Abstract
Abstract
On 1 January 2013, research using cephalopod molluscs, from hatchlings to adults, became regulated within Directive 2010/63/EU. There are significant difficulties in captive breeding in the great majority of currently utilised species. Thus, scientific research relies upon the use of wild-caught animals. Furthermore, live cephalopods are shared and transported between different stakeholders and laboratories across Europe and other continents. Despite existing European and national legislation, codes, guidelines and reports from independent organisations, a set of recommendations specifically addressing the requirements for the capture and transport of animals belonging to this taxon are missing. In addition, although training and development of competence for all people involved in the supply chain are essential and aim to ensure that animals do not suffer from pain, distress or lasting harm, the requirements for those capturing and transporting wild cephalopods have not been considered. This Working Group reviewed the current literature to recognise scientific evidence and the best practice, and compiled a set of recommendations to provide guidance on the ‘techniques’ to be used for the capture and transport of live cephalopods for their use in scientific procedures. In addition, we propose to (a) develop standardised approaches able to assess recommended methods and objectively quantify the impact of these processes on animals’ health, welfare and stress response, and (b) design a training programme for people attaining the necessary competence for capture and transportation of live cephalopods, as required by Directive 2010/63/EU.
Introduction
Cephalopods (nautilus, cuttlefish, sepiolid, squid and octopus) are the sole invertebrates listed among the species regulated by Directive 2010/63/EU for use in scientific research. The taxon counts about 800 living species, all marine, and constitutes a class belonging to the phylum Mollusca. Over the last decades, discoveries about cephalopod biological, evolutionary, morphological, genomic and physiological innovations and adaptations, as well as their neural and cognitive characteristics, have promoted a renewed interest for these animals, favouring the increase in the number of studies and species utilised for scientific purposes.1 –6 At the same time, the relevance of their welfare status7,8 and its consequences on the scientific outcome have increased in both the commercial and the scientific fields.
Article 9 of Directive 2010/63/EU specifies that animals must not be taken from the wild for use in procedures (Article 9.1) unless the relevant National Competent Authority (NCA) grants an exemption (Article 9.2) based on the scientific justification that the purpose cannot be achieved using bred animals. 9
The Directive also requires that the capture of live wild animals should be accomplished by competent people using methods which do not cause avoidable pain, suffering, distress or lasting harm (PSDLH; Article 9.3). In addition, adequate care must be taken to prevent physical injury and stress to the animals at all stages in the supply chain, including capture, transportation and acclimatisation to laboratory conditions (and quarantine when required). Consequently, the capture and transportation of animals from the wild should be well planned, meticulously prepared and effectively performed.
For the vast majority of laboratory animals – including vertebrate aquatic model species – production technology has reached a maturity that allows their breeding for use in procedures. On the contrary, cephalopod culture is still in its infancy, having faced several bottlenecks to the point that few cephalopod species are currently cultured in captivity at a limited local scale.10,11 Culture protocols of cephalopods for scientific purpose are not fully developed yet, and evidence for successful rearing of multiple generations in captivity12 –14 without altering their welfare and behaviour is still lacking, possibly with a few exceptions. Similarly, doubts and criticisms arise around the development of a possible industrial aquaculture for these animals15,16 considering their sentience, sophisticated neural organisation and cognitive capabilities.1 –3,7
Despite the increased scientific interest in these organisms, most of the research performed on these molluscs still relies upon the collection and transport of wild-caught animals. Here, we present the outcomes of the work of the FELASA Working Group ‘Capture and transport of cephalopods’ with the aim of developing recommendations about methods to be utilised and guidance about the required competence of people involved in the capture and transport of these animals for scientific research.
Cephalopods as laboratory animals: a legislative framework for capture and transport
Since Directive 2010/63/EU came into force in 2013, no gold standard method has been proposed to capture wild aquatic animals – including cephalopods – for use in scientific research. We analysed the available legislation and recommendations regarding the capture and transport of aquatic animals, and we identified several regulatory and recommendation documents of interest. A summary of the outcomes of our analysis is available in Suppl_1:(Legislative framework in the Supplemental Material), while a detailed review is provided in the ancillary work. 17
From the analysis any legislative or regulatory document with explicit mention of cephalopods were found. In addition, we found that most of the recommendations are aimed at protecting animals during transportation, with limited detail provided for the capture from the wild, at least when aquatic species are considered.
Another key and reiterated element in these recommendations is the attention given to the competence of the personnel, that is, the need for proper training for those involved in the capture and transport of wild animals. All of the recommendations point out that the main source of suffering and distress in animals being captured, handled and transported relates to the limited competence of the people involved.
