Abstract
Objective
Electronic cigarette (e-cigarette), or vaping, product use–associated lung injury (EVALI) is a novel noncommunicable disease with an unknown cause. The objective of this analysis was to describe the Minnesota Department of Health’s (MDH’s) outbreak response to EVALI, including challenges, successes, and lessons learned.
Methods
MDH began investigating EVALI cases in August 2019 and quickly coordinated an agencywide response. This response included activating the incident command system; organizing multidisciplinary teams to perform the epidemiologic investigation; laboratory testing of e-cigarette, or vaping, products (EVPs) and clinical specimens; and collaborating with partners to gather information and develop recommendations.
Results
MDH faced numerous investigational challenges during the outbreak response of EVALI, including the need to gather information on unregulated and illicit substances and their use and collecting information from minors and critically ill people. MDH laboratorians faced methodologic challenges in characterizing EVPs. Despite these challenges, MDH epidemiologists successfully collaborated with the MDH public health laboratory, law enforcement, partners with clinical and toxicology expertise, and local and national public health partners.
Practice Implications
Lessons learned included ensuring the state public health agency has legal authority to conduct noncommunicable disease outbreak investigations and the necessity of cultivating and using internal and external partnerships, specifically with laboratories that can analyze clinical specimens and unknown substances. The lessons learned may be useful to public health agencies responding to similar public health emergencies. To improve preparedness for the next outbreak of EVALI or other noncommunicable diseases, we recommend building and maintaining partnerships with internal and external partners.
During summer 2019, a nationwide outbreak of electronic cigarette (e-cigarette), or vaping, product use–associated lung injury (EVALI) emerged. By February 18, 2020, a total of 2807 hospitalized patients with EVALI, including 68 deaths, had been reported to the Centers for Disease Control and Prevention (CDC). 1 During the investigation, it became clear to public health researchers and officials that illicit tetrahydrocannabinol (THC)–containing e-cigarette, or vaping, products (EVPs) were a primary contributor to the outbreak. Multiple laboratory and epidemiologic studies eventually demonstrated a strong link between vitamin E acetate (VEA), an additive used in some THC-containing EVPs, and EVALI. 2 -5
This nationwide outbreak of a novel disease garnered a national emergency response. Although this outbreak was not the first noncommunicable disease outbreak involving multiple states to occur in the United States, the response presented various challenges to public health agencies that were unique to a noninfectious disease investigation. 6 -8 US public health agencies have less experience in responding to noncommunicable disease outbreaks than communicable disease outbreaks. One study that examined the frequency of reports of noncommunicable disease outbreaks in CDC’s Morbidity and Mortality Weekly Report revealed that only 12% of outbreak investigation reports published during January 2011–June 2018 were about noncommunicable diseases 8 ; approximately one-third of these reports were about outbreak investigations involving illicit substances.
Morbidity and mortality from the use of illicit substances have increased substantially during the past decade, and public health agencies have increasingly faced outbreaks associated with adulterated illicit drugs and other substances. Therefore, public health agencies have needed to build their capacity to respond to noncommunicable disease outbreaks. MDH is well-poised to describe its outbreak response, including successes, challenges, and lessons learned, because Minnesota was among the first states to respond to the EVALI outbreak. MDH’s experience and recommendations could help other public health agencies prepare for the next outbreak of EVALI or other noncommunicable diseases.
Materials and Methods
In July 2019, state public health agencies in Wisconsin and Illinois identified patients with symptoms that would soon become associated with this novel disease; all patients reported using EVPs before becoming ill. On August 8, a Minnesota pulmonologist alerted MDH to 4 hospitalized patients who reported using EVPs and had clinical characteristics similar to those reported in Wisconsin and Illinois. MDH issued a health advisory on August 13 requesting reports of patients suspected to have the disease that would soon be known as EVALI.
Organizing and Staffing the Response
MDH began receiving additional case reports soon after publishing the health advisory (Figure 1). From August 8, 2019, through February 28, 2020, MDH received 230 case reports and classified 150 cases as confirmed or probable according to CDC’s case definition. 9 To coordinate the response, on September 6, 2019, MDH activated its incident command system (ICS), 10 the agency’s organizational response to public health emergencies. Although the outbreak was not an infectious disease outbreak, staff members from the infectious disease division led the epidemiologic investigation, given their experience with conducting communicable disease outbreak investigations.
