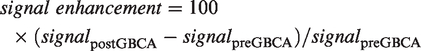Abstract
Breast cancer is the most common cancer among women worldwide. For high-risk women, contrast enhanced (CE)-magnetic resonance imaging (MRI) is recommended as supplemental screening together with mammography. The development of new MRI contrast agents is an active field of research, which requires efficacy tests on appropriate preclinical pathological models. In this work, a refined method to orthotopically induce breast cancer in BALB/c mice was developed using ultrasound (US) as a guide for the precise localisation of the tumour induction site and to improve animal welfare. The method was coupled with CE-MRI to characterise the evolution of the tumoural lesion.
Introduction
Although many therapies have been developed to overcome breast cancer, it still remains the most common cancer and the second cause of cancer-related deaths in women worldwide.1,2 Contrast enhanced-magnetic resonance imaging (CE-MRI) is recommended as supplemental screening (the primary procedure being mammography), in particular for women who carry a mutation in breast cancer genes BRCA1 or BRCA2, for women who are suspected to carry other, high-risk mutations for breast cancer and for their first-degree relatives. CE-MRI is also the first line of screening for recurrences. Gadolinium-based contrast agents (GBCAs) are widely used in clinical practice for investigating pathologies,3–6 such as the identification of tumours and inflammatory disorders, and for treatment monitoring. The number of CE-MRI procedures performed globally has grown considerably in the last decade and it is likely to rise in the future, as this kind of screening becomes available to more patients and the information obtained from a single contrast injection increases. 7 Research for new GBCAs is still active, especially recently. In the preclinical development phase, the use of animal models is essential for the evaluation of the efficacy of new GBCAs. Orthotopic tumour models are usually the pathological model of choice when testing for new pharmaceuticals, particularly because they are characterised by high translatability into clinical practice. Currently, breast cancer orthotopic mice models can be induced with two main methods: (a) orthotopic implantation of tumour cells under direct visualisation of the mammary fat pad (MFP) (orthotopic direct vision, ODV); 8 (b) percutaneous blind injection into the nipple area to attempt the implantation of cells into the MFP (orthotopic percutaneous, OP). 9 Even though the OP method is easier to perform because it is quicker and requires fewer surgical skills, it has been demonstrated to be less accurate and more confounding than ODV. ODV, however, implies the direct exposure of the MFP and thus requires surgery, which is often stressful for the animal. Moreover, this technique is often perceived as difficult since it requires specific expertise.10,11
In this study, two different breast cancer murine cell lines (TS/A and 4T1) were injected into the MFP of BALB/c mice by using ultrasound to correctly identify the inoculation site and to precisely guide the needle for cell release.
Materials and methods
Twenty-six BALB/c female mice (Charles River Laboratories, Calco, Italy), aged 7–8 weeks old (16–18 g body weight) underwent the experimental protocol. Procedures were conducted according to the national and international laws on experimental animal research (L.D. 26/2014; Directive 2010/63/EU) under Italian Ministerial Authorisation (project Research Number 821/2018-PR). At arrival and during the acclimation period, the animals were housed in polysulfone Tecniplast Double Decker ventilated cages (up to seven animals/cage). During experimental procedures, animals were housed in type IV Tecniplast cages (up to seven animals/cage). Animals were kept in limited access, air-conditioned facilities (20–24°C room temperature, 45–55% relative humidity, 15–20 air changes/h, 12-h light cycle). Food and water were available ad libitum (food: VRF1 (P), in pellets (ø 12 mm), by SDS (Special Diets Services Ltd, Witham, Essex, UK). The induction of tumours was performed after a one-week period of acclimation and observation. Animals were socially housed for psychological/environmental enrichment and were provided with items such as a device for hiding in and an object for chewing, except during study procedures/activities. The day before tumour induction, mice were first anesthetised with sevoflurane (induction, 3%; maintenance, 1% in O2) and then shaved using a chemical cream (Veet, Italy). On induction day, carprofen (Rimadyl®, Zoetis Italia S.r.l.) was administered subcutaneously (5 mg/kg) one hour before cell injection for pre-emptive analgesia. Under gaseous anaesthesia, mice were placed supine on the ultrasound animal bed and the temperature was maintained between 36–37.5°C. After identification of the nipple area of the abdominal mammary gland with the use of magnifying glasses, tweezers were used to pinch the fourth right MFP and the syringe needle (29G) was placed horizontally to the nipple, in the area of the MFP. Correct localisation of the needle into the MFP was performed by using a preclinical ultrasound system (Vevo Lazr 2100 Ultrasound System with a MS700 Transducer working at 55 MHz). Once the positioning of the needle was verified, tumour cells (4T1 (group 1 = 5 × 105, n = 7; group 2 = 106, n = 6; purchased from ATCC) or TS/A cells (group 3 = 5 × 104, n = 5; group 4 = 5 × 105, n = 8; courtesy of University of Turin) were first tested for mycoplasma then suspended in 50 µl of RPMI-1640 serum-free medium and slowly released under ultrasound monitoring. The cell number was selected for testing after comparison with previously published papers.12–15 The procedure lasted approximately 5 min; neither soreness nor bleeding were observed in the site of injection (Figure 1).
