Abstract
To provide an alternative to oral gavage for per os treatments in laboratory mice, we have recently developed and introduced the micropipette-guided drug administration (MDA) method. This procedure is based on the presentation of a palatable solution consisting of sweetened condensed milk diluted with water, encouraging voluntary consumption of the vehicle and drug formulations. In this study, we compared the MDA method in male and female C57BL/6N and BALB/c mice, two inbred strains widely used in basic and preclinical research. Administering a diluted condensed milk solution daily over a period of 30 days, we observed that male C57BL/6N mice reached the fully voluntary drinking stage within five days or less, requiring the least amount of time to drink from the micropipette. Compared with males, female C57BL/6N mice showed increased consumption times during the initial administration period, yet they all managed to reach a fully voluntary stage within seven days or less. By contrast, BALB/c mice in general, and female BALB/c mice in particular, consistently required more time to consume the diluted condensed milk solution, whether administered voluntarily (no restraint) or semi-voluntarily (with mild tail restraint). Notably, a substantial portion of BALB/c mice (12.5% of males and 62.5% of females) failed to achieve fully voluntary consumption by MDA, despite their ability and willingness to drink the condensed milk solution when using a mild tail restraint. Taken together, the present study identified significant strain and sex differences in the MDA method as applied to laboratory mice.
Introduction
The current gold standard for oral treatments in laboratory rodents is oral gavage, which refers to the involuntary administration of a substance via a feeding needle or tube into the lower oesophagus or stomach.1–3 While allowing precise dosing of the substances, the oral gavage procedure is associated with a number of disadvantages. First, it can compromise the welfare of laboratory rodents, particularly when performed by inexperienced personnel or when applied repeatedly.3–9 In addition to inducing stress responses, oral gavage can lead to injury of the oral cavity, perforation of the oesophagus and/or trachea, gastric rupture and gastric distension. These complications may in turn result in internal bleeding, throat and abdominal pain, respiration distress, reflux and weight loss,3–9 all of which can confound outcomes in basic or preclinical research.10–12 Furthermore, oral gavage fails to truly mimic the physiological processes associated with the voluntary intake of a substance via the mouth, such as swallowing and oesophageal transit. 13
To minimize such complications, we have recently developed and introduced the micropipette-guided drug administration (MDA) method as an alternative to oral gavage for per os treatments in mice. 14 In its original version, the MDA method uses diluted sweetened condensed milk as a vehicle substance.14–17 Given that this substance is highly palatable to rodents, animals orally treated with the MDA procedure are motivated to ingest the vehicle solution voluntarily. Hence, the MDA method minimizes stress and avoids injuries associated with traditional oral gavage techniques. 14 Originally established for chronic (>8 weeks) per os treatments in mice,14,15 the MDA procedure has since been used successfully in acute or sub-chronic settings, including oral application of clozapine-N-oxide in chemogenetic mouse models 16 and oral tamoxifen treatment in mouse Cre-loxP systems. 18 Interestingly, the latter study further showed that diluted condensed milk solution outperformed other palatable vehicle substances when used in the MDA procedure. 18 Moreover, the MDA method has also been applied successfully in laboratory rats 19 and adapted for oral glucose tolerance tests in mice. 20
The MDA procedure involves an initial training phase to acclimatise the animals. In the original protocol, the animals are initially scruffed and exposed to the diluted condensed milk solution via a micropipette on the first training day.14–17 On the second day of MDA training, the animals are mildly restrained by the tail and exposed to the same milk solution via the micropipette.14–17 The first training day may also be omitted, 18 while the partial tail restraining procedure used on the second day of MDA training may be continued until the animals no longer require any restraint and drink the milk solution voluntarily from the micropipette. The fully voluntary stage is usually reached within a few days, although this may vary across laboratories.14–17
The majority of previous studies using the MDA method were carried out using male C57BL/6 mice.14–18,20 Therefore, little is known about potential strain- and sex-specific differences in MDA. To address this gap, we conducted an exploratory study comparing MDA in male and female C57BL/6N and BALB/c mice, two inbred strains widely used in basic and preclinical research involving laboratory mice.21,22 Applying the original MDA protocol,14–17 we assessed several parameters that define the overall efficiency of the MDA method, including the number of training days required to reach the fully voluntary stage, the time needed to consume the diluted sweetened condensed milk solution voluntarily from the micropipette, and the proportion of animals unable to progress beyond semi-voluntary drinking. Furthermore, we monitored general welfare indicators, including bodyweight, hair coat condition and behavioural appearance within the home cage. These parameters were recorded and analysed over a 30-day period, with data collected on consecutive days.
