Abstract
Murine translational models are an important tool to understand pain pathophysiology. One procedure used frequently in murine research is the sciatic nerve block. This study sought to demonstrate the use of ultrasound-guided sciatic nerve block in a cadaveric murine model. A total of 40 injections were performed in 20 Sprague–Dawley male 18-month-old rat cadavers. Necropsy was performed to identify staining of the sciatic nerve. Staining with methylene blue occurred in 40 of 40 ultrasound-guided injections. The extremely accurate nature of this block under ultrasound guidance is favorable for future translational studies in rats undergoing sciatic nerve blocks. This method may represent a significant improvement in current methods.
Introduction
Animal research is an important tool to understand the pathophysiology of pain conditions. The murine model is used commonly in pain research. One useful aspect of this model is the ability to perform nerve blocks. In humans, nerve blocks reduce pain associated with injury or surgery. The rodent sciatic nerve block evaluates functional blockade or nerve regeneration under various conditions. The technical procedure of the sciatic nerve block is achieved with either a landmark based or open approach. 1 , 2
The landmark approach carries risk for inadequate blockade due to small injection volume (0.1–0.2 ml) and blind placement. The open approach is more accurate, but induces trauma from skin and muscle dissection. Recent studies favor the open approach. The surgery may affect the translational nature of the model due to post-surgical pain, 3 hyperalgesia, 4 and behavioral changes. 5 The ideal nerve block would be both accurate and minimally invasive. It is possible ultrasound guided sciatic nerve blockade may represent the ideal technique.
In humans, peripheral blocks are typically performed with ultrasound guidance. The benefits include decreased likelihood of injury and direct visualization of the nerve. 6 Recent studies have demonstrated success with ultrasound to identify the rat sciatic nerve. 7 A single previous study used ultrasonographic contrast injectate in rats to assess for neurotoxicity, but this was done in an open (not percutaneous) block. 8 We sought to demonstrate the efficacy of simple ultrasound-guided sciatic nerve injection in a cadaveric rat model.
Animals, materials, and methods
This study was approved by the Naval Medical Center Portsmouth (NMCP) Institutional Animal Care and Use Committee (IACUC) in compliance with all applicable Federal regulations. All procedures were performed in AAALAC International accredited facilities. A total of 20 male Rattus norvegicus Sprague–Dawley (Hsd:SD strain, Envigo, Frederick, MD, USA) cadavers of approximately 18 months of age and 450–495 g were used in the study, under a tissue sharing agreement from a separate institutional animal care and use committee (IACUC)-approved study. While at our facility, animals were pair-housed in individual ventilated cages (Optirat GenII, Animal Care Systems, Centennial, CO, USA) in environmentally controlled rooms maintained at 20–26°C, relative humidity 30–70%, and a 12-h:12-h light:dark cycle. During the 3-day acclimation period, rats were provided a commercial rodent diet (8604 Teklad rodent, Envigo, Madison, WI, USA) and municipal water (filtered and chlorinated, 2 ppm) ad libitum via an automatic watering system (Edstrom Industries, Wakefield, WI, USA). Food enrichment items such as Fruit and Veggie medley (Bio-Serv, Flemington, NJ, USA), as well as rat tunnels, nylon bones, and wood blocks. Prior to euthanasia, animals were anesthetized with urethane (1000–1250 mg/kg IP) followed by euthanisia via intracardiac injection of >150 mg/kg pentobarbital-based solution (Euthanasia Solution, VetOne, MWI, Boise, ID, USA). Cadavers were then placed in a 4°C refrigerator approximately 24 h after euthanasia. This was designed as a pilot cadaveric study, and no controls were used in this investigation.
Cadavers were placed in the lateral position with access to the non-dependent lateral hind limb. The hind portion was shaved using standard clippers without shaving cream or gel. The high frequency 5–10 MHz (Sonosite L25 Linear Probe) ultrasound (Sonosite X-Porte, Fujifilm Sonosite, Inc., Bothell, WA, USA) was placed on the lateral side of each hind limb at the mid-thigh and gel was placed to facilitate sound transmission (Aquasonic 100 Ultrasound Transmission Gel, Parker Laboratories, Inc., Fairfield, NJ, USA). The femoral trochanter served as a landmark to achieve orientation (Figure 1a). Reliable placement of the ultrasound was completed by identifying the femoral trochanter (circle, Figure 1a) by palpation, and drawing a line in a parasagittal plane caudally. The ultrasound transducer probe is then placed overlying the line, with the most cephalad portion of the transducer adjacent to the trochanter (Figure 1b). Following this technique, the sciatic nerve was identified reliably on ultrasound. Small adjustments were usually required to provide optimal image quality, most often tilting the probe ventrally, so the ultrasound beam was directed dorsally.
