Abstract
Here we list species-specific recommendations for housing, care and management of cephalopod molluscs employed for research purposes with the aim of contributing to the standardization of minimum requirements for establishments, care and accommodation of these animals in compliance with the principles stated in Directive 2010/63/EU. Maximizing their psychophysical welfare was our priority. General recommendations on water surface area, water depth and tank shape here reported represent the outcome of the combined action of the analysis of the available literature and an expertise-based consensus reached – under the aegis of the COST Action FA1301 – among researchers working with the most commonly used cephalopod species in Europe. Information on water supply and quality, environmental conditions, stocking density, feeding and handling are also provided. Through this work we wish to set the stage for a more fertile ground of evidence-based approaches on cephalopod laboratory maintenance, thus facilitating standardization and replicability of research outcomes across laboratories, at the same time maximizing the welfare of these animals.
Introduction
Since 1 January 2013 the use of any live individual (from hatching) belonging to cephalopod molluscs is regulated for scientific purposes in Member States of the European Union by the Directive 2010/63/EU 1 and the national transposed legislations (see Article 1 3(b) 1 ). Cephalopods are the sole invertebrates to have reached the same legal protection analogous to any other vertebrate species, as laboratory animals.2–4 Their inclusion represents a remarkable turning point of the current policies, while offering a unique occasion for giving greater attention to standardization of procedures for the daily care and management of animal welfare as applied to these organisms.4,5
In the current version of the Directive 2010/63/EU, and more precisely in part B of the Annex III (Requirements for establishments and for the care and accommodation of animals), 1 a list of mandatory minima for cage sizes, environmental enrichment and group housing – to mention just a few – is available for a number of commonly utilized laboratory species, namely for mice, rats, gerbils, hamsters, guinea pigs, rabbits, cats, dogs, ferrets, non-human primates, farm animals, birds, amphibians, reptiles and – to a lesser extent – for fishes. 1 Such standards, based on scientific evidence, reflect current practice in Member States of the EU – as a result of the transposed European legislation. It is interesting to note that these are also widely utilized abroad.
When considering fishes, Annex III provides only general information for animals belonging to this taxon (e.g. water supply and quality, lighting, feeding and handling). This contrasts with the previously listed organisms, and the generality of recommendations considering that more than 34,000 different fish species are currently known, and that different species have different physiological and behavioural ‘characteristics’ (see, for example, FISHBASE: www.fishbase.us/home.htm). 6
In the case of cephalopods, requirements for the care and accommodation of animals are not available yet.
Cephalopods are characterized by some remarkable features of their Bauplan, physiology and biology.7–11 They inhabit different geographical areas and diverse environments around the seas of the world. These molluscs account for a wide and diverse variety of taxa also reflecting their adaptions to different habitats in the oceans (e.g. benthic and pelagic, intertidal areas and deep sea, polar regions and the tropics) and at different depth strata.11–16 In addition to the high degree of interspecific variation, many species undergo large physical, physiological and behavioural changes as a natural part of their life cycle.17–21
Their active predatory behaviour includes different feeding strategies; 15 these are subjected to repeated changes during behavioural development after hatching for some species, and – relevant to this work – following adaptation to a captive environment. A few cases of scavenging and filter-feeding habits are also reported.15,22,23 Mating and reproductive strategies cover all possibilities, 24 spanning from the high competition for fertilizing females to the random encounter between solitary individuals in the dark deep ocean. Different spawning cycles and strategies have been observed, 24 and for some species maternal care has been reported.25,26
To partially support a description of the biological and ecological variety of forms cephalopods represent, Table 1 includes a summary of the different adaptations characterizing the species considered in this work.27–61
The species considered in this work and their main biological features. The information provided herein serves to depict biological diversity, main physiological and ecological adaptations and some behavioural requirements.
