Abstract
In 2006, the Centers for Disease Control and Prevention updated its recommendations for HIV testing of 4 population groups in health care settings: adults, adolescents, pregnant women, and newborns. Important components of the revised recommendations included opt-out routine HIV screening; eliminating prevention counseling for opt-out routine HIV screening; repeat HIV testing in the third trimester for all women at high risk for acquiring HIV and for women receiving health care in facilities and/or jurisdictions with high HIV burden; testing during labor and delivery for women with undocumented HIV status; and testing the newborn when the mother’s HIV status is unknown. To assess the integration of these testing recommendations into state laws and to inform future recommendations, we researched and assessed statutes and regulations that addressed HIV testing in the 4 population groups in all 50 states and the District of Columbia in 2018. We then classified the laws, based on their consistency with the recommendations for each of the 4 population groups. Of 31 states and the District of Columbia that had relevant laws, all addressed at least 1 component of the recommendations. Although no state had laws that incorporated all the recommendations for all the population groups, 5 states (Delaware, Illinois, Louisiana, Maryland, and New Hampshire) had incorporated all the recommendations for adults and adolescents, and 4 states (Connecticut, Nevada, North Carolina, and West Virginia) had incorporated all the recommendations for pregnant women and newborns.
In 2017, a total of 38 739 persons were diagnosed with HIV in the United States and the 6 US dependent areas (American Samoa, Guam, Northern Mariana Islands, Puerto Rico, Republic of Palau, and US Virgin Islands). 1 HIV testing and learning one’s HIV status is the entry point into care and treatment for persons with HIV. However, 14% of all persons with HIV in the United States in 2016 did not know their status, and only 56% of adolescents and young adults aged 13-24 years knew their HIV status. 2,3 HIV testing for pregnant women is essential to reduce the number of perinatally acquired HIV infections. In 2016, ninety-nine perinatally acquired HIV infections occurred in the United States. 2
In 2006, the Centers for Disease Control and Prevention (CDC) revised its HIV testing recommendations to recommend routine opt-out HIV testing for adults, adolescents, pregnant women, and newborns. 4 Some of the revised recommendations were (1) routine opt-out HIV screening for patients aged 13-64 years, (2) removing the need for prevention counseling for screening, and (3) removing the need for separate written consent because the general medical consent should incorporate consent for HIV screening. CDC further explained in its recommendations that opt-out testing included (1) notifying patients that they will be tested for HIV, (2) giving information to patients on HIV testing orally or in writing, (3) giving patients the opportunity to ask questions, and (4) allowing patients to decline testing and documenting their refusal.
Furthermore, CDC recommends routine opt-out testing for pregnant women as early as possible in each pregnancy and a second HIV test during the third trimester for all pregnant women. A second HIV test during the third trimester is advised for women who (1) received health care in jurisdictions with an elevated incidence of HIV or AIDS, (2) received health care in facilities in which at least 1 HIV-infected pregnant woman was identified for each 1000 women screened, (3) were known to be at high risk for acquiring HIV, or (4) had signs or symptoms consistent with an acute HIV infection. CDC also recommends testing women during labor and delivery when their HIV status is unknown, as well as testing newborns when the HIV status of the mother is unknown.
The purpose of this analysis was to determine whether CDC’s revised HIV testing recommendations for adults, adolescents, pregnant women, and newborns were incorporated into state laws (statutes and regulations). Unlike 2 previous studies that limited the scope of review to 1 or 2 years after the release of the recommendations, our study reviewed state laws approximately 12 years after the revised recommendations were issued. 5,6 Because it often takes state legislatures multiple years or legislative cycles to develop and pass new statutes or regulations, our analysis was intended to provide a more complete picture than studies conducted only 1 or 2 years after the release of the reccomendations. In addition, this research builds on a previous study of the consistency of state perinatal HIV testing laws with CDC recommendations by (1) expanding the analysis to include adults and adolescents and 2 (2) incorporating 2 additional years of statutory and regulatory data. 7 Findings from this study are particularly prescient given the renewed federal focus on HIV testing as part of the Ending the HIV Epidemic: A Plan for America initiative. 8
Methods
From September 18 through October 31, 2018, we searched the legal research service Westlaw Next, 9 using Boolean terms to find any statute or regulation in the 50 states and the District of Columbia that explicitly mentioned HIV testing or screening in adults and adolescents (general population), pregnant women, and newborns. We used the following search strings to capture laws for adults/adolescents and pregnant women/newborns: (adv: test! screen! /s consent! permission (opt! + 1 out) /s H.I.V. (human + 1 immuno! + 1 virus)) and (adv: TI,TE(h.i.v. “human immuno-deficiency virus” & pregnan! newborn).
