Abstract
Consideration of The Three R’s (Replacement, Reduction, and Refinement) is essential when setting Humane Endpoints; however, a common interpretation assumes that Humane Endpoints are timepoints to perform euthanasia. This interpretation is not always consistent with the three Rs. There are many available intervention options that – when used to respond to pain, discomfort, or distress – facilitate application of the three Rs while achieving experimental goals. At our institution, the term ‘Humane Endpoints’ was replaced with ‘Humane Intervention Points’, to ensure responses beyond euthanasia are given priority by laboratory animal professionals and researchers. Identification of Humane Intervention Points in the research protocol provides us with a more accurate reflection of the measures used to alleviate pain and distress in animals used for research, testing, and teaching – an outcome easily envisioned elsewhere.
Points d’intervention humains (PIH): Affinage de la terminologie d’évaluation finale pour intégrer des options d’intervention sans recours à l’euthanasie afin d'améliorer le bien-être des animaux et de préserver les résultats expérimentaux Résumé
Introduction
The concept of Humane Endpoints (HEPs) has been adopted globally for the last several decades as a means to apply The Three R’s of humane technique – Replacement, Reduction, and Refinement – proposed by WMS Russell and RL Burch, to improve treatment of research animals while advancing the quality of scientific and medical research and testing. 1 Ideally, by implementing HEPs, pain and/or distress can be prevented or alleviated whilst still meeting experimental objectives. 2 A common response to an animal reaching the HEP has been to perform euthanasia because either the animal has declined to a point that spontaneous rather than enacted death is imminent or likely, and the protocol has not specified early intervention options to prevent such a decline. The tendency to equate the term ‘HEP’ with euthanasia is incongruent with the application of the three Rs – in particular, Reduction and Refinement – and can have a detrimental impact on both animal welfare and research outcomes; thus, using terminology which describes non-euthanasia responses to potential pain and discomfort of animals on study is warranted. By contrast, the term ‘Humane Intervention Points’ (HIPs) applies to both the advancement of research and enhancement of animal welfare by describing other humane options for responding to animal distress or discomfort.
Evolution of terminology
Two issues exist with the common interpretation of the term ‘HEP’. First, the literal interpretation of the word endpoint as used in common language as ‘the final stage of a period or process’ 3 often leads to the conclusion that upon reaching the HEP, the appropriate action is to end a study animal’s life by euthanasia. Second, it is not uncommon for research and veterinary personnel to differ in their views of what determines the final stage of the