Abstract
A proper cage environment is essential for the welfare of laboratory mice, especially for females during the energy demanding lactation period and for pups during early development and growth. The most common housing system for laboratory mice is individually ventilated cages (IVCs) of which there are different layouts and ventilation strategies available on the market. The present study investigates the impact of cage environment in three different IVC types, on the maternal performance of females, and pup development and growth in C57BL/6NCrl and Crl:NMRI Foxn1nu mice. The results show differences in in-cage climate, female body weight, pup growth, feed and water consumption, and nest quality between cage types. There was a distinct effect of genotype in these differences, with the main effects found in NMRI NU mice. The results indicate that IVC systems might need to be managed differently for mice of different types and/or different physiological status. Many of the differences seen between cage systems could be drawn to the physical construction of the cage, such as location of feed hopper and location of air inlet and outlet. In conclusion, IVC in-cage climate affects the maternal performance of female mice and pup growth, but with differences between the two strains tested.
Today individually ventilated cages (IVCs) are a common housing type for laboratory mice. The development of IVCs started in the 1950s with a wish for a more controlled housing environment for laboratory rodents which led to the development of static filter-top cages. 1 This effectively protected the animals from the environment outside the cage; however the in-cage climate became poorer due to restricted ventilation. In order to improve in-cage climate, especially to reduce levels of carbon dioxide (CO2) and ammonia (NH3) in the cages, IVCs were developed in the 1970s. 1 This is a very controlled type of housing consisting of sealed cages with HEPA-filtered air released with the same pressure, speed, and temperature into each single cage within a rack. The development of IVCs has been thoroughly evaluated with focus on effective ventilation and optimal environment in the cages. With efficient ventilation it is possible to increase cage cleaning intervals and/or stocking density without impairing the in-cage climate. Several studies have assessed the levels of CO2 and NH3, relative humidity (RH) and temperature in the cages with different cage changing intervals, air changes per hour (ACH) and population densities in the cages.2–7 However, the effect on animals housed in this type of cage environment has just started to be evaluated in recent years. Studies have shown that rodents in general prefer non-ventilated cages to ventilated ones, that they prefer cages with the lowest number of ACH, and larger cages to smaller ones. 8 Further, mice have been shown to avoid cages with high air movement 8 and cages with draughts, 9 but avoidance of ventilated cages is counteracted if the mice are given nesting material. 8 They have been reported to build mounds in cages without nesting material with air intake at floor level, 3 and also reported to prefer to nest away from air inlets.10,11 Together, the results of these studies indicate that mice do perceive air movements in their cages and that they try to avoid them if possible.
The cage environment is crucial for the welfare of breeding animals. Further, lactation is the most energy-demanding physiological process a mammal can experience, 12 for example, lactation results in high heat production in the female that needs to be dissipated. 13 Also the growth and development of the pups are energy-demanding processes. For these processes to function properly, favourable environmental conditions are essential for animals. Moreover, mice are small animals with a high surface-to-volume ratio which means that they in general have a relatively large loss of body heat, 14 emphasizing their sensitivity to climate factors in their environment.
The present study investigates the impact of IVC environment on laboratory mice with a focus on maternal performance of breeding females and pup development and growth. The specific aims were to assess:
female and pup body weights; feed and water consumption; associations between pup weight and female weight, feed and water consumption; pup development; and in-cage climate
in three different types of IVC systems for mice, originating from three different manufacturers, and for two mouse strains with different genetic background and phenotypes.
Materials and methods
Animals and breeding
The experimental protocol was approved by the local committee for ethical approval of experimental studies using animals.
The study was performed in two batches, with two litters per female in each batch. Initially, 36 female and 18 male C57BL/6NCrl (B6) and an equivalent number of Crl:NMRI Foxn1nu (outbred) (NU) mice, were purchased from Charles River, Sulzfeld, Germany. They were five weeks old on arrival. Twelve females and six males of each strain were placed in each of the three different IVC systems: Allentown XJ (Allentown Inc, Allentown, NJ, USA), Arrowmight Maxiseal 580 (Arrowmight, Hereford, UK) and Green Line Sealsafe plus (Tecniplast, Buguggiate, Italy). After one week of acclimatization, males and females were placed together for mating, with two females and one male per cage. For the NU strain, heterozygous females (with fur) were mated with homozygous males, resulting in mixed litters with nude pups (homozygous) and pups that developed fur (heterozygous). Females were checked daily for vaginal plugs and after one week males and females were separated again. All females were placed in individual cages, in their respective IVC systems, and kept individually until the weaning of the pups and the start of the next mating period. At birth, litter size was limited to 10 pups, and excessive pups were culled on the day of birth. Pups were weaned 21 days after birth, and one week after weaning the first litter females and males were put together again for mating a second litter, in the same trios as in the first mating. After weaning, pups from the second litter were kept as breeding animals for the second batch, and mating started when they were 12 weeks old. The mating procedure was the same as for batch 1, with the same number of females in each IVC system. The breeding animals remained in the same IVC systems throughout the study. Both an inbred and an outbred strain were included in the study in order to make the results more generalized and practically applicable.
Housing and husbandry procedures
All IVC systems were kept in the same animal room, approximately 15 m2. The Allentown rack (ALL) consisted of 63 cages (9 [height] × 7 [width] cages), the Arrowmight rack (ARR) contained 56 cages (7 × 8 cages) and the Tecniplast rack (TEC) contained 70 cages (10 × 7 cages). Each cage in all systems was equipped with 140–150 g of Aspen bedding (Tapvei Oy, Kaavi, Finland) and 16 ± 0.2 g of nesting material (Sizzlenest; Datesand, Manchester, UK), placed at the back of the clean cages. The most centrally located cages in each rack were used to house the breeding females, In all the systems, every second cage housed a B6 female and every second cage housed a NU female. Assigned cages for each female were randomized within each strain. Important features of the different cage types were:
ALL: 500 cm2 floor area, air inlet in the back cage wall (approximately 3 cm from cage floor) and air outlet in the lid, feed hopper located at the back of the cage, height from floor to feed hopper and water nipple 5.8 cm and 6.0 cm, respectively. Half grid lid. ARR: 580 cm2 floor area, air inlet and outlet in the cage lid, feed hopper located approximately in the middle of the cage, height from floor to feed hopper and water nipple 5.0 cm and 6.5 cm, respectively. Full grid lid. TEC: 530 cm2 floor area, air inlet and outlet in the cage lid, feed hopper located at the back of the cage, height from floor to feed hopper and water nipple 7.0 and 6.0 cm, respectively. Half grid lid.
ACH were set according to recommendations from the manufacturers and were as follows: 50 ACH in ALL cages, 40 ACH in ARR cages and 75 ACH in TEC cages. All systems were set at positive pressure. The room temperature was 22.8 ± 0.8℃ and RH was 43.1 ± 9.7%, and the light period was set between 24:00 and 12:00 h.
Cages were cleaned once weekly, except around the birth of litters where cage cleaning was postponed for three days if litters were born (or expected to be born) the day before, after, or on the day of cage cleaning. In these cases the nesting material was not replaced at cage cleaning. All cage cleaning and other handling of the animals (see below) were performed in a mobile changing station (CS5 changing station; Tecniplast) in HEPA-filtered air.
Feed (CRM, Pelleted Rat and Mouse Breeder and Grower Diet; Special Diets Services, Witham, UK) was given ad libitum and feed intake was recorded on a weekly basis by weighing all feed that was added to each cage and weighing the residuals at cage cleaning. Tap water was provided in bottles, and water consumption was recorded by weighing the entire bottle with fresh water and later weighing it with residual water before renewing it. This was done twice per week but water consumption was presented on a weekly basis.
Observations of breeding females
Females were weighed once weekly during gestation and on the day of birth of the litter and again on days 3, 7, 14, and 21 during the lactation period. Feed and water intake was recorded during the lactation period, and nest scoring was performed on days 1, 3, 7 and 14 during the lactation period. The quality of the nests was assessed, according to the nest scoring system developed by Hess et al. 15 Briefly, this is a six-graded scale ranging from score 0 which is undisturbed nesting material, to score 5 which is a completely dome-shaped nest. Nest scoring was performed on the second round of litters in batch 1 and both litters in batch 2.
Observations of pups
Parameters in characterization protocol for mouse pups, assessed from birth until day 14.