After we collected the relevant statutes and regulations, 3 reviewers assessed each law for the components of CDC’s revised HIV recommendations. The reviewers checked each law to see if it contained the following components: (1) no need for separate written consent elements of opt-out routine HIV screening; (2) elimination of prevention counseling requirement for routine opt-out HIV screening; (3) the general medical consent form encompasses consent for HIV testing; (4) patient must be informed of testing; (5) health care provider must give patient specific information on HIV; (6) patient is given the opportunity to ask questions; (7) patient has the right to decline testing; (8) the law uses the terms “routine” or “opt-out”; (9) HIV testing has to take place as early as possible during each pregnancy or during the first trimester; (10) repeat HIV testing in the third trimester for all pregnant women, or for pregnant women who do not have a documented HIV test, are at high individual risk of being infected with HIV, or receive health care in facilities or jurisdictions with a high HIV prevalence; (11) a provision for testing during labor and delivery for pregnant women with an undocumented HIV test is included in the law; (12) newborn testing when the mother’s HIV status is unknown is included in the law; and (13) any novel criteria for HIV testing. We also coded whether state laws were enacted or amended after 2006, the year in which CDC revised its HIV testing recommendations.
We used laws addressing HIV testing without addressing a specific population group to answer the research question about HIV testing for the general population. We did not use laws on HIV testing in the general population to answer the research questions about HIV testing in pregnant women and/or newborns. Only laws that specifically addressed pregnant women and/or newborns were classified as HIV testing laws for pregnant women and/or newborns. We also did not include state laws that addressed testing pregnant women without specifying the timing or routine nature of the HIV test. Responses to questions from each assessor were entered on unique sheets for each of the 51 jurisdictions. We documented and resolved by consensus any discrepancies between reviewers. Finally, we entered responses into a master Excel spreadsheet for analysis. The legal citations for the regulations and statutes used in this study are available from the authors upon request.
Results
No states had laws that would hinder compliance with CDC’s 2006 revised HIV testing recommendations. However, no state had laws with all relevant components for HIV testing in both adults/adolescents and pregnant women/newborns.
Taking into account laws enacted or amended after 2006, at least 1 component of the CDC HIV recommendations for adults was mentioned in statutes in 31 states and the District of Columbia, and at least 1 component of the CDC HIV testing recommendations for pregnant women and newborns was found in statutes in 30 states. Because the CDC recommendations are not compulsory, their incorporation into state laws varies by state and depends on each state’s legislative priorities.
Adults and Adolescents
Laws addressing HIV testing for adults also included testing for adolescents but not for pregnant women or newborns. Thirty-one states and the District of Columbia had laws that mentioned at least 1 component of the CDC HIV testing recommendations for adults, 10 states had laws without relevance to the CDC recommendations (ie, a law was found using the search string but did not include any review components), and no relevant laws were found in 9 states (ie, no law was found using the search string). Twenty-six of the 31 states and the District of Columbia revised existing laws after 2006, and 3 states enacted laws after 2006 (Table 1).
