Abstract
This paper is the result of an international initiative and is a first attempt to develop guidelines for the care and welfare of cephalopods (i.e. nautilus, cuttlefish, squid and octopus) following the inclusion of this Class of ∼700 known living invertebrate species in Directive 2010/63/EU. It aims to provide information for investigators, animal care committees, facility managers and animal care staff which will assist in improving both the care given to cephalopods, and the manner in which experimental procedures are carried out. Topics covered include: implications of the Directive for cephalopod research; project application requirements and the authorisation process; the application of the 3Rs principles; the need for harm-benefit assessment and severity classification. Guidelines and species-specific requirements are provided on: i. supply, capture and transport; ii. environmental characteristics and design of facilities (e.g. water quality control, lighting requirements, vibration/noise sensitivity); iii. accommodation and care (including tank design), animal handling, feeding and environmental enrichment; iv. assessment of health and welfare (e.g. monitoring biomarkers, physical and behavioural signs); v. approaches to severity assessment; vi. disease (causes, prevention and treatment); vii. scientific procedures, general anaesthesia and analgesia, methods of humane killing and confirmation of death. Sections covering risk assessment for operators and education and training requirements for carers, researchers and veterinarians are also included. Detailed aspects of care and welfare requirements for the main laboratory species currently used are summarised in Appendices. Knowledge gaps are highlighted to prompt research to enhance the evidence base for future revision of these guidelines.
Contributors
People listed here provided data, information and comments, and contributed to different extents during the preparation of this work.
The following list is arranged by country in alphabetical order; different contributors are merged by Institution.
France
Christelle Alves, Cecile Bellanger, Anne-Sophie Darmaillacq, Céline Gaudin
Groupe mémoire et Plasticité comportementale, EA4259, University of Caen Basse-Normandy, Caen, France
Joël Henry
Physiologie de la reproduction des Mollusques, University of Caen Basse-Normandy, Caen, France
Germany
Tamar Gutnick
Max Planck Institute for Brain Research, Frankfurt, Germany
Italy
Anna Di Cosmo
Department of Biology - University of Naples Federico II, Napoli, Italy
Carlo Di Cristo
Department of Biological and Environmental Sciences - University of Sannio, Benevento, Italy
Viola Galligioni
CIBio - Centre for Integrative Biology, Trento, Italy & Association for Cephalopod Research ‘CephRes’, Italy
Anna Palumbo
Stazione Zoologica Anton Dohrn, Napoli, Italy
Perla Tedesco
Department of Biological and Environmental Science and Technologies - University of Salento, Lecce, Italy
Letizia Zullo
Istituto Italiano di Tecnologia, Department of Neuroscience and Brain Technologies, Genoa, Italy
Portugal
António Sykes
C. Mar – Centre of Marine Sciences, Universidade do Algarve, Faro, Portugal
Spain
Roger Villanueva López
Renewable Marine Resources Department - Institut de Ciències del Mar, Barcelona, Spain
United Kingdom
Ngaire Dennison
Home Office, Animals in Science Regulation Unit, Dundee, Scotland, UK
Penny Hawkins
RSPCA Research Animals Department, Southwater, West Sussex, UK
United States of America
Gregory J. Barord, Heike Neumeister, Janice Simmons, Roxanna Smallowitz
Biology Department, Brooklyn College - CUNY Graduate Center, Brooklyn, NY, USA
Jean Geary Boal
Biology Department, Millersville University, Millersville, PA, USA
Roger Hanlon, William Mebane
Marine Resources Center, Marine Biological Laboratory, Woods Hole, MA, USA
Judit R Pungor
Hopkins Marine Station of Stanford University, Pacific Grove, CA, USA
James B. Wood
Waikiki Aquarium, University of Hawaii-Manoa, Honolulu, HI, USA
1. Introduction
Cephalopods (i.e. nautilus, cuttlefish, squid and octopus) have been used for diverse scientific purposes across Europe for over 100 years.1,2 However, until recently, scientific procedures involving cephalopods have not been covered by EU regulations, with the exception of procedures using Octopus vulgaris in the United Kingdom (see discussion in Smith et al. 3 ).
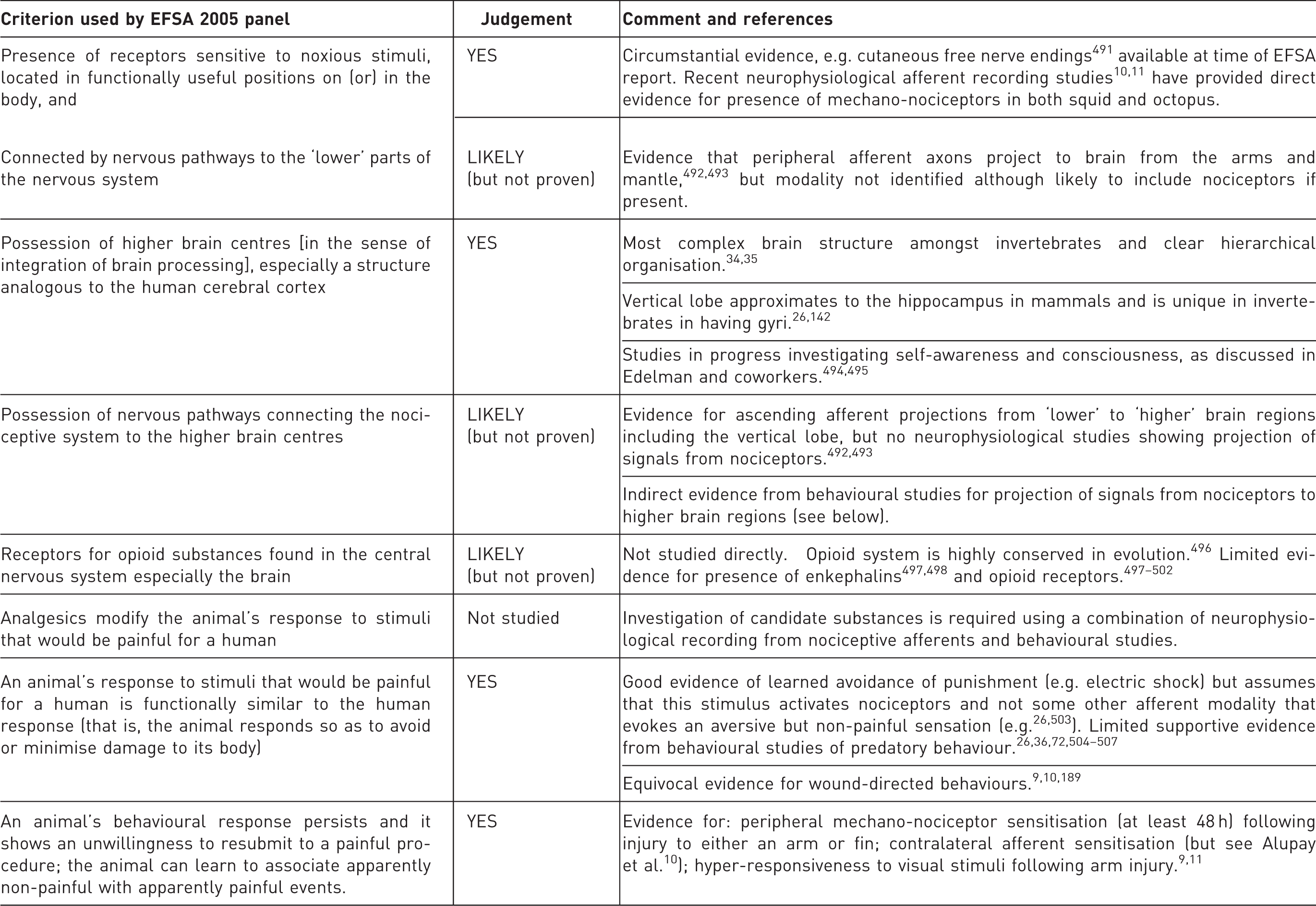
Summary of evidence for the capacity of cephalopods to experience pain based upon the criteria used by the EFSA 2005 panel (as the basis for recommending the inclusion of cephalopods in revision of Directive 86/609/EEC), and here updated with more recent studies. See also Andrews et al. for review 8 and additional references.
Prompted by the need for guidelines on these and other matters covered by the Directive, members of the international cephalopod research community have met on several occasions over the past 3 years and have produced publications aimed at cephalopod researchers, on: i. requirements of the EU Directive, implementation, ethics and project review; 3 ii. PSDLH, anaesthesia and humane killing; 8 and iii. implications for neuroscience research and the Three Rs, i.e. Replacement, Reduction, Refinement. 2
This work has led to the development of a set of consensus Guidelines for the Care and Welfare of Cephalopods in Research which aim to assist researchers in complying with the Directive, and are the subject of this paper. These guidelines have been developed as a joint initiative between CephRes (www.cephalopodresearch.org), FELASA (www.felasa.eu) and the Boyd Group UK (http://www.boyd-group.demon.co.uk/).
The Guidelines for the Care and Welfare of Cephalopods in Research, which should be regarded as a starting point for future developments, begin with a set of general principles of good practice, representing the present state of knowledge that may reasonably be applied to all cephalopods. These are followed by a tabulated set of specific guidelines (see Appendices) for typical cephalopod species, currently used in EU laboratories, which also reflect well-established principles.
1.1 What is a cephalopod?
For the purpose of these guidelines, cephalopods are defined as all living species that are members of the molluscan class Cephalopoda.4,5,12 The term ‘live cephalopod’ is not defined in the Directive, but guidance indicates that these animals are covered by the Directive from ‘when they hatch’.13,14
Cephalopods are characterised by bilateral body symmetry, a prominent head and a set of arms, including tentacles in Decapods, which are considered as muscular hydrostats and derived from the primitive molluscan foot.15–21 The class contains two, only distantly related, living subclasses: Nautiloidea (represented by Nautilus and Allonautilus) and Coleoidea, which includes cuttlefish, squid and octopuses.20,22 In the Nautiloidea, the external shell, common to the molluscan Bauplan, still exists, whereas in the Coleoidea it has been internalised or is absent. The variety of species that compose the taxon is reflected in the diversified habitats they have adapted to: oceans, benthic and pelagic zones, intertidal areas and deep sea, polar regions and the tropics.23–26
Understanding the requirements of a particular species in relation to its natural habitat is fundamental in maintaining healthy laboratory populations of cephalopods. Assumptions for housing, care and use of these animals based on fish, whilst appropriate in some circumstances, should be made with great caution as the evolutionary convergence between fish and cephalopods24,27 does not reflect the actual requirements of different species.
Generally, cephalopods have a high metabolic rate, grow rapidly and are short-lived.28,29 These animals are exothermic, highly adapted to the marine aquatic environment and are therefore unlikely to tolerate rapid or significant changes in the quality or temperature of the water they are housed in. They react rapidly to environmental changes/external stimuli with immediate physiological consequences that can be relatively long lasting. Such changes, as well as having potential welfare implications, will also impact upon experimental results.
Cephalopods are considered among the most ‘advanced’ invertebrates, having evolved many characteristic features such as relatively large, highly differentiated multi-lobular brains, a sophisticated set of sensory organs, fast jet-propelled locomotion, and complex and rich behavioural repertoires.25,30–37
1.2 What the Directive 2010/63/EU means for cephalopod research
The entry into force of the Directive 2010/63/EU (hereafter referred to as ‘the Directive')38,39 means that, from 1st January 2013, scientific research and testing involving ‘live cephalopods’ is regulated by a legal framework at both EU and Member State levels, and as a consequence all scientific projects that cross the threshold set for regulation (i.e. involve procedures that may cause PSDLH equivalent to, or higher than that caused by the insertion of a hypodermic needle in line with good veterinary practice) will require authorisation by the National Competent Authority (see the list available at: http://ec.europa.eu/environment/chemicals/lab_animals/ms_en.htm). 3
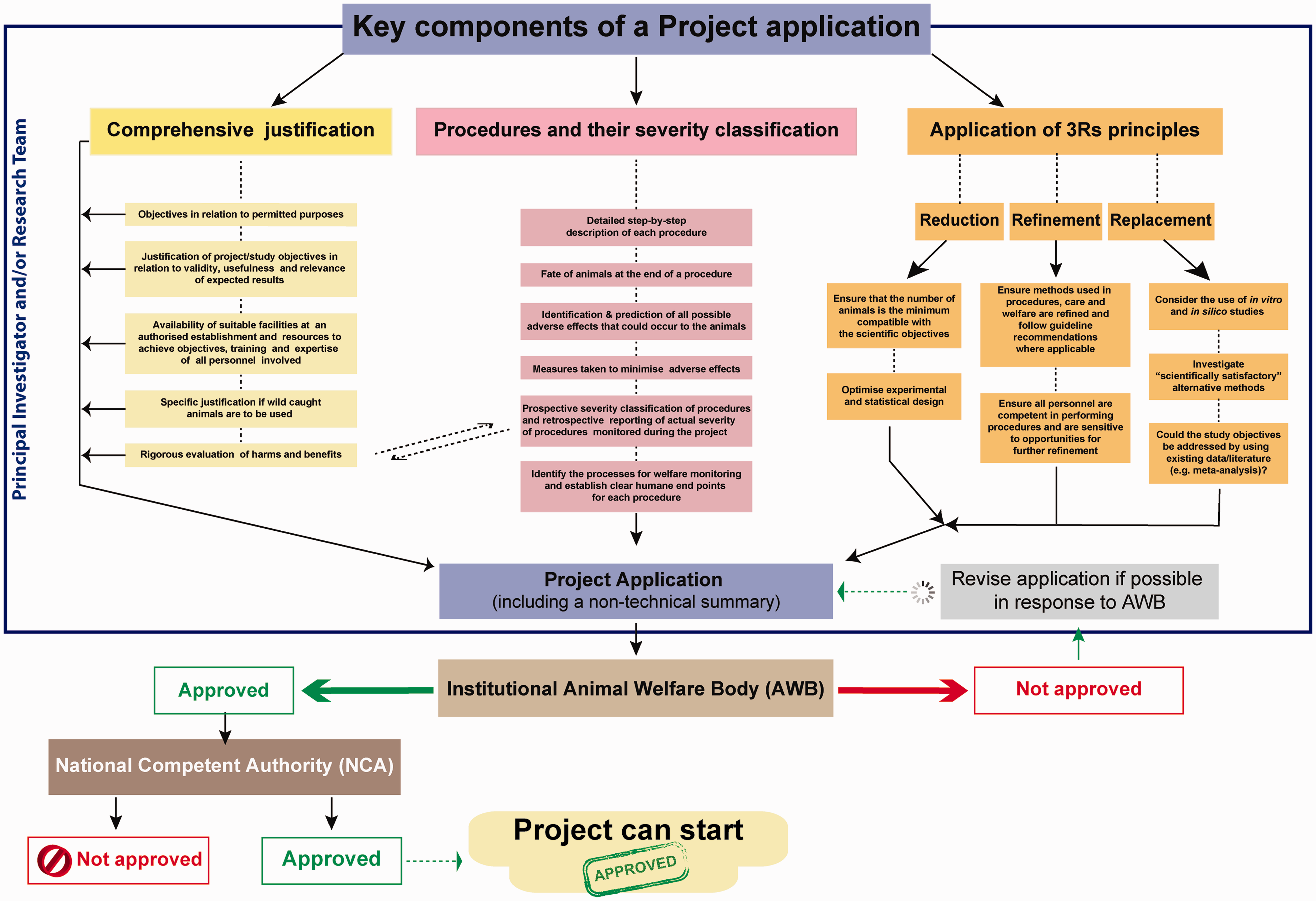
Three key aspects of the project that will need to be considered by researchers and those responsible for animal care and welfare are outlined below (see also Figure 1). Specific topics for inclusion and consideration are listed in Appendix 1, and a more detailed overview of issues relating to implementation of the Directive for cephalopods can be found in Smith et al.,
3
which also includes some hypothetical worked examples of project review, particularly in relation to opportunities for implementing the 3Rs (see 2.2.1 below).
Schematic overview summarising the major components of a project application and stages of project approval under Directive 2010/63/EU. Note that the details of the project approval process may differ across member states. For details see text and review in Smith et al
3
.
2. Project application requirements and authorisation process
2.1 Application requirements
The key requirements of project authorisation are outlined here to provide a background to the technical sections, which aim to show how these requirements can be fulfilled specifically for cephalopods (Figure 1).
Definition of key terms utilised in Directive 2010/63/EU.

Definitions of the levels of severity of procedures according to Directive 2010/63/EU, Annex VIII; see also Lindl et al. 490

An application for project authorisation must include, as a minimum, the project proposal and the items listed in Appendix 1. A non-technical project summary will be required unless waived by the National Competent Authority.
Applications must also include specific scientific justification for any requests for exemptions from certain requirements of the Directive (where permitted – see also Appendix 1). This will include requests for permission to: i. use an endangered cephalopod species where it falls within the criteria laid out in Article 7.1 of the Directive; ii. use cephalopods taken from the wild (Article 9, see also section 3.3 below); iii. carry out procedures in a place that is not an authorised users' establishment (Article 12); iv. re-use (in a different procedure) animals that have already undergone a procedure (Article 16, see also section 8.10 below); v. use drugs, such as neuromuscular blocking agents, that could stop or restrict an animal's ability to show pain, without an adequate level of anaesthesia or analgesia (Article 14§3); vi. depart from any of the general standards of animal care and accommodation outlined in Section A of Annex III of the Directive (Article 33).
For species other than cephalopods, specific justification is also required for killing animals by a method not listed in Annex IV of the Directive, or for departing from species-specific standards of animal care and accommodation outlined in Section B of Directive Annex III. However, at present, cephalopods are not included in either of these Annexes.
Once a project is authorised and underway, it should continue to be critically evaluated by the Principal Investigator and all members of the project team, using the factors listed in Appendix 1, so as to ensure that ethical considerations and opportunities for implementing the 3Rs are identified and addressed in an ongoing process for the entire duration of the project, not only at the start.
2.2 Factors to be evaluated in project authorisation and project operation
The Directive sets out the factors that must be evaluated during project authorisation and throughout the lifetime of authorised projects. These factors are listed below as a series of action points and associated questions for consideration.
2.2.1 Implement the 3Rs to minimise the harms caused to the animals
The Three Rs (3Rs) principles were first described by Russell and Burch 40 and are now internationally accepted as an essential requirement for the ethical and humane conduct of scientific studies involving animals (Recital 11; Articles 4 and 38 §2b). In addition, it is widely recognised that implementation of the principles can enhance scientific quality.41–44,45
The following are examples of questions that will need to be addressed:
Replacement
What on-going efforts will you make to identify ‘scientifically satisfactory’ alternative methods that could replace the use of some or all animals? (Article 4§1) Could you avoid the use of animals by asking different type of question, or making better use of existing data or literature to address the scientific objectives? Could in vitro studies or in silico-modelling be used to replace some or all of the animals?
Reduction
How will you ensure that the number of animals used in the project, and in individual studies within the project, is ‘reduced to a minimum without compromising the scientific objectives’? (Article 4§2) Could any further reductions be made, e.g. by taking expert statistical advice to help optimise experimental and statistical design?
Refinement
How have you refined the ‘breeding, accommodation and care of the animals’ and the ‘methods used in procedures’, so as to ‘reduce to the minimum any possible pain, suffering, distress or lasting harm to the animals’ throughout their lives? (Article 4§3) Have you considered and implemented all the possibilities for refinement described elsewhere in these guidelines? How will you ensure that all relevant personnel working on the project are adequately educated and trained, and are supervised until they have demonstrated their competence in the procedures?
2.2.2 Assess and assign the severity classification of the procedures used in the project
Each procedure outlined in a project application must be classified according to the severity of its adverse effects on the animals (Article 38§2c). This prospective severity classification sets an upper limit on the level of suffering that an individual animal undergoing the procedure is allowed to experience.46,47 The categories are: ‘non-recovery’ (for procedures carried out entirely under general anaesthesia from which the animal does not recover consciousness), ‘mild’, ‘moderate’ and ‘severe’ (Article 15§1 and Annex VIII; see also section 8 below, and Text Box 2).
The following points must be considered for all projects, in order to fully address the 3Rs and meet the requirements of the project evaluation process:
Have you tried to identify and predict all possible adverse effects that could be caused to the animals in the project? Include any pain, distress, lasting harm and other forms of suffering, such as hunger, anxiety, boredom and osmotic or thermal stress caused to the animals, which may occur at any time during the animals' lifetime; for example, a result of capture and transport to the laboratory, routine handling, housing and husbandry, or method of killing, as well as the effects of the procedures themselves. Have you taken steps to minimise each of these adverse effects as far as possible, by: i. applying the 3Rs in the design of procedures (see above), ii. using these guidelines, other relevant literature, advice from colleagues and from the institution's Animal Welfare Body to assist you? Based on the above, have you set prospective severity classifications (see section 8) and clear humane end-points for all procedures? How will you monitor the welfare of the animals used in the project, and when and how will you intervene to ensure that the animals do not suffer beyond the upper limit of severity needed to achieve the objectives of any particular procedure?
Member States must also collect retrospective information on the actual severity of procedures, after they have ended, and must make this publicly available on an annual basis (Article 54§2). The assessment of actual severity is based on day-to-day observations of the animals, and the ‘most severe’ severity experienced by the animal is reported to the National Competent Authority. For example, if records of observations indicate that suffering is moderate at the beginning of a procedure and then mild for the remainder, actual severity for reporting purposes is ‘moderate’.
Annex VIII of the Directive further explains the severity categories (see also section 8), and the European Commission has put together a series of examples* to illustrate the process of prospective severity classification, day-to-day observation and monitoring of animals and actual severity assessment.
2.2.3 Weigh the harms and benefits of the project and the individual studies within it
Taking into account all the points listed above:
What is the basis for your overall assessment that ‘the harm to the animals in terms of suffering, pain and distress is justified by the expected outcome, taking into account ethical considerations, and may ultimately benefit human beings, animals or the environment’? (Article 38§2d) Have you explained this evaluation in your project application and, where relevant, your non-technical project summary (Article 43) – the latter using language suitable for the general public? How will you ensure that the ‘weighing of harms and benefits’ is an on-going process throughout the project – i.e. part of the day-to-day practice of ‘ethical science’, and not just a one-off event at the time of authorisation?
A detailed examination of procedures for harm-benefit analysis in animal research and testing is available in a document from the Animal Procedures Committee in the UK. 48
3. Supply, capture and transport
3.1 Source of animals
Article 9§1 of the Directive requires that animals must not be taken from the wild for use in procedures, unless an exemption has been granted by the relevant National Competent Authority, based on ‘scientific justification to the effect that the purpose of the procedure cannot be achieved by the use of an animal that has been [purpose-] bred for use in procedures’.
This means that, in principle, cephalopods used for experimental or other scientific purposes should be bred and reared in captivity. However, there are significant difficulties in captive-breeding most cephalopod species (for exceptions, see2,49,50) and, therefore, this may not be feasible at the time of writing.
Development of more successful, standardised breeding procedures is urgently required. Article 38§1c indicates that projects must be designed ‘to enable procedures to be carried out in the most humane and environmentally sensitive manner possible’.