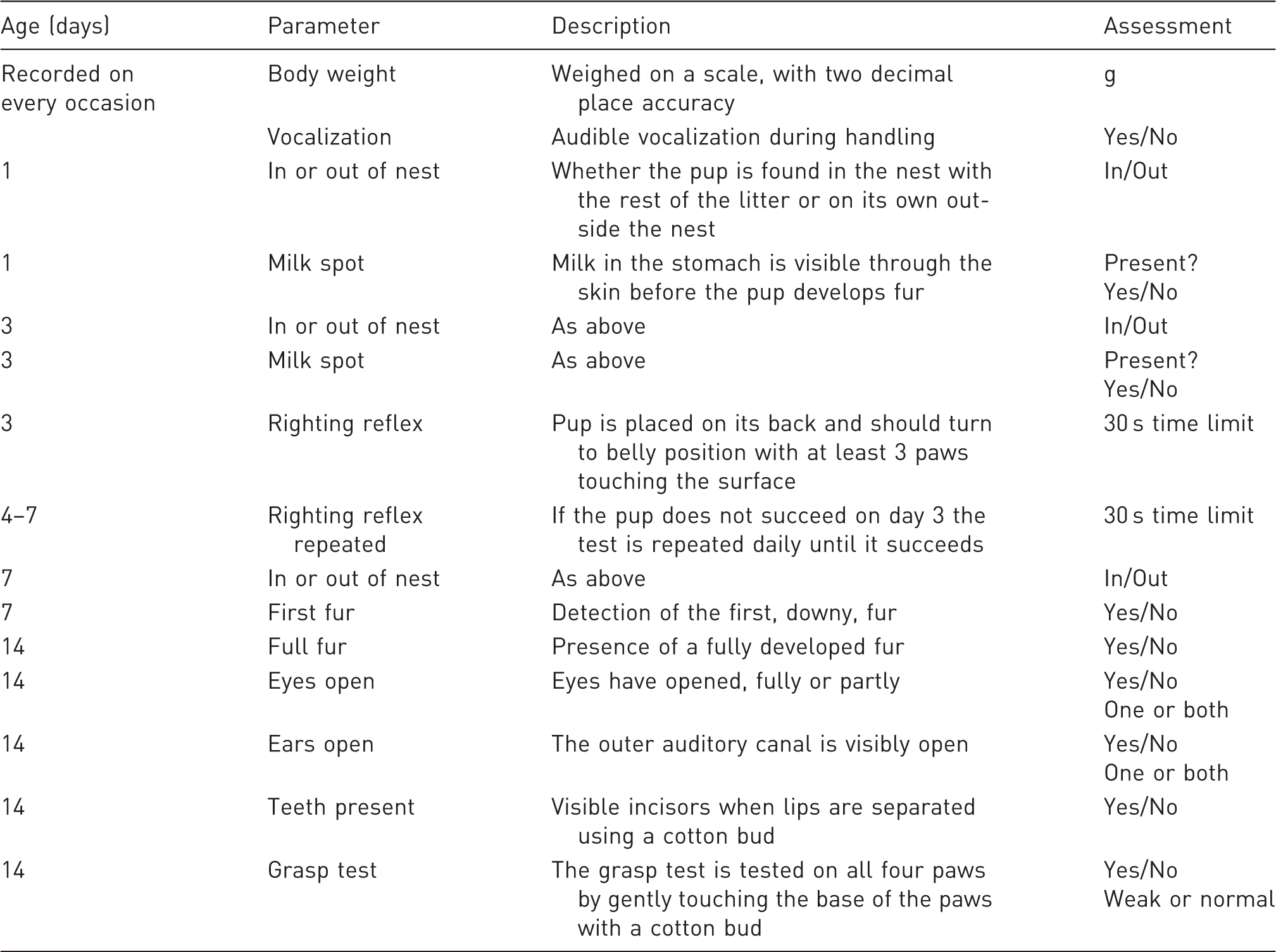
In-cage climate
The temperature and RH inside the cages were measured simultaneously in all three cage types during batch 2. A radio transmitter (ARGUS, Arrowmight) equipped with a combined external temperature and RH probe was inserted in one cage per system, and attached to the lid on top of the grid to prevent the mice from gnawing on it. Cages were chosen to have as similar a position as possible in all three systems; the third or fourth row from the floor and the third cage from the edge of the rack. All three monitored cages housed a B6 female with litter (7–9 pups) of a similar age, and monitoring was performed throughout the lactation period. A small hole had been made in one cage lid per system (by the manufacturers) for inserting the probe while keeping the lid closed. The probe was placed perpendicular to the back end of the lid, in the middle of the air inlet and outlet with the front of the probe facing the back wall. The probes were connected to small transmitters attached to the front of the cages and data were transmitted wirelessly to a base station and a computer to store data (ARGUS software, Arrowmight). Simultaneous recordings, with the same type of transmitters were also performed in the animal room. Eight recordings per 24 h (every 3rd hour) were pooled together to give a mean value for each day during the lactation period.
Statistical analyses
The statistical analyses were performed with the SAS package, version 9.2, 2011 (SAS Institute Inc, Cary, NC, USA). Residuals of all dependent variables were found to be normally distributed after examination using Proc Univariate, considering Shapiro-Wilks test for normality and a normal probability plot. Analyses were done separately for each strain (B6 and NU). Only litters with more than four pups were included in the analyses of female and pup weights, water consumption and feed consumption.
Least square mean differences between treatments (ALL, ARR and TEC) were estimated at each time point (day 1, day 3, day 7, day 14 and day 21) separately with Proc Mixed for mean pup weight in the litter, female weight, water consumption and feed consumption using the following model:
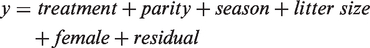
Relationships between mean pup weight in the litter and female weight, water and feed consumption were analysed for each treatment (ALL, ARR or TEC) separately with Proc Mixed. Regression analysis was performed separately for each covariate using the following model:
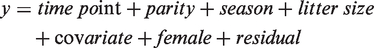
The latency (age in days) until the pups succeeded in the righting reflex was analysed with a generalized linear model with gamma distribution and log link with the following model in Proc Glimmix:
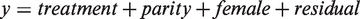
where the response variable ‘y’ was latency (age in days) until the pups succeeded in the righting reflex, and the predicting factors treatment (3 classes: ALL, ARR or TEC), parity (2 classes: 1st or 2nd), were included as fixed effects, while the individual female (n = 62) was included as a random effect.
Differences in nest score frequencies were analysed with chi-square tests in Proc Freq.
Data are presented from the day of the birth of the litters (day 1) until weaning (day 21). Feed and water consumption are presented as mean values for lactation weeks 1 (days 1–7), 2 (days 8–14) and 3 (days 15–21), respectively. Data are presented as least square means ± standard error (LSM ± STERR) if nothing else is stated, and differences are considered significant at P levels below 0.05.
Results
Initially, 103 B6 litters and 107 NU litters were included in the study, resulting in 883 and 539 (nude only) pups, respectively. These numbers represent pups after culling litters to a maximum of 10 pups per litter. Based on these conditions, the average litter size was similar in the different cage systems for both B6 litters (8.7 ± 1.13, 8.7 ± 1.29 and 8.5 ± 1.31 [mean ± SD] pups per litter in ALL, ARR and TEC, respectively) and NU litters (in total 9.2 ± 1.32, 9.5 ± 1.21 and 9.8 ± 0.54 [mean ± SD] pups per litter in ALL, ARR and TEC, respectively). In B6 mice, two litters were stillborn, one litter died on day 2 after birth, one litter was euthanized together with the mother on day 4 after birth due to the mother being injured, and one litter was euthanized on day 16 due to poor growth and severe trimming of the pups. Apart from entire litters, 29 B6 pups died before weaning and eight pups were euthanized due to injuries. In NU mice, one litter with female were euthanized on day 7 due to the female being ill, and one litter was euthanized on day 19 due to poor growth and dehydration of the pups. Apart from entire litters, 14 NU pups died before weaning and one pup was euthanized due to injury. Due to this mortality, culling and occasional missing records, information on female and pup body weights as well as water and feed consumption was recorded from 97 B6 and 103 NU litters from 61 B6 females (25 females with one litter and 36 with two litters) and 66 NU females (29 females with one litter and 37 with two litters).
Moreover, in total, 469 B6 pups and 335 NU pups were monitored in the characterization protocol and included in the analyses.
Female weights
No differences in body weight were found between cage types in B6 breeding females (Figure 1). At weaning day 21, the body weights for B6 females were 31.77 ± 0.36 g/mouse in ALL cages, 31.86 ± 0.37 g/mouse in ARR cages and 31.35 ± 0.39 g/mouse in TEC cages.