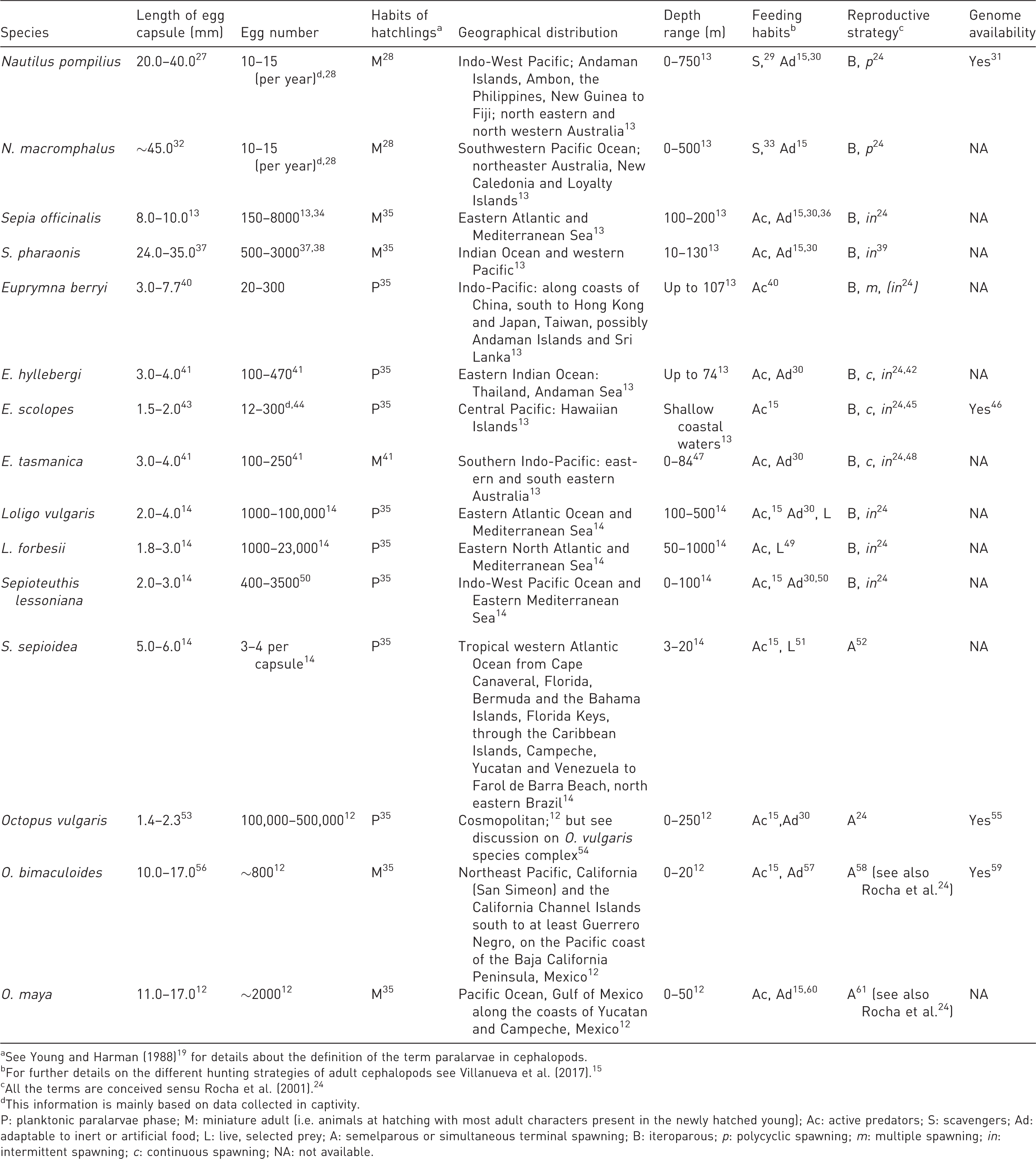
aSee Young and Harman (1988)19 for details about the definition of the term paralarvae in cephalopods.
bFor further details on the different hunting strategies of adult cephalopods see Villanueva et al. (2017). 15
cAll the terms are conceived sensu Rocha et al. (2001). 24
dThis information is mainly based on data collected in captivity.
