Abstract
In search of better predictive animal models for evaluating treatment response in lung cancer, orthotopic lung tumour models are a great step forward over traditional subcutaneous models. Crucial in the development of such orthotopic models is a reliable and reproducible instillation method. Because cells are instilled inside the thorax, the accuracy of the instillation and visualization of tumour growth demands the use of non-invasive imaging methods. We used a minimally invasive intratracheal intubation method to instill bioluminescent lung tumour cells in the lung parenchyma. Adaptation of the cell containing medium provides the possibility of tracing the exact location of the injection by means of single photon emission computed tomography/computed tomography (CT) imaging. The transplantation medium was also optimized to prevent migration of the injected substance. This results in the outgrowth of single and well-defined lung tumours at the instillation site. Finally, tumour growth was validated and longitudinally monitored with a combination of CT and bioluminescence imaging. The reported transplantation procedure enables the assessment of injection accuracy and provides a good approach for the generation of orthotopic lung tumour models for future response imaging studies.
Globally, lung cancer is the leading cause of cancer-related mortality. At diagnosis, the disease is often already well advanced and the lack of effective treatment strategies frequently results in a poor clinical prognosis. 1 Novel therapeutics that could help improve the treatment efficacy are under investigation.2–4 However, such treatment strategies have to be first optimized and validated in a preclinical setting.
Traditionally, subcutaneous tumour models using implantation of cultured tumour cells have been used for the assessment of treatment efficacy. Herein, calliper measurements provide quick and accurate data regarding volumetric changes of the tumour5,6 at different stages during treatment. Differences in treatment outcomebetween these subcutaneous experiments and the results seen in patients suggest that chemosensitivity to cytostatics could, in part, depend on the anatomic site of the tumour. 7 Substantiated by numerous efforts aimed at establishing orthotopic tumour models in rodents that simulate the clinical features of lung cancer,8–10 orthotopic lung tumour models are expected to provide a better preclinical prediction of the treatment.
In the literature, several different orthotopic transplantation methods for lung cancer have been described, where percutaneous intrathoracic implantation and intrabronchial implantation are the most relevant.11–14 The instillation of tumour cells frequently requires an invasive procedure with a heightened risk for surgical trauma. 15 Furthermore, many of the existing methods often result in multifocal lesions.11,12,14 Such multifocal lesions make volumetric response measurements extremely difficult, even with imaging modalities such as computed tomography (CT). Moreover, dependent on their location, multifocal lung tumours may rapidly decrease the health of the animals. Ideally, tumour cells are instilled with minimal discomfort and result in the outgrowth of a single lung tumour.
Crucial for accurate orthotopic transplantation models is the ability to define the location of the injected tumour cells after instillation. In cardiac stem cell therapy, tracking injected cells with paramagnetic contrast agents, radioisotopes or fluorescent dyes is a common practice. 16 In these instances, the stem cells are labelled (intracellularly) with the imaging agent, 17 while the toxicity of these labels could potentially influence cellular growth. 16 Tumour cells, however, are commonly transplanted in a buffer solution. As an alternative to labelling of the cells, the transplantation can be monitored by adding an inert imaging agent such as albumin radiocolloids to this buffer solution. With single photon emission spectroscopy computed tomography (SPECT) even minute concentrations of the radioactive imaging agent can then be traced.16,18 After tumour formation, CT imaging can be used to anatomically locate the tumour. Viable tumour cells can also be detected by bioluminescence imaging (BLI) 19 after genetic labelling with reporter genes such as luciferase.
The image described here validated transplantation procedure results in the effective outgrowth of single well-defined lung tumours that can accurately be monitored in a longitudinal manner. Moreover, the described methodology is not restricted to the use of specific tumour cell lines. Hence, it generates the possibility of creating a variety of lung tumour models that are of value for accurate response imaging during intervention studies.
Materials and Methods
Animals
BALB/cABomA-nu nude mice were bred in our own animal facility and used for intratracheal intubation experiments between 8 and 12 weeks of age. Animals were housed under individually ventilated cage (IVC) conditions with a 12:12 h light-dark cycle and ad libitum access to food and water. All animal experiments were performed in accordance with Dutch animal welfare regulations and approved by the local ethics committee.