Methods
Animals
Male and female C57BL/6N and BALB/c mice (n = 8 per strain and sex) were obtained from Charles River Laboratories (Sulzfeld, Germany) at the age of 10 weeks. Upon arrival, they were housed in individually ventilated cages (Allentown Inc., Bussy-Saint-Georges, France) in groups of four animals of the same sex and strain per cage, as described in detail before. 23 The cages were kept in a specific-pathogen-free holding room, which was temperature- and humidity-controlled (21°C ± 3°C, 50% ± 10%) and kept under a reversed light–dark cycle (lights off: 09:00 h to 21.00 h). To minimize potential bias, the age at which the animals were obtained (10 weeks) and the acclimatization period after delivery (two weeks) from the vendor were identical across strains and sexes.
All animals had ad libitum access to standard rodent chow (Kliba 3336, Kaiseraugst, Switzerland) and water throughout the entire study. All procedures had been previously approved by the Cantonal Veterinarian’s Office of Zurich, Switzerland (licence number: ZH062/2023).
MDA procedure
All animals were handled in the same way prior to MDA training, which involved daily cup handling (30-s handling sessions) for five consecutive days. The entire MDA procedure, including handling, was conducted by two equally trained and experienced experimenters of the same sex (female) throughout the study. The experimenters alternated in a pseudo-randomized manner, ensuring that each experimenter performed the MDA procedure for 4–5 consecutive days before switching to the other experimenter.
The MDA procedure followed the protocols used in its original version, with diluted sweetened condensed milk (Migros Kondensmilch; Migros) serving as the vehicle substance.14–17 The condensed milk contained unskimmed cow’s milk (59%), sugar (55 g per 100 g) and stabilizer E339. It was diluted with Milli-Q water using a 3:10 milk:water ratio and administered with a conventional single-channel p200 micropipette (Gilson Pipetteman, Thermo Fisher Scientific) using a 2 ml/kg administration volume.
Following the original version of the MDA method,14–17 all animals first underwent two consecutive training days, with one training session per day. On the first day of training, the mice were restrained by scruffing and exposed to diluted condensed milk solution, by offering the pipette tip containing the solution to the mouth until the mouse began to lick the pipette tip. As soon as the mouse began to lick the tip, a small pressure to expel the solution from the pipette was applied, allowing the mouse to drink the entire solution. On the second day of training, a looser restraint was used, such that the mice were restrained solely by the tail while being placed on the metal grid of the home cage food hopper. Again, the pipette was continually positioned next to the mice’s mouth until they started to lick and drink the content. To avoid loss of drug in facial hair, solutions were expelled from the pipette tip only when the mouse was actively licking. As of the third day, the animals were placed on the metal grid of the home cage food hopper and were offered the pipette tip containing the diluted condensed milk solution. According to previous studies applying the MDA method in C57BL/6 mice,14–17 most animals require no or minimal restraint to voluntarily drink the diluted condensed milk solution as of the third day of training. Therefore, we started the data collection and analysis on the third day of training. All parameters of interest (see below) were recorded and analysed across 30 consecutive days, during which the MDA vehicle solution was offered to the animals between 10:00 h and 14:00 h. On each day, the administration volume (2 ml/kg) was adjusted daily to the animals’ bodyweight.
For some analyses, sham control mice of the same strains (C57BL/6N and BALB/c) were included as well. These mice were obtained from the same supplier and maintained under identical housing conditions as the experimental groups (see above). The sham control animals underwent identical handling procedures as those in the MDA treatment group, with the exception that they were not offered the diluted condensed milk solution. Specifically, on the first and second days of MDA training, the sham controls were restrained by scruffing and mild tail restraint, respectively. As of the third day, they were placed on the metal grid of the home cage food hopper for a maximum of 3 min per day, which corresponded to the maximal time allowed for MDA-treated animals to consume the milk solution.
Parameters of efficiency
To assess parameters that define the overall efficiency of the MDA method, we recorded the time needed for mice to consume the diluted condensed milk solution voluntarily from the micropipette and the number of training days taken to reach the fully voluntary stage. The latter was defined as the animal drinking the milk solution from the micropipette tip without any restraint within 60 s. If MDA-trained animals failed to consume the milk solution within this time, they were subjected to mild tail restraint while being offered the MDA vehicle solution, akin to the second day of training (see above). We also recorded the number of dropout animals that refused to drink the solution either voluntarily (no restraint) or semi-voluntarily (mild tail restraint) within a maximum of 3 min.