experimental process for a research animal. Historically, laboratory animal professionals often referred to HEPs simply as Endpoints, with the assumption that the implied interpretation of the ‘humane’ element was consistent across research and laboratory animal communities. However, rather than identifying timepoints and responses to humane concerns, researchers’ interpretations of Endpoints related to terminal data collection timepoints – for example, euthanasia for tissue harvesting at the end of a treatment period. These potentially opposing perspectives of what constitutes an Endpoint generate a potential conflict between researcher and veterinarians about when to end the life of a study animal (Figure 1).
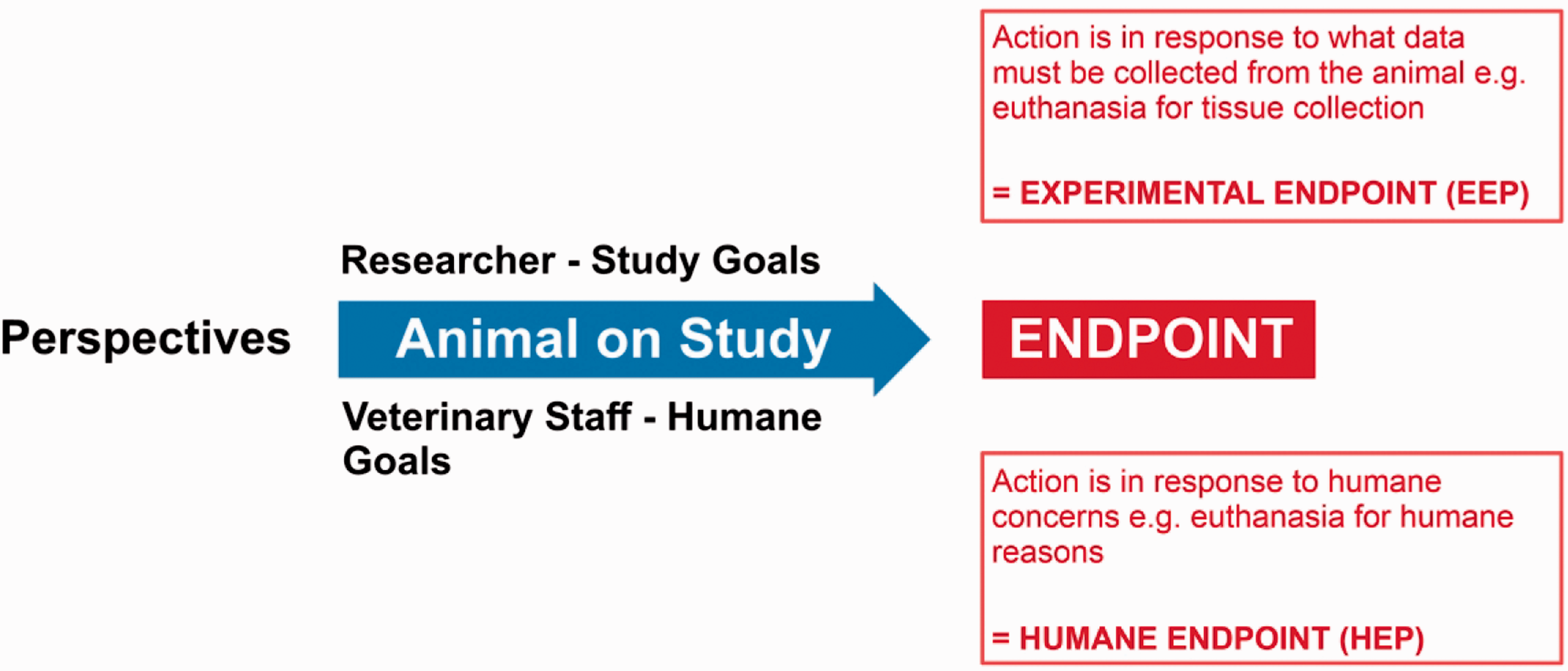
Different perspectives on the meaning of the term ‘endpoint’ according to researchers and veterinary staff. There is a tendency to equate the term with a requirement for euthanasia of a research animal. From the researcher’s perspective, the timing of euthanasia – as a response to reaching an endpoint – is based on achieving experimental goals – that is, the EEP. Whereas the veterinary staff, animal technologists, and husbandry staff interpret the need to euthanize a research animal as a response to alleviate humane concerns – that is, the HEP.
Revisions to the Guide for the Care and Use of Laboratory Animals (The Guide) (p.27)4 include the following definitions to address the different perspectives on the term ‘Endpoints’: ‘The experimental endpoint of a study occurs when the scientific aims and objectives have been reached. The humane endpoint is the point at which pain or distress in an experimental animal is prevented, terminated or relieved’. Distinguishing HEPs from Experimental Endpoints (EEPs) provides clarity to researchers on the need to plan beyond the scientific outcomes. Still, the term ‘HEP’ implies euthanasia as the go-to response to animal discomfort. For example, The Guide (p.123)4 states, Criteria for euthanasia include protocol-specific endpoints (such as degree of a physical or behavioral deficit or tumor size) that will enable a prompt decision by the veterinarian and investigator to ensure that the endpoint is humane and whenever possible, the scientific objective of the protocol is achieved.
Equating HEP with euthanasia is less consequential when the study goals are achieved in a humane manner. When the EEP is reached in the absence of humane issues, the animal remains on study until the scientific objectives are met. In ‘The guiding principles for the care and use of animals for scientific purposes’, the Singapore National Advisory Committee for Laboratory Animal Research (NACLAR) 5 uses the following language: ‘The investigator should develop humane study end-points when preparing a project application’, so that regardless of the timepoint at which the EEP occurs, there is a humane outcome. The animal has reached a Humane Experimental Endpoint (HEEP) satisfying both experimental and humane goals (Figure 2(a)).
