Abstract
Fracture healing is a complex regeneration process which produces new bone tissue without scar formation. However, fracture healing disorders occur in approximately 10% of human patients and cause severe pain and reduced quality of life. Recently, the development of more standardized, sophisticated and commercially available osteosynthesis techniques reflecting clinical approaches has increased the use of small rodents such as rats and mice in bone healing research dramatically. Nevertheless, there is no standard for pain assessment, especially in these species, and consequently limited information regarding the welfare aspects of osteotomy models. Moreover, the selection of analgesics is restricted for osteotomy models since non-steroidal anti-inflammatory drugs (NSAIDs) are known to affect the initial, inflammatory phase of bone healing. Therefore, opioids such as buprenorphine and tramadol are often used. However, dosage data in the literature are varied. Within this review, we clarify the background of osteotomy models, explain the current status and challenges of animal welfare assessment, and provide an example score sheet including model specific parameters. Furthermore, we summarize current refinement options and present a brief outlook on further 3R research.
Osteotomy models
Fracture healing is a complex regeneration process, starting with a fracture hematoma that is characterized by closely regulated inflammatory events and a hypoxic microenvironment which initiates the healing cascade. 1 The following healing phases include the formation of a fibrocartilaginous callus, the transformation to mineralized cartilage, the restructuring with woven bone, and finally the remodeling. The first phase of bone healing is particularly important, and includes the inflammatory processes within the fracture hematoma as well as optimal fixation stability. 2 To develop and test new therapeutic strategies, animal models are needed in bone healing research. Besides the physiological direct or indirect fracture healing, clinical human scenarios such as delayed union, established hypertrophic or atrophic non-union (pseudarthrosis), segmental bone defects, and multiple traumata are of great interest.
Rodent models are often used because of their advantages such as the ready availability of genetically-modified animals, high reproductive rates, low costs, easy housing, the existence of readily available research kits, and their known genome sequence. Nevertheless, there is no single animal model that replicates all the key events of a human patient, and therefore animal models should be adapted to the research question. More than half of the animals used in orthopedic research are rats (38%) or mice (15%). 3 The development of more sophisticated osteosynthesis techniques with more clinically relevant stabilization has increased the number of small rodents (including rabbits; together >80%) used in bone healing research.4,5 Most animal species show slight analogies to the human bone macro- and microstructure. The main differences in mice concern the permanent opening of the growth plate in the epiphyses of long bones leading to a lifelong skeleton modeling, the lack of a Haversian system and low cancellous bone content at the epiphyses of long bones.5,6 Mice are appropriate for basic research (transgenic modifications), while rats are more suitable for pharmacological interventions, toxicological studies and, due to their bigger size, for biomaterial approaches.7,8
Well-established osteotomy models of long bones in rodents exist with stabilization possibilities using external fixators, plates or intramedullary pin/locking nails.9–11 The most common localization is the femur, while models also exist for the tibia. Each different stabilization possibility bears the potential of different gap sizes varying between 1–8 mm in rats and 0.1–4 mm in mice.11,12 Pathophysiological processes such as bone healing disorders are created by forming an unstable fixation or by creating critical size defects, cauterization of the periosteum or removal of the bone marrow. Nevertheless, for any defined experiment each model has its own advantages and disadvantages. While the external fixation prevents the direct interaction of the device with the defect and interferes with the bone marrow only through the pins, it is especially suitable for cell-based approaches and biomaterial testing. The disadvantage is its position outside the body that tempts the animal to manipulate the device and increase the risk of infection due to direct communication between the outside environment and internal structures. Therefore, the plate features a closure of fascia and skin over the device that prevents manipulation and decreases the susceptibility to infections. Even more important is the increase in mechanical stability due to a lower offset of the fixation plate. Therefore, it is more suitable for larger defects, which prevent osteolysis at the pins. However, the plate affects the periosteum and changes the bone healing process at the site of the mounted plate. By contrast with the other two models, the intramedullary pin/locking nail more or less replaces the bone marrow with all its cells and vessels. Current understanding is that this destruction diminishes the autologous healing potential of the bone. 13 However, it is the most adequate model for closed fractures, in comparison to the clinical routine. 14
The mouse osteotomy model
The small size of mice has long been considered a hindrance in bone healing research. However fixator systems are available today for various applications, which allow experimenters to mimic clinically used stabilization techniques with systems designed and optimized for the small bones of mice. This still leaves the challenge of the small sample size of the harvested specimens, a fracture hematoma of 0.7 × 1.4 mm or 200 µL of blood serum after terminal cardiac blood sampling. However, with the high sensitivity of current analytical tools this has become a surmountable challenge.