Parameters of animal welfare
We monitored general animal welfare indicators throughout the MDA procedure, which included daily assessments of bodyweight, hair coat conditions and general behavioural appearance within the home cage. Hair coat condition and the bodyweight measures were taken immediately before MDA drinking sessions, whereas general behavioural appearance within the home cage was scored 30 min after completion of the MDA procedure. To assess hair coat conditions, we used the following scoring scale: 0 = well-groomed; 1 = scrubby fur without bite wounds; 2 = scrubby fur with bite wounds; 3 = bite wounds with bleeding. To assess general behavioural appearance within the home cage, we used the following scoring scale: 0 = active normal posture and natural reaction to activation; 1 = increased nervousness and/or exaggerated reaction to activation; 2 = stereotypic behaviours (e.g. repetitive jumping or circular running), distressed, enhanced defensive and/or aggressive behaviour. Monitoring of animal welfare parameters was by an experimenter who was blinded to the experimental conditions.
Statistical analysis
All statistical analyses were performed using SPSS Statistics (version 29.0, IBM, Armonk, NY, USA) and Prism (version 10.0; GraphPad Software, La Jolla, CA, USA), with statistical significance set at p < 0.05. Given the exploratory nature of our study, no sample size estimate was calculated to detect a pre-specified effect. Exclusion of animals was not applied. The time needed to drink the diluted condensed milk solution and bodyweights were first analysed using a full-factorial 2 × 2 × 30 (strain × sex × days) repeated-measures analysis of variance (RM-ANOVA), followed by RM-ANOVAs separately conducted for each strain. Tukey’s post-hoc test for multiple comparisons was used after RM-ANOVAs whenever appropriate. The percentage of animals reaching the fully voluntary stage across days was analysed using a log-rank (Mantel–Cox) test and displayed as Kaplan–Meier curves, whereas hair coat and general behavioural scoring scales were analysed using chi-square (χ2) tests.
Results
Time required to consume the diluted condensed milk solution across successive testing days
Analysis of the time required to consume the diluted condensed milk solution across successive testing days revealed marked strain and sex differences (Figure 1), as supported by the significant main effects of strain (F(1,28) = 35.94, p < 0.001) and sex (F(1,28) = 6.74, p < 0.05). Compared with male and female BALB/c mice, male and female C57BL/6N mice took consistently less time to drink the diluted condensed milk solution across the entire testing period (Figure 1(a)). Overall, female mice generally took longer to drink the diluted condensed milk solution than males (Figure 1(b)).
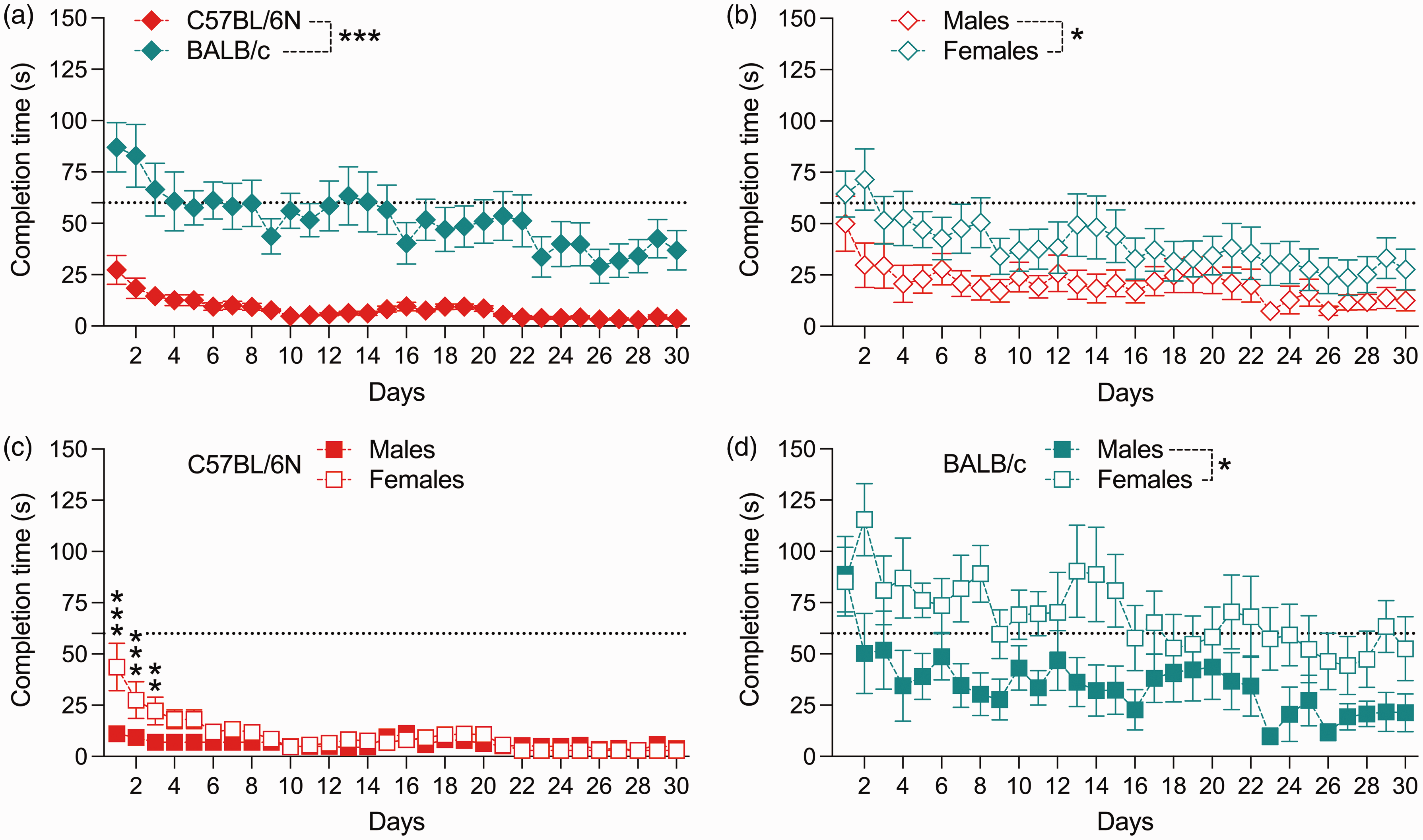