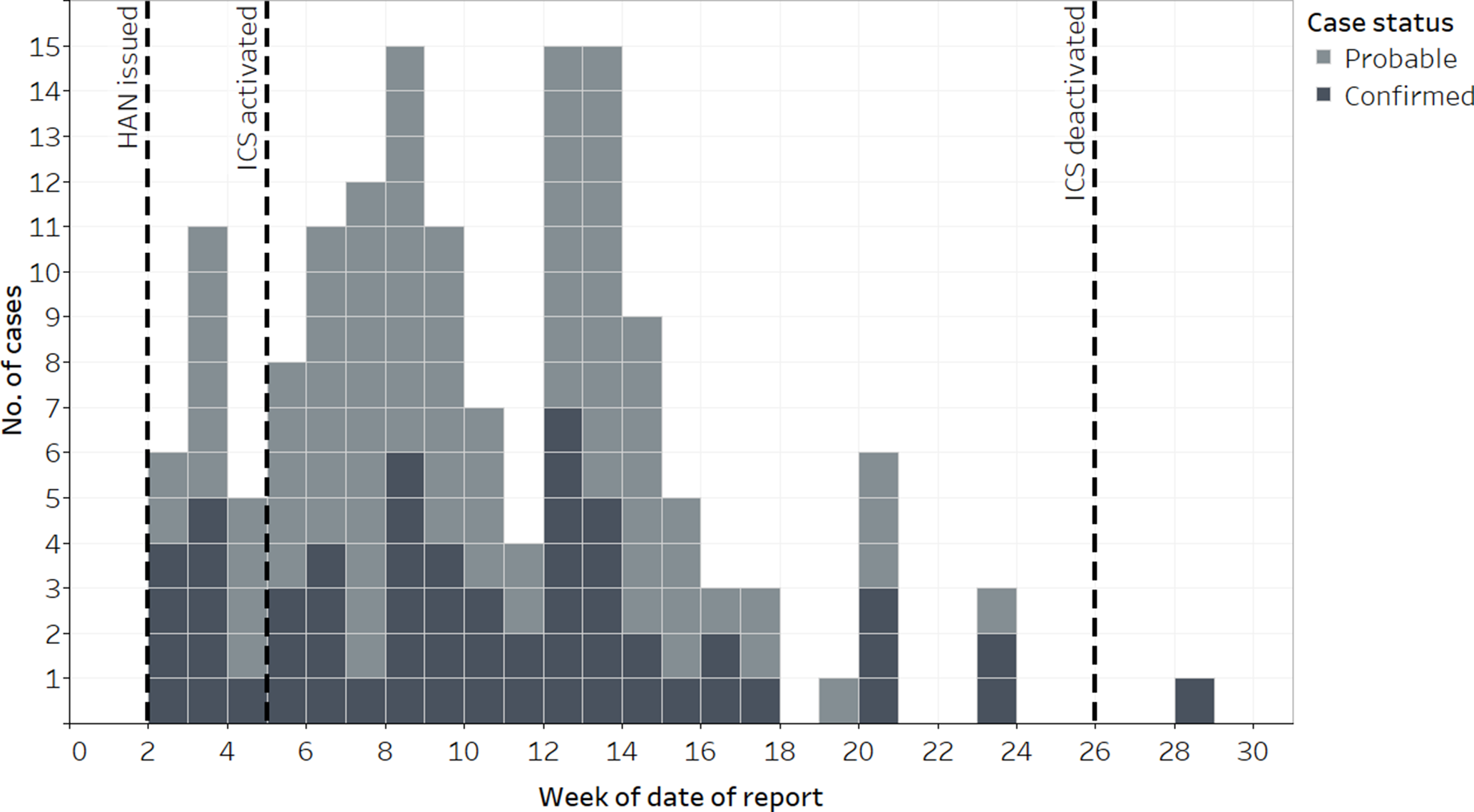
EVALI case reports submitted to the Minnesota Department of Health, by week and case status, Minnesota, August 8, 2019–February 28, 2020. Abbreviations: EVALI, electronic cigarette (e-cigarette), or vaping, product use–associated lung injury; HAN, Health Alert Network; ICS, incident command system. Data source: Minnesota Department of Health EVALI surveillance data.