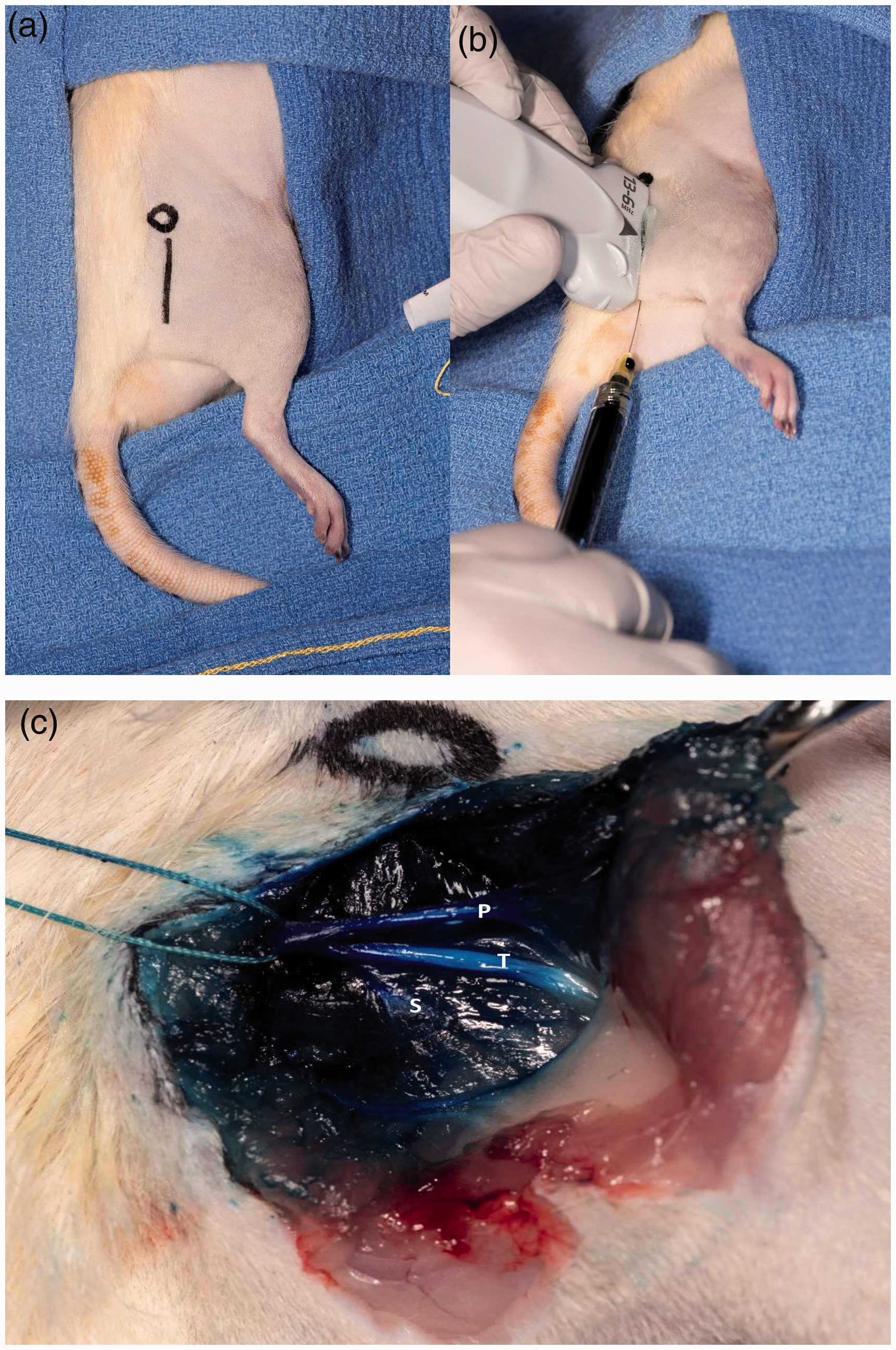
(a) Landmark anatomy of femoral trochanter (circle) and placement of ultrasound, and (b) ultrasound position and needle insertion. Typical staining pattern following injection. (c) Silk suture placed around sciatic nerve, with trifurcation visualized of peroneal (P), tibial (T), and sural (S) nerves.