Time required to consume the diluted condensed milk solution over a period of 30 days in male and female C57BL/6N and BALB/c mice. The dotted line represents the maximum time (60 s) the animals were allowed to drink the solution voluntarily without any restraint being applied. If animals failed to consume the condensed milk solution in 60 s, they were subjected to mild tail restraint while being offered the milk solution. (a) Comparison between all (male and female) C57BL/6N and BALB/c mice. ***p < 0.001, reflecting the significant main effect of strain in repeated-measures analysis of variance (RM-ANOVA). (b) Comparison between all (C57BL/6N and BALB/c) male and female mice. *p < 0.05, reflecting the significant main effect of sex in RM-ANOVA. (c) Comparison between male and female C57BL/6N mice. **p < 0.01 and ***p < 0.001, based on post-hoc tests following a significant interaction between days and sex in RM-ANOVA restricted to C57BL/6N mice. (d) Comparison between male and female BALB/c mice. *p < 0.05, reflecting the significant main effect of sex in RM-ANOVA restricted to BALB/c mice. All data are means ± SEM; n = 8 per strain and sex.
Restricted analyses conducted for each strain separately confirmed significant sex differences. As shown in Figure 1(c), female C57BL/6N mice required more time to drink the diluted condensed milk solution during the beginning of the testing period, as supported by the significant main effect of days (F(29,406) = 9.70, p < 0.001) and its interaction with sex (F(29,406) = 5.20, p < 0.001). Subsequent post-hoc tests confirmed a significant difference between male and female C57BL/6N mice on the first three testing days (Figure 1(c)). The restricted analysis conducted for BALB/c mice showed that female mice generally required more time to drink the diluted condensed milk solution than male mice (Figure 1(d)), as supported by the main effect of sex (F(1,14) = 5.59, p < 0.05). While there was a significant main effect of days (F(29,406) = 3.55, p < 0.001), the interaction between sex and days did not attain statistical significance in the restricted analysis conducted for BALB/c mice.
Number of days required to reach the fully voluntary stage
Marked strain and sex differences were also apparent in terms of the number of days taken to reach the fully voluntary stage, as reflected by distinct curves indexing the percentage of animals reaching the voluntary stage over time (χ2 = 21.64, df = 3, p < 0.001). Excluding the first two training days with scruffing or tail restraint, all male and female C57BL/6N mice reached the fully voluntary stage within three and five days, respectively. In contrast, only 87.5% of male BALB/c mice achieved the fully voluntary stage by day 27, whereas less than half (37.5%) of female BALB/c mice reached this stage within the 30-days testing period (Figure 2). These strain differences were significant in both sexes (male C57BL/6N vs. BALB/c: χ2 = 8.58, df = 1, p < 0.01; female C57BL/6N vs. BALB/c: χ2 = 9.83, df = 1, p < 0.01).