US states with statutes and regulations that mentioned at least 1 component of the 2006 CDC revised HIV testing recommendations for adults and adolescents, by whether a law was revised or new and whether the law references opt-out testing or routing testing, as of 2018 a
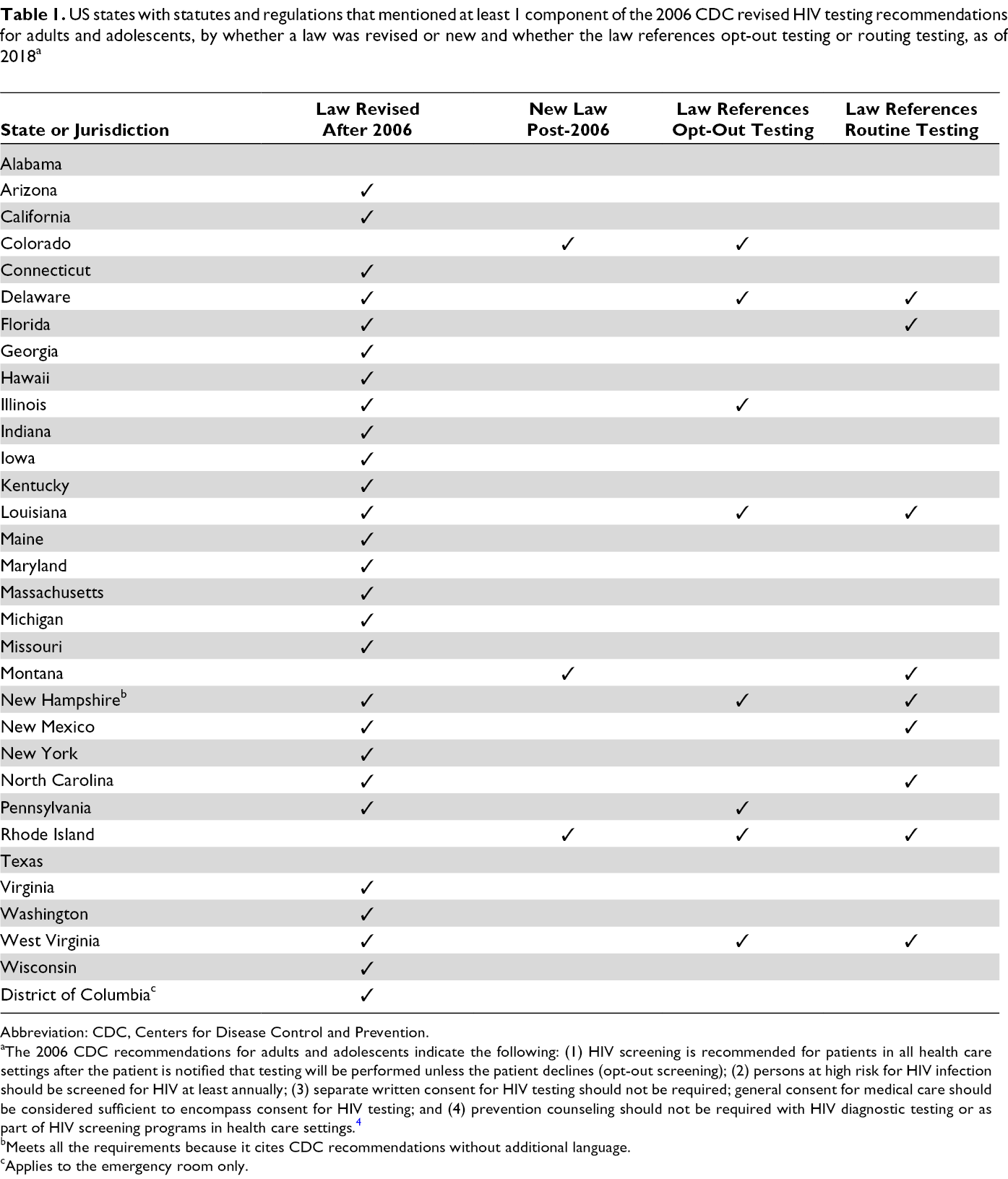
Abbreviation: CDC, Centers for Disease Control and Prevention.
aThe 2006 CDC recommendations for adults and adolescents indicate the following: (1) HIV screening is recommended for patients in all health care settings after the patient is notified that testing will be performed unless the patient declines (opt-out screening); (2) persons at high risk for HIV infection should be screened for HIV at least annually; (3) separate written consent for HIV testing should not be required; general consent for medical care should be considered sufficient to encompass consent for HIV testing; and (4) prevention counseling should not be required with HIV diagnostic testing or as part of HIV screening programs in health care settings. 4
bMeets all the requirements because it cites CDC recommendations without additional language.
cApplies to the emergency room only.