15
External fixation in the femur of the mouse can be accomplished with a gap size of 0.1 mm and up to 2 mm in a rigid or semi-rigid setting, respectively. For our research, we have established a model that does not completely heal within a time period of 21 days (osteotomy gap 0.7 mm) in the control group so that an improvement in the healing process can be easily detected in treated groups. This model only works in female animals as the bone healing process is slower in females than in males,
16
and we only use the C57BL/6 strain in our studies. This strain has good bone healing qualities: bone growth is ‘adult’ at around 12 weeks and bone reaches its peak bone mass at around 16 weeks. The external fixation system (MouseExFix; RISystem, Davos Platz, Switzerland) which we now use in multiple settings has proved to be highly reliable so that we have very few dropouts in our studies, allowing for small group sizes in our study design which reduces animal numbers (Figure 1). Surgery time for an experienced operator is around 15 min per animal. This reduces the time of anesthesia and thus the stress for the animals.
17
(A) External fixation on the left femur. (B) MouseExFix from RISystem. (C) X-ray of 0.7 mm gap post-operation. (D) Mouse 10 min after surgery, still under the red heat light with close supervision in the surgical theater, and already rearing. (E) Mouse with external fixator after observation time of 21 days, no irritation of the surrounding tissue at the external fixator, and the mouse shows all the signs of a healthy animal.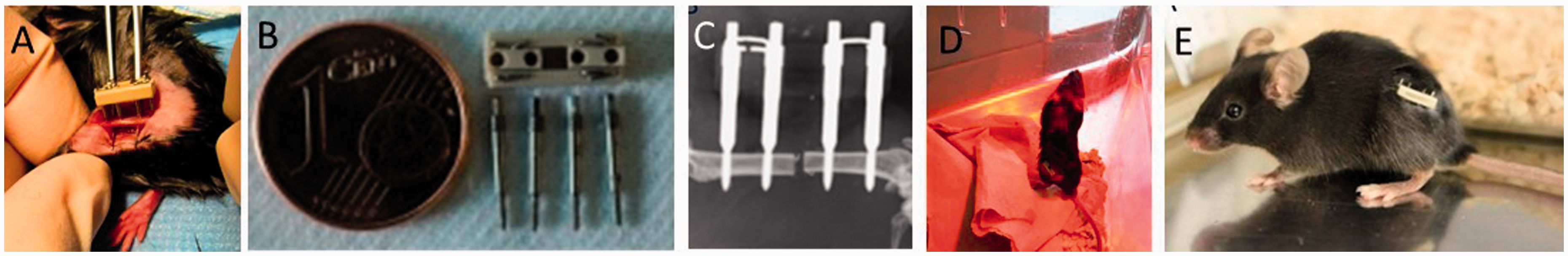
Severity assessment
Within EU regulations, Directive 2010/63/EU lists examples to aid in the assignment of severity classifications to specific procedures (Annex VIII). On this basis, severity depends on the degree of pain, suffering, distress or lasting harm expected to be experienced by an individual animal during the course of the procedure. Levels range from (i) mild (short-term mild pain, suffering or distress; no significant impairment of the well-being or general condition), to (ii) moderate (short-term moderate pain, suffering or distress, or long-lasting mild pain, suffering or distress; moderate impairment of the well-being or general condition), and to (iii) severe (severe pain, suffering or distress, or long-lasting moderate pain, suffering or distress; severe impairment of the well-being or general condition). According to Directive 2010/63/EU, the level of severity of osteotomy as a procedure depends on the degree of fixation stability. Hence, we consider a stable fixation as having moderate severity for up to three days post-surgery and low severity for an ongoing 7–10 days until the wounds are healed. Unstable fixation without restoration of functionality is normally classified as severe.