Activation of the ICS allowed for agencywide coordination of response efforts and prioritization of response needs and activities (eg, staffing, information technology, communications). The ICS developed various operational teams, including teams for managing intake of case reports, interviewing patients, collecting data from patient medical records, analyzing data, obtaining EVPs from patients, and conducting toxicology testing on products and clinical specimens. Given the situational urgency, the ICS issued an agencywide request for MDH staff members to populate these teams. For some roles, ICS recruited staff members with specific or specialized skills (eg, data analysis) to assist. The ICS also coordinated reassigning staff members to the response from their normal duties for an indeterminate period, at either full-time or part-time capacity. Employees involved represented diverse public health disciplines and departmental divisions. In total, 91 staff members from 9 of 13 divisions participated in MDH’s EVALI response. Although the total number of employees involved represented only 5% of the entire MDH workforce, some divisions, particularly the infectious disease division, had a higher percentage of staff members working on the response than other divisions did. In addition, the ICS reassigned staff members in key response roles (eg, epidemiologists, laboratorians) to work on the response full-time for the duration of the outbreak. The response required collaboration internally across divisions and externally with various partners.
Response Demobilization
Case reports began to decrease considerably in Minnesota and nationally in December 2019 (Figure 1). 1 Given this decline, MDH leadership demobilized the ICS in Minnesota on January 31, 2020. Since then, MDH has continued to conduct EVALI surveillance and receive case reports from clinicians of patients hospitalized with suspected EVALI. Although reports were sparse during early 2020, MDH detected a cluster of 12 patients suspected to have EVALI during June 1–August 3, 2020.
Results
Strengths of the Response
Collaboration with internal and external partners was crucial to the investigation. Given the substantial number of internal and external participants, effective communication and information sharing among multiple teams at MDH, external partners, and the public were essential to response success (Figure 2).
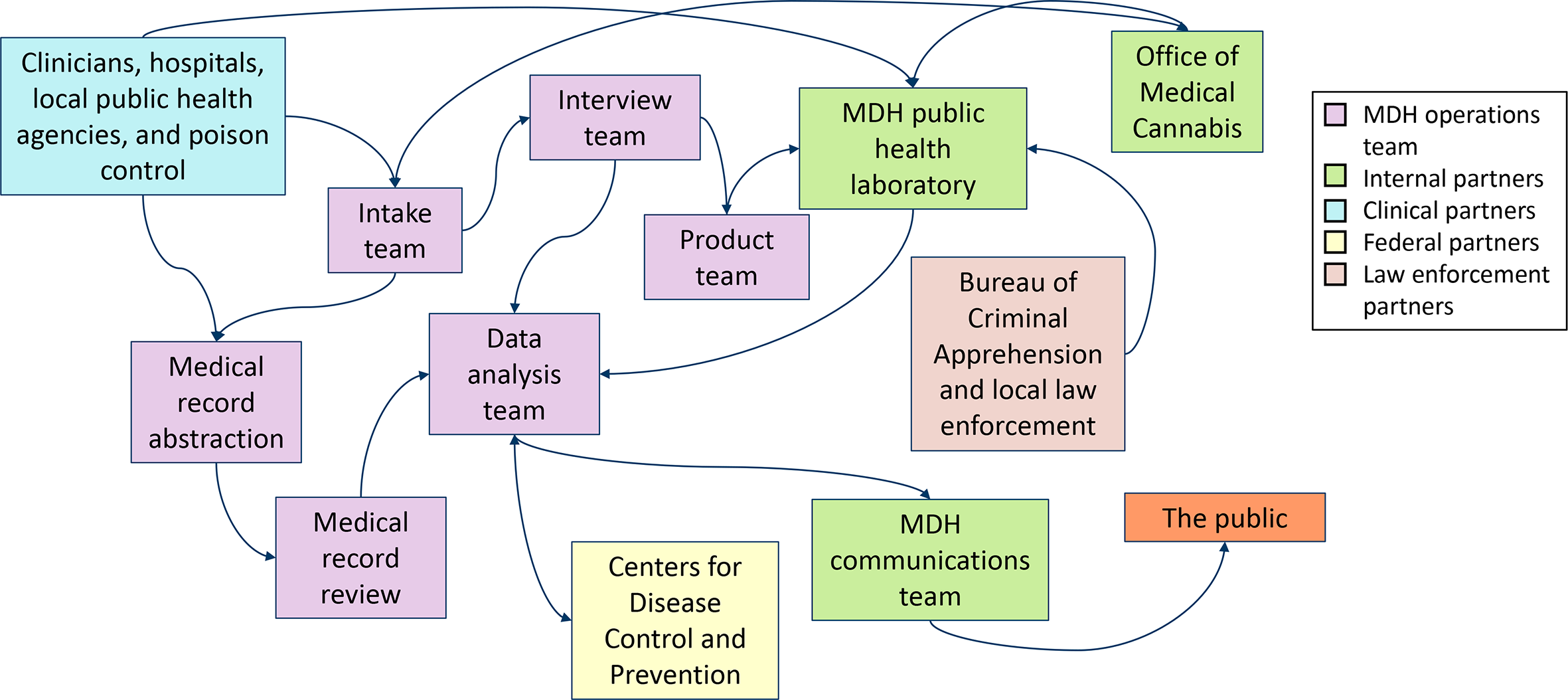
Flow of information during an EVALI outbreak investigation, Minnesota, 2019-2020. Abbreviations: EVALI, electronic cigarette (e-cigarette), or vaping, product use–associated lung injury; MDH, Minnesota Department of Health.