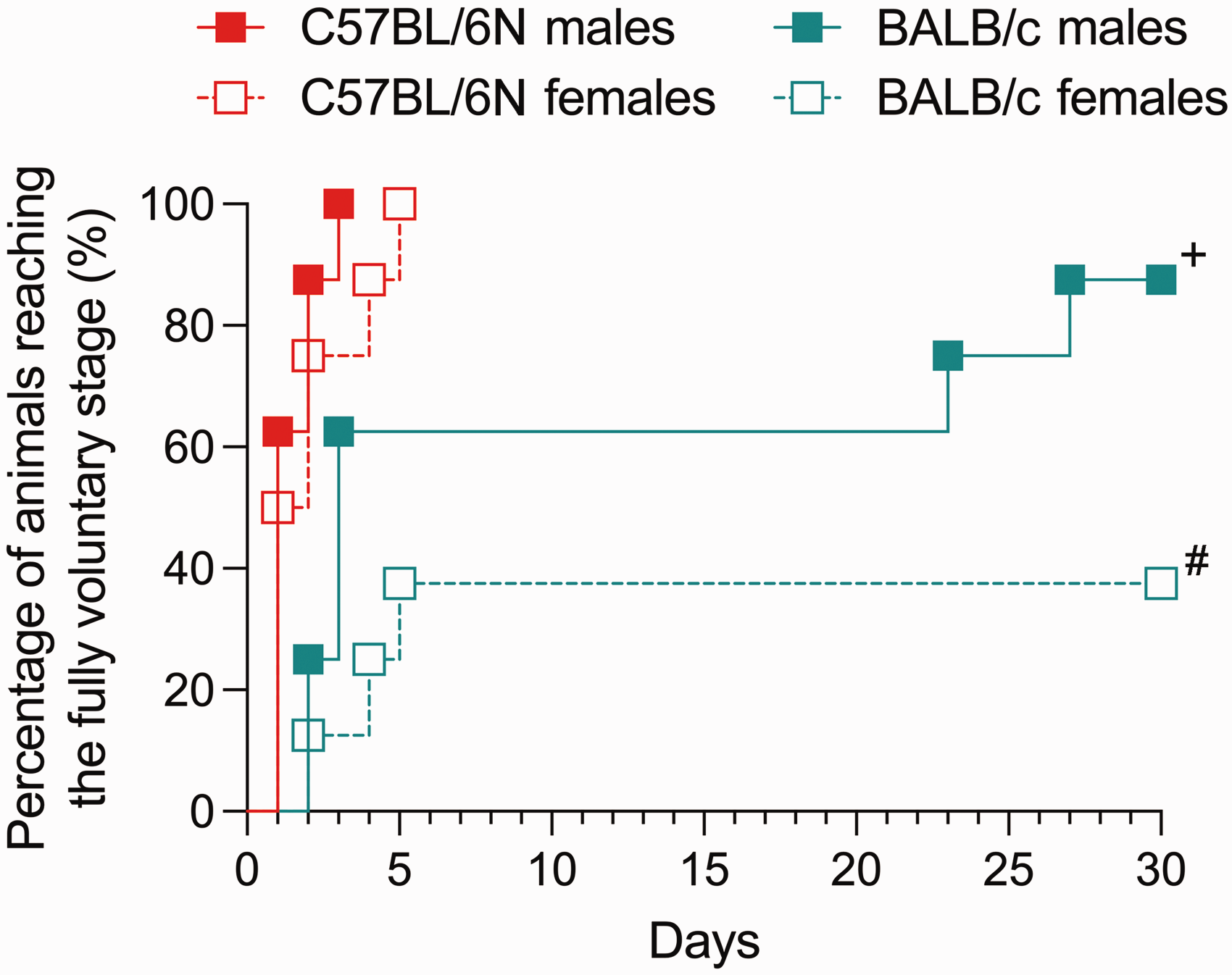
Number of days required to reach the fully voluntary stage in male and female C57BL/6N and BALB/c mice. The Kaplan–Meier curves display the percentage (%) of animals reaching the fully voluntary stage of micropipette-guided drug administration, where animals were allowed to drink the diluted condensed milk solution voluntarily without the need of any restraint. +p < 0.01, reflecting the significant difference between male C57BL/6N and BALB/c mice based on log-rank (Mantel–Cox) test; #p < 0.01, reflecting the significant difference between female C57BL/6N and BALB/c mice based on log-rank (Mantel–Cox) test. n = 8 per strain and sex.
Once the animals reached the fully voluntary stage, they consistently drank the diluted milk solution from the micropipette tip without requiring any restraint on subsequent testing days. Furthermore, animals that did not reach the fully voluntary stage within the allowed time (i.e. within 60 s) were still able and willing to drink the condensed milk solution with the use of a mild tail restraint. Hence, there were no dropout animals that refused to drink the solution either voluntarily (no restraint) or semi-voluntarily (mild tail restraint).
Parameters of animal welfare
The analysis of daily bodyweight, which included sham control mice of the same strains, did not reveal any differences between MDA-treated and sham control mice (Figure 3). This analysis revealed significant main effects of sex (F(1,56) = 557.13, p < 0.001) and days (F(29,1624) = 557.13, p < 0.001), indicating generally higher bodyweights in male mice compared with female mice, and a gradual increase in bodyweights over time (Figure 3). Likewise, the analysis of hair coat condition and behavioural scoring scales revealed no differences between MDA-treated and sham control mice (Figure 4). Regardless of strain, sex and treatment, ∼95% and 100% of the animals had a cumulative score of 0 in the hair coat scale (Figure 4(a)) and behavioural scale (Figure 4(b)), respectively.