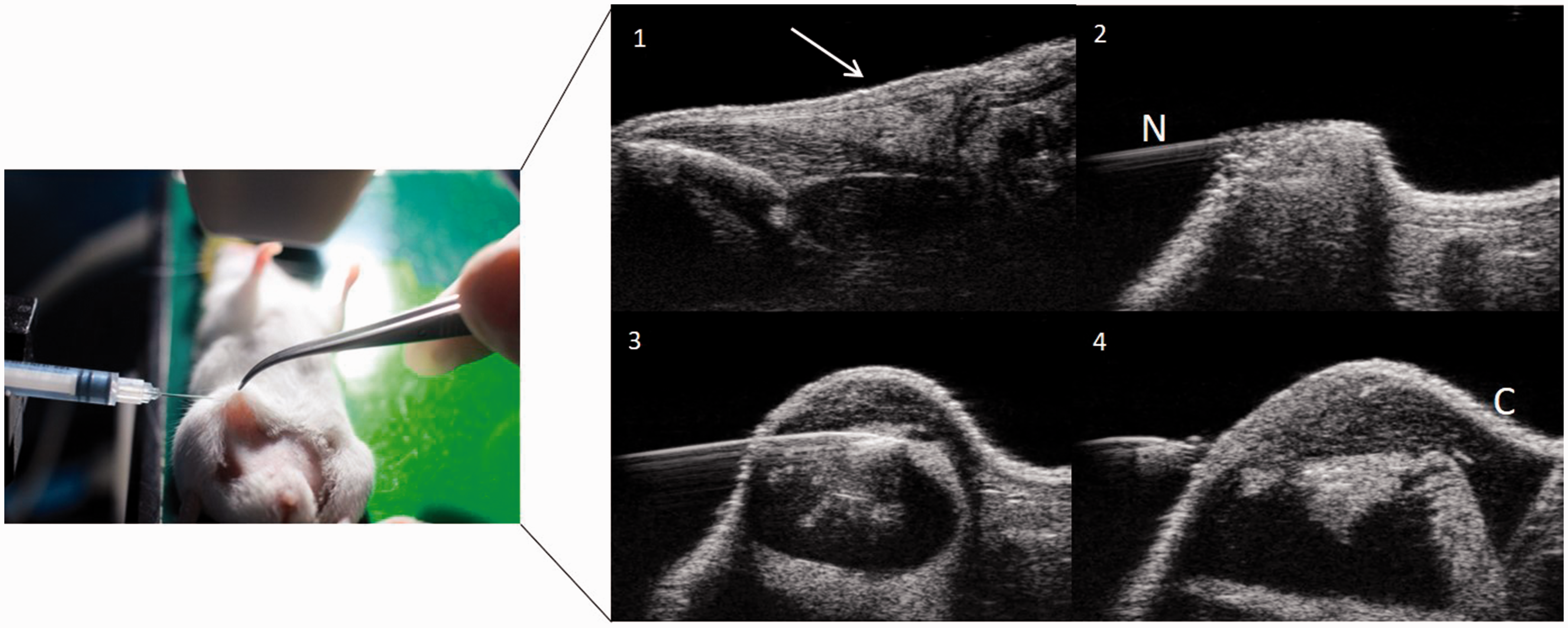
Ultrasound-guided injection of cancer cells into the mammary fat pad (MFP) of mice. 1, MFP before needle placement (arrow); 2, needle (N) placement into the MFP; 3, cell (C) injection into the MFP; 4, needle removal.