Cell line
The C204 small cell lung cancer (SCLC) cell line was derived from a spontaneous SCLC lung tumour, induced via an instillation method described by Meuwissen et al. 8 At 6-12 months after inoculation, tumours were harvested, suspended mechanically and taken into culture. 20 C204 cells were cultured in DMEM-F12 medium containing 10% fetal bovine serum, 100 IU/mL penicillin, 100 μg/mL streptomycin, 5 ng/mL insulin, 5 ng/mL epidermal growth factor (all from Invitrogen Life Technologies, Breda, The Netherlands) and 5 ng/mL cholera toxin (Sigma Aldrich, Zwijndrecht, The Netherlands). Cells were stably transfected and selected as previously described by Kemper et al. 21
Imaging agents
Clinical grade human serum albumin colloids (NanoColl, GE Healthcare, Eindhoven, The Netherlands) was radiolabelled with 99mTc using the standard clinical labelling procedure. One gram of Luciferin (Caliper LifeSciences, Hopkinton, MA, USA) was dissolved in 66.7 mL phosphate buffer saline. After filtration using a 22 μm filter (Millipore, Billerica, MA, USA), this resulted in a 15 mg/mL sterile solution.
Preparation of C204 cells containing transplantation medium
Cells were washed three times with HBSS (Invitrogen Life Technologies) and sphere-like structures were gently broken down by resuspending them using a pipette. A total volume of 20 μL containing 1 × 106 cells was injected per animal. For the SPECT-based injection experiments, the cell containing medium was made by suspending the cells in a solution holding Nanocoll (10 MBq/20 μL). In the experiments wherein the transplantation medium was enriched with matrigel (BD Bioscences, Breda, The Netherlands), 10 μL of matrigel was added to 10 μL of Nanocoll (10 MBq) before suspending the cells in this solution. The matrigel-enriched transplantation medium was kept on ice to avoid clotting.
Preparation of the canula
A 2 French polyurethane canula (UNO Roestvrijstaal, Zevenaar, The Netherlands) was placed on the 29 G needle of an insulin syringe (Terumo, Leuven, Belgium). The total length of the canula from needle point to the end of canula was 28 mm. A longer canula can puncture the outer surface of the lung parenchyma, while a shorter canula will result in instillation in the higher regions of the lung, similar to what Savai et al. 13 reported. High instillation of the tumour cells makes accurate discrimination of the tumour from other dense tissues such as the heart with CT more complicated. It must be noted that the length of the canula has to be adjusted when using animals of different age and size or possibly animals of a different strain. Initially 20 μL of air was taken up, followed by 20 μL of cell containing transplantation medium. To inflate the lungs at the instillation site before injecting the cells, another 50 μL of air was taken up.
Anaesthesia
Before initiating the intratracheal intubation and SPECT/CT and single CT imaging, mice were anaesthetized by an i.p. injection of hypnorm (0.15 mg/mL fentanylcitrate, 10 mg/mL fluanisone; VetaPharma Ltd, Leeds, UK)/dormicum (5 mg/mL Midazolam; Roche, Mijdrecht, The Netherlands)/water solution (1:1:2; 5 μL/g). BLI was performed under 2% isofluorane (Forene; Abott Laboratories Ltd, Maidenhead, UK) in an oxygen-air mixture inhalation anaesthesia.
Intratracheal intubation
The minimally invasive intratracheal intubation method presented is based on standard intubation methods for ventilation 22 and is an optimization of the method used by Savai et al. 13 It uses an intubation standard to keep the animal in an upright position and a light source to provide better visibility of the trachea.
Mice were anaesthetized and were placed in an intubation device (UNO Roestvrijstaal, Zevenaar, The Netherlands) (Figure 1a). The mouth was opened with tweezers and the canula was inserted into the trachea using the vocal cords for guidance. The luciferase expressing C204 SCLC cell containing medium was injected into a predetermined site at the base of the lung (Figure 1a) without damaging the surrounding tissues. Subsequently, the canula was removed and the animal was taken down from the intubation device.