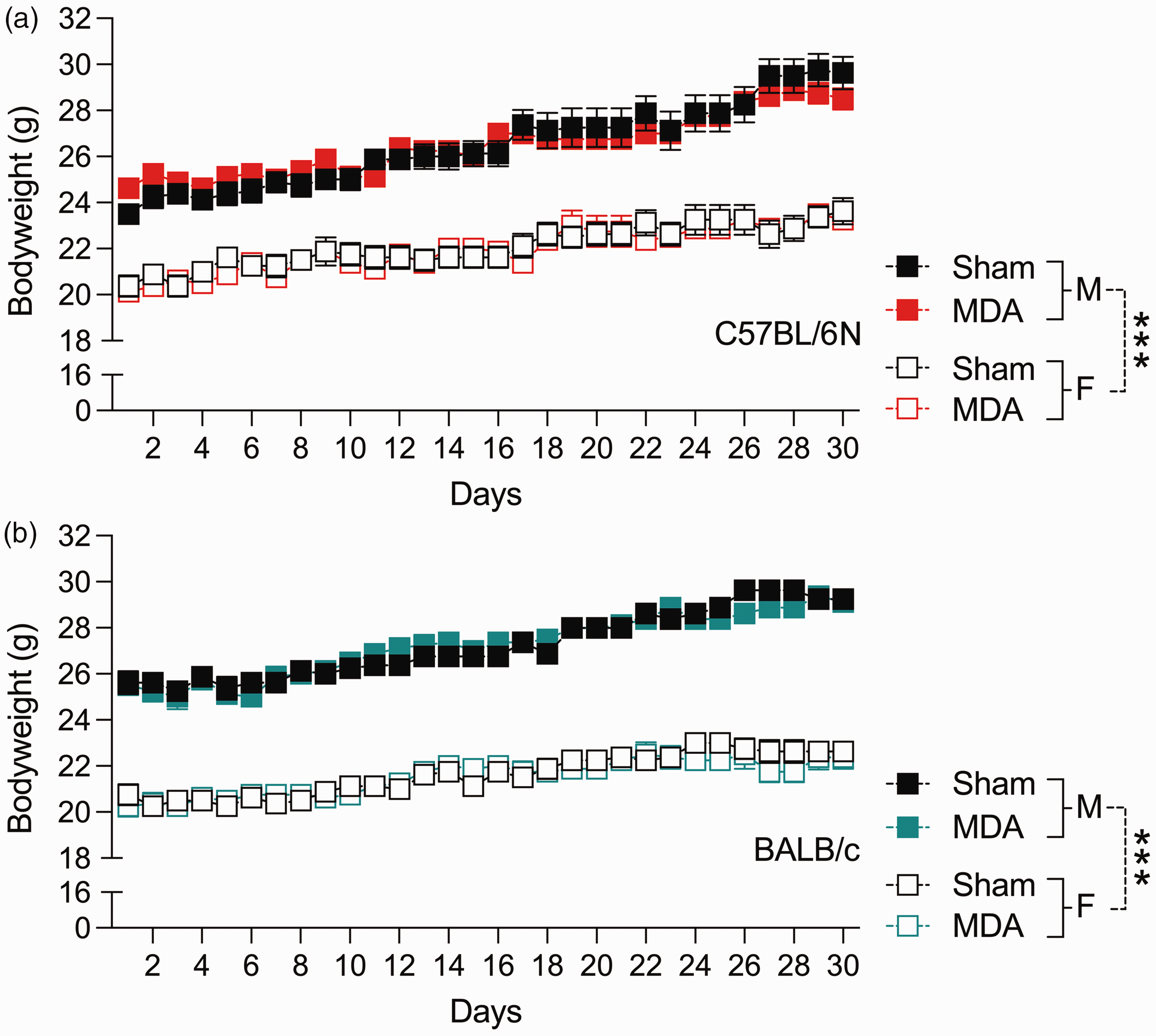
Bodyweights of male (M) and female (F) mice undergoing micropipette-guided drug administration (MDA) or sham treatment over a period of 30 days. (a) Bodyweights of C57BL/6N mice. p < 0.001, reflecting the significant main effect of sex in repeated-measures analysis of variance (RM-ANOVA). (b) Bodyweights of BALB/c mice. p < 0.001, reflecting the significant main effect of sex in RM-ANOVA.All data are means ± SEM; n = 8 in each group.