After the orthotopic injection, animals were monitored daily. The Veterinary Officer, who was available throughout the course of the study, evaluated general condition, spontaneous behaviour, reaction to handling, breathing, hydration condition and changes in body weight. Any signs of suffering were recorded according to the OECD document. 16
Animals whose health status was considered suitable for the experimental procedure by the Veterinary Officer underwent two MRI sessions per week: a first one consisting of the acquisition of a T1-weighted spin echo sequence (to simply monitor the tumour growth) and a second one, consisting of the acquisition of a series of 3D T1-weighted gradient echo scans before and after the administration of a commercial macrocyclic GBCA, Gadovist® (Bayer SpA), to monitor the kinetics of perfusion and permeability features of the tumour. MRI sessions were performed starting from day 4 after tumour inoculation until euthanasia, by using a 3T preclinical scanner (Biospec, Bruker Biospin, Ettlingen, Germany) equipped with a circular surface coil. Animals were anaesthetised with sevoflurane gas (induction, 3%; maintenance, 1% in O2) and placed on the MRI animal bed, with the surface coil positioned over the tumour area. For the administration of Gadovist®, an intravenous catheter with a 110 µl dead volume was inserted into the tail vein of the animal under anaesthesia, before positioning the animal inside the MR scanner. The GBCA was injected at a dose of 0.1 mmol/kg, corresponding to an administration volume of 2 ml/kg of a 50 mM solution at an injection rate of 2 ml/min. The injection was manually performed with the help of a timer to check the injection rate. MRI GBCA kinetics lasted approximately half an hour post injection. Animals were then monitored and heated with an infra-red lamp. After recovery, they were returned to the animal facility.
Animals were euthanised by overdose of anaesthesia and cervical dislocation at the end of the study or when one of the following endpoints was reached:
loss of weight ≥20%; severe or prolonged distress (according to checklist evaluation); mass tumour volume ≥300 mm3; tumour ulceration; within 40 days of tumour induction; necrosis of the tail, according to veterinary evaluation.
MRI data analysis consisted of the measurement of the tumour volume and the quantification of signal enhancement after the administration of GBCA. Tumour volume was measured by selecting proper regions of interest (ROIs) over the tumour on sagittal T1 weighted images by using Paravision® (Bruker, Germany) software. Signal enhancement, defined as follows:
where signalpreGBCA and signalpostGBCA indicate MR signal before and after GBCA administration was quantified by using a home-developed plug-in, running on ImageJ (imagej.nih.gov/ij/), that allows Bruker data manipulation and practical ROI positioning and signal quantification.
Results and discussion
Mammary cancer was successfully induced in 24 out of 26 mice: 11/13 for the 4T1 model (two mice, one belonging to group 1 and one belonging to group 2, did not develop any visible mass) and 13/13 for TS/A (Figures 2 and 3). Both models showed a tumour characterised by fast growth (a 100–200 mm3 volume was reached in 2–3 weeks, depending on the line and the number of inoculated cells) and by a noticeable maximum MRI signal enhancement after GBCA administration (about 70–110%, which consists of roughly double the basal signal). For 4T1 cells, a tumour mass was palpable and clearly visible starting c. 4 days after cell inoculation. The volume rapidly increased reaching the endpoint in about 15 days. No substantial differences in tumour growth were observed between groups 1 and 2, indicating that tumour growth does not depend on the number of inoculated cells. In group 1, five animals were euthanised when the tumour volume was close to the endpoint (one 10 days post induction, one 12 days post induction and three 14 days post induction), while one animal was euthanised at the end of the study 14 days post induction and one did not develop any tumour and was sacrificed 14 days post induction. In group 2, three animals were euthanised when the tumour volume was close to the endpoint 13 days post induction, two animals were euthanised at the end of the study, 9 and 15 days post induction, and one did not develop any tumour and was sacrificed 14 days post induction. For TS/A cells, the tumour mass was palpable and clearly visible 7–10 days after cell inoculation in group 3 and after 6 days in group 4. Tumour volume increased more rapidly in animals belonging to group 4 than to group 3, reaching the endpoint after approximately 3 weeks in group 4 and staying below 150 mm3 after 25 days in group 3, which was also characterised by an increased heterogeneity, in terms of mass volume (i.e. higher standard errors). In group 3, all animals were sacrificed at the end of the study (three 24 days post induction, one 22 days post induction and one 17 days post induction). In group 4, three animals were euthanised when the tumour volume was close to the endpoint (two 16 days after induction and one 19 days after induction), two were sacrificed due to tumour ulceration (one 8 days post induction and one 12 days post induction) and the remaining three animals were euthanised at the end of the study (15, 16 and 19 days after induction).