Body weight of B6 breeding females in three different cage types, presented as LSM ± STERR. ALL: Allentown cages, ARR: Arrowmight cages, TEC: Tecniplast cages. No significant differences between treatments were found.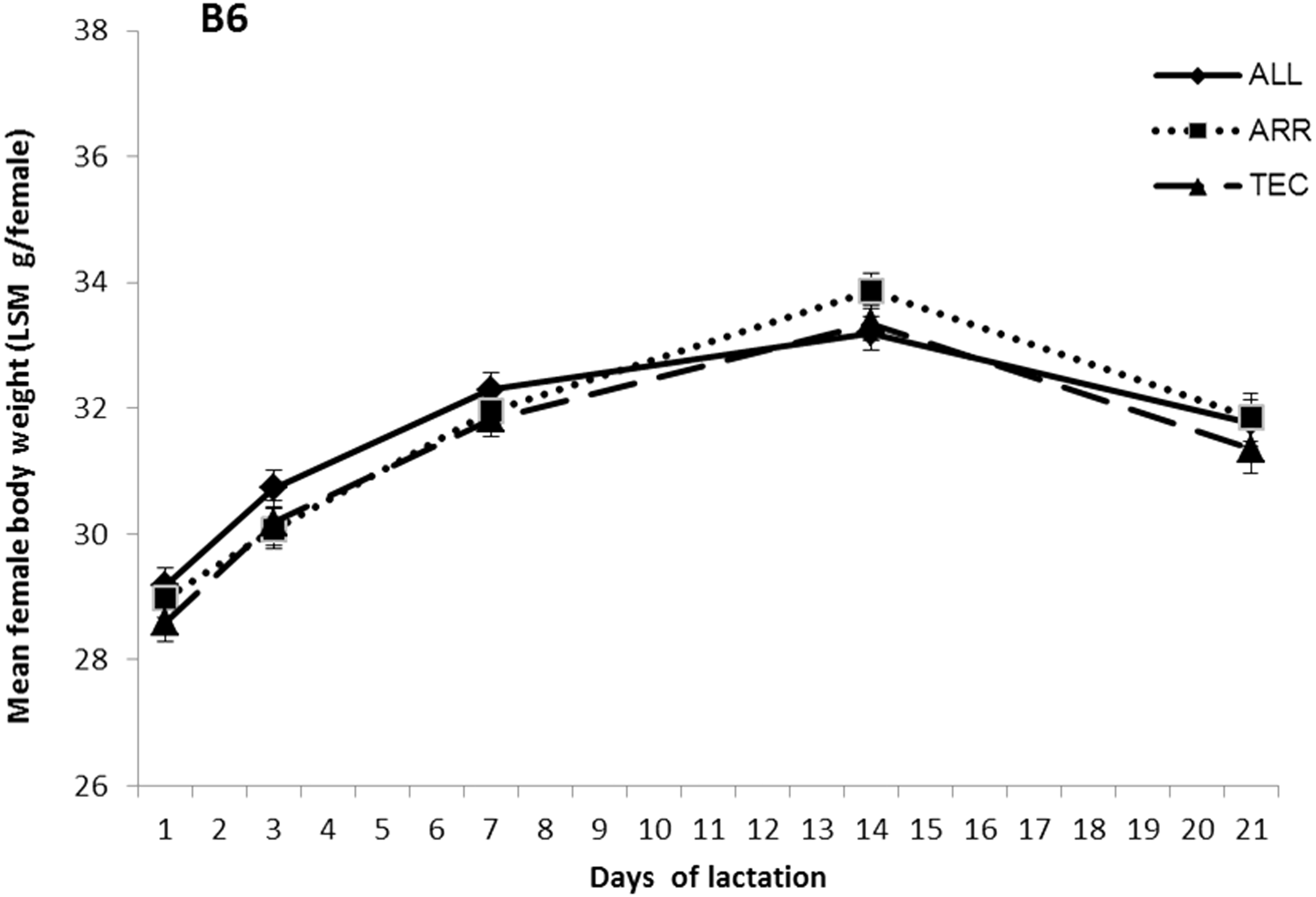
NU females in ALL cages weighed less than those in TEC cages on day 1 (P < 0.01) and day 14 (P < 0.001), and less than those in ARR cages on day 14 (P < 0.001) and day 21(P < 0.01, Figure 2). There was a tendency for a significant difference between ALL and TEC cages also on day 7 (P = 0.051) and day 21 (P = 0.052) and between ALL and ARR cages on day 1 (P = 0.057). At weaning, the body weights for NU females were 39.61 ± 0.47 g/mouse in ALL cages, 41.23 ± 0.51 g/mouse in ARR cages and 40.95 ± 0.52 g/mouse in TEC cages.
Body weight of NU breeding females in three different cage types, presented as LSM ± STERR. Different letters at each time point indicate significant differences between treatments, P < 0.05. ALL: Allentown cages, ARR: Arrowmight cages, TEC: Tecniplast cages.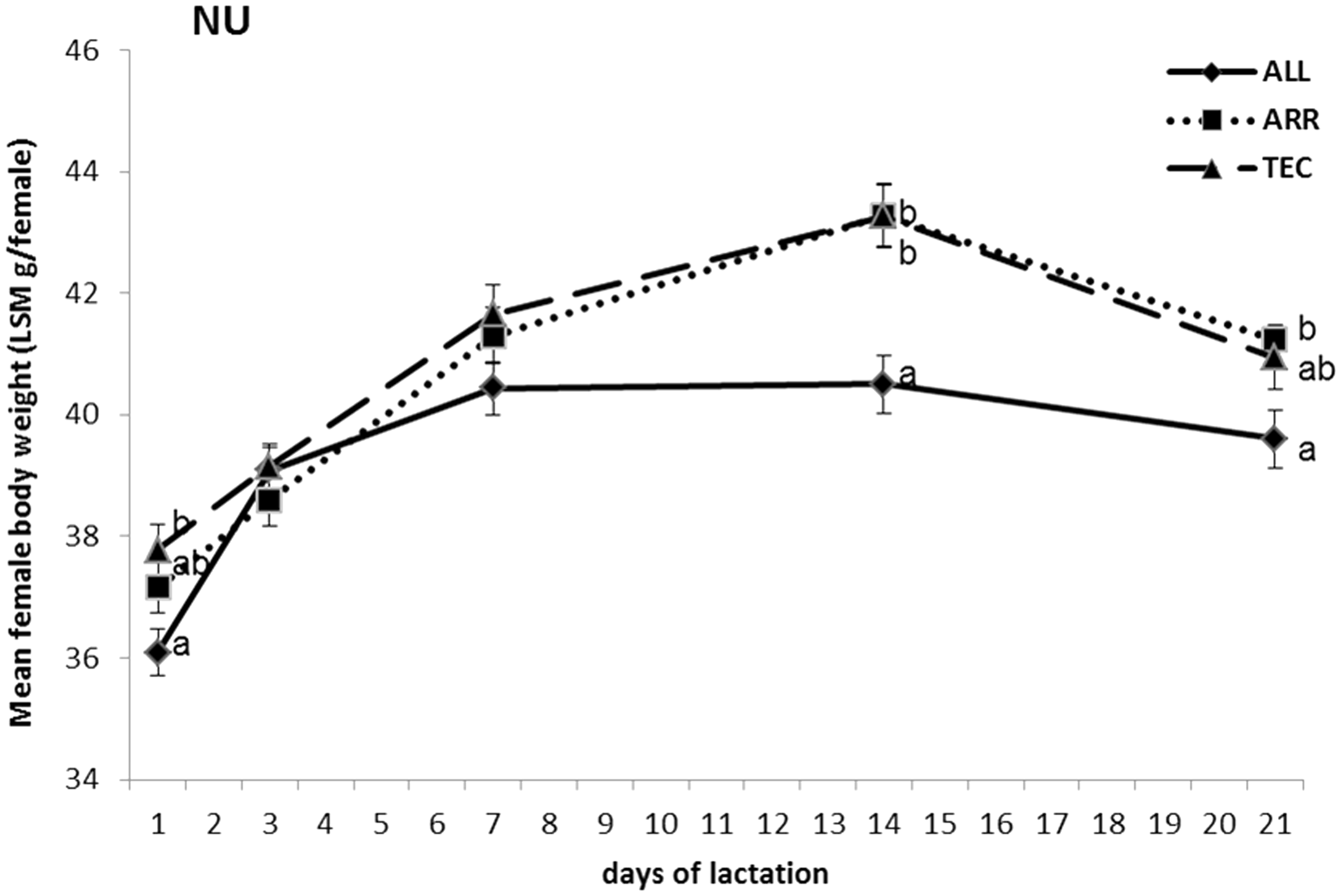
Pup weights
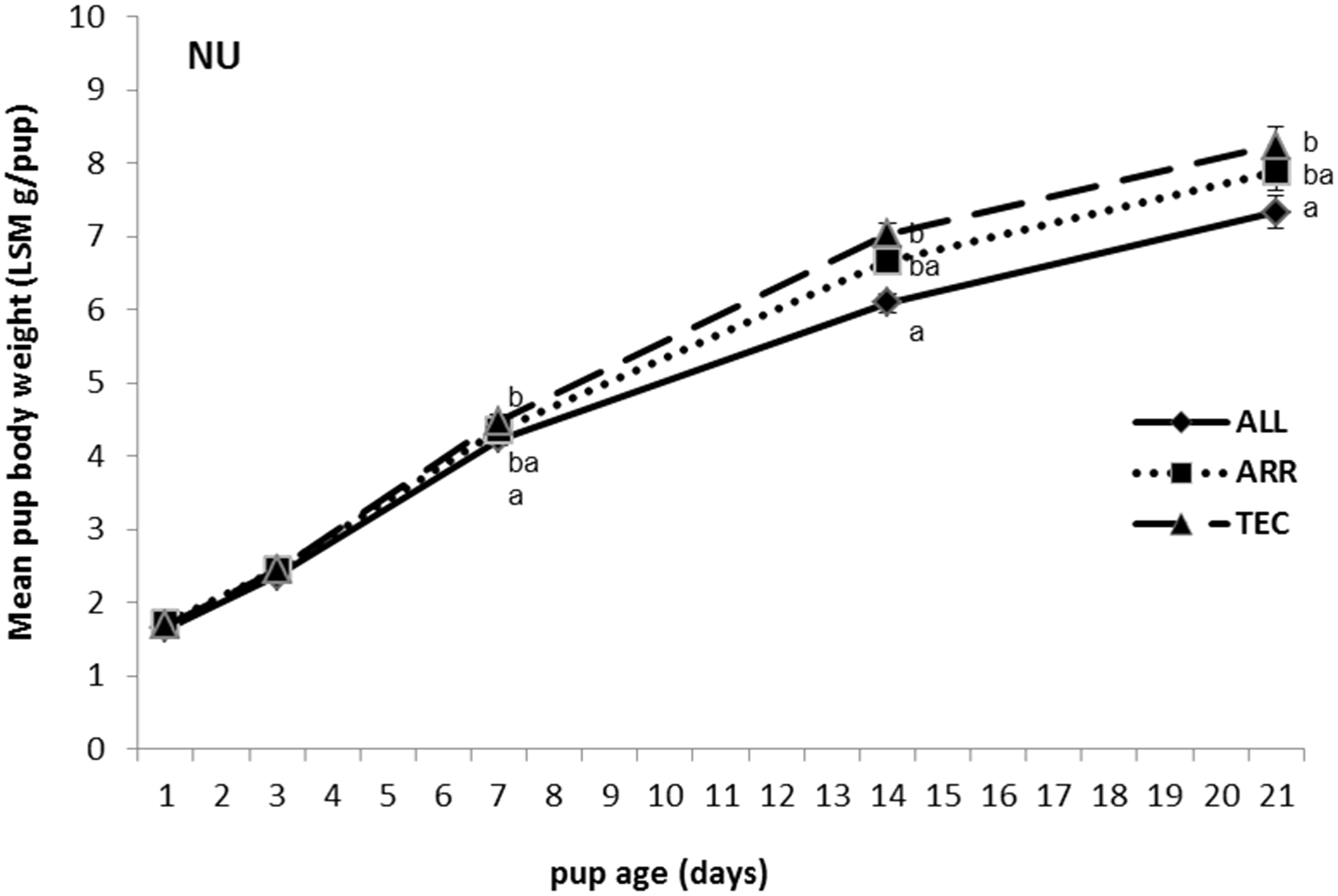
There were no differences in body weight between cage types from birth to weaning in B6 pups (Figure 3). The body weights in B6 pups at weaning was 8.8 ± 0.17, 8.4 ± 0.14 and 8.5 ± 0.18 g/pup in ALL, ARR and TEC cages, respectively. Pups of the NU strain in ALL cages weighed less than NU pups in TEC cages on days 7, 14 and 21 (Figure 4). The body weights in NU pups at weaning was 7.3 ± 0.23, 7.9 ± 0.26 and 8.2 ± 0.26 g/pup in ALL, ARR and TEC cages, respectively.