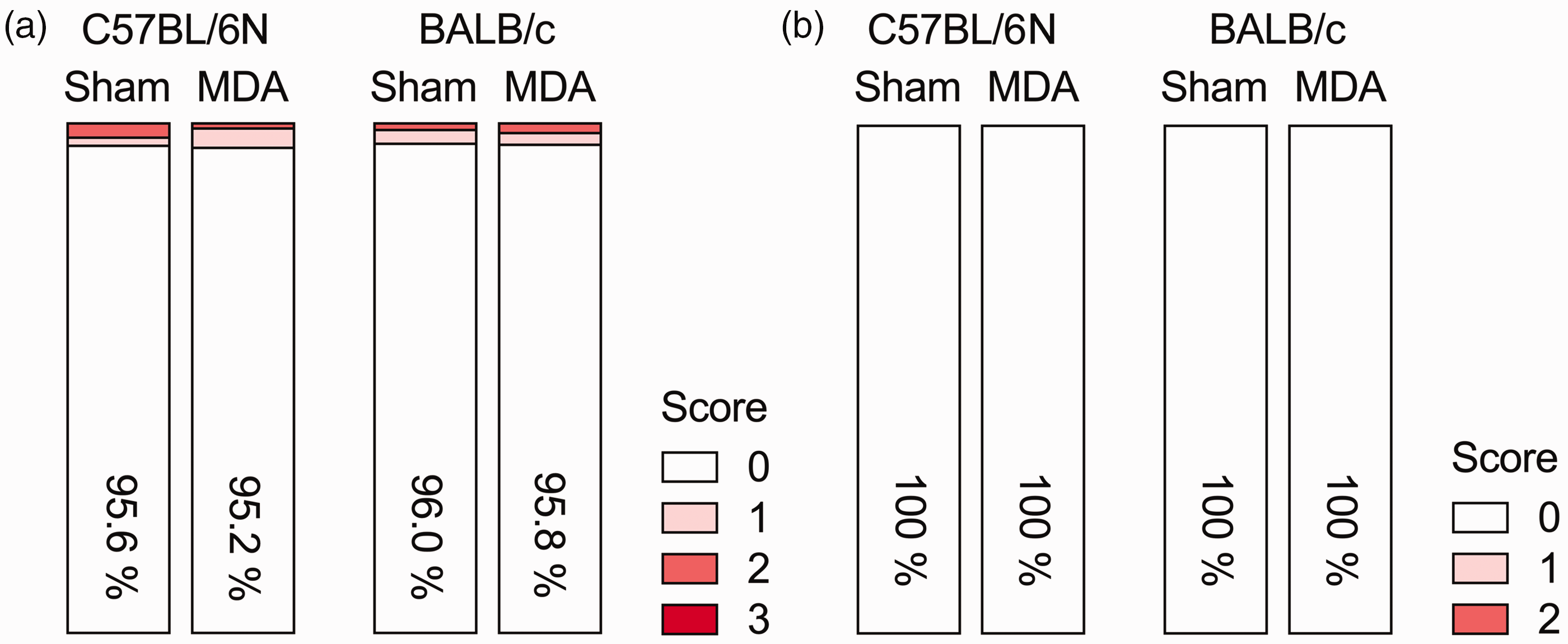
General animal welfare scores (in %) in male and female C57BL/6N and BALB/c mice receiving micropipette-guided drug administration (MDA) or sham treatment over a period of 30 days. (a) Hair coat scores: 0 = well-groomed; 1 = scrubby fur without bite wounds; 2 = scrubby fur with bite wounds; 3 = bite wounds with bleeding. (b) General behavioural appearance scores: 0 = active normal posture and natural reaction to activation; 1 = increased nervousness and/or exaggerated reaction to activation; 2 = stereotypic behaviours (e.g. repetitive jumping or circular running), distressed, enhanced defensive and/or aggressive behaviour. n = 8 in each group.
Discussion
As part of our ongoing efforts to validate the MDA method as an alternative to oral gavage for per os treatments in mice,14–16 the present study assessed its applicability in male and female C57BL/6N and BALB/c mice, two inbred strains commonly used in basic and preclinical research.21,22 We focused on parameters that define the overall efficiency of the MDA method and monitored general animal welfare parameters. Our study identified significant strain and sex differences in the number of days taken to reach a fully voluntary stage and the time needed to consume the diluted condensed milk solution across successive administration days. These differences are unlikely to result from experimental confounding factors, such as age, initial handling of the animals or the duration of the acclimatization period before MDA testing, as these factors were kept consistent between male and female mice of both strains.