Despite the existence of European Directives and various guidelines and documents (see Suppl_1: Legislative framework in the Supplemental Material, and Pieroni et al. 17 ), it is clear that regulations on capture and transport lack specific recommendations about wild cephalopod species, at both general and species-specific levels (for a detailed overview, see table 1 in Pieroni et al. 17 ). However, considerations and precautions on capture and transport of cephalopods for research purposes are included in the Guidelines for the Care and Welfare of Cephalopods in Research. 18
Capture methods.
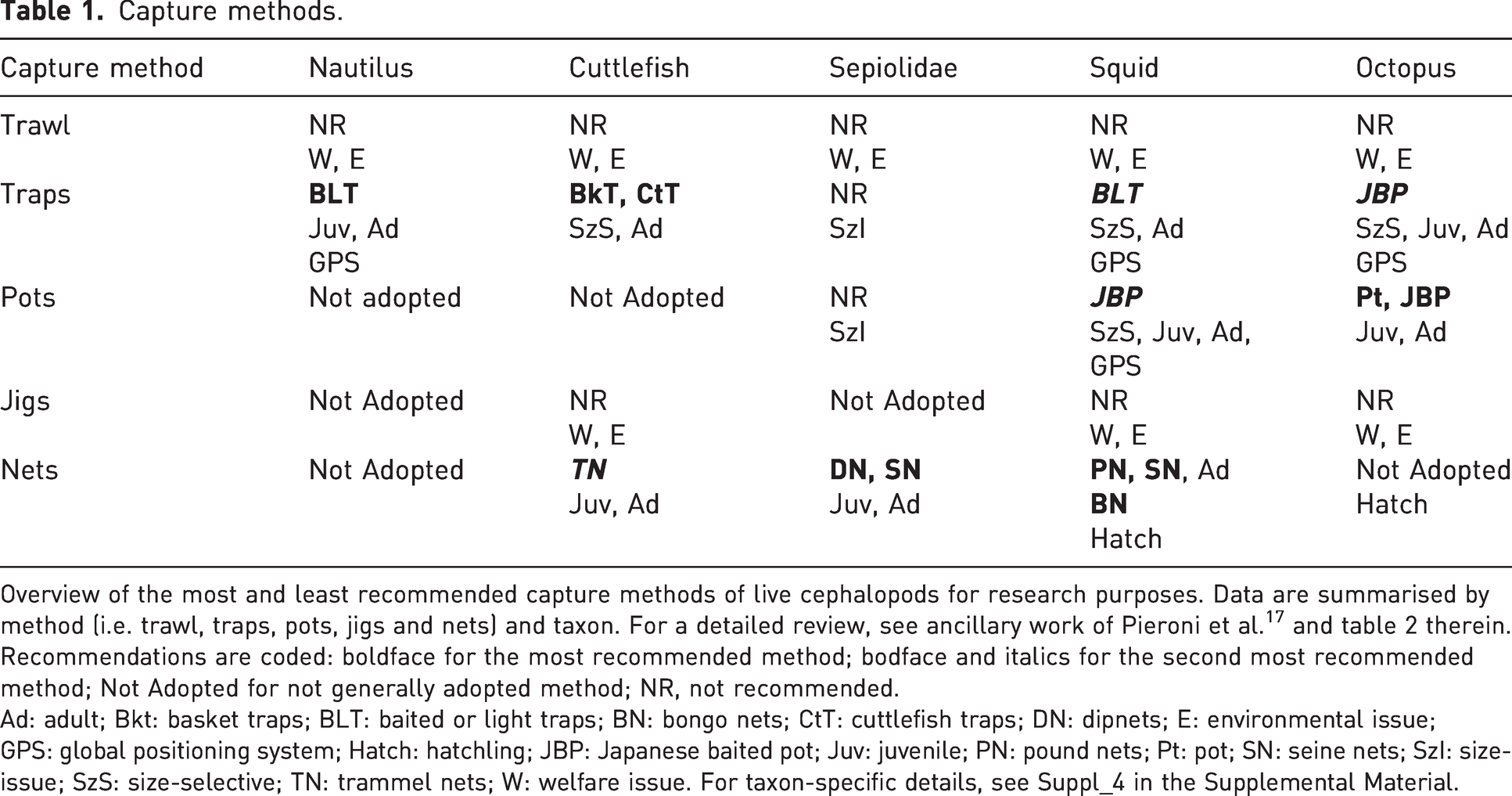
Overview of the most and least recommended capture methods of live cephalopods for research purposes. Data are summarised by method (i.e. trawl, traps, pots, jigs and nets) and taxon. For a detailed review, see ancillary work of Pieroni et al. 17 and table 2 therein. Recommendations are coded: boldface for the most recommended method; bodface and italics for the second most recommended method; Not Adopted for not generally adopted method; NR, not recommended.
Ad: adult; Bkt: basket traps; BLT: baited or light traps; BN: bongo nets; CtT: cuttlefish traps; DN: dipnets; E: environmental issue; GPS: global positioning system; Hatch: hatchling; JBP: Japanese baited pot; Juv: juvenile; PN: pound nets; Pt: pot; SN: seine nets; SzI: size-issue; SzS: size-selective; TN: trammel nets; W: welfare issue. For taxon-specific details, see Suppl_4 in the Supplemental Material.