Where there is scientific justification for using animals taken from the wild, animals may be captured ‘only by competent persons using methods which do not cause the animals avoidable pain, suffering, distress or lasting harm’ (Article 9§3). 2 All those involved must observe a strict ethic of respectful treatment of animals, take into account their conservation status (section 3.2 below), and minimise the impact on the local ecosystem (section 3.3).
Care should be taken to prevent physical injury and stress to cephalopods at all stages in the supply chain, including capture (section 3.3), transportation (section 3.4), acclimatisation to laboratory conditions (3.5) and quarantine where required (3.6). It is also important to check local requirements for transport of animals in all countries along the route.
3.2 Cephalopod species commonly used in research and conservation status
Summary of major species of cephalopod used in research together with their source. Eggs may either wild caught or captive bred (e.g. for S. officinalis). See also reviews in Smith et al. (2013)
3
and Fiorito et al. (2014).
2
The possibility of obtaining animals through a given source is indicated by (

At the time of writing, species of the Class Cephalopoda have not yet been assessed for possible inclusion in the IUCN Red List of Threatened Species ‡ and hence none is listed as endangered. However, concerns are being raised for some rare species, based on local evidence and experience. Examples of locally protected species are: Euprymna scolopes in Hawaii; Octopus cyanea, Sepia elongata, Sepia pharaonis and Sepia prashadi in Israel (N. Shashar, pers. comm.); and some Mastigoteuthidae species in New Zealand. 51 Indeed, assessments are now underway for cephalopods § under the Sampled Red List Index (SRLI) initiative**, which indicates the relative rate at which the conservation status of certain species groups changes over time, and aims to broaden the taxonomic coverage of the IUCN Red List.
3.3 Capture methods
Schematic overview of the most common methods utilised for capture of cephalopod species for research purposes. See text for comments about welfare and environmental issues assocated with each method.
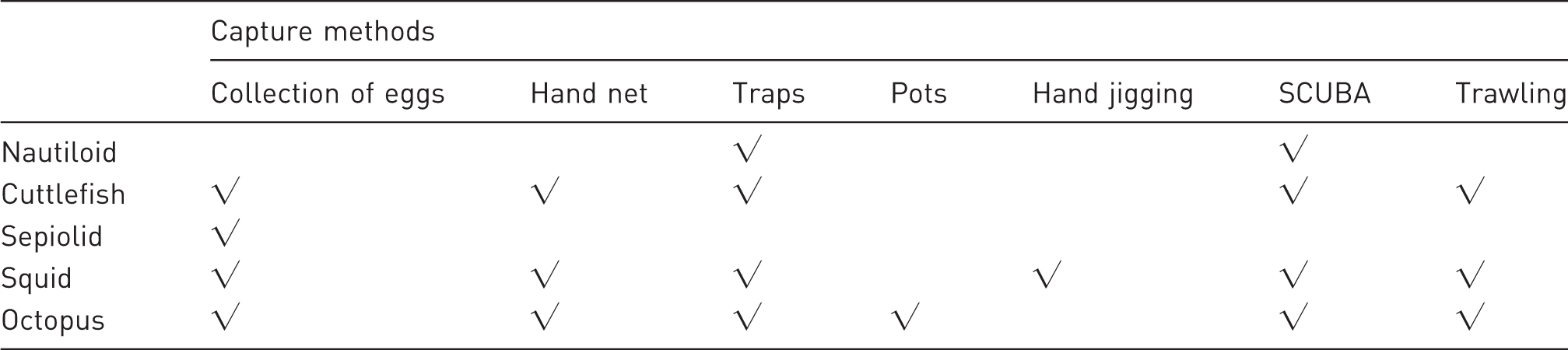
Several reviews describe commercial capture methods for cephalopod species.29,52 Current methods include, but are not limited to, nets, traps and pots (see Table 3).53,54 Environmentally destructive methods (e.g. trawling) should be avoided wherever possible. Hand-jigging is considered the ‘best’ method for capturing squid, but may not be appropriate for all squid species.51,55
Whichever method is used for capturing animals for research, it must not cause ‘avoidable pain, suffering, distress or lasting harm’ (Article 9). As noted above, animals may be captured only by competent persons. Moreover, researchers (and institutions) should only accept animals from suppliers who use appropriate capture and transportation methods; and the competence of third party providers should be evaluated based on the condition and survival of the animals supplied (see also section 3.4).
3.4 Transport (local, national and international)
Transport of cephalopods should always be in well-oxygenated seawater. 56
Whenever possible and applicable to the research project, transport of eggs is the simplest, most successful, and, hence, preferable approach. Details of methods for egg transport are available for some cephalopod species.56,57
When juvenile and adult cephalopods are transported, high survival rates should be achieved through careful selection of container type, maintenance of seawater quality in appropriate volumes and consideration of other measures to support animal welfare, such as food deprivation and cooling. The following discussion outlines general principles for transport of cephalopods, along with variations according to the duration of transport.
In the following sections, short-duration (i.e. short distance) transport is defined as requiring less than 2 hours; and long-duration (i.e. long distance) transport is for any longer period. These working definitions are based upon the consumption of available oxygen and detrimental changes in water chemistry (e.g. accumulation of ammonia and carbon dioxide and depletion of oxygen) as the duration of confinement for transport increases.
Since no specific systematic studies of transport methods for cephalopods are currently available, it is recommended that transport requirements should be based on the FAO guidelines for fish,58–60 paying particular attention to oxygen-sensitive species as they are considered to be comparable to cephalopods in their metabolic rate. Transport should also comply with the European Convention on the Protection of Animals during International Transport (ETS no 65, ETS no 193).
3.4.1 Transport containers
Since seawater is crucial for the survival of the animals, steps must be taken to ensure that water will not leak at any time (e.g. by containing animals in double bags and placing bags within a sealed container). It should be noted that several cephalopod species (e.g. octopuses) are occasionally reported to cut and bite through thin plastic bags (M. Kuba, pers. comm.), hence stronger transport containers should be used for them. In any case, the external container should be able to contain the entire water volume even if all inside containers/bags rupture.
Short-duration transport does not necessarily require animals to be placed into plastic bags, and other appropriate containers (large plastic bucket or box with a lid) may be preferred containing sufficient pre-oxygenated seawater to allow the animal to be completely immersed. Animals should be kept in dim or dark conditions, and movement and vibration of containers should be minimised.
For long-duration transport, similar to the protocol for transporting fishes 58 and depending on body size, one animal should be placed with 1/3 pre-oxygenated seawater and 2/3 oxygen-enriched air in double common aquarium bags (see also description in61–63). Once the bag is aerated, and the animal placed within it, it should be properly sealed (e.g. twisted at the top and folded over) and doubly secured (e.g. two rubber bands or cable grips). For transport periods over 12 hours, aeration and oxygenation may be necessary during transport, but care must be taken to use methods which do not add adverse conditions (i.e. bubbling) that may cause distress the animals.
Sealed holding bags (transparent to facilitate inspection if required) containing oxygenated seawater should be placed into insulated boxes (e.g. Styrofoam) to ensure that a temperature appropriate to the species is maintained during transport. The transport box should be darkened with a secure lid to keep the animals in darkness during transport, which reduces their stress. Bags should be packed with cushioning material (e.g. paper, Styrofoam pellets) to ensure they do not move during transport.
The use of specialised aerating and insulated transport containers is not yet common for cephalopods. However, the development of specialised transport containers should be encouraged (for reviews on fish transport containers see58,60).
3.4.2 Other factors for consideration
Food deprivation before shipping may be desirable, where appropriate, dependent on i. shipping distance, ii. the species and iii. size of animal. It is common practice among cephalopod researchers to withdraw food before shipping. This is to help prevent fouling of the water and ammonia build-up during transport. The duration of food deprivation should be based upon a consideration of normal feeding frequency, oro-anal transit time and renal ammonium ion excretion. However, food deprivation may require authorisation by the National Competent Authority.
Food deprivation before transport, alongside lowered water temperature, can also help to avoid build-up of toxic ammonia and carbon dioxide.64,65
With the aim of lowering metabolic rate before shipping it has been suggested that pre-cooled seawater at 2–3°C above the thermal tolerance minimum of an individual species should be used, ensuring that the temperature stays at that level for the duration of transport (see e.g. for the giant octopus 66 ). However, the impacts of this method on animal welfare are not yet known.
Sedation is not essential and is not recommended for transport of most cephalopods. However, sedation methods (i.e. cold water 67 or ‘chemical’ methods 68 ) has been used during transport in some instances with cephalopods.
It is interesting to note that in 1928 Grimpe suggested that very long duration transportation, i.e. requiring more than 2 days, should be achieved in steps allowing ‘resting’ periods in appropriate locations. 56
3.5 Acclimatisation after transport
Transport inevitably causes animals stress. Therefore it is important to allow time for them to recover from transport-related effects, to acclimatise to the new conditions including possible differences in water quality, temperature, illumination, diet, and the shape and arrangement of the environment (i.e. the tank). Allowing time for acclimatisation is vital for both animal welfare and science, as stress can confound scientific results.
Almost all cephalopods are highly stenohaline and stenotherm, and care should be provided to avoid any difference in salinity and water temperature of the container utilised for the transport and the tank where the animals will be placed. In the case of significant difference in water temperature, an adjustment of the different ‘media’ after transport (e.g. placing the container inside the final tank for slow adaptation of the temperature to the final one) should be considered.
Cephalopods arriving in a facility should be examined for injuries or other health issues, and treated and/or quarantined (see section 3.6) or humanely killed (see section 8.11) where necessary. It is also recommended that NH4 and CO2 levels in the transport water are measured. Together, these observations can help in assessing the quality of transport methods and suppliers.
Based upon species-specific requirements (i.e. individual or group living; see also Appendix 2) animals should be placed in a holding tank until they are habituated to it. The requirements and duration of this practice are species-specific (generally from 1 to 5 days) and for experimental reasons may be reduced to a minimum (e.g. when studying individual preferences towards a prey item or a stimulus), because evidence is available for contextual learning to occur in most cephalopods.26,69–71
To facilitate the required ‘habituation’ to the captive situation, it is best if the holding tanks are designed according to the same principles as the maintenance/experimental tanks for the species. Where food has been withdrawn during transport, a slow reintroduction is also recommended.
In the classic literature on cuttlefish and octopus, an adequate predatory performance is considered a sign of acclimatisation to a holding tank.26,36,72 In addition, while excessive inking upon introduction to a new tank is a sign of stress, low swimming rates, reduced likelihood of inking in response to a small disturbance near a tank, can serve as indications of successful acclimatisation (N. Shashar, pers. comm.).
When moving animals from one tank to another within the laboratory a standardised, minimally stressful protocol should be applied. In these cases, only a brief acclimatisation time may be needed (in the order of minutes to few hours). However, the experimenter should be aware of potential handling and relocation stress and its physiological consequences that may impact on the research.
3.6 Quarantine
The purpose of quarantine after reception of animals is to isolate cephalopods from the main population accommodated in the facility to allow observation and testing until animals are assessed as healthy and free from potential infectious diseases. Individuals identified as ill should either be separated for treatment, if the cause can be identified and treatment is available (see section 7), or humanely killed (see section 8.11) and autopsied (see section 6.3).
Quarantine is also useful to isolate individuals that become sick while being maintained in the facility, allowing time for sanitary measures to be put in place and ensure appropriate containment of organisms and waters.
Currently, quarantine is not the general practice in the cephalopod research community. However, further studies are required based on recent research of cephalopod diseases and diffusion of parasites. 73
The duration of quarantine should be sufficient to assure health of the individual animals. The needs of individual quarantined animals vary according to the biology and behaviour of the species (e.g. group holding maybe appropriate for gregarious species, but others may require individual accommodation; see also Appendix 2).
Quarantine should involve complete separation between animals to be quarantined and the current laboratory population; this should be achieved either by using separate rooms or equipping facilities with plastic screens to separate quarantine tanks from others. In addition, water supply should also be separate, to prevent diffusion of any potential harm to the water circulation and/or the environment. Similarly, equipment (e.g. nets) should not be shared between tanks.
During the quarantine period, animals should be monitored closely for unusual clinical signs or behaviours (see also sections 6 and 7), and detailed examinations (including autopsy; see section 6.3) made of any individuals who are considered to be ‘abnormal’. In the cases of identification of diseased animals present in the laboratory holding facility, this should be regarded as a possible indicator of disease in the entire stock/holding group, and hence particular attention should be provided, and eventually they should all be treated or humanely killed.
4. Environment and its control
4.1 Seawater supply and quality
Both natural and artificial seawater (see also below) are suitable for the maintenance of cephalopods. For fish, Annex III of Directive 2010/63/EU requires that ‘an adequate water supply of suitable quality [is] provided at all times’, and that ‘at all times water quality parameters’ are ‘within the acceptable range that sustains normal activity and physiology for a given species and stage of development’ and such requirements apply equally well to cephalopods.
4.1.1 Types of seawater circulation system
There are two principal seawater systems for keeping cephalopods: closed systems which recycle a reservoir of seawater, and open systems which either draw a continuous supply of water from the ocean (flow-through systems), or pump seawater into a reservoir and regularly replenish it with fresh seawater (semi-closed systems).
Closed systems have the advantage of enabling control of all parameters of the environment, but are more costly due to the need for additional environmental monitoring and control equipment.
Open systems rely on fresh seawater being drawn from the ocean. While this has some advantages (especially not needing expensive filters), it limits the facility to keeping animals that can live within the given water parameters. For example, non-native species cannot be kept in this type of system without considerable efforts purifying and sterilising the reflow. Naturally open systems are also limited to areas close to the shore.
Closed systems: Efficient and relatively easy-to-maintain closed aquarium systems have been developed for cephalopods.74–78 Commercially available artificial seawater preparations are considered adequate, provided they contain the necessary substances and trace elements to meet the physiological needs of the particular cephalopod species, and for this reason mixtures designed for marine invertebrates and corals should be preferred.
Water flow in recirculating systems or filtration within enclosures should be sufficient to remove suspended waste and to ensure that water quality parameters are maintained within acceptable levels. Filtering systems in recycling/recirculating seawater should be adequately planned and maintained.79,80 Appropriate processes for monitoring water parameters should be implemented, and alarms in place to ensure flow and seawater levels are adequate.
Open systems: seawater drawn from the ocean should be tested for contaminants and pathogens, and treated to remove them. The water supply should also be evaluated to ensure that there is sufficient capacity, including ability to cope with periods of maximum demand and emergency situations. To protect animals from potential contaminants, other measures, such as appropriate filtering or a reverse osmosis system, may be required.79,80
Tests to determine the chemical composition and presence of contaminants/toxins will determine the treatment necessary to make the water suitable for use. Seasonal factors such as phyto- or zoo-plankton blooms, tidal cycles, and seasonal seawater mass turnovers can have periodic effects (on a scale of hours, days or months) for seawater and these should be anticipated.
All systems: water flow should enable cephalopods to maintain normal locomotion and behaviour. Cephalopods can use rapid expulsion of water through the funnel to power jet propulsion, which results in swift movement. Cuttlefish and squid have fin-like structures on the mantle to assist in locomotion. Squid are in continuous motion due to their pelagic nature, thus water flow75,77 needs to be sufficient to ensure appropriate life-style requirements, and adequate water quality including quick removal of ink (if released).
4.1.2 Water quality
As for other aquatic species, water quality is the most important factor in maintaining the health and well-being of cephalopods. Insufficient water quality will cause stress and disease. Water-quality parameters should at all times be within the acceptable range that sustains normal activity and physiology for a given species and individual (see Appendix 2); and should remain stable, unless the life style of a given species requires changes (e.g. because of large vertical migrations during a day or seasonal changes) ‡ . Optimum conditions vary between species (e.g. deep-sea benthic octopuses are especially sensitive to changes), between life-stages (e.g. paralarvae, juveniles, and adults) and according to physiological status of the individual (e.g. females preparing to lay eggs).
Most cephalopods show little adaptability to changing water-quality conditions, and so when animals need to be moved between tanks or systems, it is important to ensure that water parameters are mirrored and maintained. If this is not possible, gradual acclimatisation will be needed (see section 3.5), as for other marine invertebrates and fishes.
Dissolved oxygen, pH, carbon dioxide, nitrogenous compounds and salinity should be monitored and maintained according to the appropriate range for each species. Appendix 2 provides a list of water quality criteria for optimum health and welfare of cephalopods; for more detailed discussion of monitoring water quality see below.
4.2 Monitoring water quality (O2, pH, CO2, nitrogenous material, salinity and metals)
Seawater parameters should be monitored (continuously by specific electrodes or intermittently by chemical methods) and recorded at an appropriate frequency (at least daily), thus allowing proactive, rather than reactive, management of water quality. Parameters that need to be measured and the frequency of measurement vary (see also Appendix 2), depending on whether the system is open or closed. For example, while there may be no need to measure nitrites/nitrate in a high volume flow-through system (depending on the source of the water), such measurements are critical with recirculation systems.
At a minimum, environmental monitoring systems should provide information on water flow, oxygen saturation and water temperature. Parameters measured should also be relevant to the health and welfare of the particular species housed in the facility (see Appendix 2). In general, recirculation systems should be monitored for a larger number of parameters, including, but not restricted to, dissolved oxygen, pH, nitrogenous material, salinity, total dissolved salts and temperature (see below). As a minimum, water quality analysis should be carried out at times of greatest demand on the system (usually after feeding) to identify potential problems.
Water and tanks should be kept clean particularly of faeces and uneaten food. In semi-open and closed systems, water should be treated to reduce potential pathologies, for example, using UV light or ozone. If ozone is used, measurements of ozone concentrations and/or redox potential of the reflow entering the system are necessary to avoid toxicity.
Alarm and notification values must be set and their significance as potential indicators of problems in the system explained to all relevant personnel. There must be an agreed, clear protocol for contacting those responsible for the facility when problems are identified outside of normal working hours.
The monitored parameters should be recorded and the information stored for at least 5 years. For all parameters considered below and for techniques of keeping animals information is also available through a recent compilation of research on the culture of cephalopods. 50
4.2.1 Oxygen
Cephalopods have high metabolic rates, so oxygen concentration should generally be kept high (close to saturation); and where tank inflow is not sufficient, supplementary aeration of the water must be provided. However, supersaturating the water is not advisable as it may cause gas bubbles to become trapped in the mucus layer of the animals, thus limiting gas exchange at the gills (for octopus: G. Fiorito, pers. comm.; for several cephalopod species: J. Rundle, pers. comm.).
Information on oxygen consumption in some cephalopods is provided by Winterstein 81 (see also82,83,83–87) and reccomended requirements are summarised in Appendix 2.
It is important to note that monitoring oxygen levels is not informative of the oxygen available to the cephalopod unless it is combined with measurements of pH (see next section).
4.2.2 pH
Due to the effects of pH on the carriage of oxygen by blood pigments, cephalopods tolerate low pH poorly.88–96 Regular measurement and careful maintenance of pH is therefore critical. Acceptable pH levels depend on many water quality factors, for example, carbon dioxide (see below), and calcium; as a consequence, control of soluble gases and water solutes is important. This may be a particular issue for establishments that use synthetic marine salts rather than natural seawater and closed filtration systems, especially where water changes are limited. Acceptable pH values for keeping cephalopods are summarised in Appendix 2.
4.2.3 Carbon dioxide
Carbon dioxide is produced during respiration and dissolves in water to form carbonic acid, thus lowering the pH. Since stability of pH is very important, accumulation of carbon dioxide should be avoided. Situations that may increase carbon dioxide levels include high stocking density and poor aeration.
Care should be taken that water supply systems, particularly in the case of groundwater-based systems, do not introduce harmful quantities of carbon dioxide to the enclosures.
4.2.4 Nitrogenous material
As cephalopods are carnivores, hence requiring a high protein diet, the accumulation of potentially toxic nitrogenous compounds can be a problem, particularly in closed systems. It is also necessary to avoid accumulations of ink (especially when keeping cuttlefish, that ink in large volumes). Timely removal of uncomsumed food, use of adequate protein skimmers and suitable water flow rates, along with careful water filtering in closed systems, will help to reduce levels of organic waste, including ammonia, nitrites and nitrates. It is also beneficial if the flow rate in the tanks can be adjusted to different situations (e.g. increased after inking). Additional water changes can provide a supplementary means of removal of waste products and substances such as ink.
Build up of nitrogenous compounds may lead to behavioural changes and/or changes in skin colouration in cephalopods. For example, at nitrate levels >80 mg/l cuttlefish become very agitated, will ink profusely and their skin tone may be dark; and larvae and hatchlings are more vulnerable to bacterial disease (A. Sykes, pers. comm.).
Levels of nitrogen compounds tolerable by different species are reviewed in Appendix 2 (see also Iglesias et al. 50 ).
4.2.5 Salinity and metals in seawater
As cephalopods are marine organisms, maintaining an appropriate salt concentration is vital. The salinity should match the natural habitat of the animals.
Commercially available artificial seawater preparations and especially any mixture designed for marine invertebrates and corals are considered adequate as they contain all the necessary substances and trace elements to keep cephalopods in good health. However, in accordance with instructions for the different brands of salt, some trace elements, in particular strontium and calcium should be monitored and added if necessary.
Copper and its alloys are considered to affect the salinity and ‘poison’ the seawater 56 and therefore should be avoided in any system holding cephalopods.
Cephalopods are reported to accumulate and be sensitive to heavy metals so care should be taken to ensure these are monitored and maintained within normal ambient ranges for the species, to limit potential damage (see above and also 2 for relevant literature).
4.3 Lighting control
Light influences, either directly or indirectly, almost all physiological and behavioural processes in cephalopods, including growth, development and reproduction. Lighting requirements vary between cephalopod species, and both wavelengths and intensity of lighting should ‘satisfy the biological requirements of the animals’, where these are known (Directive, Annex III, section 2.2a). The natural history of the species, in particular the normal living depth, can provide clues to help meet the species lightling preferences: for example there are many cephalopods that prefer very little light. There is limited specific knowledge on wavelength perception for almost all cephalopod species. 97 However, it is estimated that simulated sun-light equivalent to that normally experienced at 3–8 m depth at sea should be acceptable for the majority of cephalopod species commonly used as laboratory animals (but see further information in Appendix 2).
Photoperiod should also be maintained according to the natural requirements of the species.98–103 However, there is evidence that some cephalopods may easily adapt to changes in day/night regime.26,104–106
Where task lighting is needed for people working in the room, it should be restricted in its dispersion, and/or be placed below the level of the tank surface, to reduce disturbance to the animals. Use of automated dimmer controls that allow light intensity to be gradually increased is important and recommended (G. Fiorito, pers. comm.; see also 107 ). For example, for decapods and nocturnal octopuses sudden changes in light level may cause escape reactions, and in some cases inking, thus a simulated dusk and dawn period is desirable. Care should also be taken to ensure that animals are not disturbed by night-time security lighting entering through windows in the holding facilities. The output of fluorescent lights can be diminished by using dummy bulbs to reduce light levels.
4.4 Temperature control
Water temperature should be controlled within the natural range for the species; and, where necessary, appropriate chilling/heating equipment must be used to ensure the optimal temperature range for the animals.