Assessment of welfare
A distinct recognition of pain in experimental animals is essential for evaluation of biological effects as well as for ethical reasons. Pain can lead to wound healing and blood flow disorders, immunosuppression and increased infection risks. 18 Poorly-managed pain can have an impact on the success of a study as the animals change the load-bearing of the affected limb.19,20 Therefore, welfare and pain assessment in small rodents is of great importance. Unfortunately, there is no ‘gold standard’ for measuring or assessing animal pain, especially in rats and mice. 21 In bone research, well-described systems only exist for large animal models such as lambs and dogs.22,23 General pain indicators in rats and mice are worsening of appearance (such as neglected fur care), reduced condition (e.g. loss of body weight), prolonged re-convalescence, reduced food and water uptake, conspicuous posture, pain face (closed eyes, flattened ears and nose hairs), decreased activity, abnormal movement, vocalization, and absence of nest building. 24 The daily observation by trained personnel, video surveillance with automatic analysis and telemetric methods (e.g. heart frequency, body temperature, breath frequency) can support pain assessment.25,26 Animals should always be observed in their familiar environment and before any kind of manipulation. Furthermore, it must be recognized that rodents are nocturnal and less active during the day. Weight loss is frequently used as an indicator of poor animal welfare. 27 But severe weight loss will always be accompanied by other symptoms such as dehydration, apathy or separation from the group. 28 On the other hand, slow weight loss over days with no other symptoms might be caused by transportation, change of environment, handling or recovery from anesthesia and surgery and consequent reduction in food uptake. In such cases, the animals can be fed with soaked food, apples or jelly if they have been used as food beforehand. Rats and mice are unlikely to eat novel food when in a state of poor welfare, therefore cases of unexplained weight loss should be carefully investigated.
To evaluate pain with respect to bone injury in rodents, only models of post-fracture pain evaluation and bone cancer models have so far been used.29–31 The difference between post-fracture pain models and osteotomy models is that in these studies no pain medication is given; also with bone cancer models, pain will often exceed what is to be expected in a stabilized fracture. Nevertheless, the methods to evaluate pain could be applied to osteotomy models in rodents. To classify pain levels the following methods can be distinguished: (i) subjective assessment of spontaneous or palpation-provoked behavior of the affected limb, e.g. reduced load or prevention of use or abnormal pose of the limb (measured as guarding time span and amount of flinching);30,32 and (ii) methods such as the hot plate (thermal nociception) or the von Frey test (mechanical nociception) which indicate the development of hyperalgesia (exaggerated sensitivity to pain) that can occur temporarily after damage of nociceptors or peripheral nerves.29–31,33–35 However, both these listed methods are invasive and are associated with additional stress and pain burden for the animal. Therefore, the selection of pain assessment methods should be carefully weighed. To date, no study has been published on pain assessment in osteotomy models which provides evidence-based data on behavioral changes in mice and rats. Such studies are desperately needed and should be included in basic research. In addition, general methods to assess pain such as clinical scoring (e.g. the mouse grimace scale), and to determine well-being such as nesting score or time-to-incorporate-into-nest score (TINT), could be beneficial but need to be validated for osteotomy models. Further methods such as testing of burrowing behavior have to be considered in view of the fixation system and the possible risk of injury – for example the possibility of jamming of the external fixator on plastic housing. 36
Score sheet
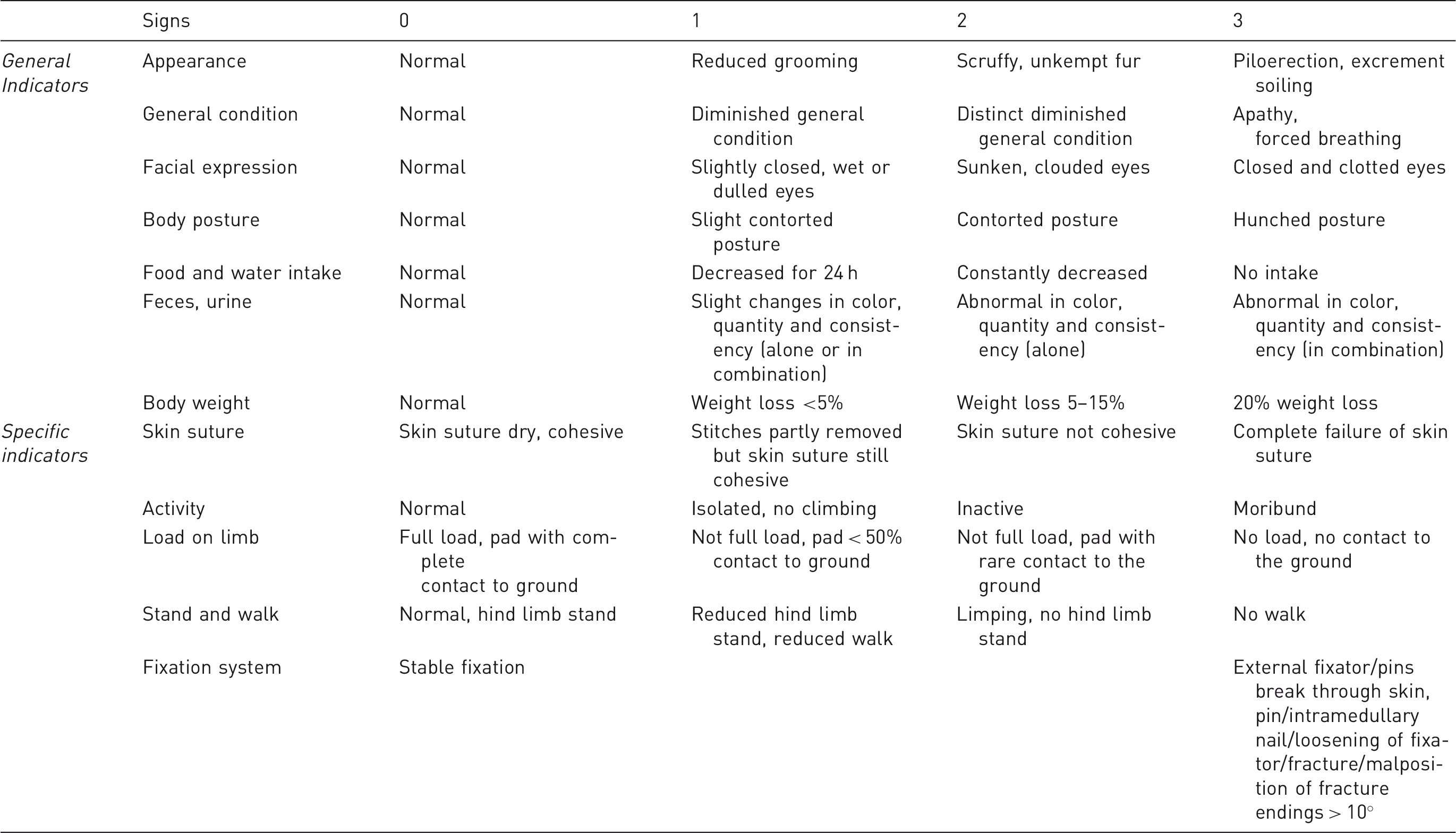
Example score sheet for osteotomy models in rats and mice.
Prescribed interventions depend on the diagnosis (for example: pain = analgesia, infection = antibiotics, circulatory disorders = infusions).
For general indicators of animal welfare, a humane endpoint would be applicable if two or more of the listed points reached grade 3 in this model score sheet. In such cases the animal must be killed painlessly and immediately. Such an animal would already be under specific observation upon reaching grade 2. The appropriate therapeutic intervention should have been applied once the animal had reached grade 2 with one or more signs, which therefore implies that interventions had failed and the endpoint has been reached. If one of the specific indicators reaches grade 3 of the score sheet, this is sufficient reason to terminate the animal immediately to prevent suffering, as either a humane endpoint has been reached or the animal can no longer be used in the experimental setting.
In any case, signs of grade 3 are undesirable and should be avoided.
Scores that are expected during a well conducted osteotomy in mice or rats from transport to sacrifice, assuming no unexpected complications, are summarized in Figure 2.