Body weight of B6 pups from birth (day 1) until weaning (day 21), housed in three different cage types. ALL: Allentown cages, ARR: Arrowmight cages, TEC: Tecniplast cages. No significant differences between treatments were found. Body weight of NU pups from birth (day 1) until weaning (day 21), housed in three different cage types. ALL: Allentown cages, ARR: Arrowmight cages, TEC: Tecniplast cages. Different letters at each time point indicate significant differences between treatments, P < 0.05.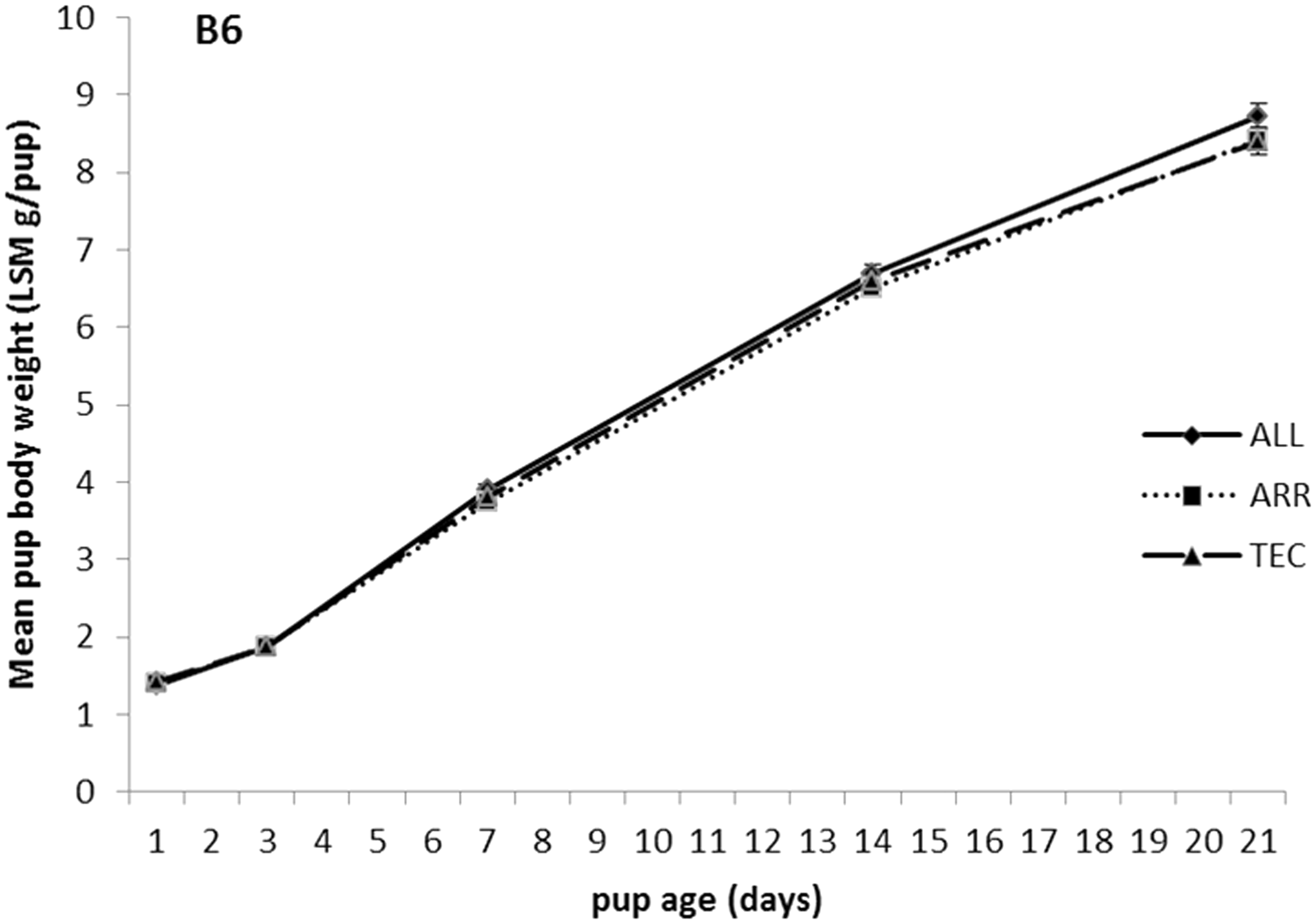
Water and feed consumption
Feed and water consumption of B6 and NU breeding females during the three lactation weeks from birth until weaning of pups, housed in three different cage types.
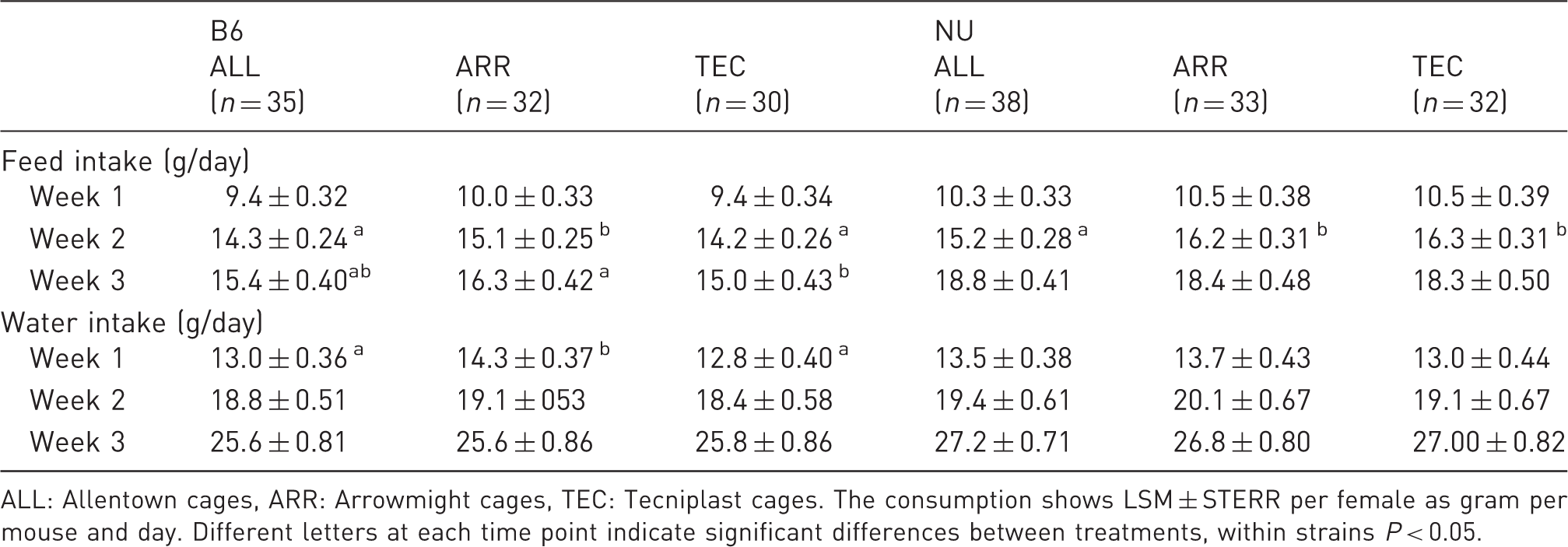
ALL: Allentown cages, ARR: Arrowmight cages, TEC: Tecniplast cages. The consumption shows LSM ± STERR per female as gram per mouse and day. Different letters at each time point indicate significant differences between treatments, within strains P < 0.05.