P: planktonic paralarvae phase; M: miniature adult (i.e. animals at hatching with most adult characters present in the newly hatched young); Ac: active predators; S: scavengers; Ad: adaptable to inert or artificial food; L: live, selected prey; A: semelparous or simultaneous terminal spawning; B: iteroparous; p: polycyclic spawning; m: multiple spawning; in: intermittent spawning; c: continuous spawning; NA: not available.
Here we provide a set of minimal requirements and recommendations for the care and welfare of cephalopods utilized in scientific research, in compliance with the principles stated in the Directive 2010/63/EU. Information given herein is considered essential for housing and care of the most commonly utilized cephalopod species in EU countries and serves to promote standardization and guidance. This work refers to the Guidelines for the treatment of animals in behavioural research and teaching and should be considered complementary to the ‘Guidelines for the care and welfare of cephalopods in research’, hereafter referred to as ‘the Guidelines'. 63
Our goal is to achieve standardisation of minimum requirements for housing, care and accommodation of each cephalopod species of interest. However, this is challenged by some degree of intraspecific variation and large physiological changes occurring as part of their life cycle (more marked in some species than in others), so that recommendations of housing, care and even of applicability of some experimental procedures should consider eventually how to cope to some extent with this variability.
Towards species-specific recommendations for care of cephalopods in research: consensus-based approach and modus operandi
Despite the long standing tradition of research on cephalopods, and availability of published accounts about care in controlled conditions, accumulating from at least the early 20th century, 5 the absence of species-specific welfare-centred guidelines for the care of these animals led to the adoption of various ‘approaches’ for their housing, maintenance and care, thus making the level of standardization available for vertebrates 64 still far from being achieved for studies involving live cephalopods (for review see De Sio et al. 5 ).
Despite the level of accuracy reached in the Guidelines, species-specific information for care and management are not included in Appendix 2 (Housing, environmental parameters, transport and feeding) of Fiorito et al. 63
To fill such a gap the COST Action FA1301 and CephRes promoted and facilitated a consensus-based initiative with the aim of producing a tabularized recommendation for cephalopods’ housing, in line with the required content of the Annex III of the Directive 2010/63/EU. This activity has been spanned in a series of meetings and working groups and was initiated in a coordinated way during the Cephs In Action meeting in Berlin (Germany, 2016). There, experts convened to: i. discuss about limitations of the current approaches available in different laboratories for housing and care of cephalopods for scientific research, ii. recognize the information included in the Guidelines, iii. discuss about the requirements and specific information included in Annex III of the Directive for other species, thus to explore a possible template as working method. During the meeting experts began to review published data and white papers in order to find relevant information with the aim to compile a set of cephalopod-care oriented data based on scientific evidence. Information was considered in the sake of the evaluation of the environmental, biological and behavioural needs of different cephalopod species thus to increase success of caring, maintenance, growth and rearing aimed to reduce stress and increase welfare in research establishments. This approach was followed by several remote meetings and exchanges between a selected number of people representing the whole group that, after more than a year, were able to prepare a collation of available information (see below) as a ground for further discussion and refinement; the basis of this compilation is included in the Supplementary material Appendices A (Environment), B (Accommodation and Care; Enrichment) and C (Thermal, oxygen and salinity tolerance ranges) online.
Coordinated by the COST Action FA1301, the experts met again to achieve consensus and finalize details of the planned work. The occasion was given by one of the last meetings of the COST Action FA1301 ( Galway, Ireland, 2017 ) also with the aim of monitoring the impact of the transposition of Directive 2010/63/EU and MFSD , in EU Member States, COST countries and abroad, facilitate and increase the improvement of the available knowledge on cephalopod biology, physiology and behavioural plasticity that may affect animals’ welfare, and contribute to the development of knowledge on care, rearing, environmental and requirements of different cephalopod species to facilitate the standardization in animals’ care and increase their welfare. The final step was to refine and discuss some relevant aspects with the Commission during a Cephs In Action Technical Workshop , held in Brussels (Belgium, 2017).