Male C57BL/6N mice adapted most quickly to the MDA method, as they required the fewest days to reach a fully voluntary stage and the shortest time to consume the diluted condensed milk solution across successive testing days. These findings are consistent with earlier findings showing that male C57BL/6N mice rapidly complete MDA training and consume the condensed milk solution from the micropipette within seconds. 14 Compared with males, however, female C57BL/6N mice showed increased consumption times during the initial administration phase, yet they all reached a fully voluntary stage within five days of successive MDA sessions. By contrast, BALB/c mice, particularly females, displayed markedly slower and incomplete adaptation to the MDA method. They consistently required more time to consume the diluted condensed milk solution either voluntarily (no restraint) or semi-voluntarily (mild tail restraint) from the micropipette. Furthermore, a substantial proportion of BALB/c mice failed to reach the fully voluntarily stage of MDA.
Despite the slower adaptation of BALB/c mice to the MDA method, all mice willingly consumed the condensed milk solution from the micropipette when offered a mild tail restraint. Thus, there were no dropout animals that refused to drink the solution either voluntarily (no restraint) or semi-voluntarily (mild tail restraint). Based on these findings, and in keeping with our earlier studies using the MDA method in C57BL/6N mice,14,15 we deem a diluted condensed milk solution a valuable and effective vehicle substance for per os treatments via MDA. Furthermore, we suggest that semi-voluntary MDA protocols, in which animals are allowed to drink the MDA vehicle substance under mild tail restraint may serve as a practical alternative for mouse strains that show a slower or incomplete adaptation to the fully voluntary MDA approach.
The underlying causes of the observed sex and strain differences remain unknown. However, several factors could contribute to these variations, including basal differences in innate anxiety-like behaviour, varying propensities to consume naturally rewarding substances, and differing stress responses. In support of this notion, BALB/c mice generally exhibit higher levels of innate anxiety-like behaviour and increased stress responses to novel or aversive environments compared with C57BL/6 mice.24–27 These traits may impede voluntary drinking in MDA tasks by creating a conflict between gaining free access to a rewarding stimulus and avoiding potentially harmful threats. 28 Compared with C57BL/6 strains, BALB/c mice also show a lower propensity for consuming sweet substances, such as sucrose and saccharine,29,30 two palatable substances activating central reward circuits. 31 Thus, the hedonic impact and/or incentive salience of sweetened condensed milk solution may be lower in BALB/c than C57BL6/N mice, which in turn may reduce their motivation to consume this vehicle in MDA tasks voluntarily. 32
Based previous studies highlighting an important role of the oestrous cycle in shaping behaviour and cognition in female rodents, 33 the potential impact of the oestrous cycle during the training of female mice should also be considered as a factor that may contribute to variability in the MDA procedure. There is, however, an increasing number of studies challenging this assumption.34–37 Importantly, a previous study has found that various behaviours of C57BL/6J females are significantly different from those of female BALB/c mice irrespective of the oestrous cycle. 38 This suggests that behavioural differences between these two strains are similarly present in both male and female animals and emerge independently of the stage of the oestrus cycle. In our study, we did not assess the stage of the oestrous cycle, which represents a limitation. However, given that both male and female BALB/c mice consistently required longer MDA adaptation times across the 30-days period compared with C57BL/6 mice, we believe that the potential impact of the oestrous cycle during the training of females was relatively small in comparison with the marked strain differences.
Assessing animal welfare parameters that are part of our routine health monitoring programme, we did not reveal any differences in hair coat conditions and general behavioural appearance between MDA-treated and sham control animals. Furthermore, daily intake of the condensed milk solution over 30 days did not affect bodyweight, consistent with previous studies involving chronic MDA treatments in C57BL/6 mice 14 and Sprague–Dawley rats. 19 Hence, the MDA procedure aligns with the principles of good animal welfare in research settings39,40 and suggests that the MDA procedure does not induce major changes in metabolic functions.14,19 However, the lack of bodyweight changes in MDA-treated animals does not exclude the possibility of other, more subtle metabolic alterations, which should be assessed in future studies. 41
In conclusion, the present study identified significant strain and sex differences in the MDA method as applied to laboratory mice. These findings emphasize the importance of accounting for strain and sex as key variables in experiments involving MDA for per os treatments. Based on the results provided here, and in agreement with previous studies conducted in C57BL/6N mice,14–16 we deem the use of the MDA method highly practical in large-scale studies when using this particular mouse strain. Using strains with slower or incomplete MDA adaptation times (e.g. BLAB/c), however, the MDA method may be more time consuming than other administration procedures. Nevertheless, even for strains that do not achieve the fully voluntary stage of the MDA procedure, a semi-voluntary MDA protocol, which allows animals to drink the MDA vehicle substance under mild tail restraint, offers a humane and effective alternative to oral gavage for per os treatments.
Footnotes
Data availability
All data needed to evaluate the conclusions in the paper are present in the paper. All original data of this study are available from the corresponding author at request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Swiss National Science Foundation (SNF grant Nr. 407940_206399) under the National Research Program (NRP) 79 (‘Advancing 3R – Animals, Research and Society’).