Cephalopod species vary in their sensitivity to changes in water temperature. In general, higher water temperatures create problems for animals from temperate climates like octopus and the cuttlefish. Transitions of temperature should not be sudden.56,108 Where water changes are performed on larger scales, temperature spikes, which may cause adverse effects, should be controlled and avoided.
4.5 Noise and vibration control
Background noise, and vibration from housing systems, such as pumps or ventilation units, should be minimised as they are likely to impact on cephalopod welfare.
Several studies suggest that cephalopods can detect sounds even at low frequencies,109–115 and other recent work shows that cephalopods are as likely as other marine organisms, to suffer from low-frequency noise traumas.116–118
In common with other aquatic species, cephalopods dislike vibrations, such as drilling or banging on tank sides, and some species, such as cuttlefish, may respond by inking. Therefore, the most important aspect of sound reduction is to minimise disruption and avoid sudden noises, which could startle the animals.
4.6 Aquatic life support systems and emergencies
Tanks should be built so that complete drainage is impossible when they are inhabited (although ability to drain tanks may be required for cleaning purposes).
Two independent sources of water movement/oxygen supply are also recommended, for example, pumps for water circulation plus extra air sources to provide additional aeration.
Electronic alarm systems help to ensure that problems in a system are detected promptly. All facilities must have an emergency plan in place should problems arise (including out-of-hours), with clear actions that are understood by all and effectively communicated to everyone. There must be a backup system to enable an appropriate response to the worst case scenario of a complete system failure, and so avoid circumstances in which animals would have to be humanely killed due to suffering from anoxia or a build up of organic waste.
5. Accommodation and care
5.1 Background and requirements of the Directive
About 50 species of cephalopod have been kept successfully in aquaria (M. Kuba, pers. comm.; see also 108 ). These range from small species such as bobtail squid (E. scolopes) to larger pelagic squid (e.g. Loligo vulgaris, Doryteuthis pealeii), octopuses (e.g. O. vulgaris, Eledone cirrhosa) and cuttlefishes (Sepia officinalis), and the giant pacific octopus (Enteroctopus dofleini).
At the Stazione Zoologica di Napoli, considered to be the first large-scale facility for the maintenance of cephalopods 56 (mostly for cuttlefishes and octopuses, A. Droesher and G. Fiorito, pers. comm.), outdoor tanks were preferred to indoor rooms, to allow animals to be kept in natural light with seasonal daylength. However, shading was provided to reduce direct sunlight to the animals. In the following years, indoor tanks were installed and artificial lighting was introduced to supplement natural illumination (A. Droesher and G. Fiorito, pers. comm.).
The knowledge accumulated in various laboratories around the world, with a variety of cephalopod species, supplemented the original studies at Stazione Zoologica and facilitated the design of closed systems for maintenance of species which adapt less readily to laboratory housing such as squid (for review and methods see 75 ).
Annex III of Directive 2010/63/EU sets out requirements for care and accommodation of animals. Section A lists general requirements pertaining to all species and section B lists species-specific requirements, for all vertebrate classes, including brief guidance for fish (but with no distinction between the different classes of fish). Some of these Section B requirements for fish might also apply to cephalopods, 2 but cephalopods are not specifically mentioned in Annex III.
5.2 Holding facilities for cephalopods
Planning design and maintenance of new accommodation facilities for cephalopods should take into consideration key points outlined by the Committee for the Update of the Guide for the Care and Use of Laboratory Animals. 119
Access to the facility should be allowed only to people who have received relevant training and have a legitimate need for access. Movements of personnel inside the facility should also be monitored and controlled to minimise disturbance to the animals and ensure biosecurity, which may require measures such as physical barriers and access restriction/control.
Walls of holding rooms should generally be of dark neutral and continuous colours. However, very dark colours may make it difficult to identify dirty areas, so specific evaluation of the appropriate colour may be required.
Cephalopods require large volumes of seawater. All facilities should have an emergency contingency capacity, capable of maintaining aerated and filtered seawater should normal systems fail. Monitoring systems including remote alarm notification should be designed and used in cephalopod facilities.
Noise should be minimised to avoid disturbing animals in both housing and experimental rooms. When applying sound-attenuating material to the ceiling or walls, always consider that it has to be sanitisable. All vibration sources (e.g. mechanical equipment, electrical switches, through ground-borne transmission) should be identified and vibration isolation methods should be used to reduce noise (e.g. by placing equipment on rubber pads). Noise-producing support functions, such as tank and filter washing, should be separated from housing and experimental areas, wherever possible.
Fire and environmental-monitoring alarm systems should be selected and positioned to minimise potential disturbance to animals.
All procedures and other manipulations of living animals should be carried out inside the facility to minimise stress to the animals, unless there is scientific justification for doing otherwsise. Therefore, a typical cephalopod facility should have available: i. adult animal housing/holding room(s) divided by species if possible, and breeding/’hatching’ room(s); ii. quarantine room(s) (if needed); iii. an area for acclimatisation of animals; iv. procedure rooms separated from holding and breeding rooms, for experimental techniques, including regulated procedures (e.g. surgery, behavioural experiments, imaging, clinical treatment, humane killing, necropsy, etc.); v. separate ‘service’ rooms for storage of food, supplies, chemicals, etc., and for waste (including biological material) storage before incineration or removal.
Shared facilities, where cephalopods are kept in water systems and rooms hosting a range of other types of marine organisms, are not recommended. Additionally prey species should never be accommodated in the same tank as their predators.
In designing holding facilties for cephalopods and selecting the construction materials, it is recommended that guidelines developed for fish are followed (for review see 79 ).
Materials used to build aquatic facilities should be non toxic. Any unavoidable use of material with the potential to be toxic should be reduced to the minimum, recorded and the information made available to staff, veterinarians and inspectors. In particular, materials that may release specific ions, chemicals or corrosion by-products from their surfaces should be avoided. The use of metals requires consideration of their interactions with seawater, and the potential effects of that interaction on the animals.
Special attention should also be given to the behavioural needs of the animals. For example, non-gregarious animals or animals that might show aggressive interactions (e.g. males during mating season) may require housing out of sight of others. Attention must also be paid to species-specific differences in terms of the level of disturbance that may be acceptable; for example, O. vulgaris appears to be quite resilient whereas cuttlefish or squid react more strongly to unfamiliar and sudden movements.
5.3 Housing
Cephalopods are strictly marine, and all require high-quality sea-water, but their varying habitats, social behaviour and especially nature and level of locomotion determine how they should be housed. Aquarium size and stocking density should be based on the physiological and behavioural needs of the individual species, and requirements for their health and welfare (see Appendix 2).
Section 3§3b of Annex III of the Directive indicates that all animals, including cephalopods, ‘shall be provided with space of sufficient complexity to allow expression of a wide range of normal behaviour’, including social behaviour, locomotion and feeding, and ‘shall be given a degree of control and choice over their environment to reduce stress-induced behaviour’.
Stocking density will vary depending on the animals’ natural history and behaviour, water flow, size, age and health. Water quality is critical (see section 4.1.2 above).
Most octopuses are solitary and should be kept in isolation. Nautilus are primarily solitary in the wild but may be housed together at low densities.
The social structures of many species, including the European cuttlefish (S. officinalis) are not known, but in general social animals including many squid, are best kept in groups. However, social interactions should be monitored to check for adverse welfare effects; animals should be grouped according to age to avoid fighting and possible cannibalism, particularly in the breeding season or where there could be territorial antagonism. Such measures should not alter the overall welfare of the animal, and, in general, should be respectful of the behavioural needs of each individual species.
Depending on the species, individuals may require dens, shelters and other devices (mostly for bottom-living cephalopods).
Enriched environments must be provided, to allow the animals to express their normal behaviour (see further discussion of enrichment in section 5.11 below).
5.3.1 Tanks
Tank requirements and stocking density vary among cephalopod species and ages (see Appendix 2 for a summary of requirements). For example, for benthic species, like O. vulgaris and S. officinalis, the available bottom area is an important requirement, while for pelagic species this is represented by the volume of water; the depth of the tank should be considered for species with known diel (diurnal) vertical migration (e.g. Nautilus pompilius).
All cephalopods should be kept in opaque tanks of neutral colour. O. vulgaris and S. officinalis may also be kept exceptionally in transparent aquaria, as long as the floor of the tank is opaque (and/or covered by sand). Tanks can be of rectangular or of any other shape, but for decapod cephalopods they should have rounded corners to minimise potential injuries (see Appendix 2).
In general (and especially for Sepia and other decapods), sharp objects and rough surfaces that can cause skin damage must be avoided. Jetting can lead to collisions with the walls of the tank if animals are startled, or there is insufficient space for escape reactions.
Tanks can be made of PVC, fibreglass, glass or any non-toxic material capable of being adapted to achieve appropriate shapes and allowing a smooth internal surface, and which is easy to clean and sterilise or decontaminate as necessary. As potentially toxic materials might have been used during initial tank assembly (e.g. silicone-based adhesives and sealants), it is recommended to wash the tank thoroughly, leave it filled with water for at least 24 hours, and then rinse with seawater, before animals are introduced.
Tanks should be equipped with a covering (e.g. tank net or rigid transparent covering) that prevents animals escaping. Lids also serve as a barrier against the accidential introduction of any foreign objects, animals or chemicals. Tanks lids may be constructed of materials such as plexiglass or clear acrylic to allow visual inspection. The distance between the water surface and lid should be enough to minimise the risk of damage, for example, in the case of squid which are capable of leaving the water using their ‘jet-propulsion’.
5.3.2 Tank labelling
Tanks must be carefully labelled, to identify and record the histories of individuals or groups of animals.
Labels should include detailed information for each individual, including origin, first dates in captivity and arrival in the laboratory, sex and morphometric measurements if possible (e.g initial body weight, dorsal mantle length), along with the number of animals in the tank (which may be an estimate depending on developmental stage).
For animals undergoing procedures, the label should identify: i. the procedures being performed (e.g. the label could refer to a detailed protocol filed for easy access by all relevant staff); ii. the date when the procedures were started; iii. the person responsible for the animals (e.g. the Principal Investigator). Records of any adverse effects shown by the animals should be also be easily accessible ‘tankside’ and should be carefully maintained (see below for further discussion). Taken together, all these points should make it is easy for animal care staff and scientists to identify animals showing signs of welfare compromise, determine the likely cause of the adverse effects (e.g. whether procedures applied, such as recent anaesthesia) could explain the abnormalities, and take action to mitigate them.
5.4 Cleaning of tanks
Water quality should be monitored daily as a minimum (see also section 4). When water changes are necessary, the smallest possible amount should be removed.
Tanks should be free of organic waste (e.g. uneaten food or faeces), otherwise water quality, and thus animal health will be harmed.
Open systems: tanks should be regularly drained and cleaned to prevent fouling and reduced water exchange. There should be no risk of back-flushing, and consequent fouling of enclosure water. The sides and bottom of enclosures should be cleaned regularly to avoid the accumulation of detritus.
Closed systems: waste material should be removed as soon as possible after feeding. Total water replacement and whole tank cleaning should be avoided, as the biochemistry and flora that develop in a mature system are essential to well-being, as known in common practice for acquaria keeping. Depending on the size (i.e. number of tanks/system) care should be given to facilitate the most appropriate conditions at equilibrium. Where complete draining out of a system is required for decontamination reasons, the system must be allowed to re-mature after the addition of clean seawater, prior to adding animals.
When cleaning of tanks occupied by animals is necessary, the process should be designed to minimise disturbance and distress; in most cases animals will need to be removed from the tank during cleaning. Capture and transfer methods should conform to the principles outlined in these Guidelines, and the time spent in a holding tank should be minimised.
Disinfectants should be used with extreme caution and only in dry tanks, which are then rinsed with clean water. Detergents should be avoided and substitutes are preferred. 120 Animals must not be exposed to any substance used for cleaning of tanks.
5.5 Methods for individual identification and marking of cephalopods
Depending on stocking density, it can be difficult to identify individual cephalopods. Marking or tagging, other than in species with external shells such as Nautilus, is difficult, owing to vulnerability to tissue damage. Individual cephalopods may have unique natural markings, and whenever possible these should be used for identification.121,122 Several marking methods have been successfully applied to different species of cephalopods (for examples see review in 123 ). Methods used with success, but which require anaesthesia for their application – and hence scientific justification and approval from the National Competent Authority – have included implanted fluorescent elastomer tags in squid and octopus,124,125 subcutaneous dye injection into the arm of octopus,123,126 and external tagging of cuttlefish, octopus and other species.127–132
Careful consideration of harms, benefits and justification is therefore needed for invasive tagging, and development of minimally invasive individual marking methods for cephalopods is an important goal.31,133
5.6 Food and feeding for adult cephalopods
Most cephalopods are carnivorous and active predators,134–136 hunting their prey using a range of strategies (review in 134 ). However, nautiloids are scavengers and some species of octopus will eat dead food items.
For many cephalopod species at different life-stages, live prey is the only known method of feeding. This prey may be fish or invertebrates, such as crustaceans, which need to be treated ethically and legally,137,138 and the feeding regime should suit the lifestyle, natural diet and developmental stage of the animals.
The duration of digestion (food intake to elimination) is 6–15 hours in the common laboratory species of cephalopods and is slower at lower temperatures in a given species,139–141 so feeding frequency (and appetite) may alter with season (temperature) in open systems.
Data on the richness of cephalopod diets in their natural habitats is limited, but known to include, amongst others, zooplankton, molluscs (including other cephalopods), polychaete worms, crustaceans, chaetognaths, sea urchins, fishes and jellyfish.26,142 An estimation of the relative breadth of diet has been attempted for some cephalopod species, including species most frequently used as laboratory animals, 26 and shows that some species’ ‘natural’ diets are restricted to certain prey items (i.e specialists), such as Spirula spirula, which feeds on detritus and zooplankton, 143 whilst others are more opportunistic species (i.e. generalists) such as S. officinalis or O. vulgaris (for review see 26 ). However, estimation of diet variety is substantially biased by research effort.
In laboratory conditions, animals usually adapt to prey on several different types of food.36,108
Nautilus requires food with a high level of calcium carbonate, such as shrimp with carapace, lobster moult shells or fish heads. Most cephalopods have a higher metabolic rate than fish, and their daily food intake which is rich in protein can be considerable: for example, 3–10% body weight per day. 144 The feeding regime, palatability and method of food presentation should ensure that animals are adequately fed. Young and/or wild caught animals need particular attention. In some cases, enrichement with favoured foods and touching the animals’ arms with food may trigger feeding. Refusal to eat can be an early sign of illness (see also section 6.1.1).
Cuttlefish and squid are especially sensitive to inadequate nutrition; the most evident signs include: protruding eyes, poor body condition and floating (especially in juveniles). Consequently, in general over-feeding is preferred, as long as excess food is removed in an appropriate time-frame for the feeding habits of the species. 145 However, ad libitum feeding of S. officinalis may cause buoyancy problems, so this is not advised (K. Perkins, unpublished data).
Use of alternatives and/or artificial/synthetic food to natural prey for rearing of some cephalopod species. The table is based on an overview of recent literature (most representative papers are included) mostly for aquaculture purposes (unless otherwise stated). For review see also Iglesias et al. 50

CPSP: namely ‘Concentrés de Protéines Solubles de Poisson’ is a concentrated fish hydrolysate currently trademark of COPALIS (http://www.copalis.fr/en/home/products-and-applications/animal-nutrition/aquaculture-breeding.html); see also Kristinsson and Rasco 528
This study is also aimed to the understanding of nutritional requirements of octopus
The frequency of feeds is important and depends on the species and water temperature in the tank. The duration of digestion also depends on the species, and other factors including the animals’ size, maturity and the type of food (for review see 148 ). In O. vulgaris, gut transit times are quite rapid (about 12 hours at 18–19°C) suggesting that crop capacity is not great and so daily feeding should be the norm.141,149,150 Daily feeding is also common practice for most coleoid cephalopods. However, other evidence from adult cephalopods, particularly cuttlefish and octopus, suggests that they may not need to eat every day.146,147,151
5.7 Food and feeding for larvae and hatchlings
Evidence is provided that different dietary needs are required for cephalopod species during the early stages post-hatching.28,152,153 For example, hatchlings of S. officinalis often have a yolk sac which provides nutrition until they start feeding a few days later; in contrast, O. vulgaris paralarvae need to feed immediately in the water column. However, not all taxa of cephalopods have been successfully reared in laboratories, and so information on the dietary requirements of hatchlings is limited (for review see 50 ). Evidence available for some species suggests that embryonic development often requires trace nutrients that are present in natural seawater,154,155 in which case development might be hindered in closed artificial systems. It has also been suggested that ‘dissolved gases and nutrients may also contribute to metabolic and nutritional requirements via absorption through the epidermis’.144,156 In addition, a close relationship between the fatty acid profile of the dietary components and of the individual at early stages after hatching has been reported. 157 This emphasises the importance of improving understanding the nutritional needs of juveniles, especially if artificial diet is being considered for rearing purposes.
5.8 Handling and moving cephalopods in the laboratory
Handling procedures should be carried out only by competent, trained personnel using techniques that minimise the potential for injury and reduce stress to the animals (see also section 10). It is recommended that laboratory coats and gloves should not be of white/pale colours, as the handler can be mistaken for a ‘predator’.
The skin of cephalopods acts as an organ 158 and is very delicate and so every effort should be made to minimise handling and removal of animals from the water. It is especially beneficial to standardise handling procedures, as anecdotal evidence indicates that cephalopods can habituate to handling.
It is preferable to move the animals in water using containers where they can be gently restricted before moving from one tank to another or any other location. Training animals to enter a container, possibly using small rewards, may reduce stress and habituate them to the transfer.
Cuttlefish and squid should be immersed at all times and a dark net should only be used to coax the animal into a container.
Nautilus is particularly sensitive to air, and repeated air exposure is anedoctally reported to have negative effects on the health of the animals (R. Smallowitz, unpublished data).
Octopuses can be moved using nets (suggested dark nylon 2-mm mesh) with a long sleeve to reduce the risk of escape; exposure to air should be minimised. A container method has been developed for O. maya 159 and, although not currently in use, represents a useful approach indicative of methods that should be developed for animal transfer within a facility.
Nets and containers should be clean, disinfected and rinsed before use. Agitation during moving should be minimised, as all cephalopods have a sensitive statocyst system. 160
Handling and other human interactions should be monitored and recorded, as the frequency and nature of the interactions can influence behavioural performance of individual animals.
5.9 Environmental enrichment
Environmental enrichment should not compromise the need for adequate levels of hygiene and the ability to observe the animals’ health (section 6 below) without causing too much disruption. The impact on health and welfare of environmental enrichment should be evaluated objectively,161–163 particularly to avoid the application of ‘environmental changes’ which may be detrimental to the animal well-being, and to ensure health or water quality are not compromised.
Section 3§3b of Annex III of the Directive states that ‘Establishments shall have appropriate enrichment techniques in place, to extend the range of activities available to the animals and increase their coping activities including physical exercise, foraging, manipulative and cognitive activities, as appropriate to the species. Environmental enrichment in animal enclosures shall be adapted to the species and individual needs of the animals concerned.’ The same section also states that ‘the enrichment strategies in establishments shall be regularly reviewed and updated’.
These provisions require on-going consideration of the effects of laboratory housing on animal welfare and efforts to enhance well-being wherever possible. Exemptions from these, and other, requirements outlined in Annex III have to be approved by the National Competent Authority, and must be for scientific, animal welfare or animal health reasons.
Environmental enrichment aims to enhance the well-being of animals in captive conditions, by identifying and providing stimuli that enable animals to express as wide a range of their normal behaviours as possible.164,165 Enrichment is proven to be effective for many species, including fishes,166–168 cephalopods and other invertebrates.138,169–175
Enrichment may be accomplished through changes in the tank environment, for example, by varying factors, such as the shape of the tank, flow of water, variety of live prey items (if these are essential), conspecifics and environmental complexity; and also by providing opportunities for animals to engage in specific activities and exercise some choice.
Enrichment strategies should be tailored to the needs of the particular species concerned. For example, open-water species may require large but less complex environments. Social animals should be housed in groups. Benthic cephalopods are better kept in complex environments with suitable substrates (sand, gravel or pebbles) and dens.
Nautiluses should have access to vertical space for movements and attachment at a variety of levels, thus meeting their natural habit of daily vertical migrations.176,177 However, not too many vertical attachments should be added, as nautiluses naturally swim up and down whilst circling around the perimeter of tanks and require space to do so. Adding texture (artificial coral reef) to at least one wall of the tank may make it more attractive to the animal (and may promote egg laying; G. Barord, pers. comm.).
In octopuses, interaction with objects is a common form of enrichment and is recommended; providing a den as refuge is not considered to be enrichment, as it is a basic requirement for octopuses, and for all benthic species that use refuges in the wild. Artificial shelters can take the form of many different objects (e.g. bricks, ceramic pots, plastic jars), but dark and opaque dens are preferred over clear ones.
Suggestions for the type of objects (artificial and/or natural) to be added in tanks as enrichment for most common cephalopod species are provided by Grimpe. 56 Recent systematic studies are missing and data available are mostly anedoctical.
Caution should be taken to avoid objects added to holding tanks that could harm or limit full expression of the behavioural repertoire of the animal. Mirrored surfaces should be avoided, since may create agonistic reactions expressed by some individuals towards the ‘ghost’ reflected image (G. Fiorito, pers. comm.). Accounts of tank design for coastal and reef squid species provide also information on environmental enrichment for these animals.76,178
6. Assessment of health and welfare
Annex III, Article 3.1 of the Directive requires that establishments have a strategy to ensure that the state of health of the animals safeguards animal welfare and meets scientific requirements. This should include regular health monitoring and plans for dealing with health breakdowns (see section 7). The starting point for fulfilling these requirements is objective monitoring and recording of the health and welfare of the animals and recognition of the factors likely to cause deviations from optimal status.
The primary factors that could cause a decline in health and welfare and which require monitoring are effects of:
▪ environmental and housing conditions (including: capture, transport, handling, stocking density, tank design); nutrition; variations in: water temperature, oxygen levels, pH, salinity and water contaminants (sections 4 and 5); ▪ infectious disease (section 7); and ▪ experimental (regulated) procedures (section 8).
Irrespective of the cause, objective criteria for assessment of the overall health and welfare status of animals are required to:
ensure that animals arriving in the laboratory are healthy; ensure that housing and care are adequate for the maintenance of good health and welfare; assess the impact of experimental procedures (section 7) in terms of severity and identification of pre-established humane end-points; identify and implement measures to rectify health and welfare problems and enhance the well-being of animals, and refine procedures so that they cause less harm to the animals; and monitor the efficacy of any therapeutic interventions.
As for other animals, the key parameters used to assess the health and welfare of cephalopods are behaviour and appearance, supplemented in some cases by measurement of a number of physiological ‘biomarkers’.