The overarching aim of this long work was thus to provide an updated supplementum to the Guidelines, 63 providing a more detailed species-specific source of information related to cephalopod care; this resulted to be the basis for the identification of mandatory minima for the housing and care of cephalopods for scientific purposes.
Target species
The biggest challenge when dealing with cephalopods is the lack of captive breeders cultured for this purpose due to several bottlenecks 65 and the consequent need for procuring the specimens from the wild following sufficient justification, as regulated by the Competent Authority (see Article 9.2 1 ). Capture and transport of live cephalopods pose many concerns for the welfare of these animals and thus impose undisputable restrictions on the number of species that can be employed in research, at least until standardized and solid protocol is implemented. The urge for defining the most suitable requirements for cephalopods derives from their particular vulnerability when kept in controlled conditions and the special ecologically demanding attention needed for their welfare when housed in a limited space with artificial conditions. As mentioned above, cephalopods are stenohaline and stenotherm, thus the range of water quality parameters and environmental settings to which they can be subjected is very narrow and could easily expose animals to distress and potential suffering if not properly monitored. For such reasons, and because animals are considered to have an intrinsic value, the EU set strict and clear rules for conducting experiments with laboratory animals, starting from the mandatory requirement of competence of the personnel carrying out procedures (Article 23 1 ).
Bearing all this in mind we focus here on the following list of species of interest (see also Tables 1 and 2) for research in EU countries: Nautilus pompilius, N. macromphalus, Sepia officinalis, S. pharaonis, Euprymna scolopes, E. berryi, E. hyllebergi, E. tasmanica, Loligo vulgaris, L. forbesii, Sepioteuthis sepioidea, S. lessoniana, Octopus vulgaris, O. bimaculoides, O. maya. The species considered in this list do not exclude other representatives, congeneric and/or belonging to the same taxa, characterized by similar lifestyles and adaptations. We are committed to expand lists and biological information included in the tabularized overviews (e.g. Table 1 and Supplementary Appendix C) based on scientific evidence.
Species-specific recommendations for the accommodation and care of cephalopods species mostly utilized, or potentially utilized, in Europe. For each species and a given body size we provide minimum values for water surface area and water depth, and required additional space in the case of group-holding; general indication on tank shape is also provided.
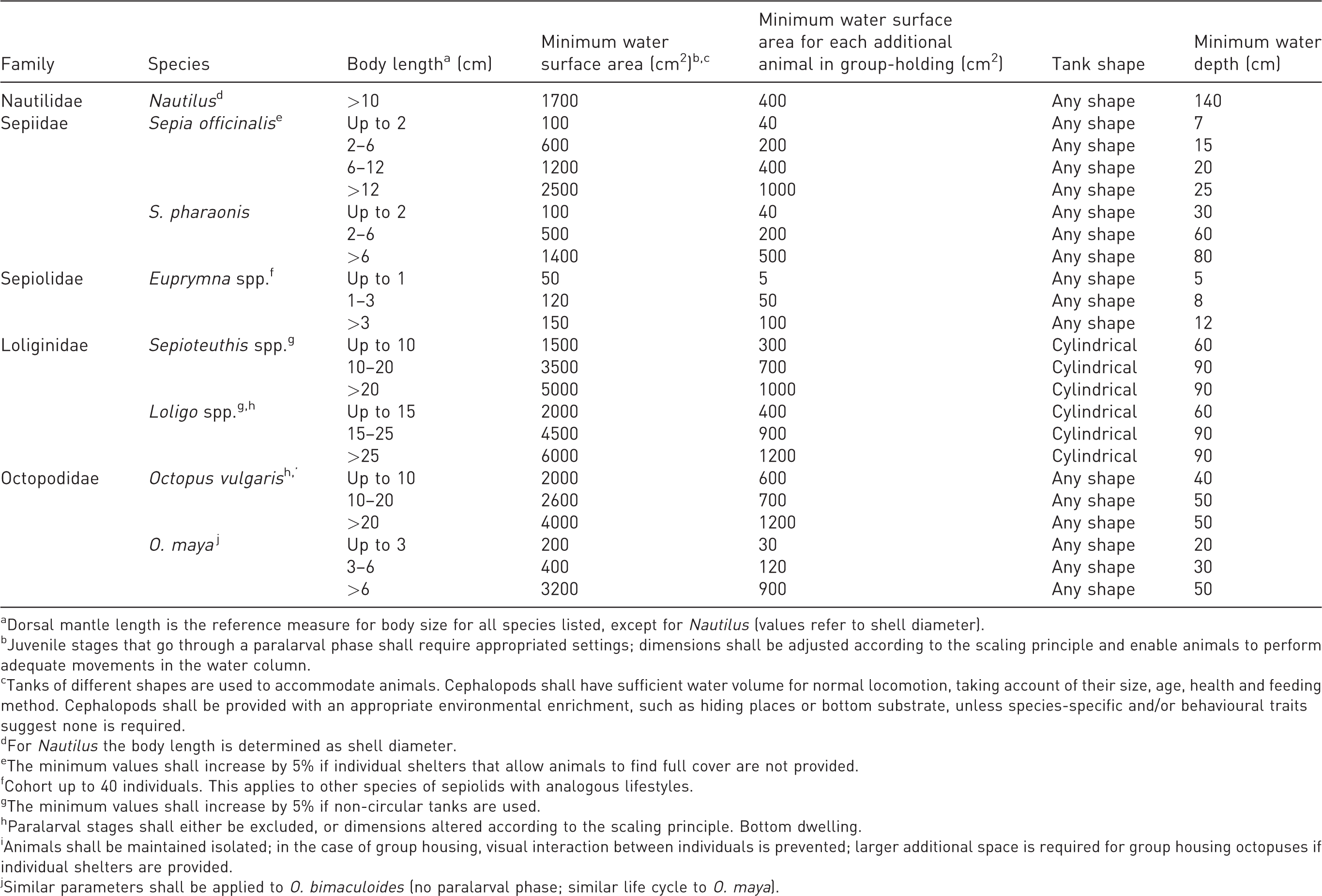