The nerve was readily identified as an oval hypoechoic area underneath the gluteal fascia (Figure 2a). The sonoanatomy revealed a thin strip of skin and subcutaneous tissue, immediately followed by the gluteus muscle. The gluteal fascia appeared as a thin white line (Figure 2a), within which the sciatic nerve reliably appeared as a hypoechoic structure adjacent to the fascia.
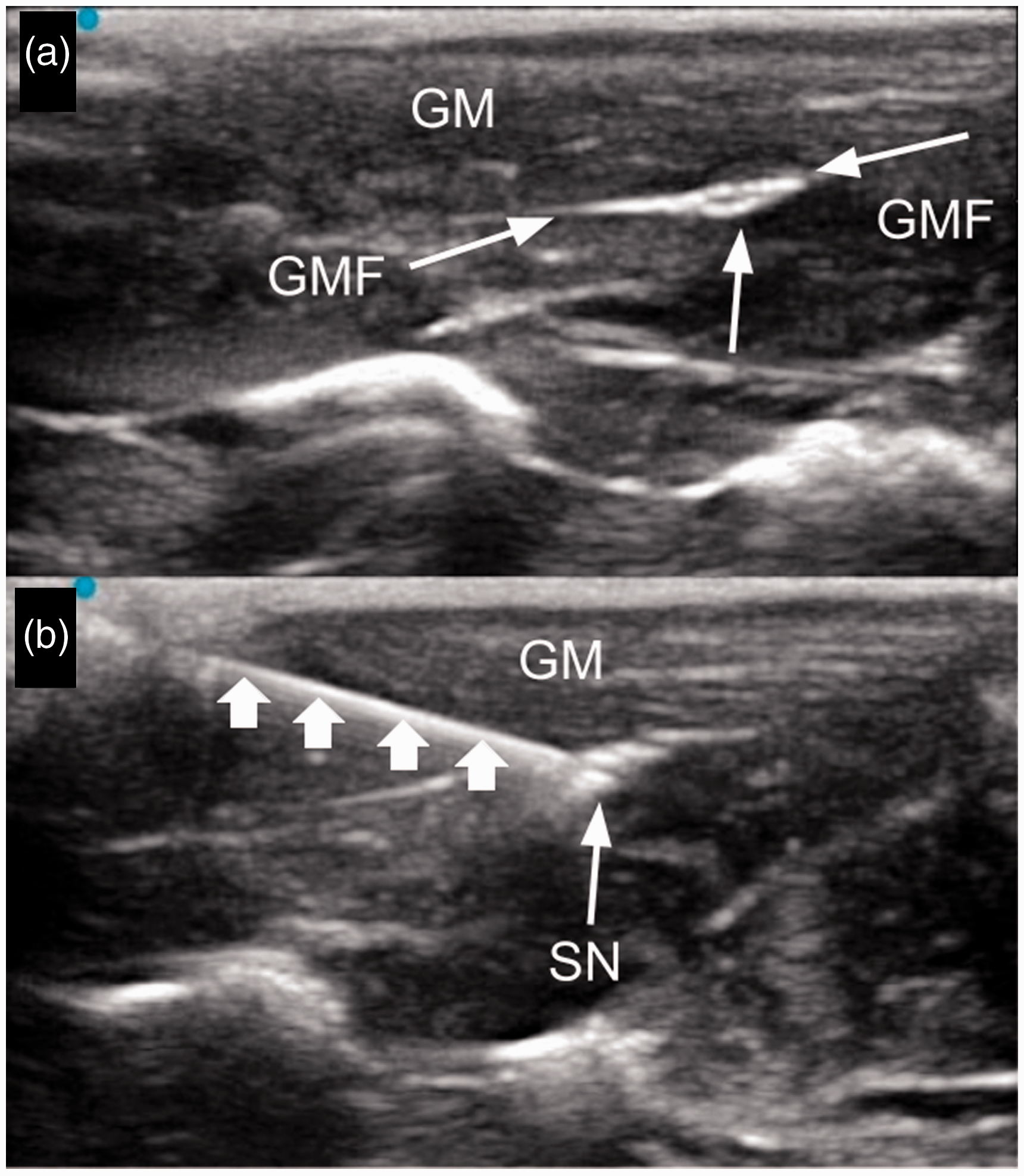
(a) Representative image of sciatic nerve (SN) laying underneath the gluteus muscle (GM). This image demonstrates the thin line of skin and subcutaneous tissue, followed by a darker band of tissue representing the annotated gluteus muscle. The thin white line immediately beneath represents the gluteus fascia (GMF), and the hypoechoic ovoid structure represents the sciatic nerve (SN). (b) Representative image of in-plane needle placed (small arrows) adjacent to the sciatic nerve (SN). Gluteus muscle is above (GM).