NU females in ALL cages consumed less feed than females in ARR and TEC cages during the second week of lactation (P < 0.05, Table 2). No other differences in feed and water consumption were found in NU females.
A general comment: from around day 14 the pups started to eat solid feed, which meant that feed intake for lactation week 3 was influenced by the intake of the pups.
Associations between pup weight and female weight, water consumption and feed consumption
Regression analyses showed no associations between pup weight and female weight for B6 animals. However, the weight of B6 pups was associated with water consumption (b = 0.04, 0.04 and 0.08 for ALL [P = 0.018], ARR [P = 0.015] and TEC [P < 0.001], respectively) and feed consumption (b = 0.07, 0.14 and 0.10 for ALL [P = 0.029], ARR [P < 0.001] and TEC [P < 0.002], respectively). This means that for example in the ALL cage system, B6 pups weighed on average 0.04 g more for every extra gram of water consumed, and 0.07 g more for every extra gram of feed consumed. Overall, the regression analysis of how female weight, water and feed consumption affected the weight of B6 pups indicated that the weight of the female (or female body condition) did not affect pup weight while water and feed consumption (of pups and females) did, and that there was no difference between cage systems.
The weight of NU pups was found to be associated with female weight when housed in the ALL cage system (b = 0.10, P < 0.001), tended to be associated with female weight in the ARR system (b = 0.05, P < 0.058) but was not associated in the TEC system. Moreover, the weight of NU pups was associated with water and feed consumption (b = 0.07, P < 0.044 and b = 0.11, P < 0.005, respectively) when housed in the ARR system, tended to be associated with feed consumption (b = 0.09, P < 0.070) in the ALL system, but was not associated when housed in the TEC system. Altogether, these results indicated that the weight (or performance) of the NU pups was less reliant on the body condition of the female and resource allowance in the TEC system than in the ALL and ARR systems.
Pup development
The development of the pups, monitored in the characterization protocol is presented in Table 3. The table gives the number of pups of each strain in each cage type that deviated from normal development, or expected results. In the righting reflex test, that reflects development of motor functions, the results show a great strain difference (Figure 5). In the NU strain, 90% (93% in TEC