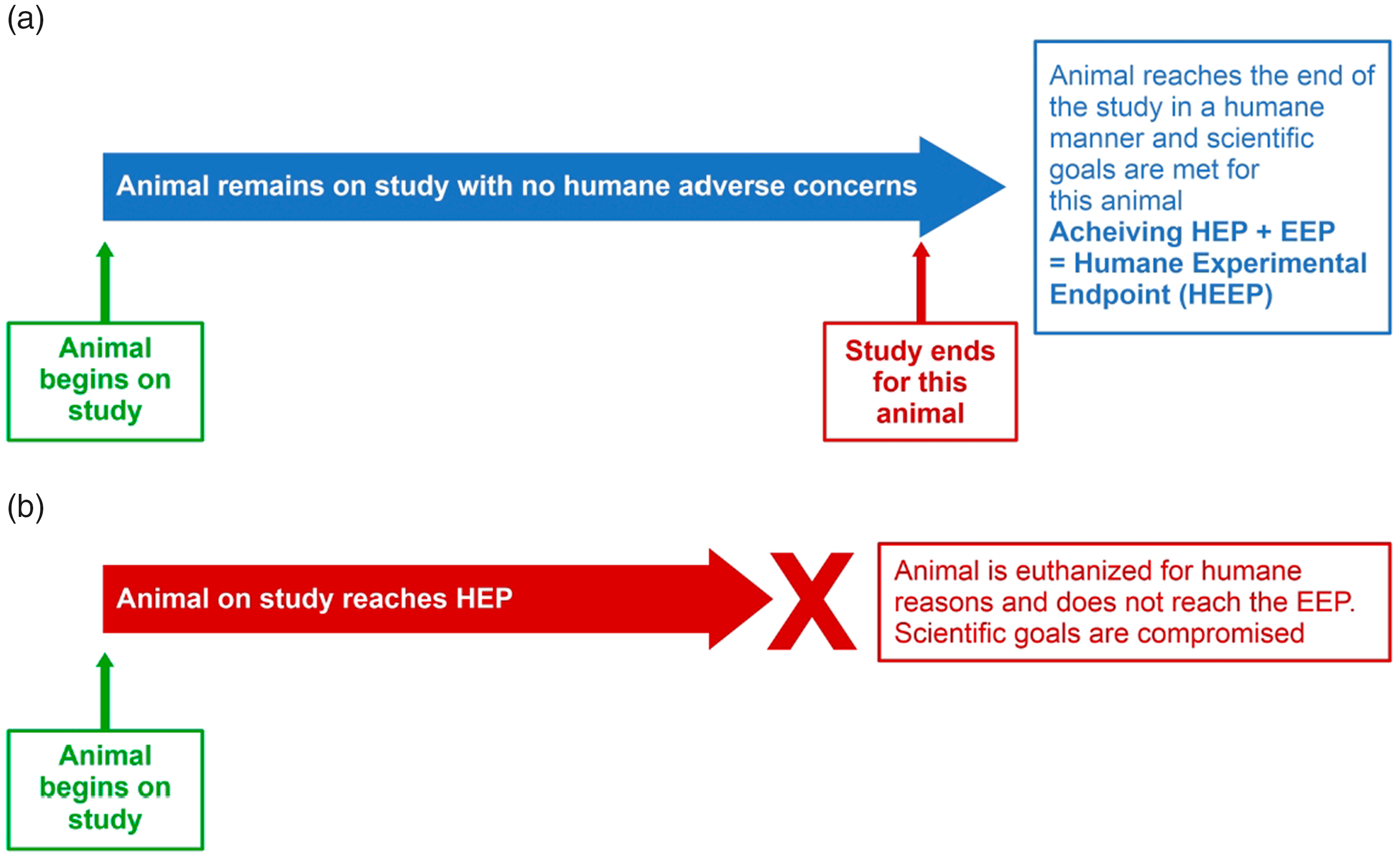
Positive outcomes may be limited when equating HEP with euthanasia. (a) Equating HEP with euthanasia is not always problematic (e.g. when an animal remains free of humane concerns throughout the course of a study and the EEP is achieved in a humane manner). In this case, achieving the EEP essentially achieves a HEEP. Designing studies to achieve HEEPs is ideal but not always feasible. (b) Equating HEP with euthanasia becomes a concern if the HEP occurs before the EEP is achieved, because the animal is euthanized prior to achieving scientific goals; therefore, the EEP is not met.
Guidelines on HEPs encourage the use of Early Experimental Endpoints (EEEPs), 6 when feasible, to avoid euthanasia of an animal that reaches a HEP before data collection is achieved – for example, the use of biomarkers as early indicators of pain or distress. However, if applying an EEEP is not feasible and we assume the common interpretation of euthanasia as the only response to reaching a HEP, animals may undergo euthanasia prior to reaching the scientific goals (Figure 2(b)).
The focus on euthanasia as the default response leaves researchers cautious about setting HEPs at earlier humane timepoints. To avoid euthanasia of animals prior to reaching the EEP, the HEP researchers identify might not be as early as necessary to be considered more humane. In addition, with euthanasia as the only specified response, animals remain on study in the absence of any intervention until the HEP is reached, which is not a humane approach for these animals.
