Abstract
Mouse colonized with human fecal microbiota is an interesting model concept with pros and cons like any other model system. The concept provides an ecologically relevant context to study food component and drug metabolism, and is an invaluable tool for phenotype transfer studies to prove the role of the gut microbiota in health and disease. The major drawbacks are the difficulties with transferring certain components of the human microbiota to the recipient mice, and immunological abnormalities observed in these mice. There seem to be unexplored opportunities for trying to reduce these limitations, but careful evaluation of pros, cons and possible alternatives is still necessary.
Introduction
There is a need for in vivo models able to capture the complexity of the human microbiome in conjunction with a biologically relevant host. For several reasons, mice are among the most popular animal models in biomedical research and they are the primary focus of this review. Mice can, with relative ease, be derived to a germ-free (GF) state characterized by the absence of microorganisms, a valuable concept for microbiome researchers. Despite some overlap, there are profound differences between which microorganisms primarily colonize laboratory mice versus humans, which may make mice with a mouse-specific gut microbiota (GM) less translationally relevant. Humanizing the GM of GF rodents is, therefore, a frequently used tool to enhance translation to humans.
The humanized gut microbiota concept
The concept of humanizing the gut microbiota (GM) of mice can take one of two overall paths: a reductionist or a holistic approach. The reductionist approach comprises mono- and multicolonizations with defined organisms and is based on control of which microorganisms are present in the host. This allows for precise mechanistic studies of function and host effects of specific organisms and their inter-organism synergies and interactions.1,2,3 The holistic approach involves transferring a complex GM from human patients or healthy donors to germ-free (GF) hosts. Mechanistic insights are more difficult to obtain from studies utilizing the holistic approach, whereas it is useful for hypothesis-generation and for proving the causative role of GM in various conditions by phenotype transfer from donor to recipients.
An alternative to using GF recipients is to diminish the existing GM with broad-spectrum antibiotics, an approach discussed elsewhere. 4 For the reductionist approaches, GF is the only meaningful solution because it allows for precise control of what organisms are present. Holistic colonization with a complex GM may utilize a host with its resident GM depleted by antibiotics, 5 as long as it is realized that the colonization success can vary according to the microbial diversity in the host prior to antibiotic depletion. 6
To the best of the author’s knowledge, there are no published reports regarding animal welfare issues related to the transfer of human GM to animals. However, though not experienced by the author, anecdotally people have experienced transient morbidity or even mortality following oral gavage with human feces to GF mice. It seems logical that an inflammatory reaction can happen in response to the intense load of foreign microorganisms, potentially leading to cytokine storm and death—a risk that is likely to be higher in young and immunological naïve GF animals compared with older and antibiotic-treated animals. The procedure of oral gavage implies a risk in itself, for example, volume-related lung obstruction was observed in GF mouse pups after gavage of infant feces. 7 Increased monitoring following transplantation with human GM is recommended, and publishing of morbidity and loss of animals related to the procedure is encouraged in order to increase awareness and set directions for possible improvements.
Opportunities
Examples of human-to-mouse GM transfer studies and relevant model features.