Five states had laws that included all the relevant components of the revised recommendations for adults and adolescents: Delaware, Illinois, Louisiana, Maryland, and New Hampshire. No state required prevention counseling in a clinical setting (Table 2).
US states with statutes and regulations that mentioned at least 1 component of the 2006 CDC revised HIV testing recommendations for adults and adolescents, by consent, whether patients need to be informed of testing, whether information on HIV should be provided, and right to decline testing, as of 2018 a
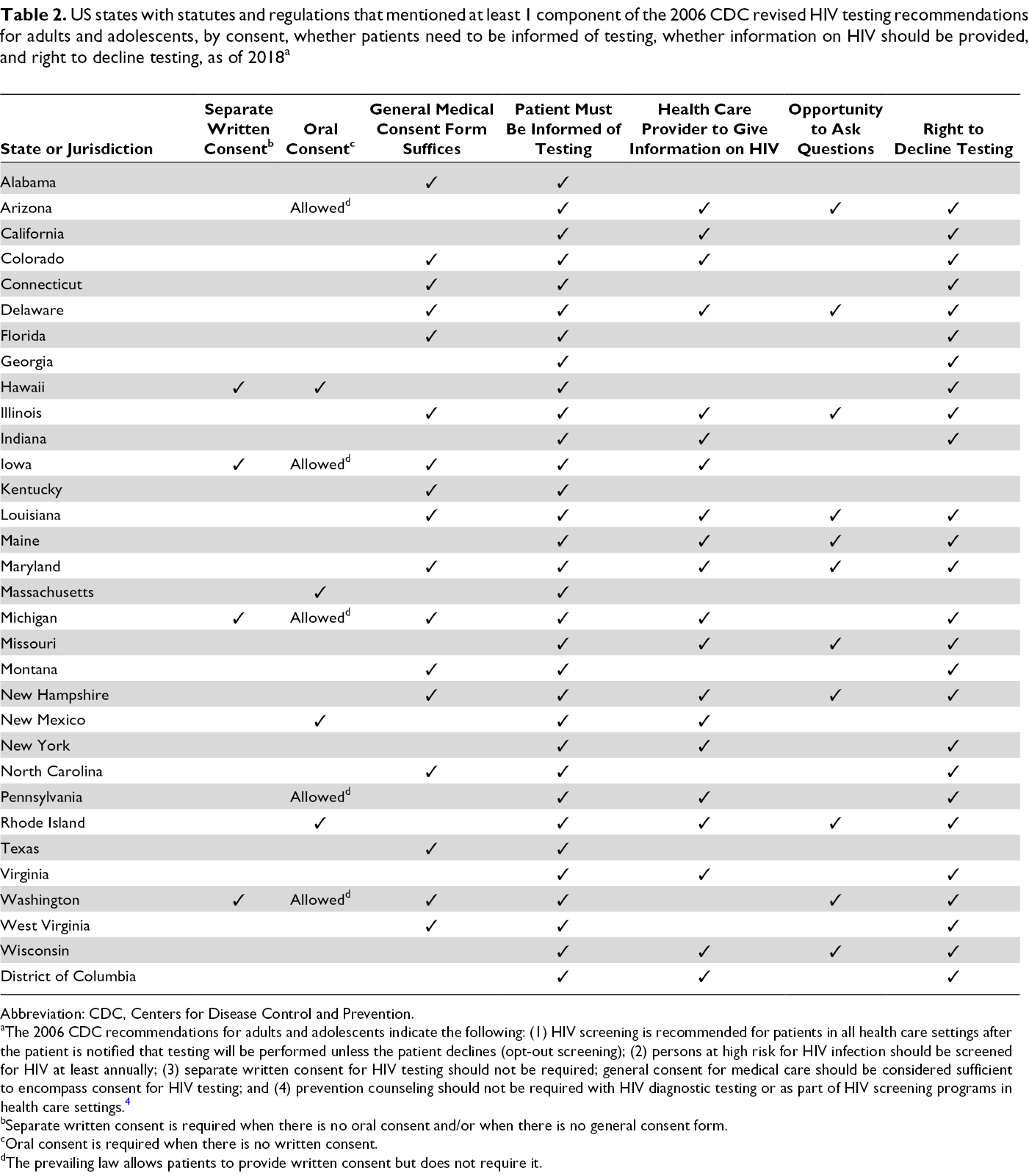
Abbreviation: CDC, Centers for Disease Control and Prevention.