If the proposed action plan for an animal reaching the HEP is euthanasia, this plan may not align with the three Rs. Rather than reducing the number of animals used, there may be a need to increase animal numbers to account for the loss of animals that were euthanized prior to completion of the study. The Guide (p.27)4 states that ‘[t]he use of humane endpoints contributes to refinement by providing an alternative to experimental endpoints that result in unrelieved or severe animal pain and distress including death’; thus, studies should be designed to include refinements that facilitate data collection in the absence of pain and distress. When euthanasia is the only specified action for animals deemed to have reached the HEP, refinement of animal use may not be achieved. Unless a protocol specifies proactive responses to prevent or treat an animal’s declining condition, the condition will be allowed to deteriorate until the HEP is reached. Only then will a response occur in the form of euthanasia (Figure 3).
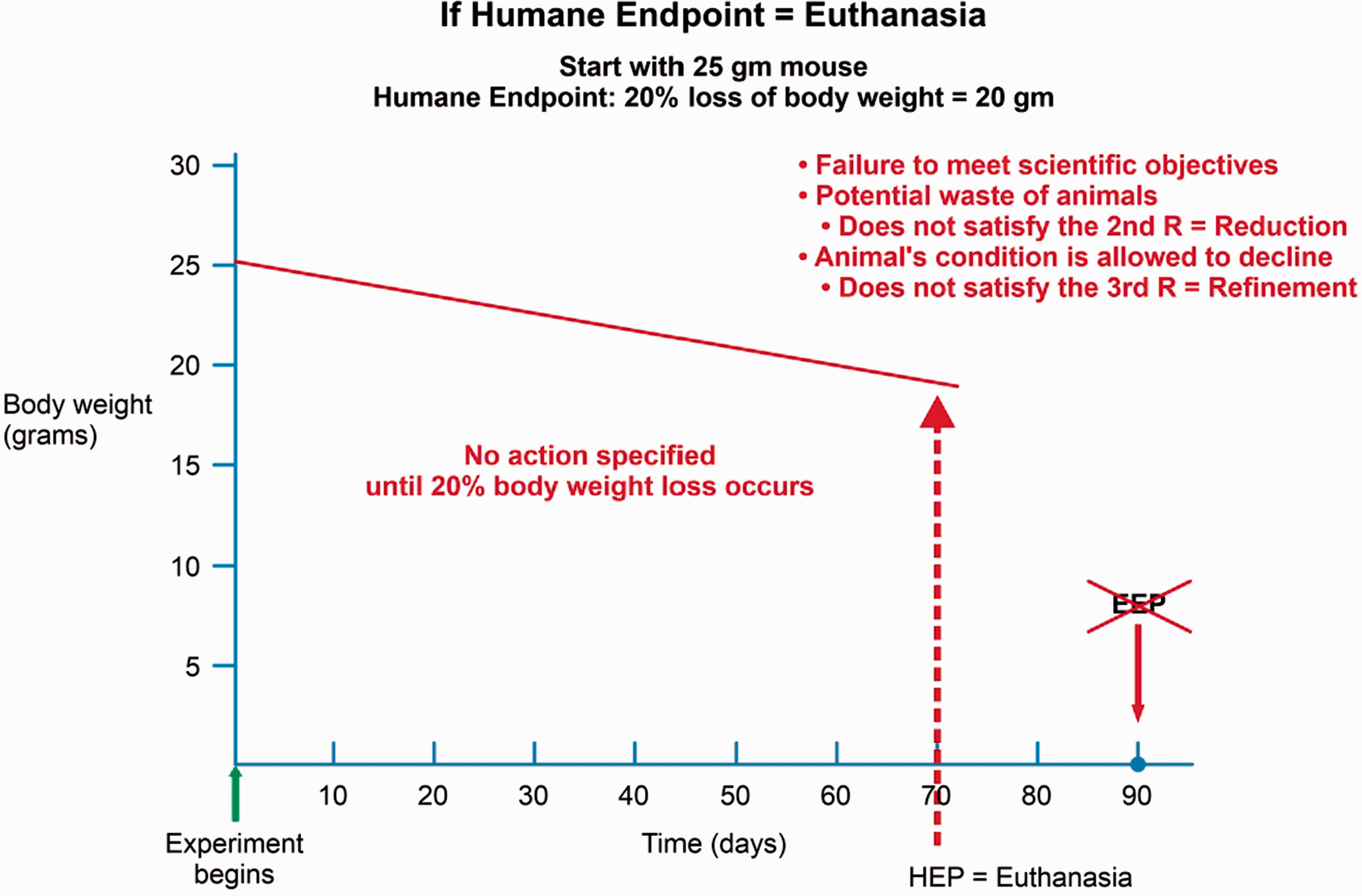
Example of how equating endpoint with euthanasia can compromise the 3 Rs. An animal reaching a HEP set at 20% body weight loss – with euthanasia as the response – will not reach the EEP. Additional animals may be required to achieve scientific goals: thus, compromising the 2nd R, Reduction. Further, the animal’s condition is allowed to decline with no intervention, until the HEP of 20% body weight loss is reached, followed by euthanasia; thus, the 3rd R, Refinement is compromised.