aDorsal mantle length is the reference measure for body size for all species listed, except for Nautilus (values refer to shell diameter).
bJuvenile stages that go through a paralarval phase shall require appropriated settings; dimensions shall be adjusted according to the scaling principle and enable animals to perform adequate movements in the water column.
cTanks of different shapes are used to accommodate animals. Cephalopods shall have sufficient water volume for normal locomotion, taking account of their size, age, health and feeding method. Cephalopods shall be provided with an appropriate environmental enrichment, such as hiding places or bottom substrate, unless species-specific and/or behavioural traits suggest none is required.
dFor Nautilus the body length is determined as shell diameter.
eThe minimum values shall increase by 5% if individual shelters that allow animals to find full cover are not provided.
fCohort up to 40 individuals. This applies to other species of sepiolids with analogous lifestyles.
gThe minimum values shall increase by 5% if non-circular tanks are used.
hParalarval stages shall either be excluded, or dimensions altered according to the scaling principle. Bottom dwelling.
iAnimals shall be maintained isolated; in the case of group housing, visual interaction between individuals is prevented; larger additional space is required for group housing octopuses if individual shelters are provided.
jSimilar parameters shall be applied to O. bimaculoides (no paralarval phase; similar life cycle to O. maya).
Source of data and extraction of information
Information about care and housing parameters adopted for cephalopods that we consider herein derives from an expert analysis of various sources (a total of 97 different journal papers, reviews and book chapters) and a few ad hoc studies reporting details considered robust enough to justify their inclusion in our analysis. As mentioned in Supplementary material, we have also taken into account the classic contribution made by Pflege, 66 which represents the first available set of guidelines for laboratory rearing and maintenance of cephalopods intended to be studied in zoology and physiology, 5 together with other reviews on care of cephalopods.30,63,67–73
Information and other data collected were collated in a tabularized overview of the ‘care and accommodation of animals’, providing important details regarding environment (water supply and quality, lighting, noise and vibration), accommodation, care (housing, stocking density and sex ratio, diet), enrichment, and temperature, oxygen and salinity tolerance ranges (see Appendices A–C, respectively, in the Supplementary material). All the information collected was accurately curated in order to be considered of relevance for the aims of this work and for the ultimate goal of producing reference to recommendations for the care and welfare of the target species.