Potential generic indicators of health and welfare in cephalopods (primarily cuttlefish, squid and octopus) that could be used in daily assessment and adapted for monitoring animals following a procedure (see section 7). For each sign (see section 5 for details and references) a guide is given to show how each can be graded (indicated from green to red) to indicate an escalation of aspects of PSDLH from mild to moderate and severe. The table makes no assumptions about the underlying cause or what the animal is actually experiencing. The table utilises the principles of health and welfare assessment developed by Morton and Griffiths385,387 and widely adopted for welfare assessment, 485 adapted for cephalopods by Andrews et al. 8 and incorporated into Directive 2010/63/EU severity assessment framework (Expert Working Group 2012: http://goo.gl/DozPKi). It should be appreciated that for practical implementation this table will need to be adapted for each species, validated by research in multiple laboratories and revised accordingly. Note that the table is included here to prompt consideration of the challenges involved in objective assessment of PSDLH in cephalopods and to stimulate research.



For animals that are being used in an experimental (regulated) procedure (section 8), the observation frequency may be increased depending upon the nature of the procedure and its anticipated impacts on animal welfare. Factors to be considered are described in detail below, but most require further validatation, including consideration of their severity (see section 2.2.3 and 8.2).
It should be noted that prompt identification of problems is essential so that i. action can be taken to reduce and preferably eliminate any suffering and ii. in the case of procedures, to ensure that humane end-points are promptly implemented where appropriate and that severity limits are not exceeded (sections 2.2.3 and 8).
6.1 Objective assessment of health and welfare
Proposed parameters are described in detail below and summarised in Table 5. Welfare assessments should be performed in the animals’ home tank and without removal of the animal from the water wherever possible. Each element of the assessment will ideally require some form of quantification to enable recognition of points at which particular parameters reach a pre-set humane end-point and to enable actual severity of a procedure to be reported.
Observation and evaluation of the following criteria can help to determine whether ‘something is wrong’ with the animals, and, considering the overall pattern of observations, can help decide strategies for rectifying any health and welfare problems.
6.1.1 Observation of spontaneous and provoked animal behaviour
a) Feeding. The common laboratory species of cephalopod have voracious appetites and eat relatively large amounts of food in relation to body weight, reflecting a relatively high metabolic rate. Therefore, altered feeding behaviour manifests as a reluctance to take food or an increase in the time to attack or subdue live prey, and is usually the first and most apparent behavioural indicator that there could be a health or welfare problem. For example, in octopuses fed on crabs or mussels, the tank should be checked for empty carapaces and shells to ensure the animals are ingesting prey and not just attacking them. In O. vulgaris the willingness to attack can be tested equally well with an artificial crab as with a live crab.
179
b) Location in the tank and water column. Each species normally locates in a characteristic place in the tank and water column. Nautiluses spend much of their time attached to the sides of tanks, and undergo daily vertical migrations (see above), so animals spending large amounts of time at the bottom of the tank other than when engaged in feeding are exhibiting abnormal behaviour. Cuttlefish alternate between hovering/swimming in the water column and resting on the bottom, partially covered by the substrate, and an animal spending a considerable period of time at or near the surface of the tank should be inspected closely for signs of physical damage to the mantle. Such changes have been observed following a pharmacological treatment, as described by Agin et al.,
180
as a consequence of cycloheximide injections. Squid rarely rest on the bottom of the tank, so their presence there for an extended period should be regarded as abnormal, as should extended periods spent at the surface. For octopuses, a ‘problem’ in the tank may be indicated if the animals spend excessive time clinging to the lid of the tank with most of the body out of the water. c) Swimming and locomotor activity and coordination. Each species has a characteristic method of moving in the tank: by walking, swimming or a combination of both.
31
Repetitive locomotion in cephalopods, such as jetting backwards continuously, or in the case of octopuses performing swimming motions while attached to a tank wall, can be a sign of stress. Any abnormalities of coordination should be noted. For example, a defect in the statocyst leads to an inability to control orientation during swimming. This ‘spinner’ behaviour has been reported in species of cuttlefish, squid and octopus.
181
Although there are likely to be minute-to-minute changes in the level of locomotor activity (see e.g. Figure 1 in Boyle
182
), each species has its own overall daily pattern of activity cued by the photoperiod.98,100,130,183–186 Rest/sleep-like–activity cycles are documented in S. officinalis,
102
O. vulgaris
101
and Octopus macropus;
103
nocturnal vertical migration is known to occur in N. pompilius.29,177 Changes could be an indication that ‘something is wrong’. d) Use of arms and tentacles. The behavioural repertoire of arm movements in cephalopod species is reviewed and described in Borrelli et al.
31
A taxonomy of arm movements for octopuses is provided by Mather.
187
An animal spending an extended amount of time with the arms curled over the body (a defensive posture), either in the den or the corner of the tank, should be monitored for other indications of distress. In Nautilus withdrawal of all tentacles into the shell with the opening obstructed by the hood is a defensive behaviour.
188
There are scattered reports of octopus using an arm to ‘guard’ an injured part of the body (I. Gleadall personal observation cited in;
8
reports of animals ‘guarding’ the mantle or cranium post-surgery by G. Fiorito, unpublished data, and also
189
), but this behaviour has not been systematically investigated and could also be linked to facilitation of healing by secreted antimicrobial peptides. Wound-directed behaviour was not observed in a study of tentacle amputation in the squid Loligo pealeii,
9
although in two species of cuttlefish (S. officinalis and S. pharaonis), the use of a partially (80–90%) amputated arm for prey manipulation and body posturing was avoided for up to 3 days post lesion.
190
In cuttlefish, reaching over the dorsal mantle (‘scratching-like’ behaviour) has been observed a few days after a transient rise in ammonia concentration leading to skin damage.
145
Tentacles in cuttlefish and squid should be retracted except when the animal is engaged in an attack. The arms are used for skin cleaning in octopus; excessive cleaning activity and/or frequent presence of sucker-rings in the tank could be indicative of abnormality. In all cephalopods, a loss of adhesion in the suckers should be a cause for concern. e) Interactions with humans and conspecifics. While cephalopods, in general, are very responsive to any novel features introduced in their tank, octopuses are especially curious about their environment. Healthy octopuses acclimatised to laboratory housing will often leave their den when the tank is inspected and will interact with a hand placed below the surface of the water (for an historical account see
191
).36,192 Reluctance to interact with humans should be a cause for concern. It should also be noted that there is some evidence that at least one species of octopus (E. dofleini) may recognise individual humans;
193
therefore, care should be taken to ensure that staff who are involved in any procedure likely to be aversive should not be involved in routine feeding or inspection as there is a possibility of inducing a conditioned aversion/avoidance. If visually exposed to conspecifics, octopuses may alter their predatory response due to agonistic interactions, but habituation resulting in a resumption of normal behaviour has been observed under controlled laboratory conditions.194,195 Squid and cuttlefish are social species and changes in social interactions with conspecifics again may indicate a welfare/health problem. f) Squirting, inking, defaecation and regurgitation. Squirting: all cephalopods use expulsion of water from the mantle via the siphon in breathing and locomotion; and this is particularly noticeable in the jetting escape reaction. Squid and cuttlefish may direct jets of water at a person attempting to capture them, and this behaviour is particularly notable in O. vulgaris where jetting is also a component of the deimatic display.31,134 Water jets directed at an observer is indicative of a moderate aversive reaction; in some cases this can also be a sign of ‘recognition’.
195
Inking: is a defensive response in cephalopods (apart from nautiloids which do not have an ink sac), so inking should always be taken as an indication that the animal perceives a threat or is stressed. However, there is individual variability in the threshold for induction of inking as some O. vulgaris will ink profusely in response to handling (M.G. Valentino and P. Andrews, unpublished data) that does not evoke the same response in other individuals (G. Fiorito, pers. comm.). In addition, inking does not necessarily result from the animal receiving a presumed noxious stimulus, such as an electric shock (I. Gleadall personal observation cited in;
8
G. Fiorito, pers. comm.). Therefore, absence of inking should not be interpreted as an absence of anxiety or distress. A continuous trickle of ink from the animal should be investigated as it may indicate a problem with the neural control mechanism or with ink duct sphincter competence. Animals should not be allowed to remain in a closed tank in which inking has occurred, and care should be taken to ensure that ink does not enter other tanks as it is an alarm signal.
196
Intramantle inking has been reported as a post-transport stress behaviour in Octopus bimaculoides.
197
Defaecation: there is insufficient knowledge of defaecation patterns and their control in cephalopods to determing whether any changes may be linked to pain, anxiety or stress, as is the case in many vertebrates. Although faecal ropes may emerge in octopus exposed to general anaesthetics, this could be due to loss of anal sphincter control. The production of faecal ropes in octopus is a useful indicator of normal digestive tract functioning in a feeding animal, but it is not known whether disease can alter the faecal fluid content or defaecation frequency (constipation/diarrhoea). However, chemical and cytological examination of fresh faecal samples can provide important insights into the health of the animal and as collecting faecal samples is non-invasive, its utility in cephalopod health monitoring should be explored. Regurgitation: there are two isolated observational reports of regurgitation/vomiting of upper digestive tract contents one in E. dofleini (I. Gleadall personal observation cited in
8
) and the other in Sepioteuthis sepioidea.
198
The location of the beak within the crown of arms would make this behaviour very difficult to detect. However, if the ability to regurgitate/vomit upper digestive tract contents is confirmed then it should be added to the list of possible indicators of illness, as is the case in vertebrates.
199
6.1.2 Appearance
a) Skin colour, pattern and texture. Skin colour and pattern are primarily regulated by motorneurones from the suboesophageal chromatophore lobes of the brain, with contributions from reflecting cells, depending upon the location on the body and the species.134,200–202 At the time of writing, there is no evidence to show that changes in the colour or pattern of the skin in any cephalopod species are specifically associated with changes in health or welfare of the animals. Oestmann and coworkers
145
caution that normally functioning chromatophores and iridiocytes may mask underlying skin defects. In Nautilus discoloration of the mantle (with loss of buoyancy) is a sign of poor health.
203
However, loss of ability to match substrate or background (see e.g.204–207) or sustained pallor with loss of normal patterning should be taken as an indication of a problem, as should excessive or inappropriate flashing in squid
208
and wandering clouds in coleoid species.
209
Note should also be taken of colour changes in response to a provocative stimulus, such as the deimantic display often observed in response to a perceived threat. Skin texture in octopuses and cuttlefish can be changed by the formation of papillae, particularly prominent above the eyes and on the mantle and is indicative of an aroused or vigilant animal.
31
b) Skin and external shell integrity. Any breach to the skin of a cephalopod is potentially problematic because of the possibility of bacterial infection (section 7.2.2) causing systemic sepsis, preventing healing, and local inflammation causing hyperalgesia. Bleeding from wounds may not be readily apparent as, although oxygenated haemolymph is pale blue (extracellular haemocyanin), it will be rapidly diluted in the tank and deoxygenated haemolymph is colourless. Healing of small wounds in octopuses, such as transection of the distal 10% of the length of the arm, appears to be rapid, with the exposed area in some animals being almost completely covered by skin in about 24 hours (T. Shaw and P. Andrews, unpublished data), but larger wounds and particularly those to the mantle appear to take longer to heal even without infection.
210
Damage to the skin most frequently occurs at the distal part of the mantle in animals (particularly squid and cuttlefish) that frequently impact the wall of the tank (‘butt-burn’: J. Rundle, pers. comm.), with four impacts per hour being recorded in a study investigating the long-term health of cultivated cuttlefish in soft-sided tanks.
211
In cuttlefish and octopus, the ventral surface of the mantle contacting the substrate should also be examined, and anecdotal evidence that ill or senescent octopuses avoid rough substrates may indicate that the skin in this region is particularly sensitive to damage. Animals showing signs of healed skin damage should be inspected closely to ensure that it is healing, as death may ensue rapidly if the lesion increases in size and penetrates the underlying muscle.
212
Breaks in the shell of nautiloids may compromise their buoyancy mechanism and so require treatment. The cause of any breach in skin or shell occurring after an animal’s arrival in the laboratory should be identified and action taken to prevent recurrence (e.g. carefully examine the tank and items in it for sharp edges, deep clean and disinfect the tanks, see section 5.6). c) Eyes. The eyes should be inspected to ensure that the cornea and lens are transparent, as opacity is one of the signs of natural senescence (see below). Pupil diameter should decrease over a few seconds in response to a sudden increase in illumination (for Nautilus see;
213
for cuttlefish and octopus;
214
for squid
215
), although there is some evidence (S. officinalis and E. cirrhosa) that the response is not consensual.
214
The classic contributions of Beer
216
and Magnus
217
should be also considered in this framework. The slit-like pupil remains close to horizontal irrespective of the position of the body, and this is particularly noticeable in octopus.
150
Both the pupil diameter and statocyst–ocular responses (nystagmus) are mediated by the brain and hence give an insight into central nervous system functionality.181,218–220 d) Body posture. Two aspects need to be considered: i. the relationship between the mantle, head and arms/tentacles (i.e. the overall appearance of the animal), and ii. the orientation of the animal in relation to the floor and sides of the tank. In Nautilus the shell should be vertical; however, air bubbles can become trapped in the eyes and under the hood, leading to adverse health effects.221,222 Information on treatments related to poor health conditions is available in Barord et al.
223
Trapped air can be released by slowly turning the animal, laterally from side to side. All cephalopods have well-developed statocyst systems to maintain body posture and coordinate body position with eye orientation
224
so an abnormal body posture may indicate a nervous system problem or a physical defect that the animal cannot compensate for (e.g. a broken cuttlebone, a fluid-filled chamber in Nautilus, gas trapped in the distal mantle of octopus). Damage to arms can also affect posture as Tressler et al.
190
reported unbalanced swimming (body axis tilted to the lesioned side) lasting up to 3 days in cuttlefish in which 80–90% of the length of third right arm was removed. The head in octopuses is particularly mobile and a raised head, particularly if moving from side to side or bobbing has been regarded as sign of ‘agitation’ in O. vulgaris by Boyle.
182
However, head bobbing and similar behavioural patterns are indicative of increased arousal, as reviewed by Borrelli and coworkers.
31
6.1.3 Biomarkers
a) Body weight. The optimum frequency with which animals can be handled for routine weighing, taking into account that anaesthesia may be necessary, is not known. It has been suggested
225
that frequent handling may impede growth, but this requires systematic investigation. Cephalopods, particularly when young, increase body weight daily (assuming sufficient food) so failure to increase weight or a loss of weight following an experimental procedure may be the earliest objective measurement of declining health or welfare, but the potential additional harms of frequent weighing will need to be assessed. Dorsal mantle length (DML) is also frequently used as an index of body size in cephalopods although the relationship to body weight (TBW) is not linear. The K-Fulton condition index, which combines length and body weight measurement and is used in fish, has been adapted for cephalopods (K = (TBW/DML3) × 100).
226
In O. vulgaris infected with Aggregata octopiana, the K-Fulton condition index decreased as the sporocyst counts in the caecum increased.
226
Consideration should be given to using this index as part of routine growth and health and welfare monitoring in cephalopods, as applied to many species of fish,227–233 and in other circumstances to other vertebrates (see, e.g.234,235). In stock animals, weekly measurement of body weight may also be a useful index of health and welfare status, providing this can be done with minimal distress to the animal (e.g. in seawater), but the percentage loss of weight over time that is indicative of illness is not known. The digestive gland has a lipid reserve;
236
Mangold and Bidder estimated as 9–13% of digestive gland weight in S. officinalis.
148
However, the impact of food deprivation on this is not known. Growth data based upon body weight are species and laboratory specific with the latter depending upon food type and feeding frequency, water temperature, stocking density, animal age, activity level (influenced by tank size and photoperiod) and parasite load. There are limited growth curve data for representative cephalopod species; for example for: Nautilus,
237
cuttlefish and squid,
238
and octopus.225,239 b) Ventilation (breathing) frequency. Ventilation frequency can be monitored by an observer provided that the animal is not disturbed, but a video system may provide an alternative in the absence of other non-invasive methodology, as standardised by Borrelli.
26
Although an increase in ventilation frequency may be an indication of physiological stress (e.g. particularly a fall in inspired pO284,240,241), it is also indicative of arousal to innocuous stimuli and ventilation frequency also correlates positively with activity levels, as for S. officinalis and O. vulgaris.182,242 Observation of breathing should also note whether the pattern is even or is interspersed with periods of apnoea/tachypnoea. Although frequency is relatively easy to monitor,
26
some assessment should also be made of depth (mantle stroke volume) as again excessively deep, shallow or laboured breathing may also indicate a problem and Smith et al. comment that maintenance of oxygen uptake (in O. vulgaris) relies more on stroke volume than increasing ventilation frequency.
240
Deep/forceful breathing may manifest as currents in the water or ripples in the surface if the animal is close. In the coleoids, the way in which the mantle distends during inspiration should be noted to ensure it is bilaterally symmetrical, that the entire mantle is involved, and inspiration and expiration are coordinated with the closing and opening of the siphon respectively. During general anaesthesia, ventilation frequency, depth and coordination all become suppressed.
243
Therefore, similar changes in a non-anaesthetised animal are likely to reflect depression of brain drive and should be investigated immediately. Stress state in Nautilus is usually expressed as a ‘rocking behaviour’ (which reflects hyperventilation), with the animal clearly rocking from front to back. c) Cardiovascular parameters. Currently there is no established non-invasive method for routine measurement of heart rate or blood pressure in an unanaesthetised cephalopod (but see244,245). In addition, studies of animals with indwelling catheters show that both parameters are very labile
150
suggesting that, even with appropriate methodology, they may not be helpful as indices of health or welfare. In particularly compliant individual O. vulgaris, it is possible to observe the beating of the systemic and branchial hearts in the mantle without anaesthesia, and to use Doppler ultrasound to investigate cardiac function and image the viscera (G. Ponte, pers. comm.). The resolution achieved is much improved from previous attempts with cephalopods.244,246–248 This potentially represents a revolution for future physiological studies with these animals (D. Fuchs and G. Ponte, unpublished data; Vevo 2100 Visualsonics, The Netherlands). Such methodology may be suitable for detailed investigation of animals showing signs of illness and could be useful for monitoring some physiological functions during general anaesthesia. d) Other biomarkers: analysis of blood. There are no validated blood biomarkers indicative of the health or welfare status of a cephalopod, and the development of such markers is hampered by difficulty in obtaining blood samples using minimally invasive techniques comparable to those available for vertebrates (but see also Table 8). Descriptions of blood sampling in the literature employ some form of general anaesthesia as, for example, done in the bobtail squid E. scolopes.
249
In animals sedated for investigation using a low concentration of a general anaesthetic (see section 8.5.5), blood sampling and analysis may be a helpful aid to diagnosis and treatment but the relative harms and benefits of undertaking this procedure solely for welfare assessment need to be considered.
The following parameters should be considered.
Haemocytes. Blood samples enable culture for bacteria and examination of smears by electron microscopy for viruses. The utility of haemocyte counts and morphology in general and phagocytes specifically as indicators of infection or stress is limited because sampling methods (especially repeated sampling involving anaesthesia) themselves seem to increase haemocyte concentration,250,251 although the concentration is increased further by bacterial infection the effect is transient (i.e. present at 4 but not 24 hours in E. cirrhosa). 126 The increase in haemocyte counts in response to intramuscular injection of Escherichia coli lipopolysaccharides in O. vulgaris begins within 4 hours of injection and is returning to control levels by 24 hours. 251 Vehicle injection (phosphate buffered saline) produced a smaller increase in haemocytes indicating that a rise in haemocytes may be a useful indicator of generalised stress as well as of infection.
Chemistry. Levels of the respiratory pigment haemocyanin can be measured by the haemolymph copper concentration. 252 Routine measurements of the common inorganic ions (e.g. Na+, K+, Ca++, Mg++, Cl− and SO4++) and protein253–257 would be helpful in establishing their utility as parameters for diagnosing disease. For example, in O. vulgaris a decrease in most inorganic elements and/or in haemocyte concentrations is observed when infection by the gastrointestinal parasite A. octopiana increases. 258 Finally, measuring activity levels of respiratory enzymes and total protein concentration may provide additional information on the health status of an animal.259,260
Humoral agents. Plasma noradrenaline and dopamine increase transiently (5 min) in response to stress induced by air exposure in E. cirrhosa.
255
However, it is difficult to envisage how such transient changes could be applied in routine health monitoring. Circulating levels of a number of hormones may give insights into health (e.g. steroids) and reproductive status, but normal ranges need to be established before these could be useful for health and welfare monitoring. Other molecules that may be of relevance for health monitoring include complement system molecules, anti-microbial peptides (AMPs) and other innate immunity-related proteins released by haemocytes, as they increase rapidly in infectious disease.261–266
e) Other biomarkers: analysis of faeces. One of the most promising methods to evaluate the physiological conditions of animals in laboratory settings is through the examination of faeces. Samples may be utilised to estimate various biomarkers including steroids (e.g. corticosteroids, estrogens, testosterone
267
), as well as to evaluate, for example, the digestibility of alternative diets as in the case of fish,267–270 or to identify possible parasite infections or cytological indicators of intestinal damage.
271
We recommend the development of faecal analysis methods to assist in evaluation of health and welfare of cephalopods in laboratory settings.
6.2 Health and welfare of ageing cephalopods: a special case?
It is very difficult to determine the age of living cephalopods. Age is a parameter that is known for almost all other species used in research and should be included in the methods section of published papers (see ARRIVE Guidelines 272 ), but which is rarely known in studies of cephalopods, unless they are laboratory reared.
Within a wild-caught population of a particular cephalopod species, in a circumscribed location and time of year, cephalopods of higher body weight are likely to be older, but the relationship between body weight and age is not linear, particularly in octopuses.238,273,274 The absence of precise age data complicates experimental design.
While there are variations due to the ecological niche of individual species, cephalopods generally live for about a year. With the exception of nautiloids, cephalopods undergo an exponential early growth phase during which they mature to adult size rapidly. However, this growth phase can be influenced by many factors, such as temperature, food availability and space, which makes body size a poor indicator of an animal’s age.238,274–276 The age of sexual maturity is variable and also appears to depend on the ecological niche of the species. As reviewed by Rocha et al., some cephalopods (e.g. Loligo opalescens among squid and O. vulgaris among octopods) are semelparous (i.e. breed and then die soon after) while others (e.g. Nautilus sp., S. officinalis among cuttlefishes, L. vulgaris among squid and Octopus chierchiae among octopods) are iteroparous (i.e. breed multiple times, generally with longer lifespan). 277 For a summary of reproductive strategies of some cephalopods species refer to Appendix 3.
In light of these considerations, ageing is relevant in the context of physical senescence (i.e. ageing changes in animals over time/after breeding, especially in females, once their eggs have hatched), but also when experimental procedures are applied to animals and age could influence the results.
Possible signs of cephalopods in senescence include reduced/absent drive to eat, poor skin quality, cloudy eyes, and changed activity pattern and behaviour.278–280 It may be difficult to distinguish this state from an animal that is showing similar signs due to disease. Good record keeping of time kept in the laboratory and age whenever possible, alongside general health records of individuals may help to differentiate the two situations.
It is unknown whether cephalopods experience any form of pain or suffering during senescence, but the precautionary principle should be applied when determining humane end-points (see section 8.3) for studies involving senescent cephalopods.