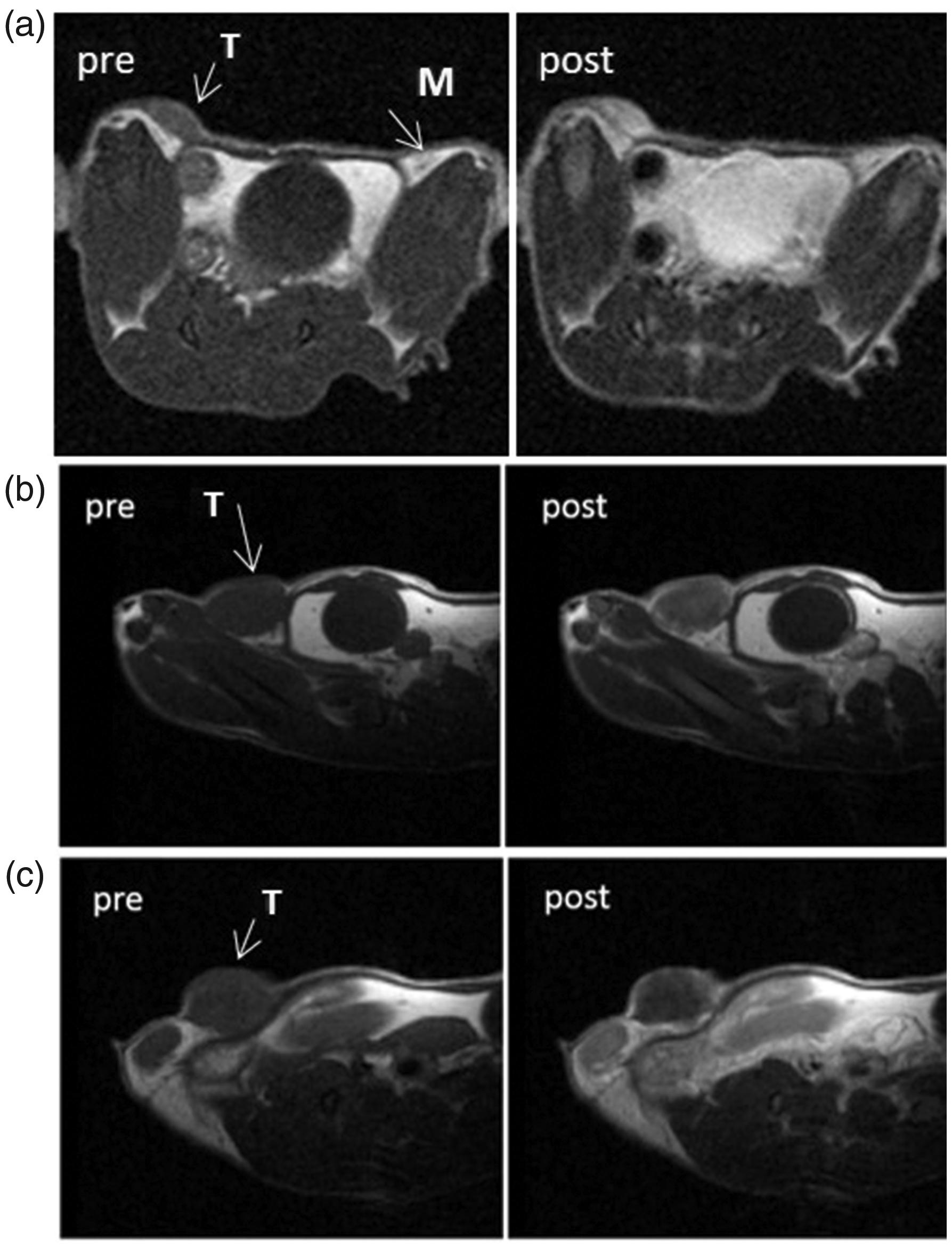
Magnetic resonance images pre and contrast injection of the fourth mammary fat pad (M). (a) External growth of a tumour (T) induced with the orthotopic percutaneous method (previous internal study); (b) 4T1 8 days post induction; (c) TS/A 8 days post induction.