cages) of the pups succeeded in the test on day 3, while only 27% (TEC cages) and up to 48% (ARR cages) of B6 pups succeeded on day 3. No NU pups needed to redo the test after day 5, while seven B6 pups had to redo the test until day 7. Two B6 pups failed even when tested on day 7. When the mean latency in age (days) to succeed in the test was compared between cage types, the B6 pups in TEC cages had a significantly longer latency to pass the test compared with pups in ARR cages (4.3 ± 0.1 days old and 3.8 ± 0.1 days old, respectively, P < 0.05). B6 pups in ALL cages did not differ from the other two systems (4.1 ± 0.1 days old). For NU pups, no significant differences in latency to pass the test were found between cage systems (ALL: 3.1 ± 0.1 days old, ARR: 3.0 ± 0.1 days old and TEC: 3.0 ± 0.1 days old).
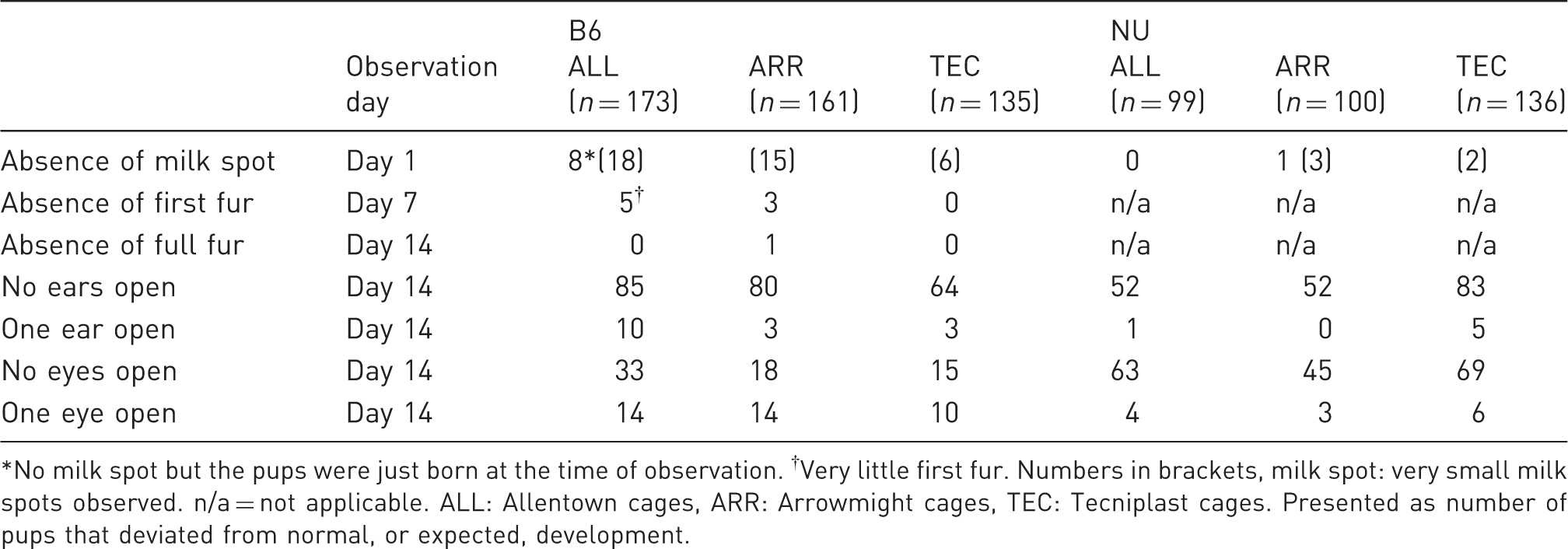
The performance in the righting reflex test presented as percentage of B6 and NU pups housed in three different cage types that succeed in the righting reflex test on day 3 to day 7. ALL: Allentown cages, ARR: Arrowmight cages, TEC: Tecniplast cages. Results from characterization protocol of development of B6 and NU pups in the different cage systems for day 1 to day 14. Presented as the number of pups that deviated from normal, or expected, development. No milk spot but the pups were just born at the time of observation. †Very little first fur. Numbers in brackets, milk spot: very small milk spots observed. n/a = not applicable. ALL: Allentown cages, ARR: Arrowmight cages, TEC: Tecniplast cages. Presented as number of pups that deviated from normal, or expected, development.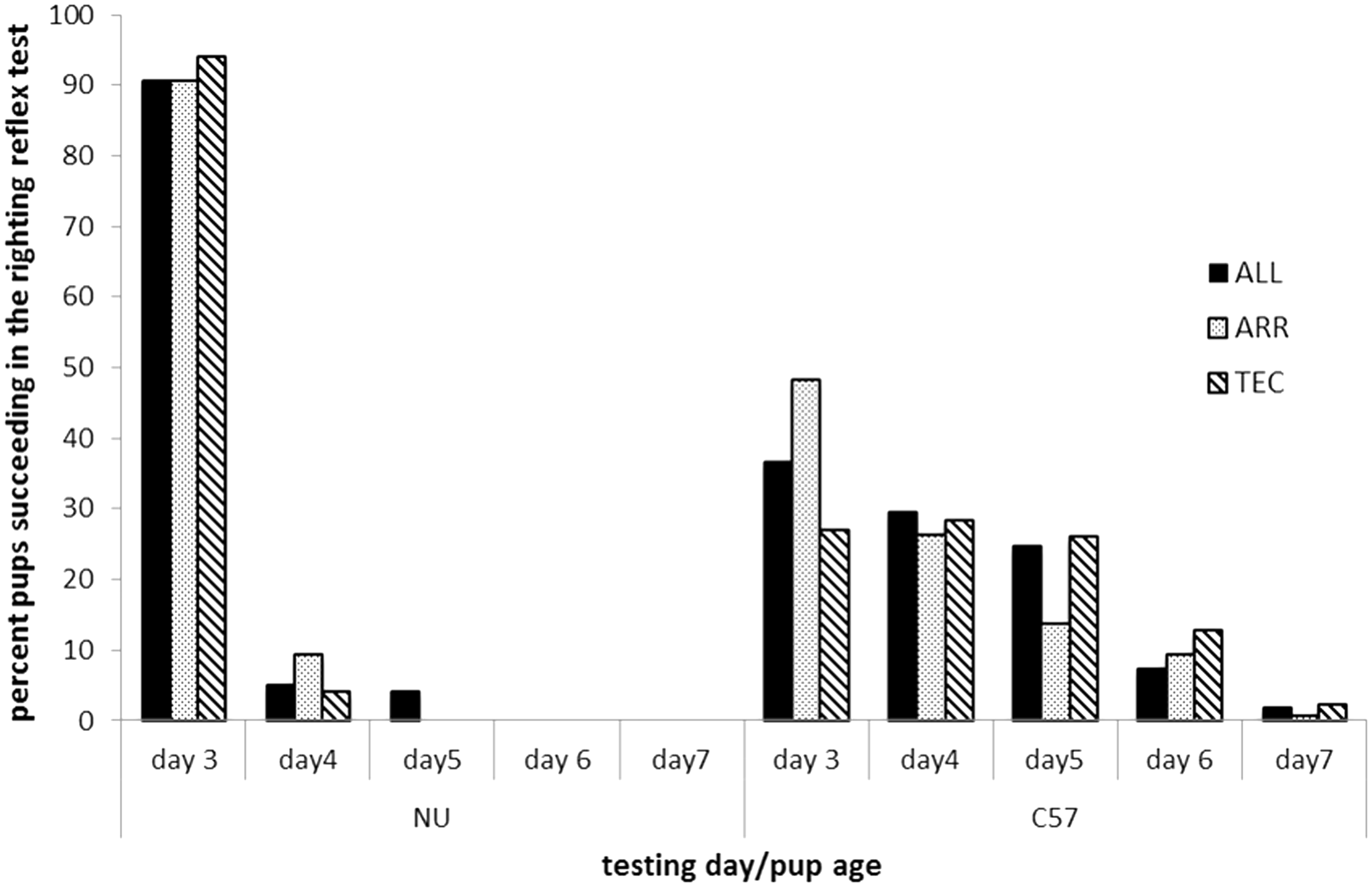
Nest score
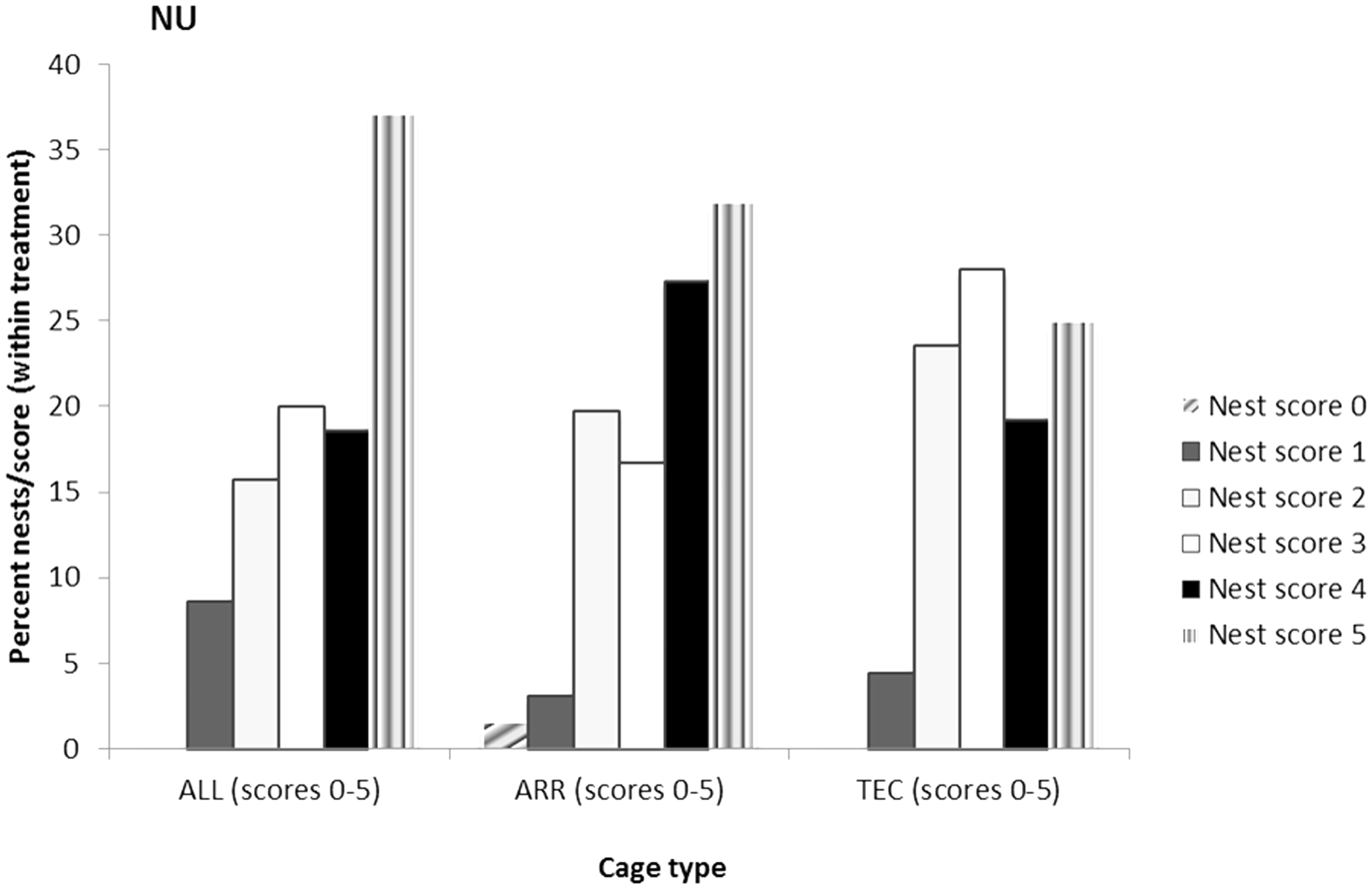
Nest score was analysed for the frequency of nests registered for each nest score. The pairwise comparisons show that for B6 females, the scores in TEC were higher than the scores in ALL (P < 0.05), recorded on days 1, 3, 7 and 14 during the lactation period (Figure 6). No differences between systems were found for the NU females (Figure 7).
Percent of nests registered for the different nest scores in three different cage types. Nests built by B6 females with litters and scored during lactation days 1–14. Score 1 represents a flat nest and score 5 represents a completely dome-shaped nest. ALL: Allentown cages, ARR: Arrowmight cages, TEC: Tecniplast cages. Different letters indicate significant differences between treatments, P < 0.05. Percent of nests registered for the different nest scores in three different cage types. Nests built by NU females with litters and scored during lactation days 1–14. Score 0 represents untouched nesting material and score 5 represents a completely dome-shaped nest. ALL: Allentown cages, ARR: Arrowmight cages, TEC: Tecniplast cages.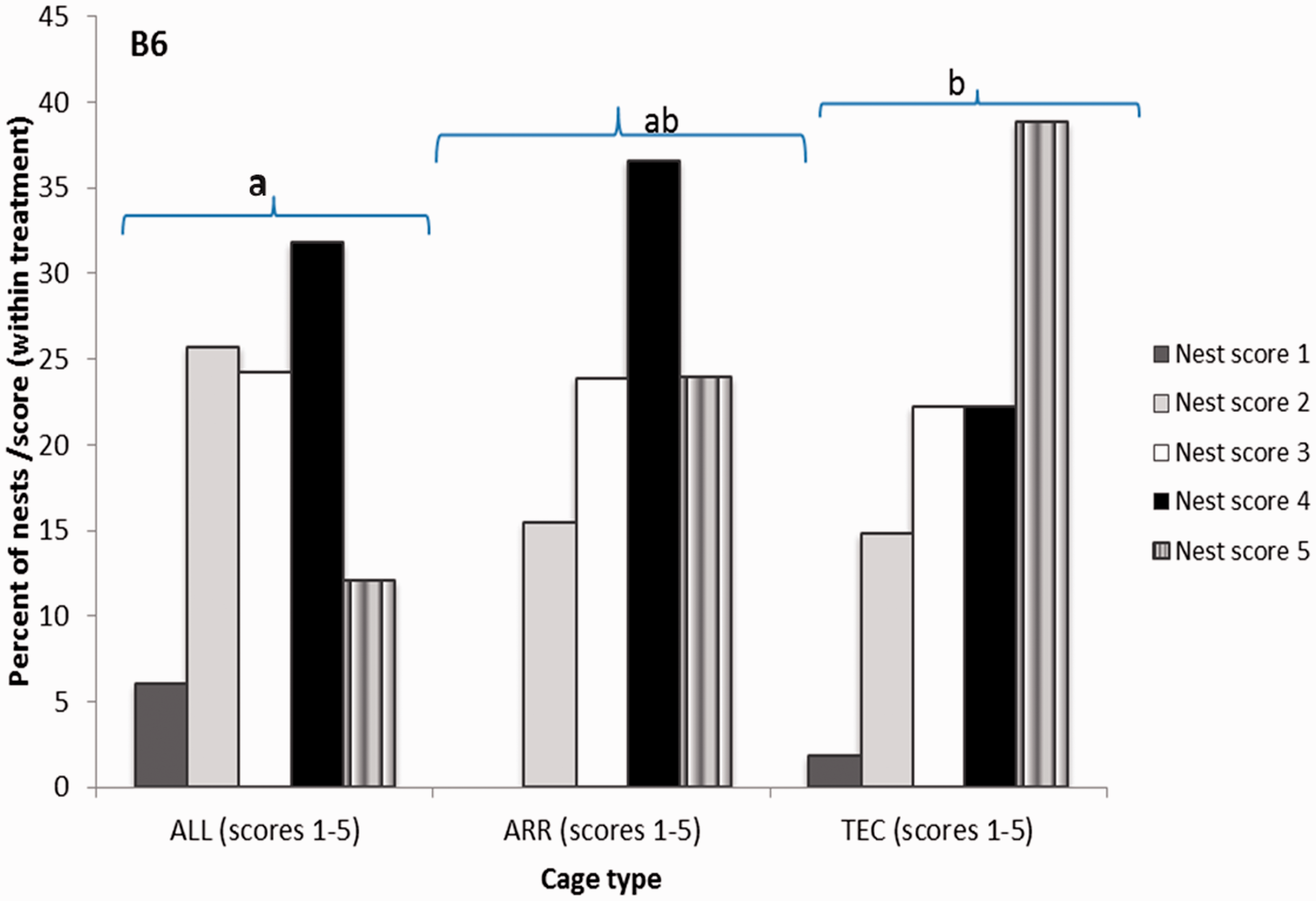