The senescent state makes animals more susceptible to a number of problems which, if they occurred in non-senescent animals, would be regarded as indicators of illness.145,278,280 These include: skin breaches including ulceration; primary and secondary cutaneous (e.g. Aeromonas sp., Vibrio sp. and Staphylococcus sp) and systemic (e.g. Flexobacter, Vibrio) bacterial or fungal (e.g. Labyrinthula sp., Cladosporium sp.) infections; increased parasite load.281,282
The senescent animals are not only more susceptible to infections, particularly of skin, 283 but they also appear to have a reduced ability to recover once infected.
In general, animals showing signs of senescence should be humanely killed, unless there is sound scientific or animal welfare justification for keeping them alive.
A discussion of ethical aspects of both caring for and using senescent cephalopods in research is available in Smith et al. 3
Careful routine monitoring of the physical condition of captive cephalopods at all life stages is essential for their proper care.
6.3 Post-mortem evaluation
Post-mortem evaluation of cephalopods is often a neglected aspect of health and welfare monitoring. It enables thorough inspection, revealing abnormalities not readily visible when the animal was living, the cause of death can be confirmed or ascertained, histological samples collected, and a database of findings can be gathered to support future post mortem evaluations.
The overall aim of such detailed evaluation is to facilitate better health and welfare assessments, and implementation of humane end-points, in future studies. Tissues such as the beak, statoliths and vestigial shells can also be collected, which may provide information on the age of the animal (for example for: O. vulgaris;273,284 Sepioteuthis lessoniana;285,286 other cephalopod species287–300).
Cephalopod tissues are rich in protease enzymes, which cause rapid tissue autolysis post mortem.301,302 Autopsies should be performed immediately after humane killing an animal, for example, on welfare grounds when humane end-points have been reached, or at the end of a novel procedure and/or when the cause of welfare effects is uncertain; or as soon as an animal is found dead (see section 8.11), but only once death is confirmed (see section 8.12).
Cephalopods do not exhibit post mortem rigidity so rigor mortis cannot be used to confirm death, and other methods need to be employed (see section 8.12).
Autopsy findings should be reported in the first instance to the person responsible for overseeing the welfare and care of the animals, and any actions needed to safeguard animal welfare in future should be agreed, recorded and implemented.
Information on likely cause of death may be required for consideration by the local Animal Welfare Body or the National Competent Authority, especially if there is unexpected mortality following a procedure but note that mortality should never be used as an end-point for a procedure.
Steps to be considered for inclusion in post mortem evaluations include:
Haemolymph sampling: when animals are humanely killed for welfare reasons, it will be possible to collect haemolymph immediately surgical anaesthesia is achieved, but before death ensues (see section 8.5.5 for techniques). A bacterial septicaemia is suspected when the haemocytes have aggregated into visible clumps. Systemic bacterial infections should be confirmed by bacterial culture of the haemolymph.
Skin examination: external lesions should be blotted to remove excess mucus, then, aseptically, samples obtained using swabs and submitted for bacterial culture. Smears obtained from the swab or skin scrapings should be air dried and stained for bacteria or fungi.
Anatomical examination: descriptions of the gross internal anatomy of the main cephalopod species can be found in the following classic references: N. pompilius; 303 S. officinalis; 304 L. vulgaris; 305 O. vulgaris; 306 E. cirrhosa. 307
In brief, following a detailed external inspection including skin breaches, abnormal colouration, damage to appendages and deformities the mantle cavity is opened by an incision, following the anatomical approach that gives best accessibility in each species.
The viscera are examined visually and particular note taken of the state of the hearts, gills and the digestive gland (hepatopancreas), which is the largest and the main metabolic organ. Organs should be inspected for abnormal colour (particularly hepatopancreas), shape, size, texture (e.g. oedema, hard lump caused by a cyst or tumour), and presence of parasites (particularly intestine) or foreign bodies.
The presence, or not, of food (digested/undigested) in the crop, stomach and caecum/intestine should be noted as well as faecal ropes in the rectum to assess gastrointestinal tract functionality. Digestive tract samples should be analysed for the presence of parasites.
The degree of filling of the ink sac should be noted as an empty one may indicate that the animal has inked profusely in the tank prior to death, which might not otherwise be apparent if the animal was found dead in a tank with circulating seawater.
Haemorrhage is impossible to detect as the blood is colourless when deoxygenated, and oedema may be hard to detect without histology.
Tissue samples can be fixed by immersion in neutral-buffered 10% formalin, and standard histo-pathological techniques applied although fixation in buffered glutaraldehyde will be required for ultrastructural studies and for identification of viruses.
Creation of a repository of data and/or reports on cephalopod pathology would provide an important resource in the effort to ensure good health and welfare in captive cephalopods used in laboratories. This is a current project of the non-profit Association for Cephalopod Research (see www.cephalopodresearch.org/projects), which is also included as goal of the COST Action FA1301 (CephsInAction; http://www.cost.eu/COST_Actions/fa/Actions/FA1301; www.cephsinaction.org).
7. Disease: causes, prevention and treatment
The major known risks to health and welfare in cephalopods are environmental parameters, and especially water quality issues, physical injury and infection (especially due to parasites), all of which may be interrelated. In addition, the psychological well-being of the animals should be considered (for a preliminary discussion see:308,309), as presented in section 6.
7.1 Introduction to the issues related to diseases of cephalopods
7.1.1 Environmental influences on disease
Host-pathogen interactions can be strongly influenced by the environment in which an animal lives. In addition, stressful conditions deriving from inadequate physico-chemical parameters (e.g. increased or decreased temperature, presence of undesirable chemicals, low oxygen saturation) may result in impaired defense responses against pathogens (review in 310 ), thus increasing the probability of a disease outbreak.
Maintenance of water quality is essential for minimising infectious diseases and tank design (including enrichment) is important for avoiding self-induced physical trauma (e.g. ‘butt-burn’, see 6.1.2b above) and reducing general ‘stress’.
Close attention to these factors should make animals more resistant to infection as well as improving psychological well-being.
7.1.2 Effects of physical trauma
Injuries may be inflicted by some methods of capture (e.g. tentacle loss by squid jig; 311 see also 312 ) and handling without appropriate care (e.g. skin damage by net reported in 313 ), or during transport as, for example, for O. vulgaris that may fight if transported together and eventually bite or cannibalise each other (see description of fighting in 31 ).
Skin damage makes the animal susceptible to secondary infections (particularly bacterial) which can be fatal if untreated. 314
7.1.3 Disease caused by feeding live food
Although feeding with live food may be preferred to an artificial diet (see section 4.2), it is important to avoid the use of species that are recognised for their role as hosts of important pathogenetic parasites. For example, coccidian Aggregata sp.258,315,316 (for review see 73 ) or viruses317–319 may infect cephalopods through food items such as crustaceans. In the case of the use of crabs as a prey item, special attention should be given to distinguish between those carrying parasites and to remove them from cephalopod facilities.
It is also noteworthy that penetrative injury to the brain has been reported to occur in O. vulgaris 320 due to the setae of ingested polychaete scaleworms (Herminone hystrix) so their presence in laboratory tanks should also be avoided.
See also section 7.2 below for discussion on infectious diseases of cephalopods.
7.1.4 Action points when disease is suspected
Animals showing signs of illness should be placed in quarantine (see section 3.6) to reduce the potential spread of the causal agent, and any animals found dead removed and autopsied (see section 6.3). Water from tanks of quarantined animals should not contaminate water supplies to other animals or the environment, and personnel handling potentially infectious animals should wear protective clothing (see section 9). Seriously ill animals not responding to treatment (where treatment is possible) should be humanely killed (see section 8.11) and autopsied immediately (see section 6.3).
Investigation of disease outbreaks should not only include identification of the immediate cause (e.g. infectious agent), but also underlying origin such as adverse water quality, contamination of food or effects caused by other animals. Failure to correct such factors will often result in further outbreak of disease.
Careful records must be kept of all occurrences of illness or mortality irrespective of identification of cause, so that patterns can be identified over time (e.g. there might be higher mortality/infection rates from certain suppliers).
7.2 Infectious agents in cephalopods
Immunity in cephalopods differs from vertebrates due to the absence of an adaptive immune response.321,322 However, these animals do have an innate (non-specific) immune response, mediated by both humoral (e.g. haemagglutinin) and cellular (haemocyte) mechanisms.73,252,323–325
As in other molluscs, circulating haemocytes are responsible for infiltration, aggregation, encapsulation, cytotoxic reactions and phagocytosis of foreign particles. Cowden and Curtis estimated that the phagocytic capacity of octopus haemocytes was low; 326 while high phagocytosis of carbon particles has been described in E. cirrhosa. 327 Phagocytic capacities of the haemocytes of the common octopus, O. vulgaris, challenged in vitro using zymosan as a test particle, 325 and those of the haemocytes of E. dofleini (see citations in 328 ) have been reported. Recently, an extensive analysis of octopus haemocytes at morphological, flow cytometry and functional level (including phagocytic capability as well as reactive oxygen species (ROS) and nitric oxide production) after challenging with different stimuli has has been carried out by C. Gestal and coworkers.73,329 In addition, several biologically active molecules likely to be involved in responses to infection and injury are known to be present in the haemolymph of cephalopods, such as lectins, proteinases, including antiprotease and lysozyme activities.126,266,330–332
The immunobiological system in cephalopods is quite effective, as reflected by the scarce reporting of illness in captivity for this class over many years, but this low incidence could also reflect under-diagnosis, particularly of systemic disease that may not have an external manifestation, or under-reporting.
7.2.1 Viruses
Viruses are the most abundant component of aquatic microbial communities (for review refer to333–335). However, there are few records of virus-inducing pathologies in cephalopods as reviewed in Hanlon and Forsythe. 336 The first known evidence of viral infections in cephalopods was provided in specimens of O. vulgaris and in the cuttlefish S. officinalis.337,338 In the octopus, oedematous, modular tumors embedded in arm musculature and tissue degeneration were observed in animals showing anorexia, apathy and often autophagy.337,339 The lesions were linked to the presence of viral particles suggested to belong to the group of iridovirus, according to their size, morphology and location. 340 In S. officinalis, virus-like particles were identified in the gastric epithelium and described as similar to reoviruses of vertebrates, but details on the symptoms induced are not provided. 338 Virus-like particles have been also reported in the epithelial cells of the tubules of the digestive gland of Loligo pealei, and in the renal appendages of several octopod species. 336
More recently, Gregory and coworkers 341 reported another possible infection of iridovirus in cephalopods (i.e. Nautilus sp.). Intracytoplasmic inclusion bodies were observed in tissues from an animal found dead in aquarium without premonitory signs of disease.
Furthermore, Todarodes pacificus (utilised as a food item in aquaculture) have been reported to be positive for Betanodavirus, 342 which is the aetiologic agent of a serious viral disease known as VER (viral encephalopathy and retinopathy) that has been detected in a wide range of vertebrate and invertebrate hosts worldwide and caused severe mass mortalities in both farmed and wild marine organisms. 343 Betanodavirus was also identified in skin lesions, in the eye and in the branchial heart of O. vulgaris. 344 Squid have been suggested also as possible vectors of zoonotic viral agents such as Norovirus.345–347
Infections from viruses may sometimes be asymptomatic (see examples from fishes:348,349) but pathogenicity may be higher if temperature increase, as in the case of global environmental changes. 350 In fishes, symptoms of viral infection may include clouding of the eye, anorexia, changes in body colour and uncoordinated swimming.
7.2.2 Bacteria
Secondary bacterial infections in skin lesions have been reported in squid, 355 cuttlefish 356 and octopus, 357 and skin lesions are considered to be the most common conditions in which infections occur. 351 Bacterial infections may spread to conspecifics sharing the tank. 77 In addition, bacteria may cause skin ulcers on mantle, head and arms, hyperplasia of the epidermis and increased mucus production (e.g. in Lolliguncula brevis; 355 in O. joubini and O. briareus; 357 for review see336,358).
While infections occurring on the skin are most commonly reported, they are not the only tissues susceptible to bacterial infection, since Rickettsiales-like organisms have been found in the gills of laboratory reared O. vulgaris, observed as basophilic intracytoplasmatic microcolonies within epithelial cells, on which they cause hypertrophia and occasionally necrosis. No significant harm has been observed in the host, but under conditions of stress or intensive husbandry, it has been suggested that these bacteria may have a detrimental effect on the host’s respiratory gaseous exchange although this has not been shown experimentally. 359
Gram-negative bacteria Vibrio lentus have been also identified in the branchial heart of wild O. vulgaris and reported to induce mortality in 50% of octopuses in the first six hours, with lesions showing a typical round pattern on the arms or head. 360
Finally, cloudy-to-opaque corneal tissue as well as opaque lenses in Loligo forbesi and S. lessoniana have been reported due to infection with Gram-positive bacteria (Microccocus sp.) found in the vitreous-induced swelling of the infected eye and causing opacity of the cornea. 361
7.2.3 Fungi
Reports of fungal infections in cephalopods are scarce and mostly relate to eggs and embryos. Hanlon and Forsythe 336 refer to infection by Labyrinthula sp. in adult O. vulgaris; in these animals grey patches of inactivated chromatophores appeared followed by progressively larger and whiter patches in which the entire epidermis and dermis was missing. Thraustochytrid and labyrinthulid fungi have also been isolated from skin lesions in E. cirrhosa, but it is not clear whether these organisms are causal agents or secondary infections. 362
Harms et al. 282 reported a case of mycotic infection in adult captive cuttlefish showing skin lesions in the dorsal mantle. Cytology revealed hemocyte granulomas surrounding fungal hyphae, and culture yelded Cladosporium sp. Infection from the same organism was also reported by Scimeca and Oestmann (1985, cited in 351 ) in octopus, while Fusarium sp. has been found infecting the chambered nautilus, N. pompilius. 351
7.2.4 Parasites
Most wild cephalopods host parasites include protozoans, dicyemids and metazoans. Generally, these are found in skin, gills, digestive tract, digestive gland and kidneys.73,363–370
Among the protozoans, one of the main parasites infecting both wild and cultured cephalopods is the gastrointestinal coccidian of the genus Aggregata, which produces severe disease in cuttlefishes and octopuses, by causing a malabsorption syndrome, diminishing nutrient absorption and reducing the immune response capability.226,258 In addition, the parasite may produce behavioural alterations in the infected host inducing excitation, impaired ability to camouflage and aggressive behaviour. 316 Mortality has been attributed to the infection, and it has been reported that a few days before dying an infected octopus became inactive in the shelters and unresponsive to stimuli. 316
Dicyemids are endosymbionts that inhabit the renal sacs of cephalopods including cuttlefish, loliginid squid and octopuses.370–372 No host damage has been reported due to dicyemids, but a possible contribution to ammonium ion elimination from the host urine has been proposed 370 . However, dicyemids could be a problem if the parasite load is elevated enough to cause physical obstruction of the renal sac.
Cephalopods are intermediate or parathenic hosts for a variety of metazoan parasites, namely trematodes, digenea, cestodes and nematodes transmitted via the food chain.370,373
Amongst these, one of the most abundant and frequent parasites are anisakid nematodes, which have been reported to cause important pathological effects in several cephalopod host species.374–376 Larvae of Anisakis simplex are pathogenic to humans when raw, under-cooked or lightly marinated fish or squid are ingested. 377 Crustaceans, such as copepods and isopods, also parasitise the gills and mantle cavity of cephalopods, affecting the body condition of the host. 378
Apart from the potentially pathogenic organisms (e.g. Vibrio, Aeromonas, Pseudomonas and Flavobacterium sp.; ciliates and dicyemids), none of these diverse organisms is known to cause severe health problems in captive cephalopods. However, as some of the mechanisms exploited by parasites to produce changes in host’s behaviour would seem to be highly conserved throughout the evolution of both vertebrates and invertebrates,379–382 it would be unwise to exclude such interactions in cephalopods without specific research.
7.3 Antibiotic treatment of infectious diseases
Substances given to some cephalopods to treat infection, but not recommended for routine use in laboratory facilities; see text for details. The table summarises for each species the treatment (i.e. dosage, route and duration) and substances tested. All drugs included in this table belong to the class of pharmaceuticals utilised as antibacterial agents, unless otherwise stated. The studies cited refer to treatment of infections, with the exception of Gore et al. 530 who investigated a pharmacokinetics. For a short discussion see also Scimeca 351 and Forsythe et al. 358 Therapeutic interventions should be discussed with a veterinarian.
Administration route: PO, per os (i.e. provided through food items); Imm, immersion in a solution; IM, intramuscular injection; IV, intravenous injection.
Antiprotozoal agent.
Despite published evidence, caution should be applied when using oral or parental routes of administration since these are stressful for animals and may be difficult to perform safely.
In any case, prophylactic use of antibiotics is not recommended because of the risk of promoting bacterial and fungal resistance, masking infection and allowing secondary infection. It should not be used to ‘prop up’ poor tank hygiene.
Cephalopods used for scientific purposes should be maintained free from infections and contact with infection sources avoided. To this end, the use of high-performing filtration systems (i.e. combining mechanical, biological and physical filtrations) is highly recommended in combination with careful screening of animals entering the facility and efficient quarantine procedures. The importance of tank design in minimising the potential for skin damage and subsequent increased probability of infection should not be overlooked.
8. Scientific procedures, severity and harm-benefit assessment, anaesthesia and humane killing
8.1 Definition of a ‘procedure’
Directive 2010/63/EU defines a regulated ‘procedure’ as, ‘Any use, invasive or non-invasive, of an animal [e.g. living cephalopod] for experimental or other scientific purposes, with known or unknown outcome, or education purposes, which may cause the animal a level of pain, suffering or lasting harm equivalent to, or higher than, that caused by the introduction of a needle in accordance with good veterinary practice’.
Selected examples of research in cephalopods which involves an intervention that it is considered would come within the definition of a procedure (see also section 8 of this work) within the Directive. Studies published recently have been selected where possible to show that the Directive will impact on current research areas. It should be noted that non-surgical interventions that may induce PSDLH fall within the definition of a procedure. The fact that a particular technique has been used in a previously published study does not guarantee that the same technique would now be permitted by the national competent authority under the Directive.
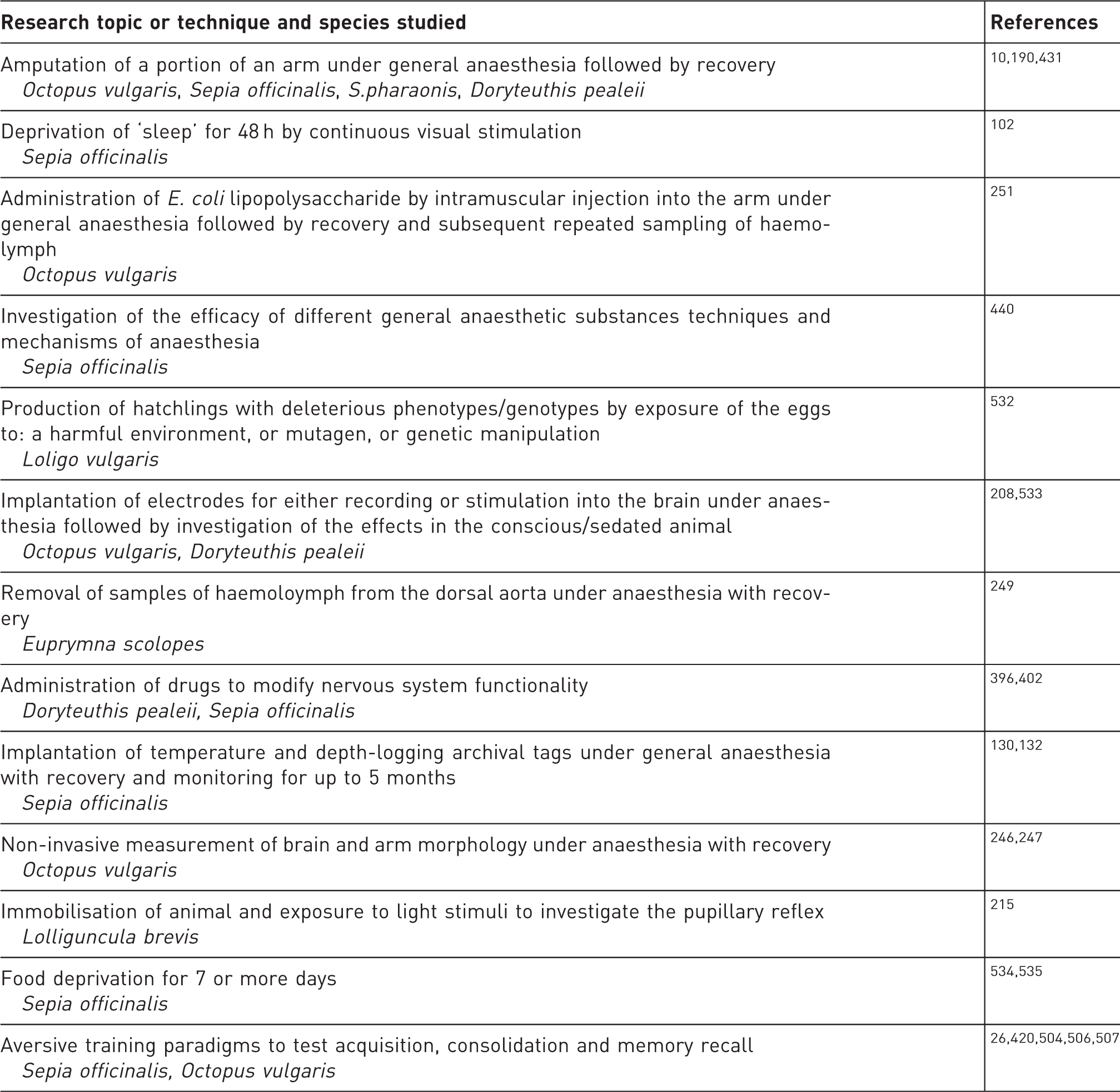
8.2 Identifying and reducing the adverse effects of procedures
All adverse effects that could be caused to animals by particular scientific procedures must be identified and predicted at the project planning stage (prospective assessment), then adequately monitored throughout the procedure. Steps must be taken to: i. refine each procedure, so as to minimise and preferably eliminate its adverse effects, and ii. alleviate any animal suffering that occurs during the conduct of procedures or whilst animals are recovering.
This is a legal, as well as ethical, requirement under Directive 2010/63/EU, which requires implementation of replacement, reduction and refinement (3Rs) strategies (section 2) wherever possible, with the aim of ‘eliminating or reducing to a minimum any possible pain, suffering, distress or lasting harm’ [PSDLH] caused to the animals. Note that 3Rs strategies should be implemented whenever feasible, at all times from birth to death of the animals including: sourcing, transport, housing and care, handling, and fate of animals, as well as the procedures themselves.
8.3 Monitoring animals undergoing procedures and setting humane end-points
Assessment of the severity of adverse effects must be carried out before, during and after procedures (see also details in section 2.2 above, which describes requirements for setting prospective ‘severity limits’ and retrospective reporting of the severity of procedures, along with questions for consideration).
Schemes for monitoring adverse effects during the procedure should cover the criteria outlined in section 6 and Table 5 for routine daily assessment of animal welfare; these should be supplemented with any other, specific, adverse effects that might be caused by the particular procedure(s). Criteria for assessment, and frequency and timing of observations, should be agreed before studies commence; then regularly reviewed as studies progress, and, wherever necessary, added to or amended.