aThe 2006 CDC recommendations for adults and adolescents indicate the following: (1) HIV screening is recommended for patients in all health care settings after the patient is notified that testing will be performed unless the patient declines (opt-out screening); (2) persons at high risk for HIV infection should be screened for HIV at least annually; (3) separate written consent for HIV testing should not be required; general consent for medical care should be considered sufficient to encompass consent for HIV testing; and (4) prevention counseling should not be required with HIV diagnostic testing or as part of HIV screening programs in health care settings. 4
bSeparate written consent is required when there is no oral consent and/or when there is no general consent form.
cOral consent is required when there is no written consent.
dThe prevailing law allows patients to provide written consent but does not require it.
Pregnant Women and Newborns
Thirty states had laws that mentioned at least 1 component of the CDC HIV testing recommendations for pregnant women and/or their newborns. Two states had laws that were not relevant to the CDC recommendations, and no relevant laws were found for 18 states and the District of Columbia. Eighteen of the 30 states revised existing laws after 2006, and 11 states enacted laws after 2006.
Ten states had laws that included at least 1 component of the CDC HIV recommendations for routine opt-out HIV testing for pregnant women: California, Colorado, Connecticut, Indiana, Iowa, Nevada, New Mexico, North Carolina, Virginia, and West Virginia (Table 3).