Implementing the term ‘HIPs’
As the terminology has evolved, many HEP definitions specify actions to alleviate, prevent, terminate, and/or treat pain or distress.6–9 Common interventions include but are not limited to provision of analgesia, supportive care, and modification of husbandry practices to support comfort – for example, increasing accessibility to food and water. If an animal reaches a point in which pain and distress cannot be alleviated by other interventions, euthanasia is a terminal intervention option. It is also important to consider that the end of an animal’s time on study need not always result in the end of the animal’s life. For animals which are clinically healthy, adoption or appropriate reuse are two options for consideration over euthanasia. The non-euthanasia responses can be described as humane interventions. These interventions occur at timepoints identified as HIPs (Figure 4).
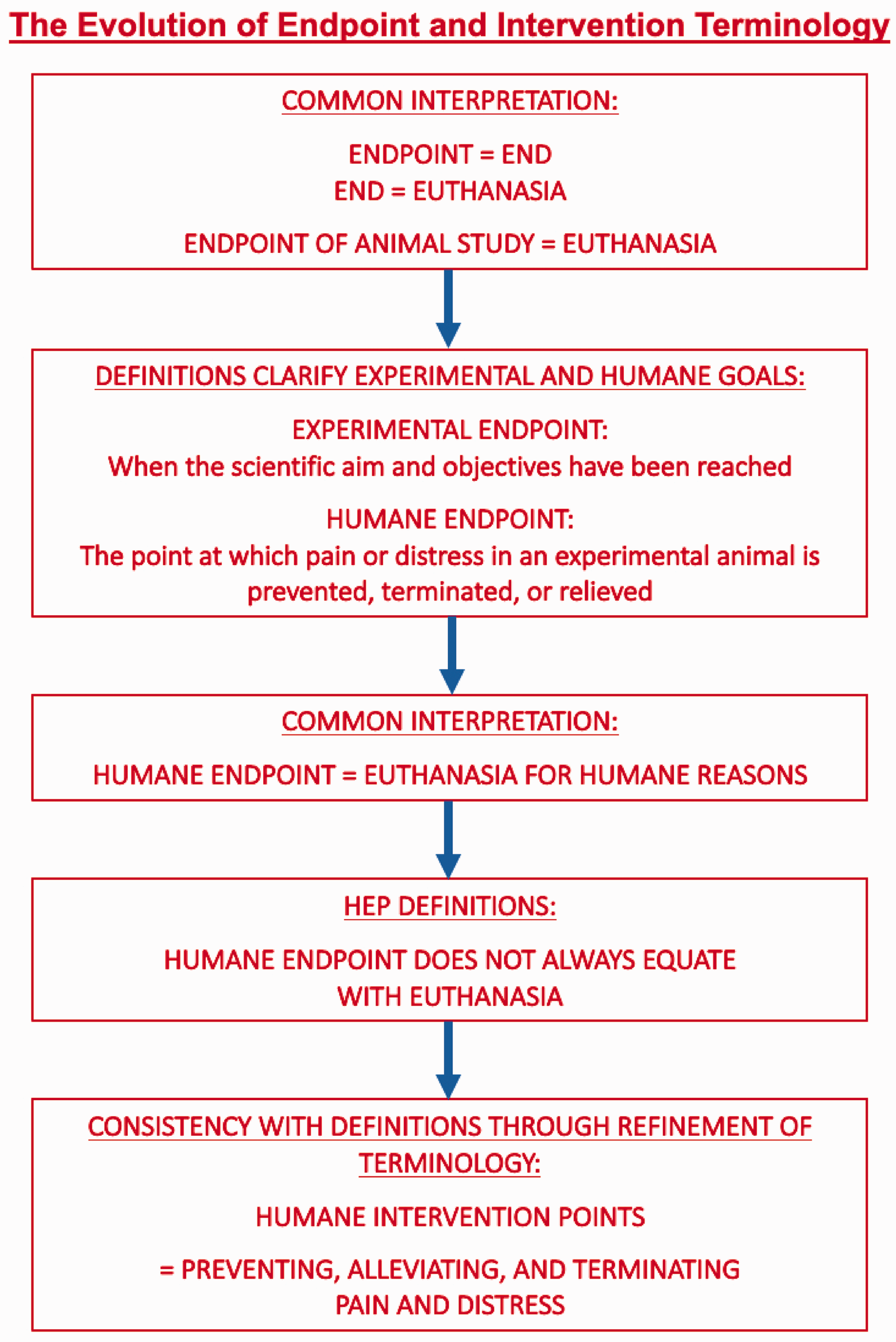
Refinement of HEP and HIP Terminology. The term, endpoint, has been interpreted as the point at which an animal’s time on study ends with euthanasia. Providing definitions of EEP and HEP distinguishes the experimental from humane goals; however, there remains an interpretation of euthanasia as the sole action upon reaching a HEP. Acknowledging that current definitions of HEP describe actions beyond euthanasia, the term, HIP provides further evolution of terminology which is consistent with non-euthanasia options for intervention.