A humane end-point must also be defined for each procedure, to describe (in terms of indicators of the nature and degree of suffering) the earliest point at which a specific intervention must be made to end an animal’s suffering, e.g. by: i. removing the animal from the study, ii. providing analgesia, iii. humanely killing the animal and/or terminating the study. The use of ‘score sheets’ for monitoring can be particularly helpful in determining when humane end-points have been reached, and when severity limits are being approached. 384 To be effective, this monitoring requires a team approach, with good planning, and appropriate training for all involved. Methods for the observation and assessment of adverse effects are relatively well developed for many vertebrate species including fish.384–387
There is a need for further development of objective criteria for assessing severity that can be used by the entire EU cephalopod community to ensure consistency. This is a current project of the non-profit Association for Cephalopod Research (see www.cephalopodresearch.org/projects).
Working on a consensus view for severity assessment of procedures is also a goal of the FA1301 COST Action (CephsInAction; http://www.cost.eu/COST_Actions/fa/Actions/FA1301; www.cephsinaction.org). An equivalent initiative has been set up for describing characteristics of laboratory mice (http://www.mousewelfareterms.org/doku.php), 8 and and for severity classification classification of scientific procedures involving fish. 384
8.4 Harm-benefit assessment
Of course, one way of eliminating animal suffering is not to carry out the procedure at all. In this context, it must be remembered that ‘procedures may only be carried out within the framework of a [authorised] project’ (Directive, Article 12.2), which is subject to a harm-benefit analysis, ‘to assess whether the harm to the animals in terms of suffering, pain and distress is justified by the expected outcome taking into account ethical considerations, and may ultimately benefit human beings, animals or the environment’.
Hence, for legal as well as moral reasons, investigators should carry out a harm-benefit analysis, as described above, prior to conducting any procedure.
In the following discussion, we cite published studies that provide evidence about possible harms caused by common procedures. These are included as examples that will help to predict and identify harms in future projects, but we are not suggesting that these studies would necessarily be considered justified according to the harm-benefit analysis conducted under the new EU Directive.
8.5 Some common procedures in cephalopod research
The following sections summarise current knowledge of the regulatory status, adverse effects and possibilities for refinement of some common procedures. It is evident that this knowledge is patchy and there is need for further work, especially to help refine procedures. Nevertheless, where possible, provisional recommendations for good practice are made.
8.5.1 Behavioural studies
Cephalopods have been used extensively for a variety of behavioural studies, as reviewed in several works.2,25,36,175,192,195,388
Classical behavioural studies have used a variety of aversive stimuli (e.g. electric shock; 72 acid solutions; 389 bitter; 390 mechanical 391 ) as part of training protocols. Because of their potential to cause distress and possible suffering, such studies would certainly fall within the definition of a regulated procedure under the Directive and should be avoided wherever possible. For example, electric shock or application of acid solutions should be unacceptable, and these and other harmful stimuli should be replaced with reward-based conditioning and, at the least, stimuli that do not cause pain.
Exposure of an animal to a stimulus known to evoke an escape response (i.e. inking, jetting locomotion, dymantic display) could also be argued to cause distress, especially if the exposure is repeated, and hence is likely to be a regulated procedure. Deprivation of food for prolonged periods, deliberate exposure to elevated noise or adverse change in water temperature, pH or chemistry, and direct exposure to a predator would also be likely to fall within the definition of a procedure, depending on the severity/degree of change.
Protocols using positive reinforcement (for example see:104,392–394) should be used wherever possible. 2
8.5.2 Administration of substances
Routes used for haemolymph sampling and drug administration in exemplar species of laboratory cephalopods species. Species are listed on the basis of the most commonly utilised for such procedures. Abbreviations: NS, not stated; NA, not applicable; R, sample removed; A, drug administered; EtOH, ethanol; SW, seawater.

Intramuscular injection of reserpine at the same dose also studied in Eledone cirrhosa and Sepia officinalis 542
Similar but more limited studies also performed in Eledone cirrhosa, Sepia officinalis and Alloteuthis sp. 401
Similar studies performed also using Illex illecebrosus 417
Cephalopods do not have readily accessible large superficial blood vessels; therefore, in general experimental agents and therapeutic drugs are given to unanaesthetised animals by bath application (assuming absorption via skin and gills), or injection via subcutaneous and intramuscular routes.
The efficacy of subcutaneous injections is not known, but drug absorption may be slow because of a low capillary density. However, this is not based on pharmacokinetic studies and only a limited number of substances have been studied.123,395
Locations for intramuscular injections include the arms (particularly proximal parts in octopus) and ‘neck’ (in cuttlefish) reported to have a high vascular density.123,180,208,251,396 Care should be taken not to damage arm nerve cords and ganglia by injection in the arms (especially in octopus), nor the cuttlebone or gladius in cuttlefish and squid respectively. The use of ultrasound may be helpful in directing injections to avoid vulnerable structures.246,247
Substances have been injected into the branchial hearts of O. vulgaris, but this requires eversion of the mantle. The animals recover rapidly from this procedure and no adverse events have been reported; however, although behaviour rapidly (<1 hour) returned to normal, 395 a systematic study of welfare of the animal has not been fully carried out.
8.5.3 Other methods
It should be noted that the welfare effects of the following methods has not been assessed. Intravenous (including vena cava) and intradorsal aortic routes have been used for substance administration by direct injection.e.g. 282 Direct administration into the brain has been successfully attempted in a number of studies. e.g.397,398. Finally, surgical implantation of vascular catheters has been used as a method of drug administration and for pressure recording.399–401
A method for gavage administration of drugs to cephalopods has been described by Berk et al. 383 and used to investigate the efficacy of a putative neuroprotective agent in blocking the effects of human tau protein on transmission in the squid giant synapse. 402 This method requires restraint of the animal and may require removal from the water and sedation. As the brain in cephalopods encircles the oesophagus care should be taken to use the narrowest gauge tube as possible, and it should also have a round end to minimise the chances of puncturing the gut particularly at the oesophageal/crop junction. Substances administered by this route will reach the crop and stomach directly.
Drugs and other substances such as nanoparticles have also been administered per os (i.e. by inclusion in the food,; 403 G. Ponte, unpublished data). Further experiments may help in better refine this approach.
8.5.4 Administration of drugs as investigational agents
When undertaking studies using pharmacological agents, the wider effects of the drug must also be considered in assessing the overall impact of a treatment on the welfare of the animal, and any unexpected side-effects should be recorded and minimised/avoided where possible. For example, Agin et al. 180 used cycloheximide, a protein synthesis inhibitor, to investigate the machinery involved in long-term memory in the cuttlefish. However, it induced positive buoyancy that interfered with animals’ ability to catch prey.
Relatively little is known about the pharmacology of central and peripheral neurotransmitter systems.1,404,405 In addition, very limited is the knowledge on the pharmacological characteristics of drug receptors in cephalopods. Therefore, caution should always be exercised when drugs, whose pharmacological properties have been defined in mammalian systems, are used as investigational agents in cephalopods.
If such studies are undertaken in group housed species, it will be necessary to identify individual animals (see section 4.1.2) so that individual variations in drug response can be identified. Additionally in group-housed species, special attention should be paid to agents that may increase aggression (e.g. by alteration of brain neurotransmitters) or that may impair the ability of an individual to escape or defend itself and action taken to reduce or eliminate these adverse effects.
8.5.5 Haemolymph (blood) collection
It should be noted that cephalopod blood does not clot, but haemocytes aggregate and vessels constrict to prevent blood loss.150,406 However, as haemolymph is pale blue (oxygenated) or colourless (deoxygenated), haemorrhage may be very difficult to detect.
A summary of routes used in several studies for haemolymph sampling is presented in Table 9. There are no systematic data on the welfare impacts of blood sampling methods in cephalopods, but in several studies behaviour was reported to return to normal following the sampling.249,251,395
In large cephalopods (i.e. E. dofleini), cuttlefish and E. scolopes, small blood samples have been obtained under anaesthesia via a needle inserted into the cephalic vein dorsal to the funnel.249,282,407,408 This method has also been successfully applied for haemolymph sampling in O. vulgaris (G. Ponte and G. Fiorito, pers comm.). In addition, small single samples of haemolymph can be taken directly from the branchial hearts in O. vulgaris (∼250 µl) and E. cirrhosa (<300 µl/100 g body weight; 1 ml/animal in animals of 500–800 g, as shown in 250 ) using a hypodermic needle, although this requires general anaesthesia to permit manipulation of the mantle to expose the hearts.250,251,255,403 In the bobtail squid (E. scolopes) single haemolymph samples of 50–100 µl can be withdrawn from the dorsal aorta by direct needle puncture under anaesthesia. 249 Where frequent sampling of blood is necessary, it is likely that this will require implantation of catheters.240,407,408 Again, the welfare impact of this procedure has not been fully assessed.
The maximum volume of blood which can be collected at a single sampling in particular cephalopods has not been assessed from a welfare perspective. In mammals, it is recommended that no more than 10% of the blood volume is removed at any one time. 409 For fish the Canadian Council on Animal Care (CCAC) ‡‡ recommends a maximum withdrawal of 1 ml blood/kg weight.
Blood volume in octopus is estimated at 5–6% of body weight,410,411 hence using the mammalian values as a guide, a maximum of 5–6 ml blood/kg weight would be advised, but, as noted above, only 1 ml/kg according to CCAC guidelines for fish.
However, in publications where multiple sampling has been used, authors either collected a reduced volume on each occasion (e.g. 10–20 µl for multiple sampling vs 50–100 µl for single samples in bobtail squid) as done by Collins and Nyholm 249 or have set a time for recovery between samples (e.g. 4 hours in E. cirrhosa). 126
8.6 Surgery
Cephalopods have been subjected to a broadly similar range of surgical procedures as have been performed in vertebrates, but in general surgical techniques are not described in detail in publications; this is no longer considered acceptable and we strongly recommend following ARRIVE Guidelines on reporting. 272
Surgical approaches and techniques, along with understanding of their welfare effects, are not as advanced in cephalopods when compared with vertebrates, particularly in relation to: i. intra-operative monitoring and maintenance of physiological parameters (e.g. blood pressure, PO2, PCO2, pH, temperature; see also section 8.8.4); ii. identification and control of haemorrhage; iii. controlled general anaesthesia and analgesia (see section 8.8); iv. optimal techniques for the repair of muscle and skin tissue and wound closure (see section 8.6.2) and healing in general; v. post-operative monitoring and special care that may be needed if, for example, feeding is transiently impaired; vi. infection risk (intra- and post-operatively) and requirement for prophylactic antibiotic cover (see section 7.3).
Although it will be some time before there is welfare guidance on all the above topics, anyone contemplating undertaking a surgical procedure in cephalopods will need to consider all of the factors, and submit relevant information, as part of the project evaluation and authorisation process, and during the conduct of any authorised surgical procedures. This will include:
ethical ‘justification’ for the procedures, in terms of harm vs. benefit of the outcomes; identification of potential adverse effects and the steps taken to refine the procedures, so as to minimise the adverse effects; processes for intra- and post-operative monitoring of the animals; timely application of methods to minimise post-operative suffering, e.g. analgesia and other specific care; and clearly defined humane end-points (section 8.3) to set an upper limit to the suffering that an animal experiences.
Some general principles for performing surgery on aquatic animals can be found in the fish literature, and these should be adapted where possible to cephalopods.412–414
As for all other procedures, anyone attempting any surgery in cephalopods must be ‘adequately educated and trained’ in the principles and practical techniques of surgery, and must be ‘supervised in the performance of their tasks, until they have demonstrated the requisite competence’ (Directive Article 23§2), for example, to the designated veterinarian or other competent person (see section 10 for further discussion).
A full description and evaluation of surgical techniques in cephalopods is outside the scope of this document but the following four specific aspects are highlighted, along with a detailed discussion of anaesthesia in section 8.8 below.
8.6.1 The operating room and environment
Surgical procedures should be performed in a dedicated room located close to the animals’ home tanks to minimise animal transport, and induction of anaesthesia should take place in the surgical procedure room. Good surgical lighting is essential, but care should be taken that this does not cause localised heating of the animal.
Although a sterile operating environment is highly desirable to prevent cross-contamination from human to cephalopod (and potentially vice versa, see section 9), and to ensure a consistent scientific baseline, maintenance of sterility is not practical unless: sterile seawater and anaesthetic solutions are used throughout the operation, and the surgeon and any assistants are fully trained in sterile technique and are wearing appropriate clothing. General information on asceptic surgical techniques can be found in a LASA 2010 report. 415
As a minimum, it is recommended that:
i. all apparatus or surgical instruments contacting the animal during surgery should be sterilised or at a minimum cleaned thoroughly with anti-bacterial and viricidal agents (e.g. diluted Halamid
§§
or Virkon
***
, F. Mark, pers. comm.); ii. all material contacting the wound site such as surgical swabs and sutures should be sterile; and iii. all personnel contacting the animal in the surgical environment should have cleaned their hands in accordance with aseptic technique and/or use sterile surgical gloves (blue nitrile gloves are recommended for handling animals, to avoid pale colours but these are not sterile and so should not be used for surgery); iv. the surgical site should be cleaned; but at present there is no information on the effect of commonly used skin cleaning preparations on cephalopod skin integrity and healing; v. the surgical site should be isolated as far as possible from any source of contamination (e.g. seawater/anaesthetic solution).
8.6.2 Maintaining physiological function
Water used to irrigate cephalopod gills during surgery should be circulated, treated and monitored to maintain appropriate anaesthetic levels, oxygen, pH, temperature and salinity, and to remove particulates. Pörtner et al. note that the sensitivity to hypoxia is greater in squid than either cuttlefish or octopus. 417
Consideration should be given to using a cooled operating table (temperature monitored) in combination with mantle perfusion with a chilled anaesthetic solution, as cooling can act as an adjuvant to anaesthesia,243,418 but should not be used as an anaesthetic alone. Cooling will also reduce metabolic rate, which may be advantageous when cardiorespiratory function is impaired as a result of anaesthesia.
The skin must be kept moist and this can be done with a sterile surgical drape moistened with sterile seawater. Care should be taken to minimise the potential for seawater to enter the wound site. Although anaesthesia to a depth sufficient to markedly suppress or stop ventilation may not be desirable from a physiological perspective, surgery involving incision of the mantle muscle and suturing may be very difficult in the absence of such marked suppression.
Whilst the efficacy of mantle perfusion with gassed anaesthetic/seawater solution for maintenance of blood gases may be questioned when there is severe suppression of cardiac function by the anaesthetic/anaesthesia, such perfusion may still be advisable to reduce anaesthetic wash out from the blood/tissues, to prevent desiccation of visceral structures and prevent/reduce hypoxic damage to critical tissues such as branchial hearts and gills. This may not be necessary for short duration (<10 min) minor surgical procedures, provided that full surgical anaesthesia can be assured.
8.6.3 Wound closure and healing
There are no studies assessing optimal materials to use for repair/closure of skin, muscle, connective tissue (e.g. hepatopancreas capsule), blood vessels or cranial cartilage in cephalopods, and publications do not often detail the materials used. The inclusion of such information in future publications is essential.
Closure techniques should follow normal practice for surgery in vertebrates, using round-bodied needles to reduce tissue trauma. Braided non-absorbable silk123,243, polyglyconate sutures 282 and cyanoacrylate adhesives132,401,419–422 have been utilised. Several authors report that cephalopods will try to remove sutures with their arms (F. Mark, unpublished data), 240 although this is not a universal finding and may reflect differences in surgical technique. Sutures sealed with cyanoacrylate have been used, but in some instances they may cause skin irritation (possibly due to being inflexible) and may not maintain integrity for longer periods of time in seawater.
An evaluation of five commonly used suture materials including braided silk, monofilament nylon and poliglecaprone has been undertaken in the mollusc Aplysia californica. 423 All materials induced similar adverse skin and subcutaneous tissue reactions, but the authors recommended the use of braided silk because it induced a less intense granuloma reaction. This result was unexpected as in studies of aquatic vertebrates monofilament sutures are less reactive than braided silk. 423
We would encourage anyone undertaking surgery in cephalopods to undertake histology on the wound site at the end of study, and include findings in any publications, with the aim of generating a database of good surgical practice. In addition to suture material, the most appropriate suture patterns (e.g. discontinuous, continuous) and needle size for skin closure in cephalopods has yet to be established, and this is knowledge especially important as the skin is particularly delicate in cephalopods.
Studies involving chronic implantation of devices such as archival (‘data logging’) tags185,417,424–426 need to consider possible chronic reaction of the tissues to the device and the impact that this may have on the normal behaviour of the animal. Mounts implanted in cuttlefish to support archival depth and temperature tags were well tolerated over a period of up to 5 months although post-mortem tissue thickening (possibly fibrosis) was observed around the implant and this increased in thickness with the duration of implantation.130,132 Chronic tissue reactions are also a potential issue for implanted identification tags (see section 5.7).
Descriptions of the natural course of wound healing following skin damage to the dorsal mantle in E. cirrhosa can be found in Polglase et al. 189 Authors also found that bacterial infection of a mantle wound impairs healing. 210 Healing and regeneration of the fin dermis in S. officinalis is described by Yacob et al. 427 and regeneration of the mantle connective in several species of octopus by Sereni and Young. 428
The arms in cephalopods have a remarkable capacity to heal and regenerate when a segment is removed by transection with the histological changes described in detail in O. vulgaris, E. cirrhosa, S. officinalis and S. pharaonis190,429–431 In O. vulgaris following transection of the distal 10% of the length of the arm as part of study to investigate regeneration, the exposed area was almost completely covered by skin in about 24 hours in some animals (T. Shaw and P. Andrews, unpublished data).
Cephalopods frequently injure arms, tentacles and fins in the wild;311,312,432–435 therefore a well-developed healing and regeneration mechanism is perhaps expected. This suggests that, provided sufficient time is allowed, surgical wounds, appropriately repaired, should heal successfully provided they do not become infected.
In addition to general anaesthesia surgery also requires the use of analgesics and this is discussed in section 8.9 below.
8.7 Tissue biopsy
Tissue biopsies for DNA isolation and PCR analysis for genotyping cephalopods have been obtained by taking small samples from the tip of an arm or tentacle with a sharp, sterile blade.436,437
The harms vs benefits of performing this procedure under general anaesthesia have not been assessed. We propose that irrespective of the use of general anaesthesia the arm tip should be treated with a local anaesthetic (in the absence of systemic analgesics). Only the absolute minimum amount of tissue should be taken (taking into account animal size) especially in cuttlefish as removal of large amounts of arm tissue transiently interferes with food manipulation and results in abnormal body posture. 190 In squid and cuttlefish, the fins may provide an alternative site for biopsy as they also appear to heal rapidly and regrow. 427 Although arms, fins and other tissues will regenerate, attention should be provided to avoid unnecessary sampling.
8.8 General anaesthesia
The Directive requires that ‘procedures are carried out under general or local anaesthesia’ unless anaesthesia is judged to be ‘more traumatic to the animal than the procedure itself’ and/or ‘is incompatible with the purpose of the procedure’ (Article 14§2). ‘Analgesia or another appropriate method’ must also be ‘used to ensure that pain, suffering and distress are kept to a minimum’ (Article 14§1), for example, peri- and post-operatively (see section 8.9 for further discussion). Furthermore, drugs that stop or restrict the ability of an animal to show pain (e.g. neuromuscular blocking agents) must not be used without an adequate level of anaesthesia or analgesia. When such agents are used, ‘a scientific justification must be provided, including details on the anaesthetic/analgesic regime’ (Article 14§3). After completion of the procedure requiring anaesthesia ‘appropriate action must be taken to minimise the suffering of the animal’, for example, by use of analgesia and special nursing care (Article 14§5).
During and after general anaesthesia and surgery, animals should be carefully and regularly monitored using a welfare monitoring scheme, and a record kept of observations and interventions to reduce or alleviate adverse effects. Of particular relevance is wound-directed behaviour but other potential welfare indicators are listed in Table 5. It is currently believed that operated animals should be housed individually at least until full recovery from general anaesthesia is assured, but this will clearly need particular consideration in gregarious species.
Many scientific procedures in cephalopods will require the use of general anaesthesia but despite more than 60 years of literature detailing studies involving anaesthesia, covering at least 17 agents, information is lacking on which agents in which species provide the most effective, humane general anaesthesia (i.e. which definitely blocks nociception and pain perception, generates no aversion, enables rapid induction and allows animals to recover quickly without adverse effects).
Recent studies have reviewed the use of agents claimed to have anaesthetic properties in cephalopods and criteria for evaluation of anaesthesia.8,61,131,418,438–440 In a recent contribution, isofluorane has been utilised to induce ‘deep anesthesia’ in O. vulgaris. 441 However, more detailed studies are required to assess the application of this agent to induce anaesthesia in cephalopods.
In some cases, a range of behavioural responses is reported to occur during exposure of the animals to anaesthetic agents (i.e. inking, jetting, escape reactions, increased ventilation);208,243,438 however, it should be noted that most studies report immersing the animal directly in the anaesthetic at the final anaesthetic concentration, and this may not be the best technique to minimise trauma (see below). It should be also noted that criteria for general anaesthesia are not consistent between studies and that behaviours observed during induction and to assess depth may differ between species. 439
Moreover, studies of general anaesthesia in cephalopods have investigated single agents and the familiar concept of ‘balanced anaesthesia’ involving more than one agent, used in mammalian studies (for reference see409,442) has not been explored in cephalopods (but see exceptions in Packard200,443).
8.8.1 General anaesthetic agents
Examples of studies using ethyl alcohol (EtOH), magnesium chloride (MgCl2) alone or in combination, or clove oil for induction and maintenance of general anaesthesia in exemplar cuttlefishes, squid and octopuses. Only publications with primary data are included; for recent reviews of other agents and species see: Andrews et al.
8
, Gleadall
438
, Sykes et al.
61
Studies utilising the same agents solely for euthanasia are not included. For each case we report: species, temperature at which the study have been carried out (T), body size (expressed in grams, unless otherwise stated), concentration of the agent utilised (expressed in %, unless otherwise stated), time to anaesthesia (in seconds), criteria for time to anaesthesia, duration of anaesthesia (in seconds, unless othewise stated), purpose of the study, recovery time (in seconds or minutes, as required), references and comments, if available. Number of subjects utilised and their gender are reported, if available from the original work, with body size. Abbreviations: F, females; M, males; NS, not stated; NA, not applicable; DW, distilled water; SW, seawater. See original works for full description. Note that other criteria for time to anaesthesia are also used to identify onset of general anaesthesia and are discussed in the text.

Juveniles (N = 6 for each concentration).
Detailed description of stages of anaesthesia provided in the original study.
10–12 weeks post hatch; N = 9.
13–14 weeks post hatch; N = 4.
DML: 18.5 cm (mean of N = 5).
DML: 7.6– 17.1 cm.
Young and sub-adult; N = 46.
M + F, N = 10.
N = 3 for each concentration.
Additional detailed description in the original study.
A study of anaesthesia only.
Although a large number of agents have been investigated for anaesthetic efficacy in cephalopods, some are now considered unacceptable on either welfare or safety grounds (e.g. urethane), 444 and so are not considered here. Similarly, we are not considering those utilised in a single experiment, and pending more evidence they are not considered herein.