Recommendations for capture and transport of cephalopods in research
The purpose of this Working Group was to carry out detailed text mining, which identified more than 100 published works. This informed a review of existing capture methods for collecting and about those adopted in transporting live wild cephalopods. The outcome of this analysis is not provided herein but is presented in detail in the ancillary work. 2
The analysis of the available information highlighted important considerations:
All the capture methods reported in various studies have been taken from fishery and readapted in a small set of cases for scientific purposes. There are no species-specific procedures but rather several ‘protocols’ and/or variants for the same method. No particular attention is given to the different life stages of cephalopods used in the studies, and this piece of information is often missing. Very little information is provided about the capture and transport procedures adopted, and in most cases, only one of the two ‘parts of the story’ is described. Some papers provide a list of recommendations which are mainly anecdotal or derive from indirect communications, and they should therefore be validated further by robust studies.
Maximising welfare during the capture of live wild cephalopods
According to the General Section 4.2 of the Council Recommendation of 18/06/2007:
Animals should be captured by ‘humane methods and by persons competent to apply them’, minimising ‘the impact of the capturing procedures on the remaining wildlife and habitats’. ‘Any animal found to be injured or in poor health should be examined by a competent person … In case of serious injury, the animal should be killed immediately by a humane method’ described in the Directive 2010/63/EU. ‘Appropriate and sufficient transport containers and means of transport should be available at capture sites, in case animals need to be moved for examination or treatment’.
19
How should we apply all of this advice to cephalopods? There is not a unique reliable method to capture every cephalopod species, but rather a small range of techniques that best fits the species-specific needs, also considering their life stage, physiology and inter-individual variability. When considering the following capture methods (Table 1), we recommend associating a severity assessment in order to try to predict the impact of a given protocol on the welfare of wild cephalopods.
Fishing: what can we learn from it?
Cephalopods are animals of great interest for commercial purposes, currently accounting for 5% of the marine capture volume worldwide, 20 with a significant increase in demand, although this was recently slowed by the COVID-19 outbreak (see Suppl_RN: Reference to the ancillary work and other notes in the Supplemental Material). For this reason, a great number of capture methods have been developed by artisan and small-scale fisheries. 21 On the other hand, many reports on cephalopods biology and fishing have been published (for a detailed review, see table 2 in Pieroni et al. 17 ), but no comprehensive summary of the numerous cephalopod capture methods is currently available for a given species and life stage. However, Rathjen 22 stated the need for ‘more resource-friendly’ fishing methods for these animals. In his work, line jigging was the most suitable fishing gear for squids (Loligo forbesii, Illex argentinus, Todarodes pacificus and Nototodarus sloanii). The technique appears selective and adjustable for the size of the specimen, thus limiting the impact on the environment or other fauna. Trawling is also much utilised for fishing cephalopods, but this results in the by-catch of other animals, as it is not species or size specific.