Scheme of scores to be expected in a well conducted osteotomy in mice or rats from transport to sacrifice, assuming no unexpected complications. SPF: specific pathogen-free.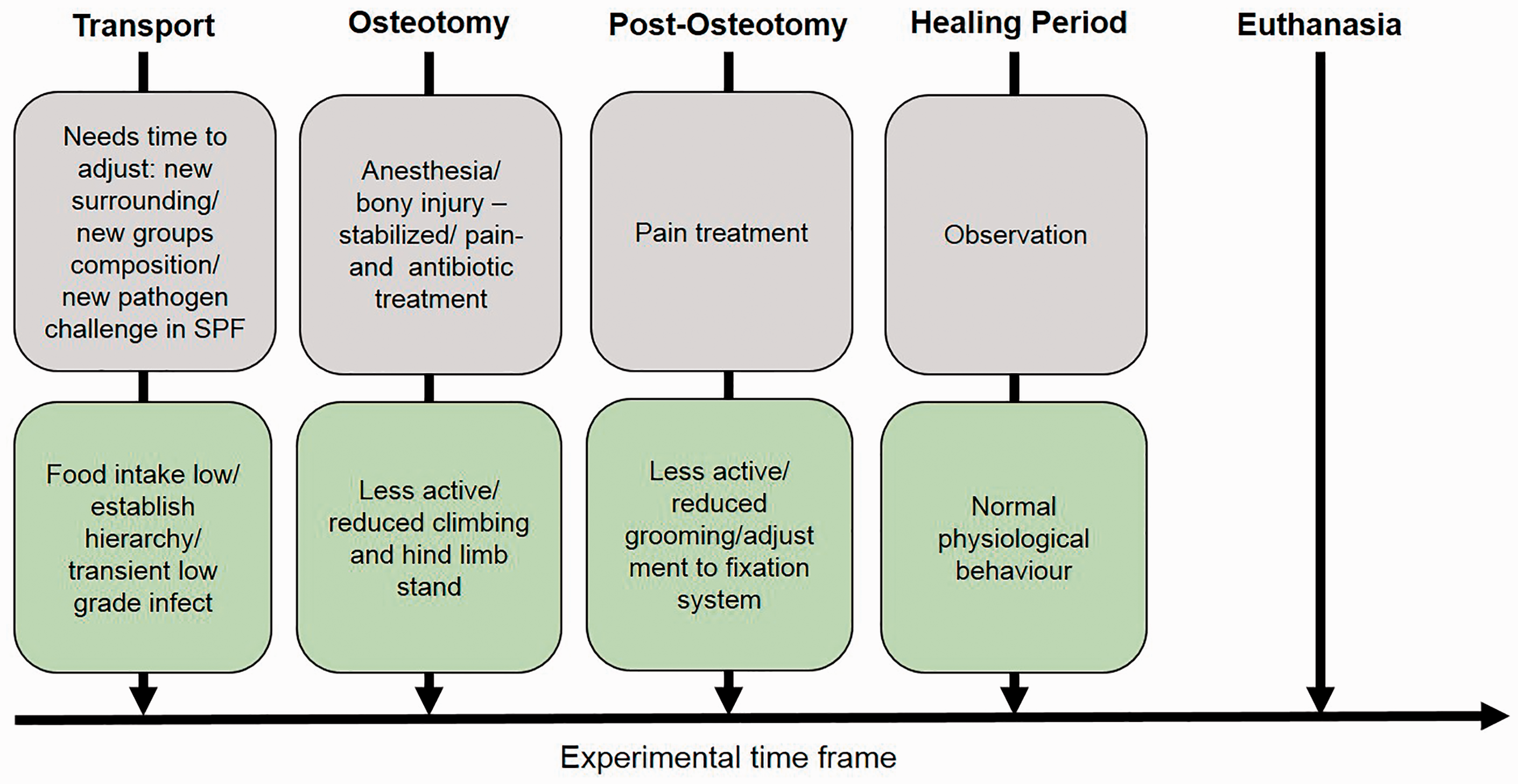
Refinement opportunities
A rigorous and established pain management plan is essential when considering refinement and animal welfare. Perioperative analgesia should begin with a pre-emptive dose of an appropriate analgesic given prior to surgical incision or pain stimulus. Therefore, the present pain is alleviated, and pain development and pain memory are avoided, resulting in a reduction in the intensity of postoperative pain. Many analgesics enable a distinct intraoperative dosage reduction of the anesthetics and therefore reduces recovery time from the anaesthesia. Postoperative analgesia enhances pain relief, accelerates convalescence, as well as preventing pain-induced symptoms.37,38 Criteria for the choice of analgesics are:
Preoperative: type of expected pain, influence on results, length of effectiveness, effectiveness of pain relief, time point of administration. Intraoperative: possible dosage reduction of anesthetics, possible additional dosage, monitoring of animals. Postoperative: length of effectiveness, effectiveness of pain relief, minimal invasive application strategies. Summary of example pain management regimen used in osteotomy models. SC: subcutaneous.