Magnesium chloride (MgCl2) is the most extensively studied and used agent, probably because it appears to be the least aversive. However, it disturbs haemolymph Mg2+ levels, and so may not always be appropriate, for example, when blood samples are required to investigate normal magnesium ion levels.
Furthermore, there has been a recurrent concern that MgCl2 may be acting at least in part as a neuromuscular blocking agent; 441,445 but see comments in Graindorge et al. 398 However to date there is no direct experimental evidence to support this concern in cephalopods, as reviewed in Andrews et al. 8 The supposition may have arisen from the original use of magnesium chloride to relax small invertebrates prior to fixation. 446
Messenger et al. 447 concluded that MgCl2 exerts an effect on the central nervous system, and subsequent studies (G. Ponte, M.G. Valentino and P.L.R. Andrews, unpub. obs.) showed that electrical stimulation of efferent nerves in ‘anaesthetised’ O. vulgaris (as per criteria below) with MgCl2 (3.5%) evoked chromatophore contraction (arm and mantle), arm extension and mantle contraction showing a lack of effect at peripheral sites of motor control.
However, more recently, Crook et al. have shown that local subcutaneous and intramuscular injection of isotonic MgCl2 suppressed the afferent nerve activity in nociceptors activated by crushing a fin. 11 This demonstrates that should such high local concentrations occur in animals immersed in MgCl2, it may have analgesic/local anaesthetic effects. Further studies of its mechanism of action are urgently required.
Ethanol is widely used as an anaesthetic agent in cephalopods, but it is not clear whether it blocks pain perception and/or is aversive. Some variability is reported in the response of animals exposed to it (see Table 10A), but this could reflect impurities in the different sources of ethanol used 439 and other factors such as temperature.
For example, ethanol is considered effective in O. vulgaris, but has been reported to produce inking and escape reactions, 243 suggesting that it is aversive. However, these reactions are not noted if the temperature is below 12°C. 448 Moreover, ethanol has been reported to be ineffective in cold water octopus species, as cited in Lewbart and Mosley following a personal communication from I.G. Gleadall. 418 However, this observation seems contradicted by other personal experience on Antarctic octopods (F. Mark, pers. comm.).
Clove oil has been the subject of limited study so it is difficult to make a judgment about its use especially as there appear to be marked species differences in the response (see Table 10D). Further studies with clove oil, and its active constituent eugenol, are required to fully assess its utility as an anaesthetic for cephalopods.
Magnesium chloride as an agent.

DML: 145–180 mm, M (N = 7).
Isotonic solution in DW with an equal volume of SW (i.e. 3.75%).
DML: 14.8 cm, average of N = 26.
DML: 7.6–17.1 cm.
In DW mixed 1:1 with SW; final concentration 3.75%.
See also Margheri et al. for similar study of arm ultrasonography. 247
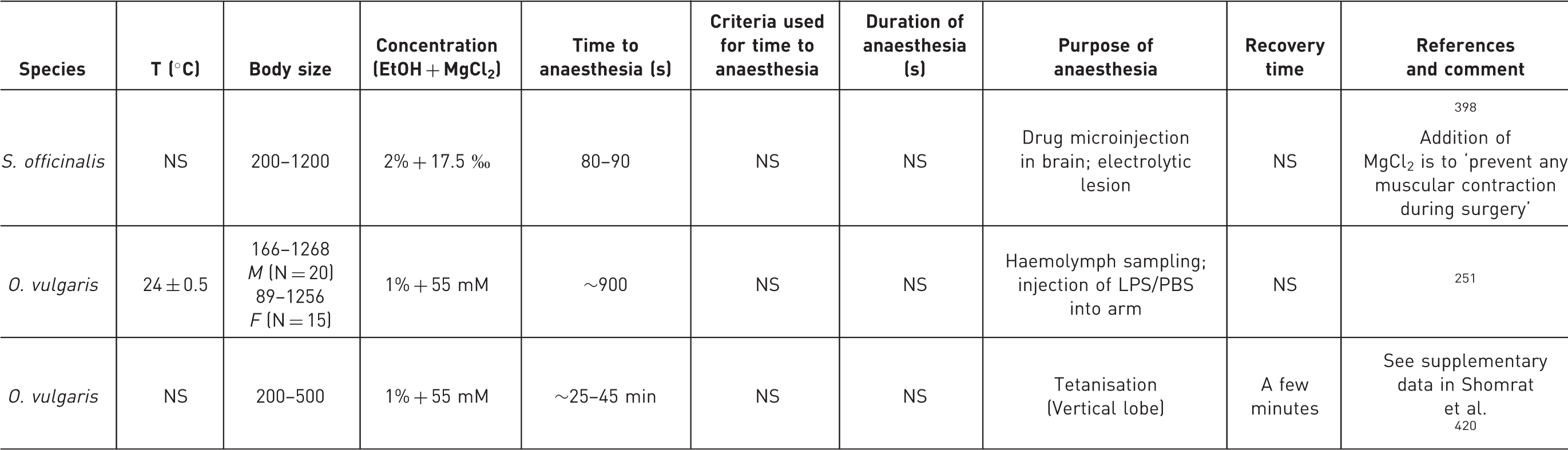
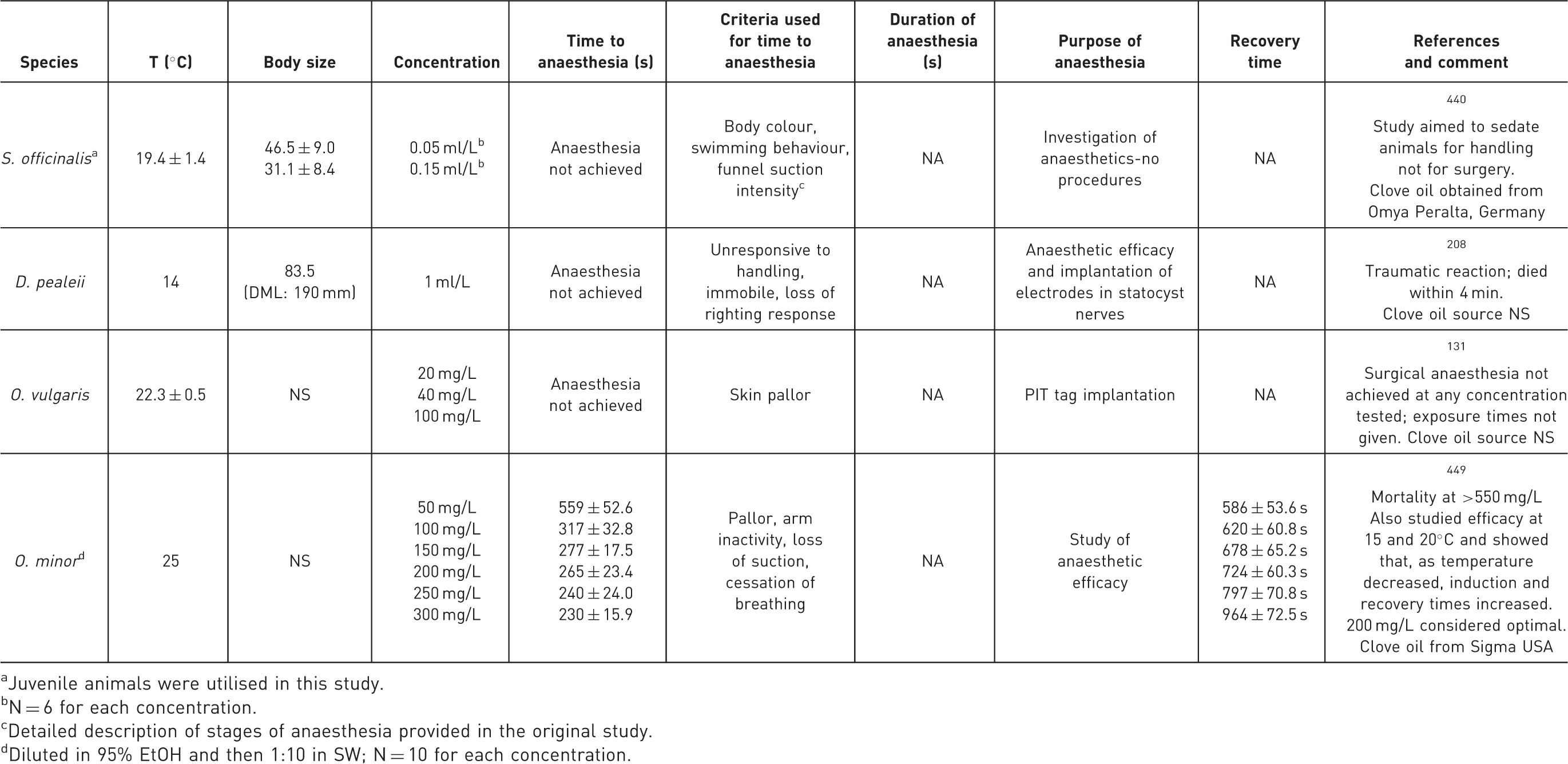
Juvenile animals were utilised in this study.
N = 6 for each concentration.
Detailed description of stages of anaesthesia provided in the original study.
Diluted in 95% EtOH and then 1:10 in SW; N = 10 for each concentration.
In addition to further research on the agents themselves, it is also apparent from the limited data in Table 10 that there is emerging evidence of differences in ‘efficacy’ of these agents according to age 102 and body weight 131 of animals; and temperature of the solution. 449
It should also be noted that the majority of systematic studies of anaesthesia in cephalopods involve the commonly used laboratory species, and particular care should be taken when attempting to anaesthetise other species, as responses to the same agent may differ markedly. For example, benzocaine (ethyl p-amino benzoate) produces a violent reaction followed by death in the squid D. pealeii, 208 but violent reactions are not reported in the octopus E. dofleini where it has been used for euthanasia. 450
8.8.2 Criteria for general anaesthesia
A detailed discussion of criteria for general anaesthesia in cephalopods, based on a review of recent studies on cuttlefishes, squid and octopuses is now available.8,440 They will only be outlined here, and further work is needed to define species-specific criteria for general anaesthesia.
Quantification of the physical (externally visible) parameters listed below might enable assessment of putative ‘stages’ or ‘planes’ of anaesthesia, and studies in cuttlefish
440
and octopuses131,243,449 illustrate this approach. In particular, ventilation depth and frequency and chromatophore activity are the parameters most amenable to continuous real time quantification to monitor the onset of ‘anaesthesia’. Currently there are no studies of electroencephalogram (EEG) activity in cephalopods under general anaesthesia, and all the parameters illustrated below rely on the assumption that the agents used to induce anaesthesia do not act only as neuromuscular blocking agents.
Decreased or absent response to a noxious stimulus. This is the most important test that the animal is sufficiently anaesthetised for surgery or other procedures to commence. Without blocking pain perception general anaesthesia cannot be considered to have been achieved. Studies have used a mechanical stimulus (e.g. a pinch) applied to the arm, mantle or supraorbital skin as a test of insensibility to a noxious stimulus.132,243,447 The selection of this type of challenge appears appropriate, as it is now known that cephalopods possess mechano-sensitive peripheral nociceptors,10,11 and in future this knowledge will enable the identification of better-defined stimuli to test for insensibility. Assuming that there is evidence to suggest that the animal is insensible, the following criteria should also be evaluated. Depression of ventilation. The initial reaction to exposure to the anaesthetic may be an increase in depth and frequency of mantle contractions with attempts to eject the solution via the siphon. Subsequently the frequency decreases progressively with time after exposure, and the coordination between the mantle and siphon become uncoordinated. With prolonged exposure, mantle and siphon contractions cease. Decreased chromatophore tone. Although animals become pale overall with increasing time of exposure to the anaesthetic, flashing colour changes have been reported on initial exposure to anaesthetic agents (e.g. in D. pealeii
208
or in S. officinalis
440
). The overall paling of the animal is indicative of a decrease in the central nervous system drive to the chromatophore motorneurones.
201
Decreased locomotor activity, arm/tentacle tone and sucker adhesiveness. The initial reaction to anaesthetic exposure may be an increase in activity (i.e. agitation, for review see Gleadall
439
), but activity gradually decreases, including swimming activity and fin movement, as for example in cuttlefish and squid.132,451 Octopuses will tend to settle on the bottom of the tank as the arms and suckers begin to lose tone and adhesion, but anaesthetised cuttlefish may float near the surface of the tank.
132
The arms should be flaccid and readily manipulated.
449
Loss of normal posture and righting response. As animals (e.g. squid and cuttlefish) become deeply ‘anaesthetised’ they lose the ability to maintain a normal position in the water column or adopt an abnormal position with the arms, head and mantle at angles not normally seen in conscious animals. For example, in squid and cuttlefish the arms and head may appear unsupported by the mantle collar muscles; octopuses adopt a flattened appearance on the floor of the tank rather than the usual posture with the head raised. In addition, animals placed on their dorsal surface make no attempt to right themselves. The righting response returns after ventilation and chromatophore activity return,
243
and this is probably a good indicator of overall recovery from anaesthesia, as both effects require complex coordination of neuromuscular activity. Absence of a response to light. The absence of a reaction to a strong light has been used as one sign of general anaesthesia (e.g. in Messenger et al.
447
), but this is poorly characterised.
8.8.3 Induction
A common practice for induction is to immerse the animal in the anaesthetic solution (made up in seawater) at its final concentration. However, to minimise trauma it is preferable to expose the animal to a rising concentration of the agent (as for example in Yacob et al. 427 ), which will also allow any adverse reaction to be quickly identified.
The animal should remain completely immersed in the anaesthetic solution for rapid effect. In addition, the use of a specialised closed anaesthetic chamber should be considered. The chamber could also be used as a transport box from the home tank to the operating room, and it may be possible to habituate at least some species (e.g. octopus and cuttlefish) to the box and train them to enter. This will reduce stress to the animals, and a closed chamber will prevent octopus escaping. As a general rule, animals should always be transported in seawater and movement should be minimised.
Anaesthetic solutions should always be freshly made, using filtered seawater, which is gassed (preferably with oxygen rather than air) and equilibrated to home tank temperature before immersing the animal. It is not good practice to anaesthetise an animal in a solution that has been used to anaesthetise another animal, as the water may contain chemical alarm signals. Animals must not be left immersed in an anaesthetic solution in which they have inked.
Although limited in scope, the pre-anaesthetic sedation technique applied by Packard 200 has not been followed by any other systematic study of methods to minimise the stress of general anaesthesia in cephalopods, as has been done for fishes. 452
Moreover, there is no general agreement about whether cephalopods should be deprived of food prior to anaesthesia. Some studies remove food for 24 hours 440 while others do not. 208
We are aware of only one report of food regurgitation by S. sepiodea during ‘anaesthesia’ in magnesium sulphate, 198 although defaecation is relatively common.
In view of the above, the anaesthetic protocol for cephalopods requires careful planning, including considering whether or not to withdraw food and, if so, for how long.
8.8.4 Maintenance and monitoring
Once the animal is fully anaesthetised (see above for criteria), it will usually be necessary to remove it from the anaesthetic chamber to perform a procedure. Anaesthesia must be maintained for the entire duration of the procedure, and physiological functions supported.282,453,454
Several authors describe apparatus (adapted from fish anaesthetic apparatus) for maintenance of anaesthesia during surgery (e.g. for cuttlefish:132,418,454 for squid:417,451), and these could also be better tested, and also adapted for octopus.
Marked suppression or cessation of ventilation (indicated by mantle/siphon contraction) is a common feature of general anaesthesia in cephalopods, so it is essential that the mantle is perfused with oxygenated seawater/anaesthetic. Such perfusion will only be effective if the branchial and systemic hearts continue to function, but little is known of cardiac function under anaesthesia. However, it has been observed that heart rate is very low in O. vulgaris anaesthetised with MgCl2 (M.G. Valentino and P.L.R. Andrews, unpublished observations) and in animals immersed in cold water. 243
Monitoring of physiological function under anaesthesia and during surgery is clearly an area requiring research so that the extent of hypoxia/hypercapnia is known and its impact on post-operative recovery and procedures can be assessed. However, O. vulgaris appears to be able to recover rapidly from protracted periods of apnea 150 and Pörtner et al. 417 comment that cuttlefish and octopus are less sensitive to hypoxia than squid (as might be expected from their different lifestyles). Doppler ultrasound (e.g. as in D. Fuchs and G. Ponte, unpublished observations; Vevo 2100 Visualsonics, The Netherlands) offers the best technique for monitoring cardiovascular function, but non-invasive methods for real time monitoring of blood gases (such as oximetry) and metabolic status (e.g. NMR spectroscopy as in Melzner et al. 455 ) need further development.
8.8.5 Recovery
The mantle should be flushed of residual anaesthetic solution and the animal then placed in clean aerated/oxygenated seawater. Ventilation can often start without intervention (depending upon the duration of anaesthesia), but gentle massage of the mantle is frequently used in cuttlefish, squid and octopods until spontaneous ventilation, as indicated by mantle and siphon movements, restarts. Other functions (sucker adhesion, chromatophore tone, righting) recover after ventilation recommences, usually in the reverse order to which they were lost.
Many studies monitor the time at which various functions return, but the time taken for full recovery of normal function from particular anaesthetic protocols is not known and, as most studies of anaesthesia do not involve surgery, the impact of surgery upon recovery is not known, but see Shomrat et al. 420 As currently assessed, recovery appears rapid (less than 15 mins, e.g. in243,420,440) and dependent on the procedure(s) performed, animals will usually take food quickly after ‘anaesthesia’ when returned to their home tank.395,420 However, further monitoring criteria are needed to ensure that animals have fully recovered from the anaesthesia and any surgical procedure. If recovery from anaesthesia is as rapid as appears to be, and the anaesthetic agents used do not have residual analgesic properties, it is vital that suitable analgesics are administered – which at this time of writing may best be done by infiltrating surgical sites with local anaesthetic (see next section below).
8.9 Analgesia and local anaesthesia
Directive 2010/63/EU, Article 14§4, requires ‘an animal which may suffer pain once general anaesthesia has worn off, shall be treated with pre-emptive and post-operative analgesics or other appropriate pain-relieving methods, provided that it is compatible with the procedure’.
At the time of writing, there is no information on the efficacy of any analgesics in cephalopods, although both ketoprofen and butorphanol have been recommended. 445
As nociceptors have been recently studied in cephalopods,10,11 it should be possible to investigate the efficacy of systemically administered substances for potential analgesic activity. In addition to identification of mechano-nociceptors, Crook et al. also showed that injury to a fin in squid induced spontaneous activity and sensitisation at sites distant from the lesion including the contralateral body; 11 similar sensitisation of both the wound site and at distant sites has been reported in octopus. 10 This implies that potentially surgery at any surface site (but possibly anywhere including the viscera) could evoke a more general sensitisation of nociceptors. If this is the case, it is essential that suitable analgesic agents are quickly identified.
In the absence of the availability of systemic analgesics, it is recommended that local anaesthetics are used to produce localised analgesia either by infiltration into a wound site or local nerve block. Xylocaine (2%) and mepivacaine (3%) have both been shown to be effective in producing a block of transmission in the arm nerve cord lasting at least 1 hour (G. Ponte, M.G. Valentino and P.L.R. Andrews, unpub. obs.).149,456 It should be noted that local anaesthetics acting on the fast tetrodotoxin (TTX) sensitive voltage gated sodium channels may not be effective in species such as the blue-ringed octopus which possesses endogenous TTX. 457
Selective nerve block with infiltration of a local anaesthetic should also provide an interim means of preventing more generalised nociceptor sensitisation (see above), as it has been demonstrated that afferent nerve block by injected isotonic MgCl2 prevented both local and distant sensitisation. 11
8.10 Fate of animals at the end of a procedure
At the end of a procedure as defined in section 8.1 above, a decision about the fate of the animal is required. The Directive identifies three possibilities:
Humane killing: Article 17 requires that ‘at the end of a procedure’ an animal must ‘be killed when it is likely to remain in moderate or severe pain, suffering, distress or lasting harm’. Other animals should also be humanely killed at the end of procedures, unless ‘a decision to keep an animal alive’ has been taken ‘by a veterinarian or another competent person’, when the possibilities described below should be considered. Methods of humanely killing cephalopods are discussed in section 8.11 below.
Release or re-homing: An animal ‘may be… returned to a suitable habitat or husbandry system appropriate to the species’, provided that: ‘its state of health allows it; there is no danger to public health, animal health or the natural environment; and appropriate measures have been taken to safeguard [its] well-being’ (Article 19). This could include release to the animals’ natural environment, transfer to public aquaria, educational or other competent holding facilities. However, in general, cephalopods should not be returned to the wild, except in studies where, following a procedures, animals are released immediately at the exact location where they were captured, during a relevant season for migratory species, having been certified fit by a veterinarian or other competent person (Articles 17§2 and 19). Any requirements of other national and international legislation regarding the release of animals to the wild must also be met.
Re-use: Animals may also be considered for re-use, provided certain conditions are met.
Article 16 defines re-use as ‘use of an animal already used in one or more procedures, when a different animal on which no procedure has previously been carried out could also be used. By definition, re-use applies to situations in which the objectives of the first and second procedures are unrelated. †††
Reuse is only permitted when:
‘the actual severity of the previous procedure(s) was ‘mild’ or ‘moderate’; the animal’s ‘health and wellbeing has been fully restored; the further procedure is classified as ‘mild’, ‘moderate’ or ‘non-recovery’ (see section 8.1); and the proposed re-use ‘is in accordance with veterinary advice, taking into account the lifetime experience of the animal’.
‡‡‡
8.11 Methods of humane killing
The ultimate fate of cephalopods in the majority of studies will be humane killing. All personnel involved in humane killing should be trained and be familiar with the principles of good practice, such as those set out by Demers et al. 458
Article 6 requires that, whenever animals are killed:
it should done with minimum pain, suffering and distress by a competent person; and one of the methods of killing listed in Annex IV (section 3) of the Directive should be used, followed by confirmation of death, using one of the methods listed in Annex IV section 2.
However, whilst methods listed for fish might be applied to cephalopods, Annex IV offers no specific guidance on methods for humanely killing cephalopods. Although the CCAC 459 take the view that the priority is a rapid loss of consciousness, these guidelines concur with the view of Hawkins et al. from a discussion of CO2 killing in vertebrates that ‘it is more important to avoid or minimise pain and distress than it is to ensure rapid loss of consciousness’ (p. 2). 460
We have described some possible methods below, but the efficacy, and level and nature of any suffering caused, have not been comprehensively evaluated for all of these techniques, and further research is needed. Animals should not be killed in the rooms used to house other animals nor within sight of conspecifics. It must also be ensured that blood or chemical alarm signals cannot be detected by other animals (e.g. by entering the water system). Whichever method is used in a given species, the potential impacts of factors such as body weight, age, sex, season and water temperature on the efficacy of the method must be considered.
8.11.1 Chemical methods
Chemical methods for killing cephalopods are based on an overdose of anaesthetic agents, by using either a higher concentration and/or longer exposure time than that needed for anaesthesia. As also outlined above, these methods all have the potential to cause adverse effects prior to unconsciousness, such as skin or eye irritation, or sensation of asphyxia. To reduce and avoid any such suffering, animals should be exposed to a gradually rising concentration of the anaesthetic, and not directly immersed in a solution at the full concentration needed to cause death. In tropical and temperate species, cooling may be used as an adjuvant to the anaesthetic for humane reasons, and could also reduce post-mortem tissue damage if tissue is required for in vitro studies.