MDH’s public health laboratory was a crucial internal partner throughout the investigation. This laboratory is 1 of 10 Level 1 laboratories in CDC’s national Laboratory Response Network; these laboratories are tasked with responding to chemical-related incidents of public health importance. 11 As such, MDH’s public health laboratory has developed the highly specialized capacity to perform testing on various substances for contaminants of emerging concern. In addition, the laboratory routinely collaborates with MDH epidemiologists to investigate drug overdoses, including performing toxicology testing of clinical specimens through the Minnesota Drug Overdose and Substance Use Pilot Surveillance Activity. Therefore, MDH laboratorians had the necessary expertise and equipment to test clinical specimens from EVALI patients for multiple commonly misused drugs, prescription medications, and novel psychoactive substances.
MDH’s public health laboratory rapidly developed procedures to test EVPs and identified several chemicals of interest for which to purchase analytical standards for method development and quality assurance. They tested products obtained from 12 patients and clinical specimens from 51 patients. The laboratory also provided subject matter expertise on multiple chemicals of concern identified throughout the investigation, including medium-chain triglycerides and VEA.
Another internal response partner was MDH’s Office of Medical Cannabis. This office coordinates the state medical cannabis program and provides access to medicinal cannabis, which includes THC-containing EVP for patients with qualifying medical conditions. It coordinated acquisition of EVPs containing medicinal cannabis for testing at MDH’s public health laboratory for comparison with illicitly obtained THC-containing EVPs.
Collaboration with external partners was also crucial to response effectiveness. MDH contacted the Minnesota Poison Control System, a longtime partner for poisoning-related events, for information about ingredients in EVPs and toxic effects. In addition, the Poison Control System assisted with reporting cases when clinicians contacted them requesting treatment guidance for EVALI patients.
MDH also consulted with other medical toxicologists, infectious disease physicians, and pulmonologists. The clinicians’ subject matter expertise improved understanding among MDH staff members of the potential pathogenesis of EVALI. In collaboration with these health care providers, MDH developed a step-by-step guide for clinicians to use in assessing and reporting patients with symptoms of EVALI. 12 MDH requested assistance from the Minnesota Hospital Association to coordinate clinical webinars for health care providers in the state. In collaboration with the Minnesota Hospital Association and 2 pulmonologists, MDH held 3 clinical webinars during the response. These webinars included investigation updates and presentations by pediatric and adult pulmonologists on clinical signs and symptoms, as well as recommendations for patient management.