US states with statutes and regulations that mentioned at least 1 component of the 2006 CDC revised HIV testing recommendations for pregnant women and infants, by whether law was revised or new, whether patients must be informed of testing and can ask questions, whether health care provider should give information on HIV, right to decline testing, and testing provided as part of routine care, as of 2018 a
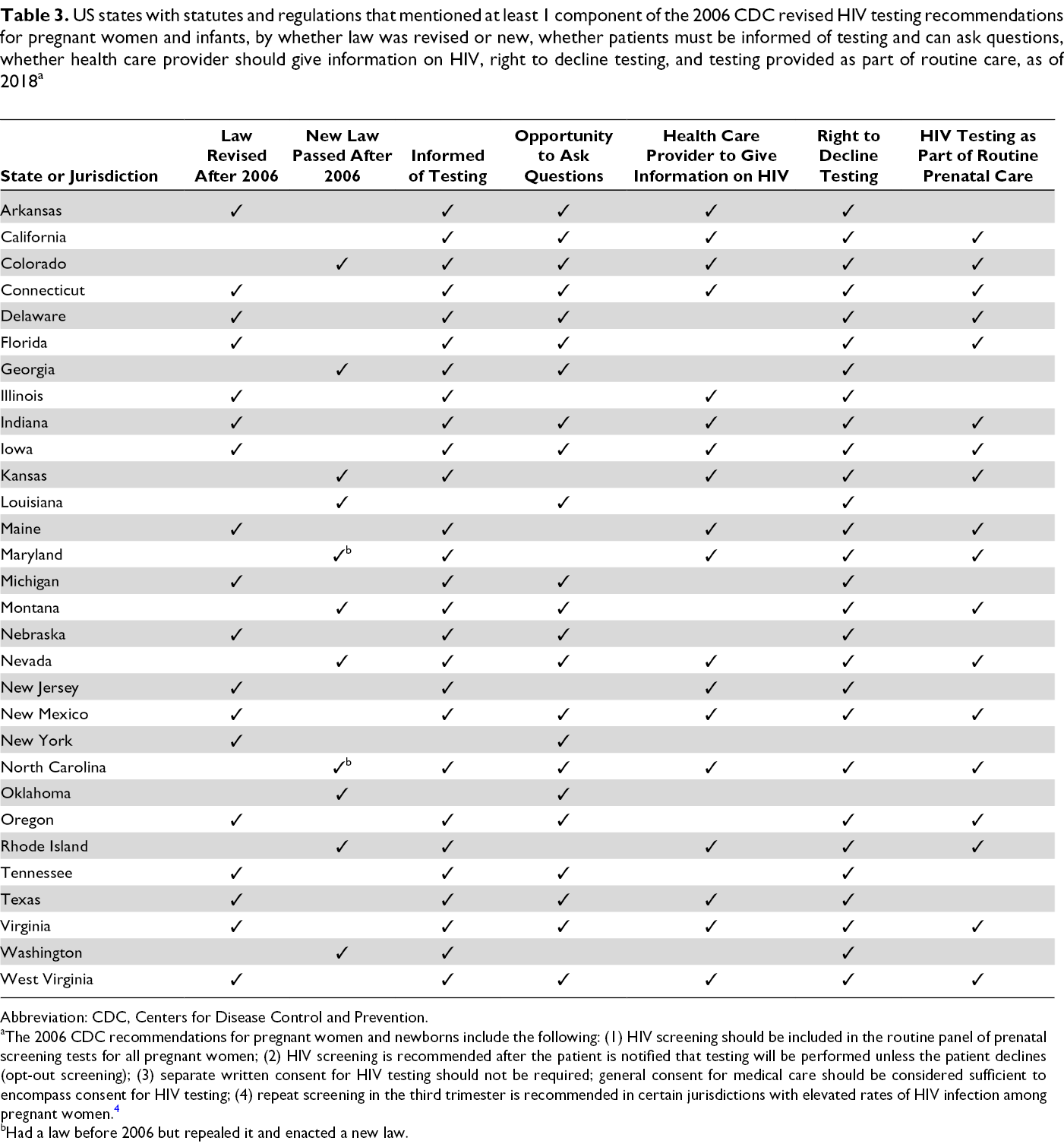
Abbreviation: CDC, Centers for Disease Control and Prevention.
aThe 2006 CDC recommendations for pregnant women and newborns include the following: (1) HIV screening should be included in the routine panel of prenatal screening tests for all pregnant women; (2) HIV screening is recommended after the patient is notified that testing will be performed unless the patient declines (opt-out screening); (3) separate written consent for HIV testing should not be required; general consent for medical care should be considered sufficient to encompass consent for HIV testing; (4) repeat screening in the third trimester is recommended in certain jurisdictions with elevated rates of HIV infection among pregnant women. 4
bHad a law before 2006 but repealed it and enacted a new law.
Eight states included all the recommendations for testing pregnant women in the third trimester and during labor and delivery and for testing newborns: Connecticut, Illinois, Kansas, Nevada, New Jersey, North Carolina, Texas, and West Virginia (Table 4).