At first, it needs to be pointed out that there are substantial deficits in knowledge about pain treatment in small rodents (e.g. uncertainty regarding dosages, route of administration, efficacy, and duration of action, i.e. injection interval). The selection of suitable analgesics for pain management in osteotomy models is limited, since concerns have been raised about potential adverse effects of non-steroidal anti-inflammatory drugs (NSAIDs) during the initial and inflammatory phase of bone healing.
39
Therefore, researchers normally use morphine, buprenorphine or tramadol in these models. Nevertheless, empirical data on the effectiveness of these analgesics for osteotomy models in rats and mice are missing. That is why varying dosage details, application methods (injection vs drinking water and food) and intervals as well as different treatment durations are found in the literature. With our studies, osteotomized mice and rats are treated for at least three days with pain medication postoperatively. There are only a few studies on fracture pain showing evidence for morphine as a short-term treatment.30,32,40 Buprenorphine is a fast acting and potent opioid, and to date it is the most commonly used analgesic in treating postoperative pain in small animals.41–44 Different dosages have been specified for subcutaneous or intraperitoneal injection ranging between 0.05 and 0.75 mg/kg for repeated doses, respectively,45–47 or for administration via drinking water (9 mg/L) for mice or via food (jelly) at 0.15 mg/kg.48,49 There are new studies on the application interval of buprenorphine which indicate that the pharmacokinetic half-life is short (<6 h).
50
In order to avoid additional stress for the animals from regular handling during injection, non-invasive application over drinking water and food is preferable. This is of special interest for tramadol, a synthetic opioid analog, which has an extremely often-ignored short half-life (<2 h).
51
Therefore, tramadol should only be applied via drinking water. The German Society for Laboratory Animals Science recommends 1 g/L (the former recommendation was 25 mg/L)52,53 for the application via drinking water without any data-based justification. Therefore, the need for evidence-based and empirical data for the dosage and effectiveness of analgesics in bone research is apparent and further studies need to be carried out. In our osteotomy models, we administer 0.03 mg/kg Buprenorphine s.c. pre-operatively and 25–100 mg/L (mice) and 25 mg/L Tramadol (rats) via the drinking water for three days post-operatively.17,20 To provide evidence-based data for our pain management regimen, we are now performing a specific study on this topic and results will be published soon. Table 2 lists the normally used pain medications in small rodent osteotomy models. Nevertheless, it should be taken into account that pain management regimens can vary between different mouse and rat strains, and should be adjusted accordingly.
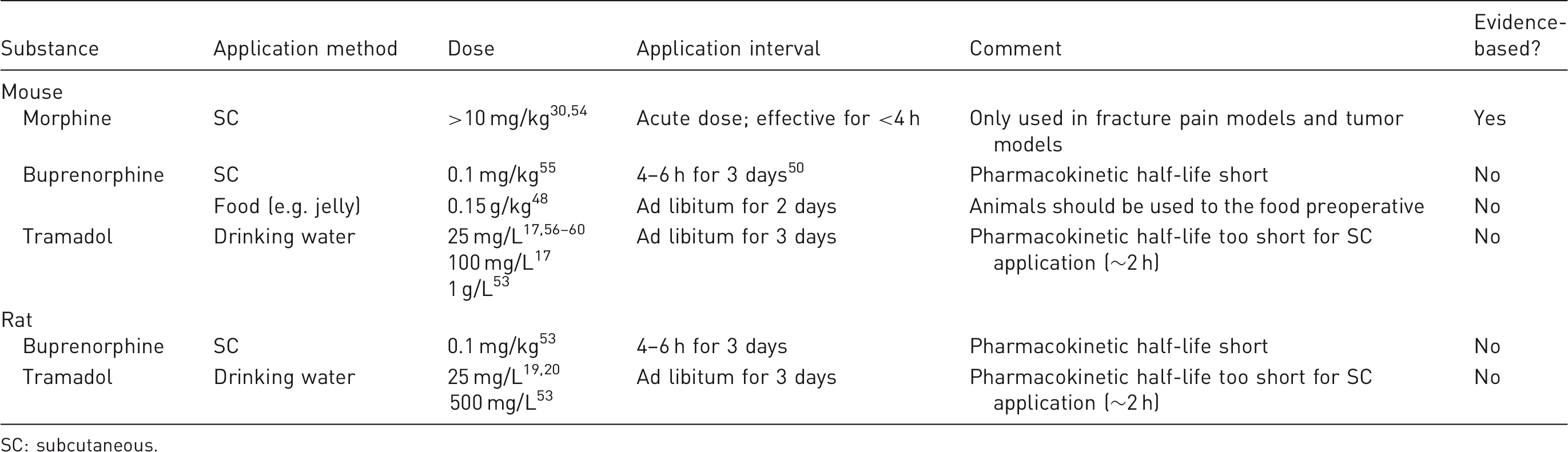
Furthermore, an effective pain management regimen should include: (i) knowledge of the pain pathology, (ii) knowledge of the pharmacological and non-pharmacological analgesic strategies, and (iii) the ability to identify pain. 21 Therefore, researchers should be informed and be adequately trained to recognize pain and choose the appropriate treatment. A short surgery time without additional infliction of injury is essential to avoid stress and pain in research animals, therefore surgeons should be properly trained. Surgical training can be acquired at a first stage while performing together with an experienced surgeon using animal cadavers from other experiments, and at a second stage through assisting with an actual surgery. Once the handling and the familiarity with the drill and saw are assured, the trainee can perform the surgery independently. The surgical procedure should include standard precautions such as gentle handling and positioning as well as heat supply. Furthermore, the use of standardized and validated fixation systems is of great importance and reduces failure rates. For rodents, it is always advisable to use single stitches to prevent reopening of the wound as the animals are liable to nibble at the sutures, and eventually remove them themselves.