In adopting the term ‘HIPs’ at our institution, our goal was to maximize the chance of an animal reaching the scientific goals in the most humane manner possible. Researchers and laboratory animal veterinarians collaborate to identify 1) the relevant indicators (e.g. clinical signs, biomarkers, pre-symptomatic euthanasia) to mark the most appropriate timepoints for implementing interventions specific to a study, and 2) the most beneficial interventions for achieving experimental goals as humanely as possible, thus preserving the integrity of the science and the welfare of the research animals. Through the Institutional Animal Care and Use Committee (IACUC)/Ethics Committee protocol preparation and review process, research and veterinary personnel work together to predict events that may adversely impact the welfare of the animals (Figure 5). When it is feasible to anticipate the events causing pain or distress (related to the research model, study design, and experimental manipulations), it is also conceivable to plan responses to pain and distress using predetermined intervention criteria and plans; these HIPs are described in the research protocol. Should an animal on study reach a predetermined HIP, the predetermined intervention is applied as per the IACUC/Ethics Committee protocol, using humane measures that will facilitate the animal staying on study to meet the scientific goals (Figure 6).
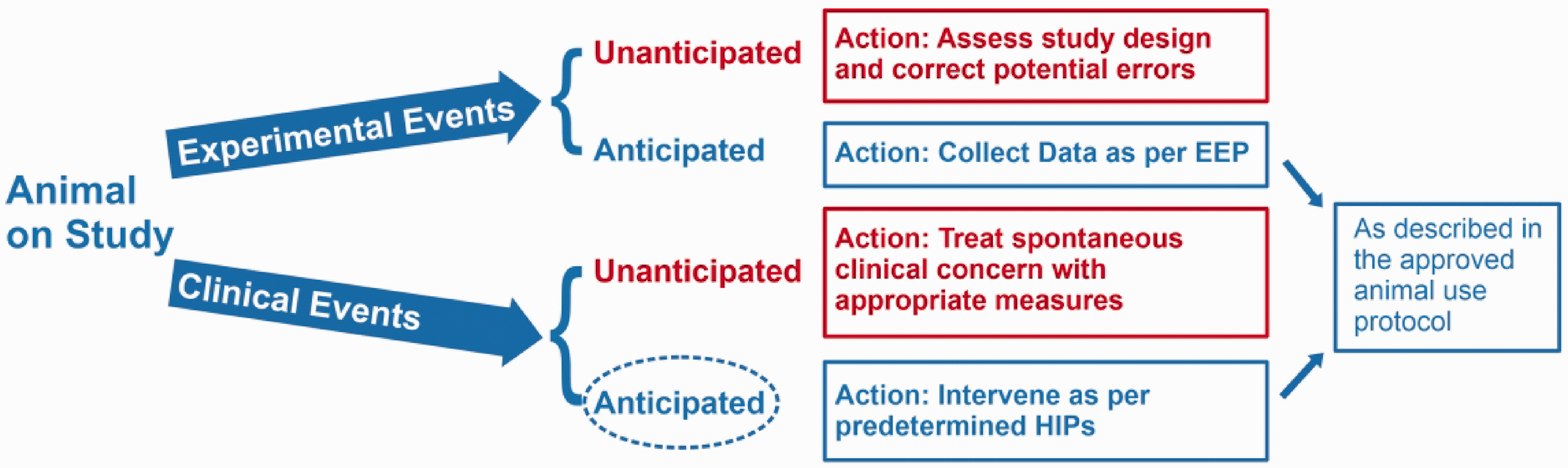
Implementing HIPs to manage anticipated clinical events while preserving scientific goals. During a study, animals experience experimental or clinical events which initiate actions from researchers or veterinary staff. These actions may differ depending on whether the events are anticipated or unanticipated. Identifying anticipated clinical events facilitates identification of criteria and time points for intervention. Like the anticipated experimental events, the anticipated clinical events and intervention responses are addressed in the animal use protocol as HIPs. Figure 7. Example of implementing HIPs to facilitate positive outcomes for animal well-being and data collection. Using HIPs in place of HEPs, interventions are set to occur at multiple points, starting at 5% body weight loss. The interventions prevent further decline in the animal’s condition, increasing the likelihood a humane experimental outcome.