US states with statutes and regulations that mentioned at least 1 component of the 2006 CDC revised HIV testing recommendations for pregnant women and infants, by timing of testing during pregnancy, testing during the third trimester and during labor and delivery, and newborn testing, as of 2018 a
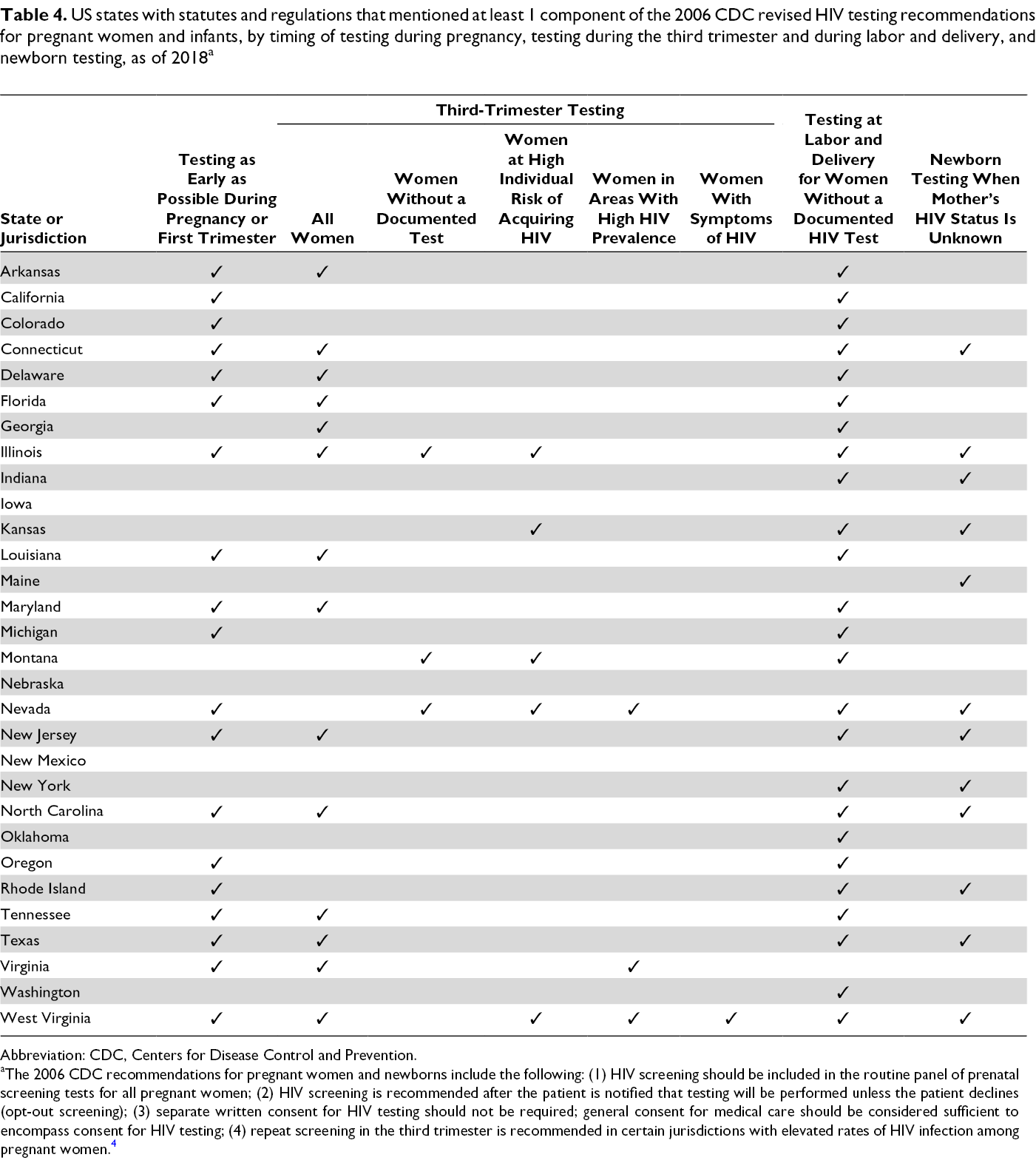
Abbreviation: CDC, Centers for Disease Control and Prevention.