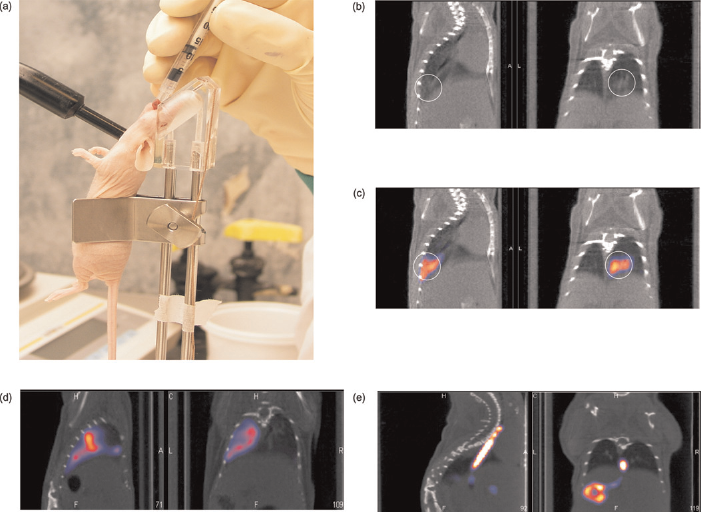
Orthotopic transplantation of C204 tumour cells. (a) Using an intubation standard and light source, the cell containing medium is slowly injected via the trachea into the lungs. (b) After implantation computed tomography (CT) images faintly show the presence of some fluid in the lungs. (c) Single photon emission computed tomography/CT imaging clearly shows a radioactive region in the lungs correlating to the volume faintly detected with CT. (d) Fast injection of the medium results in distribution over the entire lung. (e) Accidental injection via the oesophagus into the stomach is easily detected
SPECT/CT imaging
To provide a non-invasive readout of the accuracy of the instillation procedure, cells were injected together with a small amount (10 MBq) of 99mTc-Nanocoll. Imaging was performed on the SPECT/CT within 30 min after injection, while the animal was still fully anaesthetized. Mice were placed individually on the SPECT/CT (NanoSPECT/CT; Bioscan Inc, WA, USA). A region of interest (ROI) was selected based on a sagittal tomographic planning CT image incorporating the complete thorax. CT imaging of this ROI was performed using the integrated cone-beam CT (total scan time: 4 min). For SPECT imaging, a scan (6 min) of the same field of view was performed using the multi-pinhole camera. Following reconstruction, the CT and SPECT data were automatically co-registered.
Visualization of the tumour and volumetric monitoring of tumour growth over time were done by CT imaging alone. Data-sets were analysed in the InVivoScope postprocessing software (Bioscan, Inc). All tumour sizes were measured on two separate occasions by two people simultaneously and the average of the two values was used. Tumour volumes were calculated using the following formula for ellipsoid spheres: 4/3π(1/2a)(1/2b)(1/2c). Here a, b and c are the diameters in the x-, y- and z-axes of the tumour.
Bioluminescence imaging
Four weeks after instillation of the cells, tumour presencewas evaluated bymeans of a bioluminescence scan. Fromthis point on, the animalswere scannedweekly to followthe growth progression of the tumour. Animals were injected i.p. with 150 mg/kg luciferin. Mice were anaesthetized and placed in a temperature-controlled (37°C) IVIS 200 camera (Xenogen Corp, Alameda, CA, USA). Bioluminescent images were recorded between 11 and 15 min post-postluciferin administration. After imaging, the animals were placed back in their cage where they recovered within minutes from the anaesthetics. The weekly scans had no adverse effect on the welfare of the animals. The bioluminescent signal intensity in the ROI (photons/s/cm2) was quantified using Living Image software.
Histology
When tumours reached a size of approximately 80 mm3 or when an animal showed signs of breathing difficulties, the animal was taken out of the experiment. The animal was killed by means of cervical dislocation, after which the lungs were flushed with 4% formalin solution. The lungs were thenembedded in paraffin and cut into 4 μm sections. For validation of the presence of singlewell-defined tumours, haematoxylin and eosin (H&E) staining was performed according to standard protocols.