Based on the above collection and through the joint effort of researchers providing various expertise, we produced a consensus-based set of standardized values and parameters required to accommodate the selected cephalopod species here considered. Data were revised by experts working with each selected species and then agreed upon during scheduled designated meetings. Whenever species-specific information was missing, we completed it with proper indications based on the working experience of the experts and the current good practices for each specific cephalopod taxon employed in research. The recommended notes here included are based on accurate data mining from our source of information (see Appendices A–C in the Supplementary material) and discussion between experts at dedicated meetings. The above-mentioned appendices represent background information on the species-specific proposal of recommendations for care of cephalopods for scientific purposes.
The following notes are inspired by the criteria adopted for Urodeles and Anurans amphibians as included in Annex III of the Directive. The approach we followed is similar to that included in the Background information on the species-specific proposals for reptiles presented by the Expert Group on Amphibians and Reptiles 74 provided to the Council of Europe in order to implement the species-specific section about amphibians and reptiles in the Annex III of the Directive 2010/63/EU. 1
Requirements for housing and care of live cephalopods
Table 2 lists the recommended minimum requirements for housing live cephalopods under research settings. Values included are based on our data source (see Appendices A–C in the Supplementary material) and other relevant publications, including considerations about health monitoring and post-mortem evaluation of cephalopods. 73 It is interesting to mention here that hatchlings’ viability, appropriate water quality and food items provided to cephalopods and suitable living space were considered essential requirements by Pflege 66 and years afterwards by von Boletzky 72 for the aim of achieving the best possible conditions for the care and management of live cephalopods for scientific research purposes.
General recommendations
Part A – General section – of Annex III of the Directive 2010/63/EU includes general recommendations about the overall care and accommodation for laboratory animals. Such guidance highlights that the welfare of the reared species depends first of all on facility and holding rooms status together with the environment and general care in which all subjects are housed, and finally on the individual husbandry conditions (Part A, Annex III 1 ).
When referring to cephalopod molluscs, it is easy to understand that a crucial element to consider is the system that supplies seawater of adequate quality. In general, either open or closed systems are adopted. Each one has its pros and cons and the choice of one over the other should be properly and attentively made (see Supplementary material). Water temperature should be set and regulated according to the natural range for the species and the geographical area in which it was captured (if from the wild); the life-stage should also be considered.
Regardless of the cephalopod species, a daily monitoring of the welfare state should be carried out by competent personnel in appropriate rooms where ‘simple diagnostic tests, post-mortem examinations, and/or the collection of samples can be performed’ (see 1.3(a) of Part A, Annex III 1 ); ‘these checks shall ensure that all sick or injured animals are identified and appropriate action is taken’ (see 3.1(b) of Part A, Annex III 1 ).
Noise and other interference should always be kept to a minimum to avoid or reduce any cause of potential distress in animals. 63 The lighting conditions should be regulated as well, to respect and emulate the biological and physiological needs of cephalopods.
As for animals housing, concerns should be directed towards the species-specific ‘social’ needs (i.e. group- or solitary-living habits of the species) and also their behavioural requirements, providing access to adequate environmental enrichment that fosters visual, tactile and cognitive stimulation and at the same time attempts to reproduce the motivation standards to which the animal is exposed to in nature. Resting and ‘sleeping’ areas should be a minimum requirement to help specimens feel protected while inactive, together with presenting dens in which to hide, or sandy areas to dig themselves into whenever they feel threatened by potential external menaces.
Concerning feeding it is required that animals should have access to a diet that meets their nutritional and behavioural needs in form, content and presentation. The food shall be palatable and non-contaminated with chemical, physical and microbiological traces (see 3.4(a, b) 1 ). General principles about the type of food items to provide to animals is included in Fiorito and coworkers. 63
Each animal shall be able to access food, allowing to exhibit the species-specific predatory behaviour and sufficient feeding space to limit competition and ‘cannibalism’. We recommend that ad libitum feeding should be avoided to prevent motivational decline and reduce risks of counter effects in the efficiency of the systems assuring adequate quality of seawater.