Transport methods.
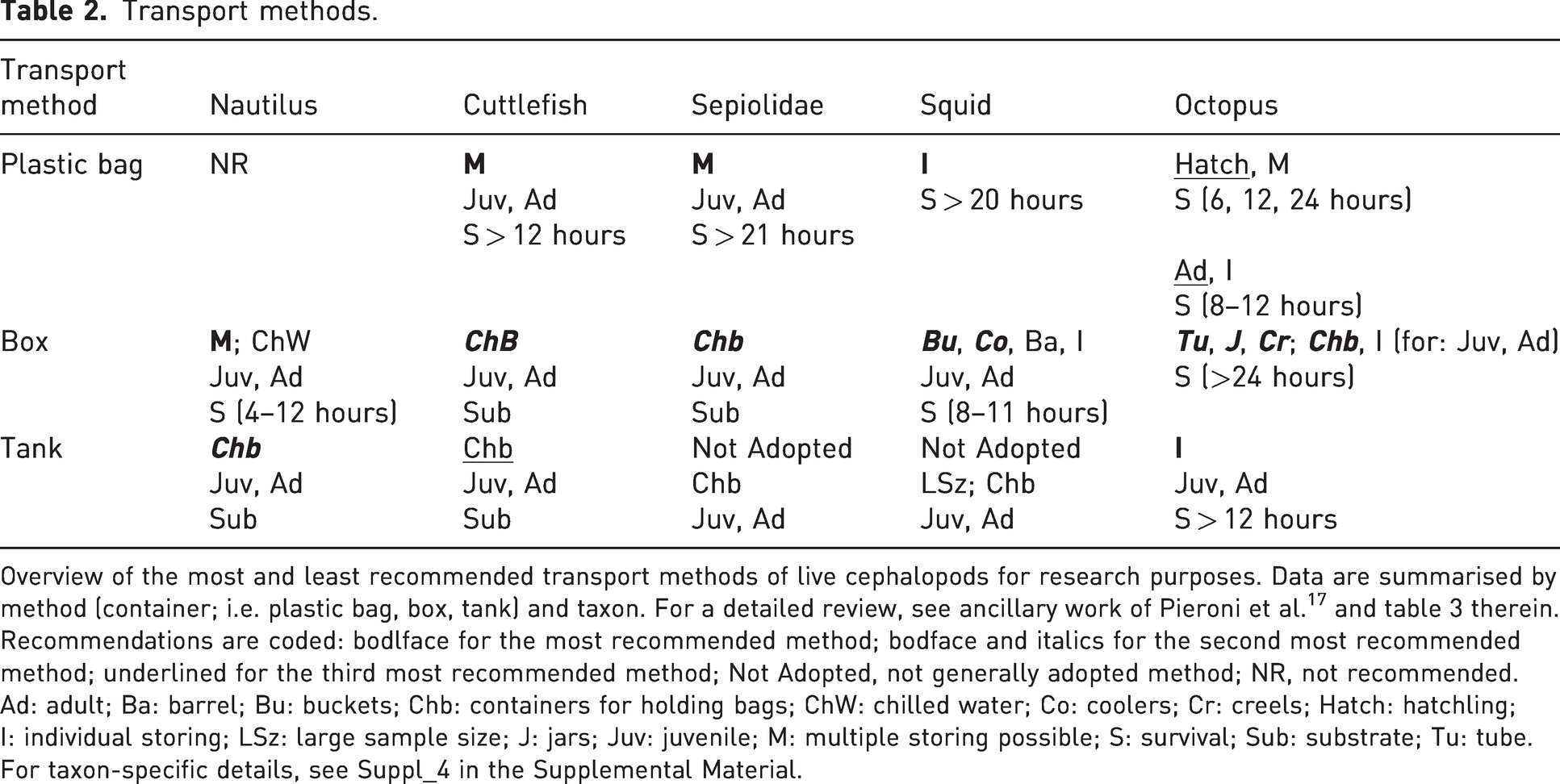
Overview of the most and least recommended transport methods of live cephalopods for research purposes. Data are summarised by method (container; i.e. plastic bag, box, tank) and taxon. For a detailed review, see ancillary work of Pieroni et al. 17 and table 3 therein. Recommendations are coded: bodlface for the most recommended method; bodface and italics for the second most recommended method; underlined for the third most recommended method; Not Adopted, not generally adopted method; NR, not recommended.
Ad: adult; Ba: barrel; Bu: buckets; Chb: containers for holding bags; ChW: chilled water; Co: coolers; Cr: creels; Hatch: hatchling; I: individual storing; LSz: large sample size; J: jars; Juv: juvenile; M: multiple storing possible; S: survival; Sub: substrate; Tu: tube. For taxon-specific details, see Suppl_4 in the Supplemental Material.
Traps and pots are utilised in many geographical areas and represent traditional gears for fishing cephalopods. These rely on the natural trend of some species to search for dens and hidden refuges. Spearing, multiple hooks and trolling are still used, although with local adaptions and relatively small variations.
In our opinion, the fundamental question for the scientific community would be whether we can adjust some of the currently available fishing methods to render them suitable for collecting cephalopods used for research purposes. The answer is yes, providing that coordinated, cooperative interaction between different stakeholders is established.
Capture of live cephalopods for research purposes
An overview of the best recommended methods for the capture of live cephalopods for research purposes is provided in Table 1.
Nautiluses
Collectors and scientists unanimously agree upon the use of baited or light traps as the best method for capturing live wild nautiluses. A prototype originally reported by Carlson 23 is still utilised with some variations, including monitoring systems.
Cuttlefishes and sepiolids
From the little information available about the capture methods for cuttlefishes, the most feasible technique seem to be traps, in particular basket or cuttlefish traps, very similar to those employed for squid but larger and lighter. Under these conditions, juvenile and adult cuttlefishes are captured uninjured. These size-selective gears may allow for the addition of the seabed which can be used as an attractive spawning substrate for broodstock destined for aquaculture purposes. 24 Large nets, such as trammel nets, are also suitable for catching both juvenile and adult animals without excessive constraint and without resulting in any environmental issue as is the case for trawling. Seine nets and dipnets are mostly used for adult forms of sepiolids (e.g. Euprymna scolopes and E. tasmanica) destined for research and are considered the less traumatic method for these small cephalopods. 25
Squids
For squids, one of the most frequently employed capture method is the jig lure with barbless hooks operated mechanically or by hand. 26 Analyses of the impact of this capture method revealed that it induces some injury to the animals (see Table 1 and Suppl_4: Supporting info to the overview of capture and transport methods in the Supplemental Material). Alternatively, several kinds of nets have been utilised to capture squids for laboratory use: pound nets, bongo nets, seine and dip nets which all proved to be harmless. These nets are large, and squids are able to swim before they get caught, forming a consistent sample size.17,27 Furthermore, these appear suitable for capturing specimens of any life form, but attention must be paid to the by-catch of egg masses.