Results
SPECT/CT imaging visualizes the radioactivity component used during instillation of the cells, giving a direct measure for the distribution of the injected medium and providing insight into the efficacy of the instillation procedure. When the cells are slowly injected into the lung, only a single ‘hot’ spot can be seen (Figure 1c). However, after fast injection, distribution of the injected medium throughout the lungs and trachea was observed (Figure 1d). Misplaced injection into the oesophagus and stomach did occur sporadically and were directly detected, resulting in the exclusion of these animals from the study (Figure 1e).
Next to the injection location and speed, the cell containing medium also influences the instillation accuracy. When cells were instilled without matrigel, the injected volume could be traced over time from the instillation site to several other sites along the injection track (Figure 2a). This resulted in the formation of multifocal tumours. On the other hand, cells instilled in combination with matrigel predominantly remain at the instillation site (Figure 2b). The adhesive properties of the matrigel prevent the cells from migrating, resulting in the generation of single and well-definable orthotopic lung tumours.
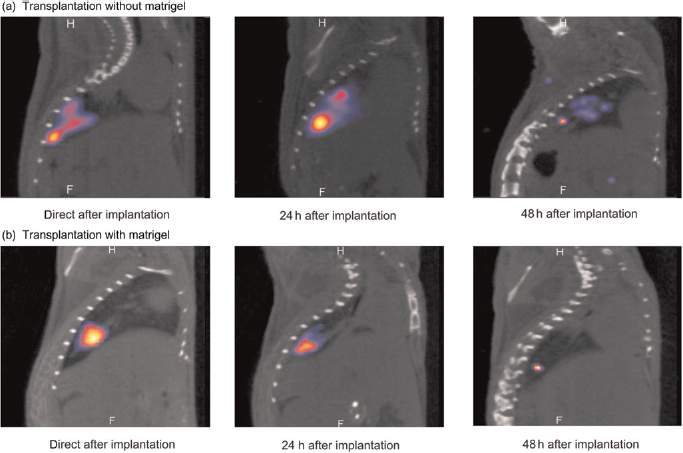
Dynamics of the implanted cell suspensions: (a) Orthotopic transplantation without matrigel revealing a migration of the radioactive signature over a 48 h time period. (b) Transplantation with matrigel containing cell suspensions gave retention of the radioactive signal intensity at the initial transplantation site during a 48 h time period
The genetic modification of C204 cells with the luciferase gene enables longitudinal monitoring of the tumour progression in vivo (Figure 3a). Tumour formation mostly required several weeks and the first tumour-positive bioluminescent images could only be generated 3-4 weeks after injection. With BLI a small amount of viable tumour cells could already be made visible and, in this case, the highly luminescent C204 cells provided a positive signal two weeks before the tumour could be detected on CT (Figure 3b). Positioning of the animal during the CT procedure proved to be of importance for the accuracy of the volumetric measurements. Placing the mouse in a slightly stretched position enabled a better differentiation between the tumour and the liver.
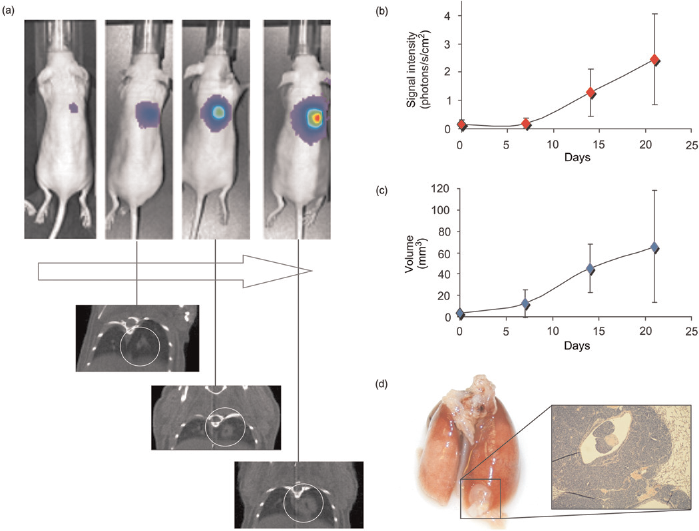
Tumour progression analysis. (a) Monitoring of tumour growth using bioluminescence imaging and computed tomography (CT) in the same animal. (b) Over time, a longitudinal increase in bioluminescent signal could be seen serving as a measure for the increase in the amount of viable tumour cells. (c) Longitudinal increase in tumour volume could be measured by CT. (d) The tumour can be detected in (inflated) dissected lungs and the presence of tumour cells was histologically validated via haematoxylin and eosin staining
Using the relative increase in bioluminescent signal intensity, the increase in the amount of viable tumour cells can be semi-quantitatively followed for several weeks (Figure 3a). 23 In parallel, the longitudinal increase in volume of a single lung tumour can be monitored by CT (Figure 3b). Based on the SPECT/CT data obtained directly after the instillation, it is relatively easy to locate the primary tumour. At the end of the monitoring period, most tumours eventually developed micrometastasis in other regions of the lung. These micrometastasis could not be detected with CT, but appeared at histology.