Species-specific requirements
As mentioned above, there are no cephalopod species-specific requirements included in part B of Annex III of the Directive 2010/63/EU. Also, information provided for fish is very general and not adequate to translate to cephalopods.
Here we include (Table 2) a ‘minimum water surface area’ per animal for each of the species considered. This has been defined as ‘a constant function of the “footprint” area of the animal, at least within a given species’. The animal footprint area refers to the square of any linear dimension, creating a plot of the amount of space allocated to animals, able, on a proportional basis, to take into account both large and small subjects. 74 In addition, this minimum space requirement allows for the introduction of environmental enrichment for a given specimen. When the species in question can be group-held, a value for the minimum water surface area for each additional animal has to be defined, taking into account the biological tolerance in terms of inter-individual space requirements of the target species.
In identifying tank shape and the minimum water depth we also considered the species’ physiological and behavioural needs such as their benthic or pelagic biology or the daily vertical migration (with the unavoidable constraint of captivity).
In more detail, parameters included in Table 2 referring to a specific taxon are proposed taking into account different body size (such as dorsal mantle length for a coleoid, and shell diameter for Nautilidae). Furthermore, the minimum water surface area refers generally to life stages that do not include the transition from the paralarval form as that may require additional considerations 75 and therefore any value should be readapted according to a scaling principle.
As for the tank, any shape can be chosen with few exceptions that may concern animals’ lifestyle. As can be seen from the ‘minimum water depth’ reported in Table 2, Nautilidae need a tank that is consistently much higher than wide, compared with coleoids, as they usually perform daily vertical migrations. 76 Benthic species usually require large ground areas, low water column and rounded edges, while pelagic species need circular (or oval, as for squid) tanks, smaller areas, with sufficient space to swim or high-water column, thus allowing the animal to display its natural behaviour. In this regard, families such as Octopodidae need dens and refuges as a minimum requirement, in order to allow the animals freedom to express their natural behaviour, including hiding in a den. Other cephalopods, and in particular female specimens, require bottom substrate as a spawning ground or surfaces suitable for attaching egg masses. In addition, environmental enrichment and feeding protocols best fitting the behaviour of these animals should be provided in order to reproduce as much as possible their natural environments while keeping their motivation high (see General recommendations above).
Little information is currently available on the minimum water surface area for single and multiple specimens and the proposed values derive from the critical analysis of data available in the scientific literature. In some instances, information derived from studies carried out in other contexts (i.e. aquaculture or public display) where the maximising of the growth rate, and the stocking density or longevity are the main focus. As a consequence, we discussed about the available information and attempted to best fit cephalopods’ biology requirements and maximize the achievement of the Five Freedoms.77,78
The number of specimens that can be held in the same tank depends on species-specific social behaviour, age and size of the animals and individual space requirements in terms of water surface area. For instance, O. vulgaris is generally a solitary-living species 79 and therefore should be kept isolated; if group housing is inevitable, visual interaction must be avoided or maximally reduced with at least a shelter per individual together with larger space as indicated by the parameters under the entry ‘minimum water surface area for each additional animal in group-holding’. Nevertheless, the lack of scientific data makes it difficult to define categoric recommendations, which are therefore based on prolonged practical experience in successful rearing of cephalopods.
Closing remarks
Cephalopods, though accounting for less than 2% of the phylum of Mollusca, are considered as the class of invertebrates endowed with the largest nervous system underlying levels of plasticity comparable to those of some vertebrate. Their sentience (sensu Broom 80 ) is at the basis of the decision of including them as the sole invertebrate taxon listed in the Directive 2010/63/EU. A recent framework for evaluating scientific evidence of sentience based on eight criteria provided further support. 81
Very little is known about inter-specific compatibility, which appears to be a possible scenario in public display, when multiple species are housed in the same enclosures. Studies are required with focus on animal welfare, and these can be facilitated by works carried out in the wild, but also when accurate welfare monitoring is applied to public aquaria.