Later in the investigation, MDH’s public health laboratory contacted personnel from local law enforcement and the Minnesota Bureau of Criminal Apprehension. These agencies shared confiscated illicit EVPs that had been seized during raids of unregulated EVP manufacturers and distributors in 2018 and 2019. 3 Laboratory testing results of these products provided important insight into the role of various toxicants, especially VEA, in the outbreak. Testing showed that EVPs confiscated in 2018 did not contain VEA, whereas products confiscated in 2019 contained VEA. In addition, laboratory testing of THC-containing EVPs obtained from patients detected VEA in products submitted by 11 of 12 patients. 3 These findings were further corroborated by a CDC study that examined lung fluid of people with EVALI and people without EVALI. This study found VEA in the lung fluid of 48 of 51 people with EVALI and no VEA in the lung fluid of people without EVALI. 2 Another study found that inhaled VEA induced EVALI-like pathogenesis in the lungs of mice. 13 Together, these findings illustrated a strong association between VEA and EVALI.
Finally, MDH collaborated with CDC, the Council of State and Territorial Epidemiologists, the Association of Public Health Laboratories, and the US Food and Drug Administration to discuss the outbreak and review protocols and findings.
Challenges to the Response
Minnesota’s EVALI investigation faced multiple challenges, most of which are not typically encountered in outbreak investigations of infectious disease.
A poorly understood behavioral phenomenon
Behaviors associated with EVALI were new, evolving, and unfamiliar to both clinicians and MDH staff members. Although MDH’s Tobacco Prevention Unit provided subject matter expertise on nicotine-containing EVPs, use of THC-containing EVPs was initially unfamiliar to most response personnel. MDH staff members worked to improve their understanding of EVP-related behaviors, products, brands, flavors, ingredients, and terminology. Before the availability of CDC standardized interview forms, initial development of interview questionnaires and use of appropriate terminology were challenging, given the lack of familiarity with the subject among MDH staff members.
A variety of substances, devices, brands, and flavors
Another challenging aspect of this outbreak was the diversity of products used. MDH staff members quickly learned about the various nicotine-containing and THC-containing EVPs, devices, flavors, and brands. In addition, many patients reported trying multiple flavors, brands, and products, and using more than 1 EVP device. To further complicate matters, investigators discovered that, unlike brands of legal products, brands of illicit THC-containing EVPs rarely corresponded to actual manufacturers and supply networks. Instead, these pseudo-brands were cartridges of illicit THC-containing EVPs created by unknown manufacturers that purchased branded packaging through the internet. Because these products were made separately from the packaging in which they were sold, brand names did not indicate product source, and ingredients listed on the packaging were not consistent with the ingredients contained in the products. These inconsistencies and inaccuracies meant that some standard disease investigation activities, such as product traceback, were impossible.
An illicit activity
Another challenge was that many EVPs used by patients were illicit. Eighty-five percent of patients with EVALI in Minnesota reported using THC-containing EVPs. Recreational marijuana is not legal in Minnesota, and most patients interviewed reported using THC obtained from informal sources (ie, family or friends). Although most patients were willing to report illicit THC use and describe the EVP used, some patients hesitated to admit they used THC products, and patients were often unwilling to disclose where they had obtained THC-containing EVPs.
Depending on the patient’s age, purchase of nicotine-containing EVPs was also illegal. Before the outbreak, the minimum legal age to purchase any tobacco products (including nicotine-containing EVPs) in Minnesota was 18 or 21 years, depending on the community. However, 35% of all patients classified as having confirmed or probable EVALI in Minnesota who reported using nicotine-containing EVPs were aged <18 years, which indicates underage acquisition of products from informal or commercial (stores or online) sources. We suspected that some patients aged <18 years may have been unwilling to disclose their EVP use to their parents or health care providers upon initial consultation for fear of negative consequences. This lack of disclosure likely delayed identification of patients with EVALI by health care providers.
The illegality of the exposure might have affected the overall interview completion rate: of 150 patients classified as having confirmed or probable EVALI, only 86 patients agreed to be interviewed (interview completion rate of 57%). In addition, patients might have been unwilling to give their illicit EVPs to MDH for testing out of fear of criminal prosecution. MDH staff members informed patients interviewed that the information they shared was considered private medical data and would not be shared with law enforcement agencies, consistent with Minnesota law governing the privacy of health information. Although all interviewed patients were asked if they would be willing to provide MDH with the EVPs they had used, only 28 patients provided products to MDH for testing.