A previous review of this topic proposed the following protocol as suitable for S. officinalis, D. pealei, O. vulgaris and E. cirrhosa: ‘At least 15 minutes immersion in MgCl2, with a rising concentration [optimal rates to be determined], ending with a final concentration of at least 3.5% in the chamber used for humane killing; possibly enhanced by using chilled solutions or with the clove oil’s active ingredient eugenol, followed by immediate mechanical destruction of the brain’ (Andrews et al., p. 61). 8
It was further proposed that if the brain was needed, the immersion period should be extended to more than 30 minutes to ensure unconsciousness as required in Directive 2010/63/EU (Annex IV, 1(a)) prior to removal of the brain. If the brain is not removed for study, confirmation of permanent cessation of circulation (see 8.12 below) is also considered as a possible method for completing killing according to Directive 2010/63/EU (Annex IV, 2(a)).
8.11.2 Mechanical methods
When carried out by highly skilled operators, death by mechanical destruction of the brain takes only a few seconds, but the nature and degree of any suffering is unknown. For this reason we take the view that cephalopods should not be killed by this, or any other, mechanical method without prior sedation/anaesthesia. However, it may be possible to utilise a ‘mechanical method’ if it can be justified and is authorised by the National Competent Authority as a specific regulated procedure within a project application.
Electrical methods such as ‘Crustastun’ used for humanely killing crustacea, such as lobsters and crabs, 461 might also be considered and evaluated for their suitability in terms of animal welfare. It will be particularly challenging to develop humane methods for use when the brain is required intact, but where the use of anaesthesia may be a confounding factor.
8.12 Confirmation of death
Use of a method for confirming death following humane killing is mandatory, and options are listed in Annex IV§2 of the Directive. Two of the methods listed, i.e. ‘dislocation of the neck’ and ‘confirmation of onset of rigor mortis’ are impossible for cephalopods – the latter because it does not occur in cephalopods.
This leaves three possible methods: i. confirmation of permanent cessation of the circulation; ii. destruction of the brain; or iii. exsanguination.
Confirmation of permanent cessation of the circulation and exsanguination. Octopuses, cuttlefish and squid have two branchial hearts that move blood through the capillaries of the gills. 462 A single systemic heart (the only one in nautiloids) then pumps the oxygenated blood through the rest of the body. The heart(s) may continue beating for some time after permanent cessation of breathing, so transection of the dorsal aorta/vena cava may be used. Transection of the dorsal aorta/vena cava will be effective in inducing exsanguination if the systemic heart is able to pump effectively (i.e. the anaesthetic used does not supress cardiac function); also note that the systemic heart is distension sensitive. 463 Finally, the possibility of transection of the branchial aorta afferent to the heart, at the level of the auricle, should be further explored considering the easy access to them through the mantle cavity nearby the gills (G. Ponte and G. Fiorito, pers. comm.).
The effectiveness of exsanguination as a method of killing is not known.
Freezing (below −18°C for several hours) after killing may be a further means of confirming cessation of circulation and hence death that does not necessarily entail destruction of the body.
Destruction of the brain may be difficult to ensure in some species because of the location and relatively small size (e.g. Nautilus sp.) although this can be overcome by training and a detailed knowledge of the cranial anatomy of the relevant species.
The methods used for humane killing and confirmation of death should always be included in publications.
Article 18 of the Directive requires member states ‘to facilitate the sharing of organs and tissues of animals killed’ where appropriate. Researchers should be encouraged to use tissue from animals killed in other projects for in vitro research (e.g. tissue bath pharmacology), rather than killing an animal specifically/only to obtain tissues; and consideration should be given to setting up banks of frozen and fixed tissue to optimise animal use.
9. Risk assessment for operators
This section will focus on the potential risks from the direct handling of cephalopods in a laboratory setting, but will not cover the more generic risks associated with working in either a laboratory (e.g. tissue fixatives, reagents) or a marine aquarium environment (e.g. tank cleaning agents, slipping, electricity in a wet environment). However, all personnel involved in research should be appropriately educated, trained and competent to perform any task relevant to the research. Personnel should be actively involved in risk assessment and management, and incident reporting encouraged.
9.1 Personnel to consider
The following personnel should be considered and their risks assessed:
a. Fishermen, divers or others responsible for the capture of cephalopods in the wild. Although such people may not be employees of the institution/facility, as far as possible, the institution/facility should be assured that safe practices are being employed. The Directive specifies that, in cases where justification is provided, to obtain animals from the wild ‘competent persons’ (Article 9) should be involved. As part of competency assessment issues related to health and safety practices of cephalopod capture should be explored. b. Animal carers and technicians. This includes anyone involved in cleaning and feeding animals; cleaning animal rooms and equipment (e.g. tanks, filters). c. Animal technologists or research laboratory technicians. These are personnel who may be involved in manipulating animals during experiments, sampling biological fluids, euthanasia and necropsies. d. Principal and other Investigators: All personnel involved in performing research experiments including in vivo regulated procedures and in vitro handling of live tissue. e. Designated veterinarian (or other suitably qualified expert). f. Personnel responsible for the disposal of animal remains.
9.2 Risk identification, prevention and protection
9.2.1 Physical risk
Bites of cephalopods are produced by the hard beak-like jaws associated with powerful musculature of the buccal mass located at the center of the arm crown. Such bites do not always penetrate the skin of human beings (e.g. see p. 68 in Wells). 150 The effects of penetrating bites are exacerbated by enzymes, venoms, other bioactive substances and microorganisms in the saliva. The possibility of injury should not be overlooked even when animals are transported in a plastic bag. 464
The arms of all cephalopods are relatively strong, and this is especially the case in larger octopuses where the grip is enhanced by the numerous suckers. Areas of erythema may be induced if attempts are made to detach an animal by pulling in air, rather than allowing it to leave the arm naturally under water. Special precautions should be used if handling members of the teuthoid family as their arms/tentacles have hook-like appendages. Staff should be trained in how to remove animals that have become attached to their arms using the minimum of force and without inducing the animal to bite.
The use of Personal Protective Equipment (PPE), such as gloves or gauntlets resistant to penetration, may be suggested, but care should be taken to ensure that the gloves do not harm the delicate skin of cephalopods and that safe handling is not impaired by wearing gloves. In addition, the typical behaviour of the animals should be well recognised and mostly for signs of imminent aggression, escape attempt and other putative abnormal behaviours.
9.2.2 Chemical risk
Direct contact with mucus, faeces and biological fluids
Biological materials can represent a risk of allergy, intolerance and/or toxicity particularly with repeated exposure. Exposure to mucus, faeces and ink may occur during routine cleaning and handling and other fluids during autopsy. Faeces and blood (haemolymph) are also potential routes of infection. We are not aware of reports of reactions to these biological fluids, but in case of doubt the use of waterproof gloves is recommended; in addition, in case of inadvertent contact with any biological substances, hands should be properly washed immediately.
Handlers and those undertaking autopsies should also be aware of any experimental procedures previously undertaken involving the administration of potentially hazardous substances (e.g. infectious agents, radioactive material, drugs, nanoparticles) to the animals so that assessment of potential risks can be undertaken.
Venom, enzymes and other pharmacologically active substances
The secretion from the posterior salivary glands of coleoid cephalopods (see Table 4.1 in Wells 150 ) is injected into prey via the salivary papilla to immobilise and digest it with a mixture of venoms (i.e. cephalotoxin 465 ), enzymes (e.g. chitinase, carboxypeptidase, hyaluronidase, phospholipase A2) and other biologically active substances (e.g. 5-hydroxytryptamine, dopamine, substance P) as reviewed by Ruder et al. 466
A localised or systemic response could be induced by one or more of these substances particularly in sensitive individuals, but documented examples of systemic reactions to bites are rare except in the case of the blue-ringed octopus.
In fact, the venom of blue-ringed octopuses (Hapalochlaena spp.) can be fatal 467 unless there is prompt medical attention.467,468 The toxin involved is the potent sodium channel blocker TTX 469 that is found in the posterior salivary glands, skin, branchial hearts, gills and Needham’s sac, 470 so care should also be taken with handling these animals post mortem. Recently, TTX has been found in the eggs with the levels increasing after laying; 471 therefore, the risk with this species does not only come from adults. Other data show that the venom is produced by symbiotic bacteria (Aeromonas, Bacillus, Pseudomonas and Vibrio) found in the salivary glands. 472 Clinically, the bite of the blue-ringed octopus is most often painless but freely bleeding. Erythema and edema at the bite site usually occurs, but the most important effects are those that are systemic. Most severe envenomations are characterised by generalised weakness, slurred speech, circumoral paraesthesias, respiratory difficulty and dysphagia. Such symptoms may last for 12–24 hours. There is no antivenom, therefore treatment includes pressure immobilisation and immediate transport to a medical facility while monitoring respiratory and neurologic status. 473
No reactions specifically attributable to the venoms from other species of cephalopod have been described, but there are several reports of localized reactions to bites from cephalopods although these seem rare. Wells reports having been bitten himself 10 or 20 times without event, but also describes a reaction to an O. vulgaris bite on the forearm in a student who had previously never been exposed to cephalopods. 150 The response included swelling of the forearm (comparable to a bee sting) and overnight pain, both of which resolved the next day. A local skin reaction probably related to proteolytic activity has been described after red octopus (O. rubescens) bite. 474 Haemolytic activity against mammalian red cells has been reported in vitro with low concentrations of saliva from E. cirrhosa, 475 but we are not aware of any evidence for haemolysis in vivo, although caution should be exerted.
Reactions to a bite will depend upon the sensitivity of the individual and this may be a particular issue with atopic individuals or those who have become sensitised by repeated exposure. The reaction to a bite may not be immediate as indicated by a case report of giant cell-rich granulomatous dermatitis/panniculitis one month after a bite from an octopus (species not given) on the wrist. 476 Bites are also a source of infection (see below) and again the reaction may be delayed.
Precautions should be the same as for the protection against bites (see above). All occurrences of bites by cephalopods should be recorded in the laboratory safety book, medical/paramedical advice sought particularly in cases where the skin is broken and the clinical outcome monitored. Anderson et al. reported anecdotally that immediate hot water treatment was effective in neutralising the localised effects of the bite of O. rubescens. 474
Seawater and ink
Cephalopods in general and octopuses in particular can forcibly squirt seawater and/or ink directly at handlers. In addition to melanin, ink contains an array of bioactive substances including enzymes (e.g. tyrosinase, tyrosine hydroxylase) and other chemicals (e.g. dopamine, 5,6-dihydroxyindole).477,478 There is a theoretical risk that the seawater may be contaminated by pollutants or infectious agents in open systems but seawater or ink in the eye could cause irritation and also distract the handler from their task increasing the risk of a bite or animal escape. Wearing eye or face protection should be considered when handling cephalopods.
9.2.3 Biohazards
The animals
Animals, especially those taken from the wild, can transmit infectious agents (zoonoses) to humans and as cephalopods host a number of bacteria (Gram + and −), viruses and parasites; this is a potential risk for anyone in contact with cephalopods. The major risk of transmission is via accidental ingestion or a bite.
This section will not cover the potential pathogenicity of different agents, however, mention should be given to, for example, Anisakis and Aggregata that are known to be the cause of the zoonotic disease, as reviewed by Yang et al. 479
Most information regarding infection with Anisakis comes from human consumption of uncooked cephalopods, but poor hand hygiene and laboratory practice – especially when undertaking autopsies of fresh animals – means that hand-to-mouth transmission is a possibility. An additional potential source of parasites (and possibly bacteria, viruses and toxins) is the fish, crustacea and molluscs used as food for cephalopods so wearing gloves and hand hygiene should also be considered.
The main documented risk is bacterial infection from bites although the bacteria may originate from contaminated water as well as the animal itself.480–482
Particular attention should be paid when handling cephalopods with skin lesions as these wounds are often infected (see section 4.4).
Seawater
Seawater itself is a potential reservoir and transmission vehicle for infectious organisms, chemical pollutants and toxins (e.g. from algae) particularly in open systems. In seawater, many pathogens can be responsible for infections, e.g. Staphylococcus aureus, Streptococcus pyogenes, Mycobacterium marinum, Vibrio vulnificus, Erysipelothrix rhusiopathiae, Aeromonas hydrophila, Pseudomonas aeruginosa, Prototheca wickerhamii. 473
These represent a potential hazard for both animals and humans who have contact with the seawater but the risk should be minimal if the water quality is monitored and maintained within strict limits. Additional safety precautions may need to be put in place if there is a breakdown in water quality management especially for those who may need to decontaminate and clean the tanks.
Allergens
Repeated exposure to animals, chemicals (e.g. some disinfectants) and some disposables can result in the appearance of several clinical forms of allergic reaction (e.g. contact dermatitis or urticaria). To minimise the allergy risk operators should have a medical assessment before starting work and at regular intervals. Cephalopod mucus can be an irritant and has been occasionally described as an allergen (A. Affuso, pers. comm.). Since there are cases of an acquired allergy to cephalopod eggs, it is recommended that they are handled wearing gloves. Arginine kinase from Octopus fangsiao has been shown to react with IgE in the serum of octopus-allergenic subjects, 483 emphasising the importance of identifying individuals who may be especially sensitive to cephalopods before they begin work so that the risk can be assessed and managed.
9.3 A summary of practical advice
9.3.1 Assess and manage the potential risks
Each person involved with cephalopods should be assessed, and monitored for potential risks, notes taken of the species involved and the work to be undertaken before any work assigned. For example, the risks in moving a potentially lethal blue–ringed octopus or a 30 kg E. dofleini between tanks are different from moving a 200 g cuttlefish. Protocols of good laboratory practice (GLP) – Standard Operating Procedures (SOPs), should be developed in conjunction with the Health and Safety officer or other person, incorporated into training programmes (see section 9) and common protocols (e.g. handling, cleaning, humane killing) displayed or kept in a file in the facility and in the room dedicated to procedures. Risk assessment will also need to comply with local ‘out of hours’ policies. All working areas should be equipped with required PPE (overalls, hypoallergenic gloves, safety glasses, eye wash) and have a telephone with an emergency number displayed.
9.3.2 Operator health
Most staff undergo some form of health assessment at the beginning of employment. Anyone working with cephalopods should be asked if they are allergic to these animals or if they are atopic so that risks can be managed. All staff with regular exposure to cephalopods should be monitored regularly for signs of sensitisation.
9.3.3 Recording incidents
All incidents (including ‘near misses’) should be recorded partly to facilitate development of improved protocols. For example, skin-penetrating bites must be recorded and reported immediately to the relevant person so that appropriate action can be taken and any delayed reactions (e.g. infections) documented.
The ‘incident book’ should be reviewed regularly and protocols and policies modified as required. Lessons for the wider cephalopod community should be posted on the CephRes web site (www.cephalopodresearch.org) and if possible published.
9.3.4 Dealing with incidents
Staff should be familiar from training and laboratory SOPs/GLP with action to be taken in particular circumstances. The most likely incident requiring immediate action is a bite in which the skin is broken and the wound infiltrated with secretions from the posterior salivary glands and/or seawater. A protocol for this and other eventualities should be drawn up in consultation with the trained on-site first responder and a medical practitioner.
10. Education and training: carers, researchers and veterinarians
The Directive requires that all staff involved in regulated research are adequately educated and trained for tasks they are required to perform (Article 23) including: i. carrying out procedures on animals; ii. designing procedures and projects; iii. taking care of animals; or iv. humane killing of animals.
Education and training therefore includes all staff involved in daily care (including veterinarians; since courses on marine invertebrate medicine are rare), researchers performing procedure and principal investigators designing studies. Anyone having direct contact with the animals will need to be able to demonstrate that they are practically competent in performing tasks or regulated procedures and have this competence assessed periodically. There may also be an argument for providing limited theoretical training for members of an institutional project/ethical review committee about novel species.
Trained individuals should understand and be able to demonstrate the importance of the animal welfare regulations and guidelines for housing, care and use, assessment of animal welfare including signs of illness, PSDLH and their palliation or treatment.
Developing training programmes to meet these requirements is a particular challenge for cephalopods as although knowledge of general care and welfare is relatively well developed (as reviewed in Fiorito et al. 2 ), knowledge of PSDLH and their application to assessment of animal welfare aspects of a project are less well established.3,8 In addition, there are multiple species of cephalopod with a variety of care (see also Appendix 2) and welfare requirements. Many aspects of training will need to be delivered by animal technologists, researchers and veterinarians who have gained experience prior to the regulation of research involving cephalopods, but it would be desirable to involve some trainers with expertise in working with aquatic vertebrates (including expertise in ethical review).
Courses will need to align with specific national requirements, and will need to be recognised by the National Competent Authority (http://ec.europa.eu/environment/chemicals/lab_animals/ms_en.htm) of Member States as fulfilling the requirements of the Directive 2010/63/EU for all persons involved in the use, care and breeding of cephalopods for scientific procedures. Recognition at EU level will facilitate movement of personnel between member states. The content and delivery of the modules should be validated by a university or other competent awarding body, and linked to Continuing Professional Development (CPD) programmes of professional bodies.
Training modules should be designed taking into account of the EU Commission working document of a development of a common education and training framework to fulfil the requirements under the Directive.
Ideally courses should be offered at EU level but as many aspects of training are ‘hands-on’ courses will need to be based in facilities with aquaria and access to several species. It is proposed that a training course should be structured into three modules; basic training, a species-specific module, procedures, PSDLH assessment and management module. These modules cover the key welfare assessment competencies and welfare training topics outlined in Tables 12 and 13 of Hawkins et al. 484
Delivery and assessment of modules are not discussed here.
10.1 Indicative content of modules
10.1.1 Basic module: an introduction to cephalopods in research
This should cover: i. national and EU legislation on protection of animals used for scientific purposes; ii. a brief introduction to cephalopod biology; iii. why cephalopods were included in the Directive; iv. the philosophy of the institution regarding animal care and use; v. the requirement to comply with all national regulations and institutional guidelines; vi. the key differences between undertaking research in a legally regulated and an unregulated environment; vii. record keeping; viii. the requirement to respond immediately to any PSDLH issues; ix. reporting animal care and use concerns at institutional and national levels; x. health, safety, risk assessment and security; xi. roles of the institution and project (‘ethical’) review committee, veterinarian (or other competent expert), animal care, and research staff in the animal care and use programme; xii. public engagement.
10.1.2 Species-specific module
This focuses on the species utilised for research in a particular institution and should include the following species-specific topics: i. biology and behaviour; ii. supply, capture and transportation (including any additional permits and regulations); iii. environment (tanks, water, enrichment) and control; iv. signs of health, welfare and disease; v. assessing when an animal should be killed humanely; vi. specialised techniques for identification of individuals; vii. anaesthesia and humane killing; viii. tagging and marking; ix. genotyping; x. analgesia/anaesthesia/euthanasia/confirmation of death.
10.1.3 Procedures, PSDLH assessment and management module
This module has three main elements:
a) Project/licence and ethical committee applications. This will cover: i. experimental design from a 3Rs perspective (see section 2.2.2; see also Smith et al.
3
); ii. principles of harm-benefit analysis (e.g. Bateson cube); iii. principles of severity assessment (prospective, actual and retrospective); iv. setting humane end-points; v. writing a lay summary of the project/ethical application; vi. public engagement. b) Recognising PSDLH and their management. This component deals with these aspects in depth as this module is intended for those designing projects or performing procedures some of which may never have been performed in a cephalopod previously. The following topics will be covered: i. evidence for the capacity of cephalopods to experience PSDLH (see review and discussion in Andrews et al.
8
); ii. recognising PSDLH and techniques to minimise and treat them in the context of a regulated procedure; iii. special considerations regarding senescent cephalopods. c) Procedures. This covers: i. what is a procedure within the meaning of the Directive?; ii. an introduction to basic surgical techniques and post-operative monitoring and care; iii. non-surgical procedures, techniques and assessing their impact on the animal. d) Reporting studies under the Directive. Annual statistical return (Article 54) and an introduction to the ARRIVE Guidelines (www.nc3rs.uk/ARRIVE;
272
) and the Gold Standard Publication Checklist.485,486
Education and training of all personnel involved in the research programme at whatever level is essential to ensure the optimal care and welfare of animals and for the standards to improve with time by identification and dissemination of examples of good practice. These Guidelines and recent publications1,2,487 establish the core material required for the delivery of the modules outlined here as a basis for education and training of personnel involved in research now regulated by the Directive. The next step will be to develop the above outlines into a document that can be used for accreditation of a course compliant with FELASA recommendations for laboratory animal science education and training, as outlined by Nevalainen et al. and recently updated.488,489
11. Concluding comment
This paper represents the first attempt by members of the international cephalopod community to develop guidelines for Care and Welfare of cephalopods utilised in scientific research. Although the guidelines primarily address the requirements of Directive 2010/63/EU, we anticipate they will also be utilised by the wider cephalopod research community outside the EU. It is recognised that in contrast to equivalent guidelines for vertebrates the evidence base for some aspects of these guidelines is not strong.
It is hoped that this paper will prompt research directed specifically at the Care and Welfare of cephalopods in the laboratory to provide a solid evidence base for future revisions of these guidelines.
Footnotes
Disclaimer
These Guidelines represent a consensus view, but inclusion in the authorship or contributor list does not necessarily imply agreement with all statements.
Funding
CephRes, AISAL and FELASA support this work. This publication is also supported by COST (European Cooperation in Science and Technology).
Acknowledgements
This work originates from a joint effort to develop the first Guidelines for the Care and Welfare of Cephalopods in Research according to Directive 2010/63/EU made by the Association for Cephalopod Research - CephRes, The Boyd Group (UK), and the Federation for Laboratory Animal Science Associations – FELASA. This has been initiated and subsequently developed following April 2012 CephRes Meeting in Vico Equense (Italy). We would like to thank Drs John Messenger, Andrew Packard and Sigurd von Boletsky for their contributions to the initial meeting where ideas for Guidelines for the Care and Welfare of Cephalopods were discussed. We are also grateful for comments and contributions by Piero Amodio (CephRes, Italy), and by Ariane Dröscher (University of Bologna, Italy) for historical data.
PA and GF conceived and edited this work as well as a making a major contribution to the authorship. GF and GP organised the meeting; GP and AC provided assistance to the leading authors on behalf of CephRes.
This manuscript provides the basis for the activities of the FA1301 COST Action ‘A network for improvement of cephalopod welfare and husbandry in research, aquaculture and fisheries (CephsInAction; http://www.cost.eu/COST_Actions/fa/Actions/FA1301; ![]() )' aimed to facilitate and support the cephalopod community in meeting the challenges resulting from the inclusion of cephalopod molluscs in Directive 2010/63/EU.
)' aimed to facilitate and support the cephalopod community in meeting the challenges resulting from the inclusion of cephalopod molluscs in Directive 2010/63/EU.
We are grateful to Professor Torsten Wiesel for his enthusiasm and genuine interest for cephalopod biology research.

