, the blunt of FAP surface lapping with acid slurries are more obvious than that with the alkaline slurries, and the MRR of lapping with alkaline slurries are higher than that of lapping with acid slurries. These well explain the experiment results in Section 2.2 and 2.3.

The effect mechanism of slurries on lapping process.
Conclusion
The roughness value of sample increases with the pH value of lapping slurry when lapping the Si3N4 ceramics with W7 FAP. When the lapping slurry pH = 1, the surface quality of lapping sample is the best and its roughness is only Ra = 37.8 nm. When the lapping slurry pH = 13, the surface quality of lapping sample is the worst and its roughness reaches to Ra = 58.3 nm
The MRR decreases with the increase of pH value of slurry when the pH is less than 7, and the MRR is the lowest, only 40.21 nm/min when pH = 7. On the contrary, the MRR increases with the increase of pH value of slurry when the pH is more than 7, and the MRR is the highest and reaches 95.4 nm/min when pH = 13.
The acid lapping slurries in the experiment could speed up the chemical reaction as the catalyst and facilitate the forming of the soft layer SiO2, which helps removal the materials on work piece surface.
The OH– ions in the alkaline lapping slurries could react with the copper particles added in the FAP, which could improve the self-conditioning, reduce the blunt of FAP, and thus improve MRR.
All the FAP surface appear a certain degree of bluntness and abrasion after lapping for a long time, the processing property of FAP becomes worse and chip pockets get smaller. According to the bluntness of FAP surface after lapping, the surface quality and the MRR with different slurries, the optimal lapping slurries is the alkaline slurry of pH = 13.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is financially supported by the National Science Foundation of China (U1804142), Project funded by China Postdoctoral Science Foundation (2020M672220), Science and technology plan projects of Henan province (192102210216) and the young backbone teachers’ project of Henan province (No.2019GGJS167, No.2019GGJS171).