Octopuses
Undoubtedly the best existing capture method for octopuses is the pot.21,28,29 Pots, like traps, are generally made of natural, non-toxic materials with non-abrasive surfaces and exploit the natural tendency of these animals to search for a den. Octopuses spontaneously settle in these gears which are very likely to catch undamaged specimens. As reviewed, 17 pots should have a dark tone, narrow entrance and large interior that allows the animal to settle and see outside without exposing itself to danger. A series of adjustments have been proposed, such as the insertion of a GPS monitoring system or the addition of a removable lid that might also be useful for transportation. Pots are alluring for both juveniles and adults, and very often they can be chosen by females as substrate for laying eggs, which, if not required for the aim of the project, should be returned to nature afterwards. A combination between pots and traps are the so-called Japanese baited pots, 30 combining shelter and bait (Table 1).
Transport methods of cephalopods: maximising welfare from sea to the lab and between labs
With regard to capture, the best transportation methods should avoid (or at least limit) PSDLH and should not increase the stress associated with the capture technique. Again, most likely, there is not a unique protocol to transport cephalopod species (for a detailed review, see table 3 in Pieroni et al. 17 ); inter-individual variability and species-specific features (e.g. body size, physiology and biological requirements for every life stage) must be considered when preparing the animal for the journey.
A detailed list of considerations for transportation of live cephalopods for research purposes is provided by Fiorito and colleagues 18 which in turn is mostly based on available guidelines for the transportation of live fishes, 31 in compliance with the codes and regulations for European and international transport of live animals (see Suppl_1.2: Principles for transport, documentation and planning of the journey in the Supplemental Material). The work could be therefore considered as the groundwork for building upon the best recommendations for cephalopod transport summarised here in Table 2.
In the first known guidelines on the rearing of cephalopods for scientific use (Grimpe’s ‘Care, treatment and rearing of cephalopods for zoological and physiological purposes’),32,33 several insights about the methods of transportation and maintenance of the animals during the journey for different species of cephalopods are provided. These can be considered for the development of good practice. In Grimpe’s words, a vital aspect is always to keep cephalopods in well-oxygenated seawater throughout the journey, as these animals have a high metabolic rate that rapidly produces large amount of carbon dioxide and ammonia.34 –37 For this reason and to limit distress (e.g. agonistic attacks and aggression, inking), animals should be kept individually in separate bags. Some cephalopods are shipped in Styrofoam fish boxes (cooled or heated according to the species), although a temperature slightly below the optimum has been suggested because it reduces the animals’ metabolic rate, allowing the shipping water to hold more oxygen and reduce waste production. 10 However, containers should be kept in the shade when being transporting (e.g. by boats), or air conditioning should be used when transporting by car or other vehicles (see Suppl_3: Other recommendations for transport of live cephalopods in the Supplemental Material).
Currently, there are no specific aerated containers designed for cephalopods, and neither is there an open system for their transport, but these could be obtained by adjusting those available for live fish transportation.31,38 Containers can be provided with substrate only during short journeys, as the addition of organic material can be subjected to decomposition processes, thus reducing water quality and oxygen, potentially compromising animal welfare during longer journeys; sand poses the risk of H2S release from fouled sediments. Sedation (e.g. cold water, MgSO4 or MgCl2) is not essential and is not recommended for the transport of most cephalopods, despite being suggested in some cases. 39
With regard to capture methods, we reviewed the literature concerning the transportation of cephalopods (see Pieroni et al. 17 and table 3 therein) in order to extrapolate general indications that might be useful for creating some species-specific recommendations. Here, we summarise the recommended methods for the transport of live cephalopods for research purposes (see also Table 2).
Nautiluses
Boxes or insulated chests with chilled seawater have been recommended for both juveniles and adults of different species of nautiluses and are preferred to plastic bags, as these can wear out, putting the welfare of the animals at risk. More specimens can be contained in the same box, providing each animal with at least 4 L of seawater. 23
Cuttlefishes and sepiolids
The transportation of cuttlefish can be challenging; animals should be transported in plastic bags or barrels, using large containers to store them. It has been suggested to transport fewer animals per plastic bag or barrel according to the size. 40 Our recommendation is to transport animals individually and not as ‘group’ of individuals. As for cuttlefishes, sepiolids are mainly transported in plastic bags containing few individuals according to body size and the relative final volume of the container. It is recommended to put these bags in larger insulated boxes that ensure no leakage or asphyxiation of the animals. 13
Squids
During transport, squids should be individually placed in plastic bags (preferably laid horizontally), barrels or buckets filled with seawater and oxygen in appropriate proportions (Table 2), sealed and placed in larger tanks or Styrofoam boxes.