Discussion
Studies regarding the progression and response of lung tumours in their natural environment require that the tumour cells are instilled into the lungs and that well-defined tumours grow in the lung parenchyma. For the accurate detection and longitudinal follow-up of tumour growth, a validated and reliable instillation procedure is critical.
After conventional percutaneously intrathoracic implantation, tumours are frequently located in the pleural space and in the chest wall, 14 where tumours are difficult to monitor. Although previously described intrabronchial methods result in tumours that grow in the lung parenchyma, 11,12,14 the invasive nature of the procedure increases the discomfort for the animal and the possibility of surgical trauma. 15 The transplantation procedure described here provides tumours situated in the lung parenchyma, and the minimally invasive nature of the procedure minimalizes tissue damage and thus reduces the amount of discomfort for the animal.
The ability to non-invasively monitor the accuracy of the implantation with SPECT/CT gives a big advantage over the previously reported transplantation procedures,11–14 which do not provide any early-stage readout for the accuracy and location of the instillation site. Unfortunately, the bioluminescent properties of the injected cells cannot be used as a preliminary screening method to study the effectiveness of the instillation, due to the poor spatial resolution and high tissue attenuation encountered during BLI. However, the addition of the radioactive tracer 99mTc-Nanocoll enables accurate monitoring of the instillation accuracy with SPECT/CT. Widespread distribution over the lung or misplaced injection into the oesophagus and stomach can easily be detected. The animal can then immediately be excluded from the study, preventing unnecessary discomfort. Furthermore, it saves valuable time and resourses. As some of the injected radioactive signal (t1/2 = 6 h) can even be detected after longer time periods, evaluation of the instillation validity does not necessarily have to be performed within 30 min after instillation but can be postponed.
The fluid injected in the lungs only gives a faint signal on CT (Figure 1b), which provides the anatomical detail required to assess the location of the injected substance. Detection of freshly injected medium with CT, however, is less accurate than with SPECT (see Figures 1b and c). Similar to BLI, SPECT imaging alone gives little or no anatomical information. Hence, the addition of anatomical data obtained with CT is crucial in the interpretation of SPECT results and provides a clear image of the location of the instillation. Vice versa, CT alone is not able to accurately detect the small injected volumes and requires a SPECT signal to monitor the instillation accuracy.
In conclusion, the reported minimally invasive instillation procedure of a tumour cell containing medium (enriched with matrigel and Nanocoll) results in the outgrowth of single primary tumours, located in the parenchyma at the base of the lung. The accuracy and location of the instillation can be validated with SPECT/CT, while longitudinal progression of the lung tumours can be monitored using BLI and CT. Moreover, the instillation method and follow-up of the tumour described are not restricted to the use of a single cell line, and can be adapted for other cell lines that grow in the lung.
Footnotes
Acknowledgements
This research is supported, in part, by the Technology Foundation, applied science division of NWO and the technology program of the Ministry of Economic Affairs (STW BGT 7528 Veni). We thank Renato Valdes-Olmos and Bert Pool of the department of nuclear medicine at the NKI-AVL for their support. Furthermore, we thank Anton Berns, Joaquim Calbo Angrill and Olaf van Tellingen for setting up the SCLC tumour model and supporting our efforts with the C204 cell lines.