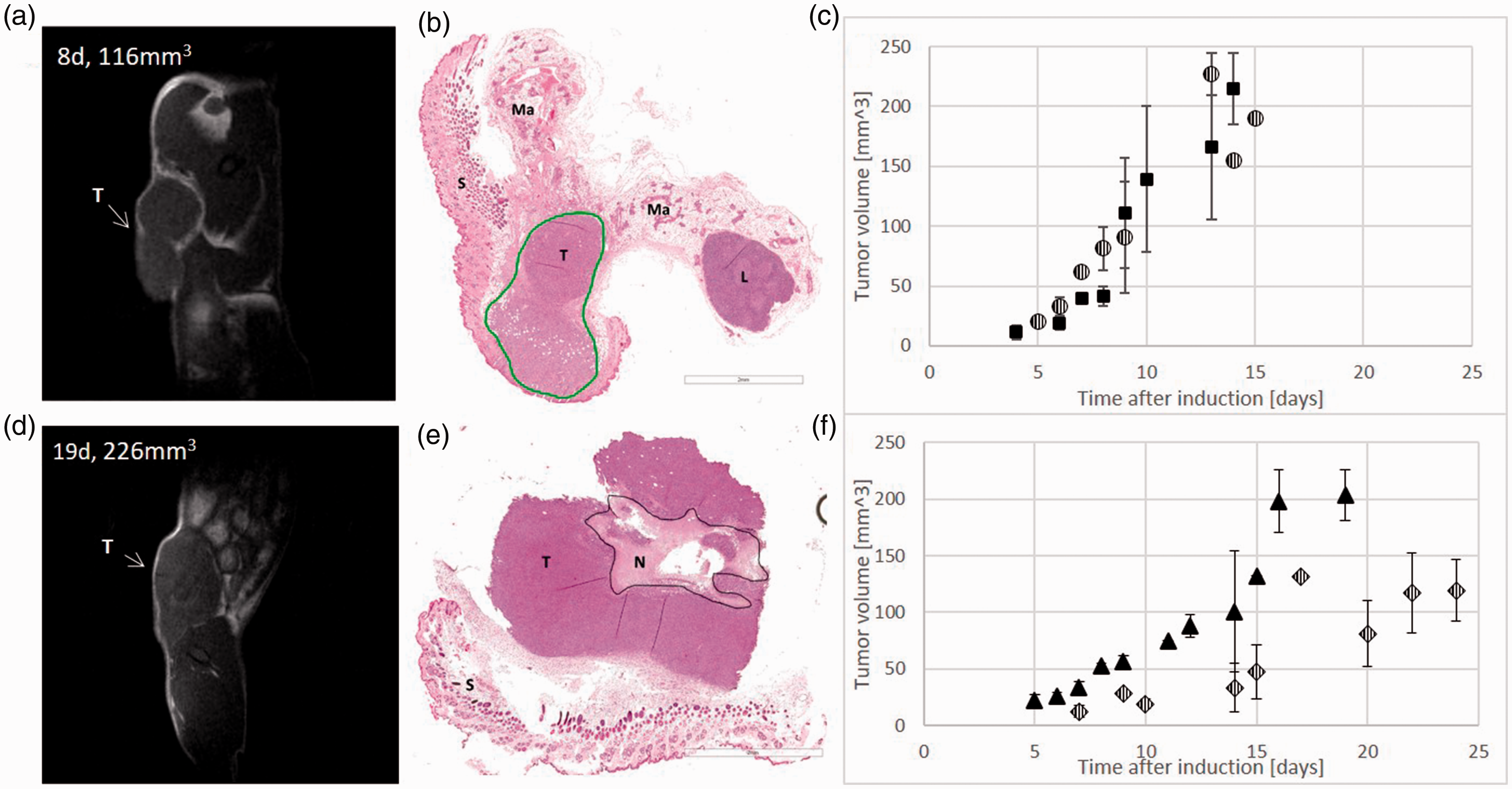
Qualitative correlation of magnetic resonance images and histology. (a) 4T1 tumour in a T1w image 8 days post induction, before euthanasia. (b) Histology of 4T1 tumour (T, tumour; L, lymph node; S, subcutis; Ma, mammary fat pad). (c) Tumour growth curve of 4T1 tumours for groups 1 (six out of seven animals with developed mass, square) and 2 (five out of six animals with developed mass, circle). Error bars represent standard error; group 1: n = 3 (day 4), n = 2 (day 6), n = 1 (day 7), n = 4 (day 8), n = 2 (day 9), n = 3 (day 10), n = 2 (day 13), n = 4, (day 14); group 2: n = 1 (day 5), n = 4 (day 6), n = 1 (day 7), n = 3 (day 8), n = 2 (day 9), n = 3 (day 13), n = 1 (day 14), n = 1, (day 15). (d) TS/A tumour in a T2w image 19 days post induction, before euthanasia. (e) Histology of TS/A tumour (T, tumour; S, subcutis; n, necrosis); scale bar 2 mm. (f) Tumour growth curve of TS/A tumours for groups 3 (diamond) and 4 (triangle). Error bars represent standard error; group 3: n = 2 (day 7), n = 1 (day 9), n = 5 (day 10), n = 3 (day 14), n = 2 (day 15), n = 1 (day 17), n = 4 (day 20), n = 4 (day 22), n = 3 (day 24); group 4: n = 3 (day 5), n = 6 (day 6), n = 4 (day 7), n = 4 (day 8), n = 5 (day 9), n = 1 (day 11), n = 7 (day 12), n = 2 (day 14), n = 1 (day 15), n = 3 (day 16), n = 2 (day 19).