Octopuses
The personal experience of Grimpe 32 with Octopus vulgaris, Eledone moschata and other species made the author suggest the use of enamel pots placed in Demijohn baskets with stuffed hay between them in order to reduce potential insults related to the transport method. 33 These have a cylindrical base and are conically tapered at the top. Only the lower part of the pot (containing between 20 and 80 L, depending on the size of the Demijohn) should be filled with seawater; the rest must be air, the circulation of which must be assured by multiple holes in the cork. 32 Pots, such as those utilised for the capture of octopuses, may be employed to facilitate transportation (see Table 2). These should be placed in a larger container or tank, as in a modern version of Demijohn baskets. Containers must be filled with seawater (recommended from the collection site) saturated with oxygen. Our suggestion based on knowledge of the biology of these animals is to keep each in individual separate bags or pots and not together with other specimens. Grimpe’s original recommendation32,33 has been readapted in different ways, but his approach for successful transport of live cephalopods is still valid and has been widely applied.
Eggs as a possible alternative
The collection and standardised transport of eggs for target cephalopod species have been proposed as an alternative, since these appear easier to manage. For ‘classic’ laboratory species (e.g. fish), moving embryos, sperm or eggs between research facilities has increased and is now well established. This approach is also considered as a way to comply with one of the 3Rs – Refinement – since transport appears to be stressful for live animals and could impact their welfare, while for early life stages, it seems to have limited effects (although no specific studies are known). On the other hand, the use of captive animals raised from eggs would allow experimental requirements to be fulfilled, such as controlling previous experiences.
With regard to cephalopods, consideration should be given to offer a standardised method of egg collection to reduce the impact on natural resources (e.g. collecting those stranded on the coastline or those originating from by-catch) and to minimise the harm to the egg masses. By-catch as a source of eggs should be the recommended option. Of course, this is not possible for all species currently used in research. Although cephalopod eggs need to be handled delicately, accurate temperature and salinity control during all phases of the process, including transportation, are equally important.41,42
In terms of eggs, it is preferable to obtain egg clutches in the middle stage of development because the survival rate is the highest, whereas during the early stages, death of the embryos is more likely to occur. On the other hand, eggs collected at advanced stages of development may prematurely hatch during transport, as their metabolism is very high, 43 and such conditions should therefore be attentively taken into account to fulfil any welfare requirement properly.
For some cephalopod species, maternal care is needed (e.g. incirrate octopods or oceanic squids44,45), and attempts to simulate properly are mandatory. Maternal care is a critical factor for proper embryonic development and for limiting the risk of premature hatching.10,46 –49 The utmost attention is required in these cases, and specific standardised protocols require further development, despite some attempts.32,42,50
Directive 2010/63/EU considers the protection of this taxon from hatching. However, several studies are based on the collection and culture of egg masses from the wild which – although easier to obtain and then transport in terms of the size of containers and water volume – require particular care, being very sensitive even to small changes in temperature, pH and salinity. 51 Storing conditions during embryonic development should also be monitored because the health state of the eggs will consequently affect the ‘quality’ of the hatchlings.
Methods for transport of eggs of several cephalopod species have been developed, achieving some standardisation; examples are available for cuttlefish, squid and octopus eggs.32,40,42
Future needs and how to achieve them
Two main actions are suggested for the future: (a) collaboration between fishermen and scientific research to standardise and implement further the best recommended methods for the capture and transport of live cephalopods for research purposes; and (b) a training programme that may help to increase the acquisition of the required competence for the people involved. Here, we briefly discuss these two actions.
Developing an experimental approach to validate recommended methods
What emerges from the analysis of the scientific literature, various recommendations, technical reports and unpublished data (for a review, see ancillary work 17 ) is the need for more in-depth studies on the capture and transportation methods that are specifically able to address the best way to handle these animals and their welfare under such circumstances. Body size, life stages and the biological and physiological needs of the species are all fundamental aspects to consider. The vast majority of methods currently utilised are based on personal experience and interaction with local fishermen, but relatively little scientific systematic studies have been carried out for the purpose of assessing the best capture and transport methods for cephalopods.
Approaches to assess the stress response (indicator of animal welfare) of aquatic animals to the capture and transport methods have been applied to fish and a few crustaceans, supporting the identification of the most appropriate catching gear to be used for these animals. These studies also provide useful approaches for the design of taxon-specific containers suitable for different modalities of transportation (for a review, see Pieroni et al. 17 ). With regard to cephalopods, only recently, Araújo and co-workers 52 studied the effects of a simulated long-journey transportation at high density on live Octopus vulgaris. No mortality was recorded at the end of 48 hours of transportation at the different densities of animals considered, and no significant changes in the physiological parameters were found. In another study, Barragán-Méndez and colleagues 53 evaluated the impact of some octopus species after capture (i.e. trawl; see also Pieroni et al. 17 ). Despite these works, studies are still required to facilitate informed guidance on species-specific capture and transport methods based on the assessment and control of stress-induced levels in live cephalopods. This Working Group promotes ad hoc studies that will help to achieve this goal.