Additionally, animals should be kept under permanent surveillance after surgery until they recover their mobility fully. Housing should be in groups with two individuals that are familiar (same group as before) and of the same character (activity). 61 Cages should be well-filled with soft bedding and must not include houses or pipes due to the risk of injury if an external fixator has been used. Nesting material (e.g. tissues, towels or paper shavings) has the potential to improve rodent well-being. The nesting material should be provided prior to surgery so the animal can build a good quality nest before they may be disabled and unable to engage in effective nest building behavior.62,63 Easy to reach soft, palatable food (in the form of a gel) and water (via a longer sipper tube) can be quite suitable for postoperative animals that are reluctant to move or put weight on their hind limbs to engage in rearing positions. Furthermore, note that it is not necessarily to operate on all planned animals in order to achieve a statistically relevant number of animals. Pilot studies can provide first evidence of the effect and continuous analysis of the results during experimentation, which can lead to a reduction in the number of animals used if statistical relevance is achieved before the full group is used. Nevertheless, it is important to avoid underpowering, and to control the type 1 error rate and follow statistical recommendations. 64
For further reading, we recommend the review by Auer et al. (2007) as well as the web-based search for general refinement approaches from specific institutions (e.g. NC3R, Norecopa) that can be adopted to the used animal model. 21
Recommendations and further 3 R research
Before starting a study using the osteotomy model in rats or mice, the following considerations are strongly recommended: (i) perform an extensive literature research, (ii) choose the most suitable species and fixation system to answer the clearly formulated research question, (iii) define the pain management strategy after consultation with experienced researchers in this field, (iv) determine the appropriate score sheet (including humane endpoints), (v) instruct competent animal keepers of the goal of your experiment and keep them involved during the duration of the study, and (vi) carefully train the surgeon before starting the trial. After accomplishing the study, it is recommended to describe in detail the strategies of pain assessment and analgesia employed as well as unusual findings and failures in the publications according to the ARRIVE guidelines.
Nevertheless, further 3 R research, especially in refinement is needed. To mention one example: rats are known to gnaw at the external fixator, necessitating the use of a suitable collar to prevent this. Such a collar is associated with stress for the animals. The development of a bite block that is fitted onto the external fixator could solve this problem without impeding the animal.
In addition, the standardization and harmonization of pain assessment strategies in rats and mice are necessary as well as the enrolment of efficacy studies for analgesics. This could be realized without the use of additional animals if these studies are embedded in basic experimental studies.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Our work is supported by the DFG (SCHM2977), BfR (RefineMOMo), BCRT and BSRT.