The induction procedure proved to be viable, reproducible and overall, easy to perform. Animal welfare improved, especially if compared to other invasive procedures such as ODV, which causes a lot of stress to the animal and loss of body weight due to the recurrent removal of sutures and the invasiveness of the procedure (all five animals induced on a previous internal study experienced a loss of body weight ranging between 5 and 15%, data not shown). Animals which were induced with the ultrasound guided method showed very moderate clinical signs, including redness of the tumour mass (n = 2) and tumour ulcerations (n = 2), which are likely to be related to the pathology itself rather than to the method of tumour induction. Moreover, the induction procedure proved to be better than OP, which in our experience resulted to be less reproducible and more operator dependent, with tumours growing on the external border of the MFP or just in the subcutis, as shown in Figure 2(a), with images acquired from a previous internal study with an Icon scanner (Bruker, Germany) operating at 1 T. Conversely, the ultrasound procedure showed correct localisation in 100% of animals.
Histology from both cell lines confirmed the carcinogenic nature of the lesion and the localisation of the tumour in the MFP. Both tumours (4T1 and TS/A) displayed features of poorly differentiated breast cancer, with the tumour correctly localised in the mammary gland. When reaching larger volumes, both tumour types exhibited areas of necrosis, and this was more frequent for TS/A. Since 4T1 cells are commercially available and their growth was more homogeneous, with substantially no outliers, these cells were selected as the best choice for syngeneic breast models. The observed failure on two animals among 13 inoculated was considered less relevant compared to the other factors (homogeneity, animal welfare, presence of necrosis or ulceration). The best compromise between optimal development, tumour enhancement and animal welfare is thus represented by group 1 between 5 and 8 days after development, that is when the diseased mass volume ranges between 20 and 60 mm3.
The protocol proposed in this study, already introduced in rats, 17 is here applied for the first time on mice, a species for which the identification of inoculation site and the avoidance of cell dissemination is much more critical. With the described method, in addition to the proper identification of the MFP, the real-time recording of the injection by the US device can be used to visualise whether the injection of cells succeeded or if there was any dissemination during the injection. Moreover, animals do not have sutures (therefore recover quickly) and there is no risk of puncturing the peritoneal cavity during the overall procedure.
In this study, MRI was used as a non-invasive method to characterise the model under investigation in terms of growth rate and tissue permeability features. As the growth of the tumour progresses, the tissue exhibits a change in contrast enhancement, which is in turn linked to a change in perfusion and permeability. These lesion features are very important for follow-up and decision making in therapy. The relationship between CE-MRI and changes in tumour perfusion and permeability has already been discussed in a previously published article. 18
MRI, coupled with histology, helped us to confirm the correct localisation of the tumour in the MFP. After administration of a contrast agent, CE-MRI served as a way to evaluate total perfusion and permeability of the tumour by studying its enhancement properties. Thus, it allowed the assessment of this model’s potential use in efficacy studies that can bridge the gap between clinical and preclinical research. Moreover, the model is perfectly designed to fit into a scientific context increasingly driven by the principles of the 3Rs (Replacement, Reduction, Refinement). 19
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: FLC received a PhD funded by Bracco Imaging SpA at the time of the study; FLC, AFM, SCS, ADV, CC, PO, AC, CB and LM are employees of Bracco Imaging SpA
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.