We recommend a set of experiments designed to evaluate the physiological effects of different combinations of capture and transport methods on both sexes of juveniles and adults of the most commonly utilised cephalopod species in scientific research. The idea behind this is to adopt a collaborative effort with selected geographically distributed fishermen communities using a significant number of individuals for each species (and possibly in two separate seasons), utilising a couple of capture methods chosen for comparison with those claimed to be the most recommended ones such as: (a) nets versus traps in cuttlefishes, (b) jigs versus traps in squids and (c) pots versus traps in octopus. Target species should be those mostly utilised in research to facilitate the focus of these studies. Following capture, different transport conditions will be tested (e.g. individual vs. multiple ‘storing’ of animals kept in standard, open, large, darkened buckets or plastic bags). After the transportation phase (3–4 or 24 hours, testing duration of transport), the welfare of the animals will be assessed using a panel of indicators selected among those included in table 5 of the FELASA guidelines. 18 Monitoring individual animals should be performed (a) immediately after capture, (b) immediately after transportation and (c) on day 1 and on day 4 or 5 after capture to measure how much time the animals take to recover and acclimatise to the estimated baseline levels of these physiological indicators (for additional details, see also Pieroni et al. 17 ). Once preliminary studies have proven effective in guaranteeing the survival of the cephalopods, other investigations will follow in close collaboration with fishermen to reach a consensus on the benefits and expanding the study to a larger group and conditions (geographical, boats, etc.).
We aim to find the best conditions for capture and transport that could be used to improve animal welfare in different circumstances (e.g. capture and transport of different life stages, intercontinental journeys, etc.). In addition, we are convinced of the importance of the active involvement of fishermen and transporters as scientific suppliers for live cephalopod species.
Increasing competence and good practices: towards ad hoc training
Article 23 of Directive 2010/63/EU specifies the need for competent personnel when (a) carrying out procedures on animals, (b) designing procedures and projects, (c) taking care of the animals and (d) killing animals in order to limit and/or avoid the induction of PSDLH in the animals. 9 Both the capture and transport of cephalopods are part of the phases that relate to the ‘life’ of live animals for scientific purposes and must be performed by trained and expert personnel. As such, in our view, the ‘working document on the development of a common education and training framework to fulfil the requirements under the Directive’ should be addressed also to people specifically involved in the capture and transport of living cephalopods. Annex IV of EU Council Regulation No 1/2005 has already provided instructions concerning the training for transporters which shall include notions on animal physiology and their needs, handling and impact on stress and welfare. 54
Here, we propose a training programme (see Suppl_Box 1 in the Supplemental Material) designed for the suppliers of live animals to improve their competence in dealing with cephalopods with the aim of assuring compliance with species-specific biological, physiological and behavioural needs and welfare requirements.
We are convinced that by focusing on the training of fishermen – whose expertise and practical knowledge of the sea are undoubtable – we will be able to develop the best practice for capturing cephalopods in the most humane way. Of course, fishermen’s beliefs and behaviour depend upon the economic and social structure within which they are operating (see discussion in Pieroni et al. 17 ). We want to rely firmly on the experience of proud lifetime cephalopod artisan and small-scale fishers, and we are interested in their holistic analysis of the context. What we want to achieve is joint action between trained personnel, which will ensure that wild-caught cephalopods will be properly captured and transferred to their destination without experiencing unacceptable pain or suffering.
The most challenging part is to approach fishermen and transporters and persuade them to take part in the training and education process. The biggest resistance could be due to the loss of potential work by undertaking the training without having beneficial personal profit. It is therefore our aim to find a general balance between their needs and the obligations necessary for fulfilling competence, as required by the increasing attention on all aspects of animal care due to the inclusion of live cephalopods in the regulations for scientific research purposes. In our view, incentives should be provided for attendees who will partake in becoming suppliers of live wild animals for the laboratories, such as gaining a greater income and the possibility of working throughout Europe and not necessarily just in their own country or for the local supply. Moreover, since the successful trainees will receive a certificate of completion, this may help them to access fishing licenses more easily according to national (and possibly international) legislation. A coordinated effort between different stakeholders, including non-profit organisations and local and national governments, will then be required. The training framework should be accessible, affordable and – with joint effort from the member states – hopefully free, as well as being flexible to meet the working time or shifts of the trainees.