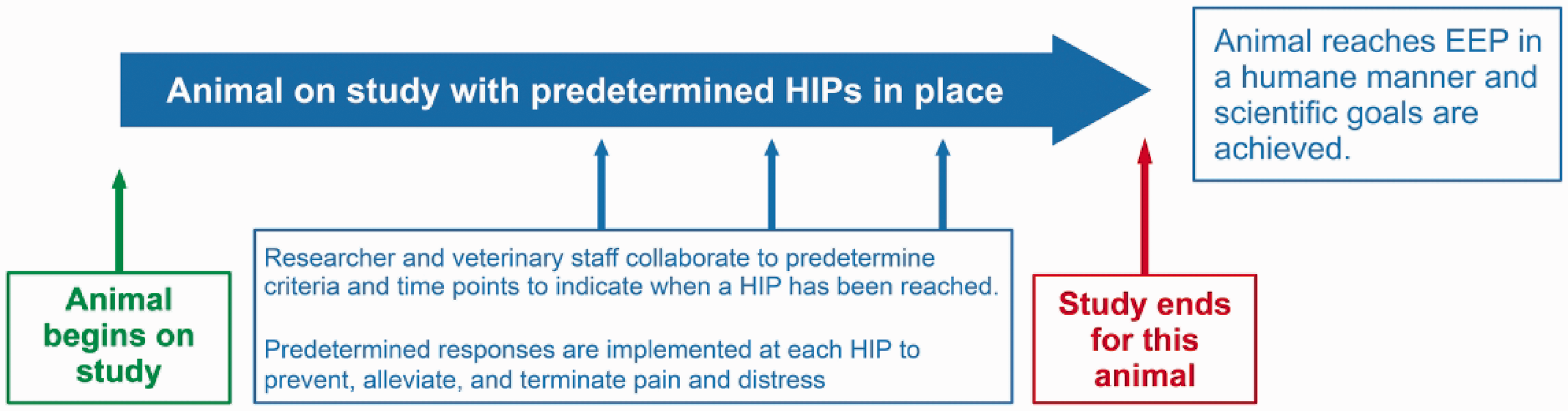
Implementing HIPs to achieve humane data collection. Together, researcher and veterinary staff work to predict possible events that may adversely impact the welfare of the animal. This collaborative effort serves to create HIPs which are most likely to ensure animal well-being throughout the study to facilitate achieving the EEP with minimal confounding of data.
The predetermined agreement on such interventions helps to prevent discrepancies and resistance from the researchers or the veterinary staff intervening as described in the approved research protocol. Incorporating HIPs into the protocol potentially shifts the focus to determining responses which alleviate pain and distress, rather than waiting to terminate the lives of animals that experience pain or distress as HEP implies (Figure 7). It is both reasonable and important to continue to assess the HIPs to ensure they are serving the purpose of preserving the welfare of the animal and integrity of the science. Where practical, all animals in all groups should be provided with the same interventions (e.g. moist chow) at the same time and in the same amount per cage, even if clinically normal. Researchers should include relevant information about the HIPs in their journal manuscripts for publication. The value of replacing the term ‘HEP’ with ‘HIPs’ goes beyond our institution and we encourage that HIPs be added to the terminology more widely around the globe.