Laboratory characterization of EVPs
The public health laboratory faced numerous challenges in testing EVPs obtained from patients. First, what substance(s) in EVPs were causing illness and whether EVALI was associated with THC-containing EVPs, nicotine-containing EVPs, or both were unclear. Although preliminary testing conducted by other laboratories identified VEA as a potential toxicant of concern early in the investigation, the role of other toxicants with EVALI was unknown. 14 The MDH public health laboratory used this early identification of VEA along with internet-based research of chemicals commonly used as EVP diluents to focus its list of compounds of interest. Laboratorians then rapidly developed and validated testing procedures for different products and compounds, which necessitated development of novel analytic methods. Although laboratorians were familiar with testing unknown substances, they had to determine how to best open various EVP pods, cartridges, and devices, and retrieve what was sometimes only a few hundred microliters of remaining e-liquid.
A novel disease
Finally, one of the biggest challenges was that EVALI was a novel disease. The exact pathogenesis of EVALI was unknown, and no confirmatory diagnostic tests or distinct clinical findings were available to identify patients with EVALI. Because no national case definition existed, CDC took several weeks to develop a case definition to classify cases. 9 The resulting case definition was complex and one of exclusion.
Practice Implications
Because Minnesota was one of the first states to begin investigating cases of EVALI, we have described MDH’s experience responding to an atypical outbreak. We outlined successes and challenges so that other public health agencies can better prepare for the next outbreak of a noncommunicable disease. We summarized our recommendations from lessons learned (Box).
Minnesota Department of Health recommendations for public health agencies to prepare for future outbreaks of noncommunicable diseases
Build and maintain strong partnerships with internal partners, including state health laboratories. Assess capabilities of laboratories to analyze substances, including unknown or illicit substances and clinical specimens for commonly misused drugs, prescription medications, and novel psychoactive substances. If the state laboratory is unable to perform certain types of testing, identify protocols for how testing would be arranged with other state or private laboratories.
Build and maintain strong partnerships with external partners, such as the state poison center and law enforcement. For a multistate outbreak, consider engaging with other involved states and, depending on the type of outbreak, the Centers for Disease Control and Prevention, the Council of State and Territorial Epidemiologists, the Association of Public Health Laboratories, and the US Food and Drug Administration.
Ensure the state public health agency has legal authority to conduct noninfectious disease outbreak investigations, including obtaining private health data for patients with nonnotifiable conditions.
Develop an agencywide all-hands-on-deck protocol to ensure sufficient resources are available from across the agency working on the investigation to conduct the response effectively, while not disproportionately burdening individual divisions.
Consider implementing a state surveillance program that uses laboratory testing to monitor illicit drug use or substances.
Internal Partnerships
The most important success of the response was collaboration with partners. Collaboration among MDH epidemiologists and laboratorians proved invaluable to advancing understanding of additives in EVPs and added to evidence of the timing and relationship of VEA to EVALI. 3 MDH epidemiologists routinely collaborate with the MDH public health laboratory to investigate clusters of severe outcomes associated with drug use and gain important situational awareness. Clusters of drug overdoses attributable to contaminated drugs have become increasingly prevalent in the United States, whether they are associated with fentanyl-laced pills or adulterated synthetic substances. 6 -8 Laboratory testing of both substances and clinical specimens provides information to determine potential toxicological causes of these outbreaks. Because not all state laboratories have the same analytical capacity, a formal list of state and private laboratories should be established to provide information about the analytical capacity and available methods at each laboratory. Having such a list could expedite the testing of substances or clinical specimens during outbreaks. In the absence of such a list, we recommend that jurisdictions reach out to their public health laboratories and learn about their capacity to analyze substances, including unknown or illicit substances, and clinical specimens for various chemicals. If the laboratory is unable to perform this type of testing, collaborative or contractual arrangements for testing with other laboratories should be considered.