Suppl_Box 1 in the Supplemental Material summarises the organisation of the proposed training programme. It is our aim that fishermen and transporters of live cephalopods should be actively encouraged to face the challenge of improving the well-being of the animals they work with, by achieving awareness about the concept of welfare, the biological needs of the species and ethical approaches when dealing with cephalopods as animals destinated for scientific work. In order to reach the maximum degree of commitment, a special version of a dedicated training course for cephalopods will be designed so as to allow people coming from different cultural backgrounds to undertake an initial induction course that will help them to reach the same level when approaching the objectives of the main training programme (see Suppl_Box 1 in the Supplemental Material). Once successful, collectors and shippers may start working under the supervision of an expert group for a period of time to monitor further if the required skills match the animals’ and stakeholders’ benefits, and to assure animal welfare will be always the top priority.
Concluding remarks
In this report, the Capture and Transport of Cephalopods FELASA Working Group wanted to highlight the current limited knowledge about protocols for the capture and transport of cephalopods destined for scientific research. European legislation and recommendations are not sufficient and do not directly address cephalopods (see the review in Pieroni et al. 17 ), while regulations concerning the wildlife capture methods are poorer in taxon-specific information, even for vertebrates. Different organisations and few other countries include cephalopods among the animals whose welfare should be protected during the capture and transport and are mainly bound to international transport and shipping rules. From the text mining carried out in this Working Group (see also the ancillary work 17 ), some considerations emerged about the need never to omit fundamental information in scientific work, as also recommended by PREPARE55,56 and ARRIVE57,58 guidelines. We attempted to define the capture and transport conditions likely to be the most suitable for specific taxon (Tables 1 and 2) with attention to the target life stages needed for experimental studies (see Pieroni et al. 17 ).
General considerations that can be drawn from this analysis are:
The best capture method is any harmless tool that exploits the natural behavioural tendency of the animal (e.g. seabed substrate for spawning cuttlefish, octopus’s preference for dens) and that considers its daily cycle and diet composition – according to the species and its life stage – to catch it more efficiently. A good capture method should be classified as a mild procedure, and therefore the target cephalopod should experience only short-term distress. All the large-scale non-selective methods (e.g. trawl) must be avoided because of the impact on the welfare of the animals (and on the environment59
–62). The best transportation method is the one able to avoid or reduce further stress related to the capture procedure. The key factor in preventing the animals from experiencing PSDLH is planning (duration, resting place and number of health checks), thus avoiding any factor that may compromise animal welfare. Environmental requirements (e.g. oxygen, pH, salinity and temperature) must be monitored throughout the journey, and they should fit the welfare requirements of the different cephalopod taxa. Depending on the duration of the journey, particular attention should be paid to the type, size and equipment of the means of transport, as well as the containers used while on board.
These two processes are not independent from each other. To transport the animals better, the capture procedure should be done properly to avoid any handling and exposure to aversive conditions from the collecting site to the container. With this in mind, we propose pilot studies that could be carried out in a collaborative fashion with select geographically distributed fishermen communities to compare the effect of different combinations of capture and/or transport methods on the survival rate, physical conditions and physiological milieu of the captured (and transported) individuals belonging to the most common cephalopod species (e.g. cuttlefish and octopus).
Finally, the competence of the personnel carrying out these activities is pivotal in the success of both capture and transport methods. For this reason, we propose a special version of the education and training programme for cephalopods (CBC FELASA accredited training programme) dedicated to fishermen and transporters and designed to consolidate the required competence and attention to animal welfare.
Supplemental Material
sj-pdf-1-lan-10.1177_00236772231176347 - Supplemental material for FELASA Working Group report: Capture and transport of live cephalopods – recommendations for scientific purposes
Supplemental material, sj-pdf-1-lan-10.1177_00236772231176347 for FELASA Working Group report: Capture and transport of live cephalopods – recommendations for scientific purposes by Antonio V Sykes, Viola Galligioni, Juan Estefanell, Stuart Hetherington, Marco Brocca, Joao Correia, André Ferreira, Eleonora M. Pieroni and Graziano Fiorito in Laboratory Animals
Footnotes
Acknowledgements
This Working Group would like to acknowledge Dr João Pereira, Dr Graziano Fiorito and Dr David Smith as the proposers of this initiative to FELASA. Dr G. Ponte provided advice and networking experience based on the COST Action FA1301. The Working Group is also grateful to the Association of Cephalopod Research ‘CephRes’ and to FELASA for their support.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Attendance at the meetings of this Working Group was supported by travel grants from FELASA. AVS was supported by Fundação para a Ciência e a Tecnologia through Programa Investigador FCT 2014 (IF/00576/2014), and Plurennial funding to CCMAR (UID/Multi/04326/20132016). EMP was supported by Association for Cephalopod Research ‘CephRes’ and the HSA-Ceph 1/2019 grant to CephRes. GF is supported by intra-mural funds of the Stazione Zoologica Anton Dohrn.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