For the first time, we attempted to provide species-specific minimum requirements for housing, accommodation and care of live cephalopods with the aim to support inclusion of required information in Annex III of the Directive. This work represents a step forward towards the improvement of cephalopods’ welfare. Recommendations proposed are intended to enhance the current available knowledge on this taxon’s biology, husbandry and care to guide both experienced and early career scientists, students, caregivers, technicians and veterinarians towards the achievement of best practices for increasing the success in cephalopods’ maintenance, growth and rearing for scientific purpose in research establishments.
Requirements listed in Table 2 are derived by a joint effort of a group of experts – based on the supporting information (see Appendices A–C in the Supplementary material) – coordinated for matching and adapting the available data on space requirements with the experience and knowledge of the behavioural and physiological needs of cephalopods, keeping animal welfare as our priority. The consensus-based approach here adopted represents one of the main outcomes of the COST Action FA1301 and originates from the whole series of meetings starting in late 2014.
Here we reviewed all the available literature and collected relevant data about the use of cephalopods as experimental animals. We also provided supplementary information regarding water quality requirements, feeding and environmental needs of the selected species in order to guide researchers and caregivers in fully achieving animals’ welfare while attempting to implement the Five Freedoms principles in cephalopods.
The guidance list of minimal requirements for housing and care of selected cephalopod species included in this work has to be considered as complementary to the general Guidelines. 63 It represents a further step towards a more intense series of studies providing adequate, experimentally based data helping to refine the requirements here included.
Although they might be demanding for researchers and establishments, the minimum accommodation needs represent the basis to ensure animal welfare and consequently improve the quality of research data by making it more transparent, replicable and thus reliable.
Supplemental Material
sj-pdf-1-lan-10.1177_00236772221111261 - Supplemental material for General and species-specific recommendations for minimal requirements for the use of cephalopods in scientific research
Supplemental material, sj-pdf-1-lan-10.1177_00236772221111261 for General and species-specific recommendations for minimal requirements for the use of cephalopods in scientific research by Giovanna Ponte, Katina Roumbedakis, COST Action FA1301, Viola Galligioni, Ludovic Dickel, Cécile Bellanger, Joao Pereira, Erica AG Vidal, Panos Grigoriou, Enrico Alleva, Daniela Santucci, Claudia Gili, Giovanni Botta, Pamela Imperadore, Andrea Tarallo, Lars Juergens, Emily Northrup, David Anderson, Arianna Aricò, Marianna De Luca, Eleonora Maria Pieroni, Graziano Fiorito in Laboratory Animals
Footnotes
Acknowledgements
This work is the result of a joined effort of the community of researchers and experts including participants to the COST Action FA1301 (CephsInAction; https://www.cost.eu/actions/FA1301/; ![]() ). The work has been coordinated also by the non-profit organization Association for Cephalopod Research ‘CephRes’. We are grateful to Dr David Smith for his continuous support and advice. The Federation of European Laboratory Animal Science Associations (FELASA) also provided valuable advice to these activities. We would like to express our gratitude to Sigurd von Boletzky, who inspired our action. We are grateful to Professor Torsten Wiesel – who continually inspired the action of some of us – for his enthusiasm for and genuine interest in cephalopod biology research.
). The work has been coordinated also by the non-profit organization Association for Cephalopod Research ‘CephRes’. We are grateful to Dr David Smith for his continuous support and advice. The Federation of European Laboratory Animal Science Associations (FELASA) also provided valuable advice to these activities. We would like to express our gratitude to Sigurd von Boletzky, who inspired our action. We are grateful to Professor Torsten Wiesel – who continually inspired the action of some of us – for his enthusiasm for and genuine interest in cephalopod biology research.
Data availability
This work is based on previously published works and extensively refers to those. No primary data have been utilized in this review. Supplementary material online includes detailed information and references for original data.
Declaration of conflicting interests
The author(s) have no conflicts of interest to declare.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