External Partnerships
Besides collaborating with internal partners, coordinating with external partners was important to MDH’s response. The MDH public health laboratory used preexisting relationships with law enforcement agencies in the state to obtain confiscated illicit THC-containing EVPs. This collaboration was essential for elucidating the link between VEA and EVALI. Throughout the investigation, MDH was in regular contact with the Minnesota Poison Control System, clinicians, and the Minnesota Hospital Association to engage subject matter experts and organize communication efforts. Public health agencies should consider building and maintaining relationships with external partners, including nontraditional partners such as law enforcement, during similar investigations. It was also important to work with other states, CDC, the Council of State and Territorial Epidemiologists, the Association of Public Health Laboratories, and the US Food and Drug Administration.
Legal Authority to Conduct Noncommunicable Disease Outbreak Investigations
Public health reporting of infectious diseases has a solid foundation in the United States and is widely accepted. Although EVALI is not a reportable condition in Minnesota, the state commissioner of health has the authority to conduct an epidemiological investigation to identify and describe a public health problem, 15 including the authority to collect health data. 16 The experience of responding to this outbreak and previous outbreaks or clusters of illicit drug use has clearly demonstrated that public health agencies will continue to face outbreaks of noncommunicable, nonnotifiable diseases. Therefore, health departments should ensure they have the legal authority to conduct these outbreak investigations appropriately, including the authority to obtain private patient data. Previous outbreaks of novel diseases, such as the outbreak of brodifacoum-laced synthetic cannabinoids in multiple states, 8 illustrate the need for agencies to establish this authority in advance of public health emergencies and educate health care providers about reporting of noncommunicable diseases during such events.
All-Hands-On-Deck Protocol
The EVALI outbreak escalated rapidly in Minnesota and nationwide. As case reports began to increase, MDH staff members working on the response quickly became overwhelmed. The distribution of responsibilities across multiple divisions and staff members was necessary. Although staff members were reassigned from other divisions to help with the response, some divisions were disproportionately affected, and some staff members were overburdened until additional staff members were available. The allocation of additional staff members to work on outbreak investigations is often challenging. One lesson learned was to develop an agencywide protocol for requesting additional staff members from all divisions and to understand the agency’s surge capacity. When an agency’s ICS is activated, an all-hands-on-deck protocol would ensure a sufficient workforce from across the agency working on the investigation to conduct the response effectively, while avoiding disproportionately burdening individual divisions or staff members. Our experience has been helpful in navigating MDH’s COVID-19 response.
Surveillance of Illicit Substances
A final lesson learned is that real-time surveillance of illicit substances and products would provide valuable information for outbreaks associated with illicit drug use. Modernized surveillance systems that allow for rapid responses to noncommunicable disease outbreaks would complement traditional public health reporting mechanisms. Specifically, laboratory surveillance could provide information about the association of substances or adulterants with outbreaks. Public health agencies and laboratories should consider implementing surveillance that uses laboratory testing to monitor illicit drug or substance use in their jurisdiction. 17,18
Conclusion
Minnesota was among the first states in which health care providers reported suspected EVALI cases, and MDH responded quickly and effectively to the EVALI outbreak. Despite multiple challenges, MDH’s response had many successes, primarily collaboration with internal and external partners. Minnesota’s experience with the EVALI outbreak provided lessons learned that can be used to strengthen future responses to outbreaks of noncommunicable diseases. Although US public health agencies are well versed in handling outbreaks of infectious diseases, now is the time to ensure adequate capacity to respond to other types of outbreaks.
Footnotes
Acknowledgments
The authors thank Deborah Anderson, PharmD, Minnesota Poison Control System; Elisabeth Bilden, MD, Saint Mary’s Medical Center; Anne Griffiths, MD, Children’s Hospital; Muneera Hassan, Public Health Associate Program, Centers for Disease Control and Prevention; Sakina Naqvi, MD, Fairview Health Services; Travis Olives, MD, Hennepin Healthcare and Minnesota Poison Control System; staff members and leadership of the Minnesota Department of Public Health; Forensic Drug Chemistry Department, Minnesota Bureau of Criminal Apprehension; Anoka County Sheriff’s Office; Minnesota clinicians; and interviewed lung injury patients.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Support for this project was provided by the Council of State and Territorial Epidemiologists through the Applied Epidemiology Fellowship Program.