In-cage climate
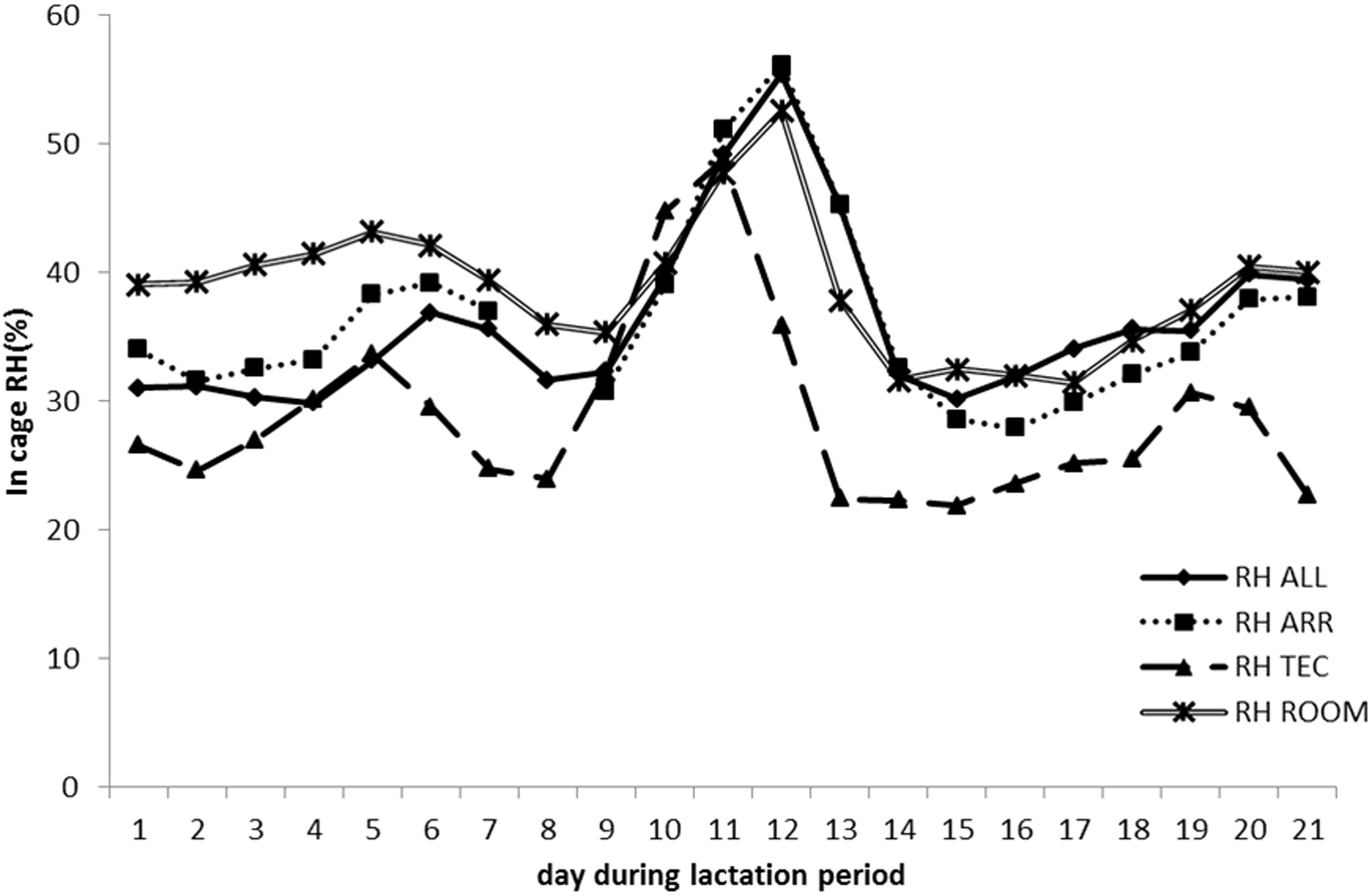
The mean temperature during the lactation period in the three different cage types were 24.8 ± 0.69℃ in ALL, 23.8 ± 0.61℃ in ARR, and 24.4 ± 0.86℃ in TEC. The corresponding levels of RH were 36.2 ± 6.72%, 36.5 ± 7.29% and 28.8 ± 7.18%, respectively. Around peak lactation (days 10–14), there was a slight increase in temperature in the cages (Figure 8) and a large increase in RH (Figure 9). The mean levels of temperature and RH for the peak lactation period were 25.6 ± 0.52℃ and 44.3 ± 8.94% in ALL, 24.5 ± 0.69℃ and 44.8 ± 9.34% in ARR, and 25.5 ± 1.05℃ and 34.8 ± 12.3% in TEC.
Temperature inside one cage per system housing one lactating B6 female with litter. Each day represents a mean of eight recordings, every 3rd hour. ALL: Allentown cages, ARR: Arrowmight cages, TEC: Tecniplast cages, ROOM: animal room. Relative humidity (RH) inside one cage per system housing one lactating B6 female with litter. Each day represents a mean of eight recordings, every 3rd hour. ALL: Allentown cages, ARR: Arrowmight cages, TEC: Tecniplast cages, ROOM: animal room.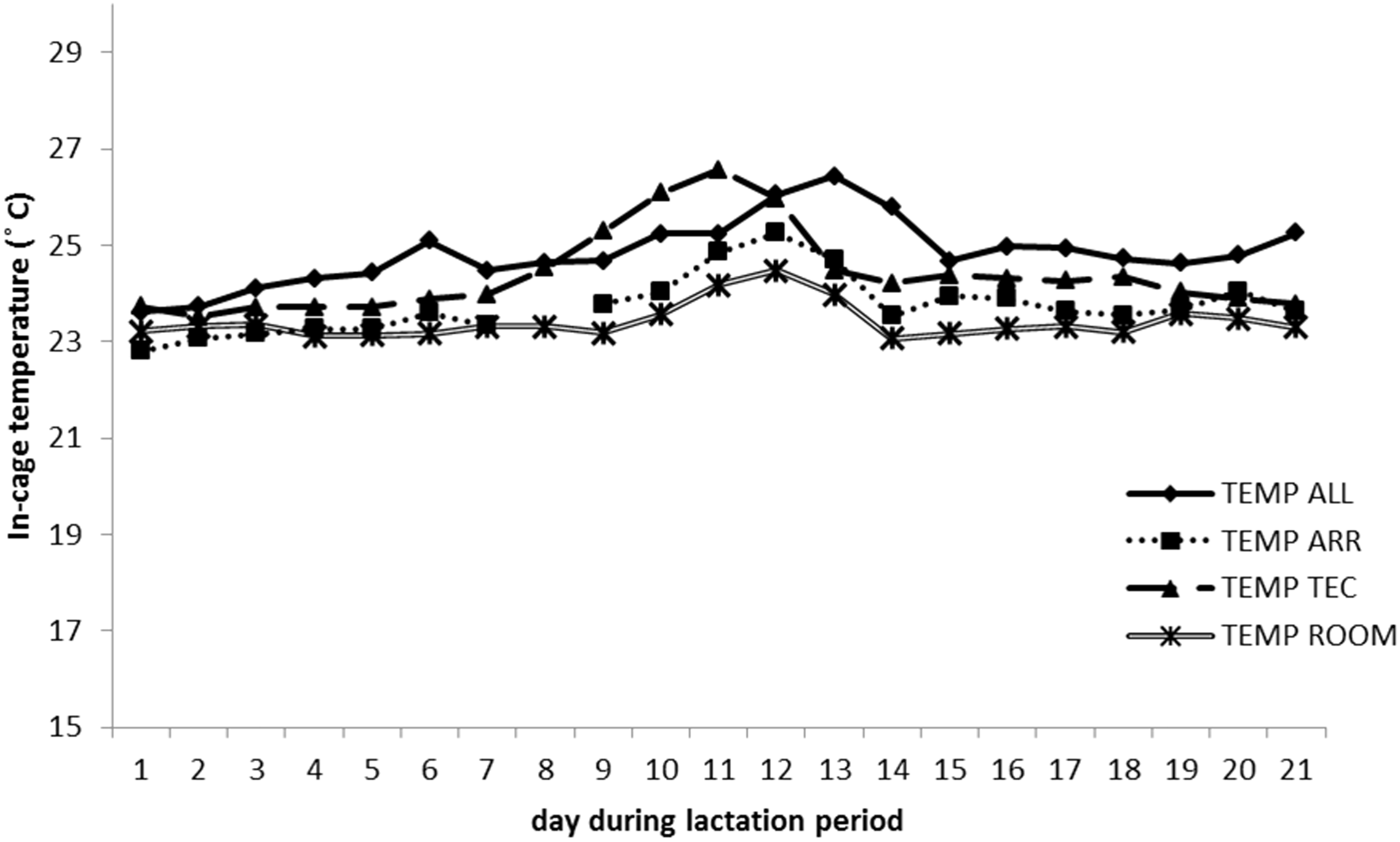
Discussion
The present study evaluated the effect of three different IVC housing types on maternal performance and pup development in C57BL/6NCrl and Crl:NMRI Foxn1nu female mice and their pups. The main findings were that NU females in ALL cages had a lower body weight during the lactation period, compared with NU females in ARR and TEC cages. This was also reflected in a lower body weight of NU pups, from seven days of age. In addition, there was temporarily lower feed intake in NU females in ALL cages during the second lactation week. The in-cage temperature and RH recorded during the lactation period revealed, above all, a peak in RH around days 10–14, in all cage types, which coincided with the period of peak lactation in mice. There was also a slight increase in temperature. This shows that peak lactation is a period when the females produce excess heat. Anecdotally, we observed the females often rested elongated (with maximal exposure of body surface) outside the nest during this period, which indicated a greater heat production.