Once the nerve was identified, a 30G needle was inserted in an in-plane approach (Figure 2). When the needle tip was adjacent to the nerve, 0.2 ml (1:4 0.5% methylene blue and 0.9% saline) was injected. This was repeated on the contralateral hind limb after repositioning to the opposite lateral position. Immediately following injection, necropsy was used to confirm staining of the sciatic nerve. Lateral incision over the hind limb was performed, with dissection of the gluteal and femoral biceps muscles to reveal the sciatic nerve (Figure 1). The injection was considered adequate if methylene blue staining was present at the sciatic nerve.
Results
A total of 20 Sprague–Dawley rat cadavers underwent bilateral ultrasound guided nerve injections, for a total of 40 injections. The sciatic nerve was identified on ultrasound in 40 of 40 procedures; 40 of 40 sciatic nerves were identified on necropsy. Of the 40 injections performed, all 40 stained the sciatic nerve (100% accuracy). The staining pattern reliably covered the sciatic nerve as it trifurcated into the sural, peroneal, and tibial nerves.
Discussion
This study evaluated the accuracy of performing non-invasive ultrasound guided sciatic nerve blocks in a murine model, and serves as a refinement over currently used techniques. The consistency suggests it is a reliable method for future studies. Reducing surgical stress in animal models that require sciatic nerve blockade, an ultrasound-guided sciatic nerve block may help limit confounding variables. Similarly, because of the ability to see the spread of the injectate surrounding the nerve, volumes less than 0.2 ml are likely to be effective, and may reduce toxicity associated with local anesthetics.
The technical performance of this ultrasound-guided procedure was not challenging and could be performed by individuals with experience in ultrasound-guided needle placement techniques. The proceduralist in this study has significant experience in ultrasound-guided regional anesthesia techniques in humans; however, this was not a technically challenging procedure. Ultrasound guided nerve blocks are increasingly being used in veterinary medicine, 9 and this represents a reasonable extension of prior large animal studies. Adaptation for this procedure for use in mice may be difficult or impossible due to the decrease in size. This would be a reasonable further investigation which may help refine the mouse sciatic nerve block as well, and may require the use of an ultra-high frequency ultrasound, such as those in the 22 MHz, 33 MHz, or 70 MHz range.
There are several important limitations for this study. The use of a cadaveric model is limited in functional response. No assessment of block density or duration could be assessed. Similarly, there was no control group as this merely an attempt to suggest reliability of the de novo technique, future studies should compare functional outcomes, comparing ultrasound with landmark studies. That said, cadaveric studies using dye are often used to predict block performance. 10 The staining pattern seen with this study reflects other cadaveric studies, and the methylene blue staining of surrounding tissue likely reflects the natural spread that occurs in vivo of local anesthetic. In vivo nerve blocks have a wide spread of local anesthetic with little clinical impact. This has also been used in veterinary medicine for the same purpose. 11 , 12 While functional studies are necessary to validate this as the preferred method for murine sciatic nerve block, it provides potentially significant improvement to current methods.
Animal welfare statement
The experiments reported herein were conducted in compliance with the Animal Welfare Act and Regulations and per the principles of the ‘Guide for the Care and Use of Laboratory Animals,’ Institute of Laboratory Animals Resources, National Research Council, National Academy Press, 1996 Research data derived from a tissue sharing agreement with a Naval Medical Center Portsmouth IACUC approved protocol, #NMCP.2019.0060.
Footnotes
Data availability
All data can be made available by contacting the corresponding author at
We are military service members. This work was prepared as part of my official duties. Title 17 U.S.C. 105 provides that “Copyright protection under this title is not available for any work of the United States Government.” Title 17 U.S.C. 101 defines a United States Government work as a work prepared by a military service member or employee of the United States Government as part of that person’s official duties.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