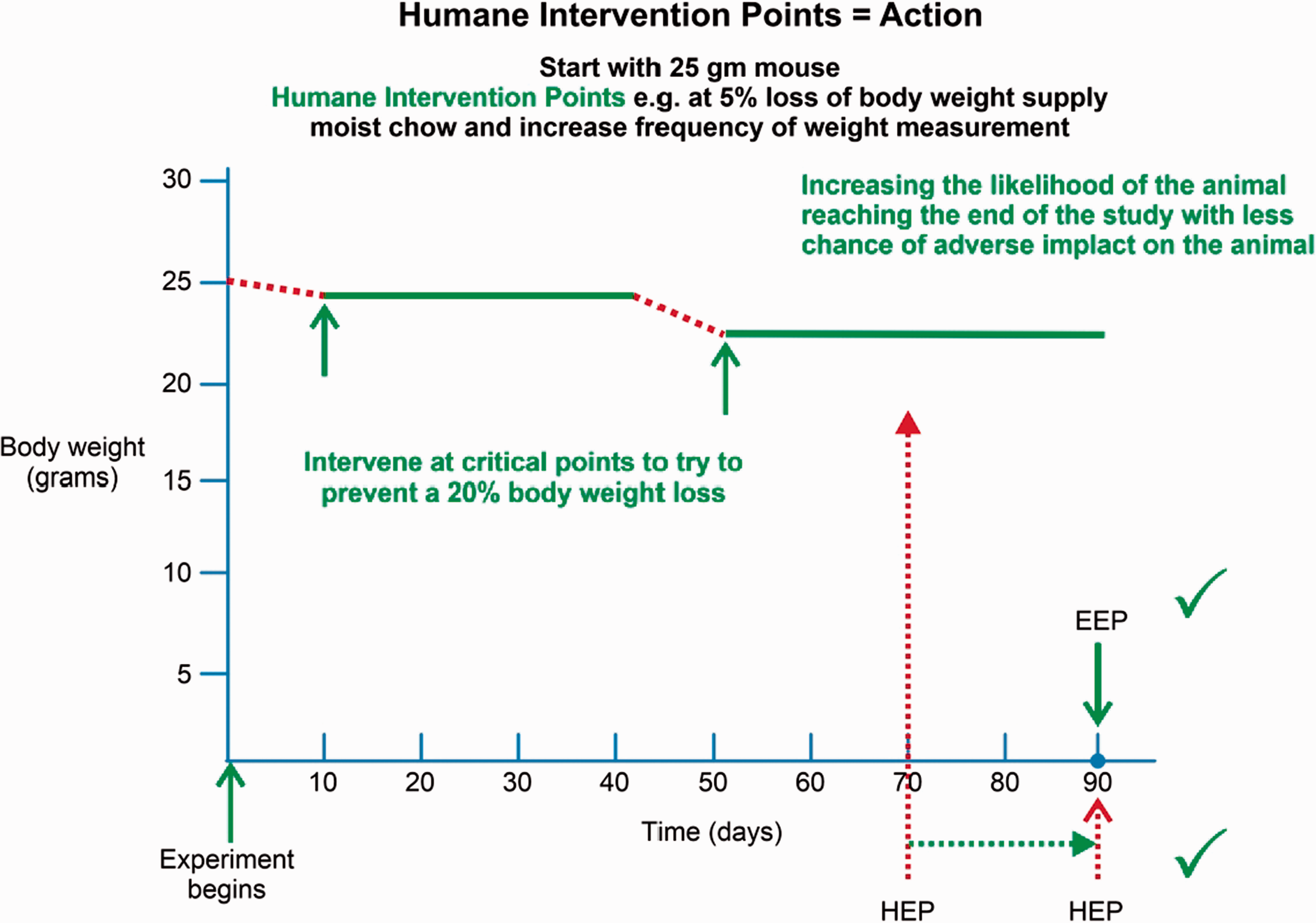
Example demonstrating that implementing HIPs improves the chances of a positive outcome for animal well-being and scientific data collection. In Figure 3, the example showed HEPs set at 20% body weight loss, such that no action was required until 20% body weight loss occurred, then euthanasia was set as the only option for action. In a similar example, using HIPs in place of HEPs, the intervention actions incorporated into the research protocol are specified at multiple time points, and start at a lower percentage of body weight loss (e.g. 5%). The resulting actions are intended to prevent further weight reduction from occurring. Rather than allowing the body weight to decrease to the HEP of 20% body weight loss, the intervention responses are designed to prevent decline in the condition of the animal and from reaching a point at which euthanasia is required for humane reasons. Intervening to prevent further decline helps increase the likelihood of that animal reaching the end of the study while decreasing the chance of adverse impact on the animal. The result is a humane experimental outcome through the implementation of HIPs. Where practical, all animals in all groups should be provided with the same interventions (e.g. moist chow) at the same time and in the same amount per cage, even if clinically normal. The implemented HIPs should be addressed in the published manuscripts.
Conclusions
Refinement to the current EEP and HEP terminology is appropriate and necessary, specifically including the term ‘HIPs’ to describe a variety of protocol-specific responses to the humane concerns that animals experience during an experiment, and to encourage consideration of non-euthanasia options for intervention. By implementing the term ‘HIPs’ in place of ‘HEP’, researchers have a better understanding of expectations surrounding responses to anticipated clinical events that arise during their studies. Using HIPs promotes joint ownership in defining humane outcomes for animals on study. Through appreciation of the researchers’ perspectives, veterinary staff are better informed to respond to concerns by offering guidance and services to assist the researchers in setting HIPs that can improve outcomes for both animals and research studies.
Footnotes
Acknowledgements
The authors extend their gratitude to Drew Kirby of the Center for Animal Resources and Education at Cornell University for his assistance in formatting the original figures.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