There is a limit to feed intake in lactating female mice even if the litter size increases, which results in a decreasing mean pup mass with increased litter size. 17 According to Król and Speakman18,19 the limit is centrally controlled by the maximal capacity of the female to dissipate the heat produced when producing milk and processing the feed. The authors found effects of ambient temperature on female body mass, feed intake, milk energy output, litter size, mean pup mass and pup growth rate.18,19 Thermoneutrality (30℃) resulted in a decrease in the above listed parameters, compared with a cooler (21℃), or a cold environment (8℃). Shaving the fur on the back of lactating mice resulted in increased feed intake and milk production and heavier pups at weaning, compared with non-shaved females (at 21℃). 13 Lactating MF1 mice have been reported to have a 1.5℃ higher body temperature compared with non-reproductive mice. 20 In summary, lactating females in environments which are too warm risk hyperthermia if they do not decrease their milk production. The results of NU mice in ALL cages in the present study is in line with these findings (female and pup body weight, feed intake) which indicated that the ALL cage environment was experienced as warmer, especially at peak lactation, than the ARR and TEC cages. In accordance, the regression analysis indicated that the weight of NU pups was more reliant on the body condition of the females and resource allowance in ALL cages than in TEC cages, indicating that a physiological challenge on the females during this sensitive period also affected the pups. By contrast, the regression showed no effect of female body condition on pup weight in B6 mice. Apart from maintenance and growth, NU pups also need energy for thermoregulation. 21 Hence their feed intake likely needs to be greater than that of age-matched pups that have fur, such as B6 pups. This puts a greater demand on the female to produce enough milk for their growth, which could explain why ALL in-cage climate affected body weight and feed intake in NU females and not B6 females. In addition, B6 females are slightly smaller which gives a higher surface-to-volume ratio, i.e. they should in general have a greater heat loss.
Regarding the in-cage climate in ALL and TEC cages, the temperature was similar but the RH was higher in ALL cages. In addition, the number of ACH was 50 and 75 in ALL and TEC cages, respectively (as recommended by their manufacturers). The experienced temperature is affected by air movements/speed and the level of humidity. Air movements create heat loss through convection, and high levels of humidity will make the experienced temperature higher than it actually is. The higher rate of ACH in TEC cages should have resulted in a greater movement of air which the females might have experienced as a cooler environment. The lower RH in TEC cages should also have added to the cooling effect, further supporting this hypothesis. Kostomitsopoulos et al. 10 found increased water consumption in growing C57Bl/6 J male mice housed in forced-air IVCs (60 ACH), compared with those housed in motor-free IVCs (25 ACH), indicating that a higher ventilation rate gives drier air.
It is possible that the differences in body weights would have been eliminated if ALL and TEC had had the same number of ACH. Reeb-Whitaker et al. 5 found higher pup mortality at 30 ACH compared with 100 ACH in C57BL/6 J breeding pairs housed in 337 cm2. This is possibly another example of a too warm cage environment (30 ACH) for lactating females, and hence reduced milk production and poorer pup growth and survival. From an animal welfare perspective it is relevant to consider what in-cage climate is appropriate during breeding. Especially since it is common to house breeding trios (with double litters) in cages of this size, and not single mothers with one litter as in this study. We recorded in-cage climate in breeding trios during one mating period with levels of 23.7–24.9℃ and 44.3–59.4% RH in the three cage types. The in-cage climate is easily adjusted by changing the ACH,4,5 and maybe this should be considered during lactation. If the females had been exposed to additional challenges (experimental factors or altered basal physiology due to genetic modifications) the problem might have been even greater. However, it is also important to consider the optimal microclimate for the growing pups. Reeb-Whitaker et al. 5 found that, although more pups survived, they had lower weaning body weights at 100 ACH than at 30 ACH, which might indicate a too cold environment for them. These pups did not have access to nesting material which is important for modulating the microclimate and likely reduces the cooling effect on them. An alternative explanation could be that litters were larger (hence with lower body weight/pup) in 100 ACH conditions, but there were no difference in pups born or pups weaned per dam between 30 and 100 ACH. 5
Apart from the temporarily lower feed intake in NU females in ALL cages, B6 females showed increased intake in ARR cages. This difference in B6 mice could be correlated to the height of the hopper from the floor, which was lowest in ARR cages (5.0 cm). In addition, the feed hopper was V-shaped and placed in the middle of the cage which gave access to feed from both sides of the hopper and on several levels. The smaller B6 females could reach the feed hopper more easily in the ARR cage and therefore took the opportunity to eat more, while the NU females were slightly bigger and therefore reached the feed hopper equally well in all three cage types. ALL cages also have a V-shaped feed hopper but it was placed at the back of the cage resulting in more limited access to the feed. In TEC cages, the feed hopper was also located at the back of the cage and it had a flat bottom. There was much space available for eating but if there was little feed in the hopper the pellets might have just rotated or moved away when the mice tried to gnaw on them. In both TEC and ALL cages it was difficult to see the quantity of feed in the hopper without taking the cage out of the rack.
The main effect on pup development was the one of strain. The poorer growth of NU pups in ALL cages was not severe enough to affect their development (days 1–14 and righting reflex test). It is interesting that the highest number of NU pups (although not significant) that passed the righting reflex test on day 3 were housed in TEC cages, while the results were the opposite for B6 pups. Further studies would however be necessary to deduce the reason for this. This type of early characterization is relevant to find potential indicators of poor welfare 22 as well as abnormal development that might have consequences for the (future) animal model.
Nest building is an important fitness component and is positively correlated to the number of young born and weaned. 23 The quality of the nests is affected by the type of nesting material provided to the mice. 15 When given access to two different types of nesting material, mice have been observed to mix them, and to use paper or Kleenex tissue to line nests built of cellulose fibres or paper stripes.15,24 The nesting material used in the present study (Sizzlenest, shredded paper) made it possible for the females to build dome-shaped nests. The nests of B6 females were of a higher quality in TEC cages compared with those in ALL cages. If, as discussed above, the cage environment in TEC cages was perceived as a cooler environment, the B6 females might have found it necessary to build more complex nests to protect the pups from the frequent air movement in the cages. Gaskill et al. 25 have shown that mice with access to nests radiate less heat and have lower feed intake. This shows that nesting material is an important resource for mice which helps them to modulate their microclimate.
In conclusion, we found IVC in-cage climate has an effect on maternal performance in Crl:NMRI Foxn1nu female mice, which has consequences for pup body weight. The results presented here do not however make it possible to favour or discard any of the IVC systems tested. The optimal housing climate in IVCs seems to be dependent on the type of animals (e.g. age, reproductive state, sex or genotype) that are housed in the cages and, importantly, also on their physiological status. From an animal welfare perspective this is a management issue. IVC racks are usually large, holding more than 100 cages at times, and often housing different groups of animals. These are exposed to the same environment (in terms of ACH) all the time, which might be beneficial for some but disadvantageous for others. This should be taken into consideration when managing IVC housing, and the simplest and quickest way to improve the situation is probably to give all mice access to nesting materials. They can then build more or less complex nests depending on their needs, and can thereby modify their microclimate.
Footnotes
Acknowledgements
Thanks to Allentown, Arrowmight and Tecniplast for providing the equipment that made this study possible. Thanks also to Monica Aronsson at St Eriks Eye Hospital, Stockholm, and students Sofie Sundgren and Malin Wirf for assisting in the study.