aThe 2006 CDC recommendations for pregnant women and newborns include the following: (1) HIV screening should be included in the routine panel of prenatal screening tests for all pregnant women; (2) HIV screening is recommended after the patient is notified that testing will be performed unless the patient declines (opt-out screening); (3) separate written consent for HIV testing should not be required; general consent for medical care should be considered sufficient to encompass consent for HIV testing; (4) repeat screening in the third trimester is recommended in certain jurisdictions with elevated rates of HIV infection among pregnant women. 4
Seven states included all the recommendations for testing pregnant women as early as possible during each pregnancy, in the third trimester, and during labor and delivery, and for testing newborns: Connecticut, Illinois, Nevada, New Jersey, North Carolina, Texas, and West Virginia (Table 4). Four states had laws including both (1) routine opt-out testing for pregnant women and (2) all the recommendations for testing pregnant women as early as possible during pregnancy, in the third trimester, and during labor and delivery, and for newborn testing: Connecticut, Nevada, North Carolina, and West Virginia.
Discussion
Our analysis shows that the 2006 CDC revised HIV testing recommendations have been integrated into the laws of most US states. Our results demonstrate a robust integration of the recommendations into state laws, unlike previous studies that indicated only a moderate uptake of HIV testing recommendations. However, this analysis brought to light 2 legal nuances that may have consequences for public health practice.
First, testing adolescents who are un-emancipated minors could require parental consent. The 2006 recommendations include routine testing starting at age 13, and the HIV testing laws for adults include adolescents. However, some adolescents might still be considered minors in their respective states and would need parental or guardian consent to be tested for HIV. Generally, adolescents can consent to health care services without parental or guardian consent when they have reached the age of majority or when they are emancipated. The age of majority varies by state (18, 19, or 21 years). 10 Some minors are emancipated and no longer need parental consent. 11
Several states allow un-emancipated minors of a specific age to consent to specific health care services. 10 In addition, most states allow all or some minors to get tested for HIV without parental or guardian consent, based on age (12, 14, or ≥16 years). 10 All states allow all or some minors to receive services for sexually transmitted diseases without parental or guardian consent, based on age (12, 14, or ≥16 years). 10 These services include sexually transmitted disease testing, diagnosis, and treatment, some of which may include HIV testing. Nonetheless, it might not be possible to test some minors aged <14 years or 16 years for HIV in some states, when they are unable or unwilling to obtain parental or guardian consent. 10 Therefore, it is hypothesized that the uptake of HIV testing for some minors who are un-emancipated and at risk for acquiring HIV might be low.
Second, some states took a different legal approach to implementing opt-out HIV testing. CDC recommends that patients be tested unless they decline (implied consent). However, 9 states allowed or required express oral consent for testing adults, and 4 of these states required documentation of oral consent. Future research should examine the potential barriers or facilitators to HIV testing uptake and normalization when express oral consent is required in conjunction with a state’s opt-out approach.
Limitations
This study had 1 limitation. We used Westlaw Next 9 exclusively to find state statutes and regulations and their dates of enactment or amendment. Had we used additional or different legal databases, we might have gotten different results. Some dates may not be accurate because states may have had laws before 2006 but repealed them to enact new statutes. In this analysis, these states were categorized as not having laws before 2006.
Conclusion
Because the CDC recommendations do not have the force of law, it is encouraging to find that state laws have changed to align with the 2006 CDC revised HIV testing recommendations. In addition, our findings elucidated the progress of states in using the law as a tool for structural change to advance public health and HIV prevention efforts. However, it is important to note that enacting laws is not always indicative of whether the recommendations are being implemented. States are encouraged to identify and correct any deficiencies in how CDC recommendations are being incorporated into state law. More importantly, states should assess how these laws are implemented and determine whether these laws or other policies are a good vehicle for implementing the recommendations.
Future research should examine the association between a state’s uptake of the recommendations and HIV testing rates. Nevertheless, laws that include the relevant components for HIV testing of adults, adolescents, pregnant women, and newborns set the norms and further support the implementation of HIV testing activities in that state. Future HIV testing recommendations could be informed by the findings of this study.
Footnotes
Authors’ Note
The findings and conclusions in this article are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work of S.S.V. and J.C. was supported by funding from the Oak Ridge Institute for Science and Education.