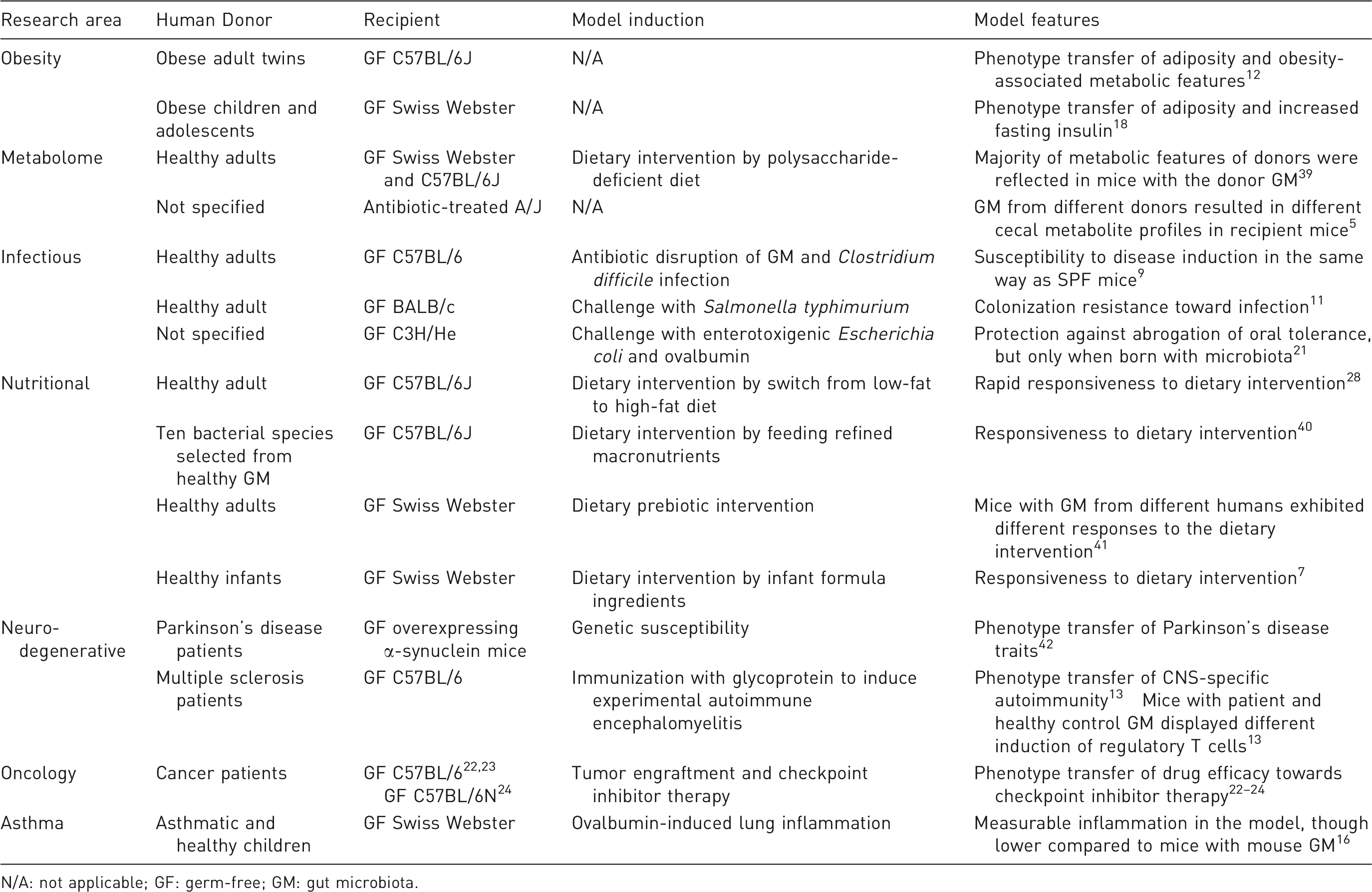
N/A: not applicable; GF: germ-free; GM: gut microbiota.
For discovering associations between host phenotype and GM, animals with human GM aid in elucidating mechanistic pathways and possible new therapeutic targets. A famous example of phenotype transfer is that GF mice with GM from an obese twin gain more weight than mice having the GM from the lean counterpart twin. 12 Studies like this have helped improve our understanding of how the microbiome can be causal in the development of metabolic disorders. Linkage of GM composition to phenotype is not restricted to conditions in the gastrointestinal tract. GF mice colonized with GM from patients with multiple sclerosis (MS) had exacerbated symptoms of experimental autoimmune encephalitis compared with mice with GM from healthy controls, indicating a causative role of the GM in MS which was supported by fewer IL-10+ regulatory T cells (Tregs) in the mesenteric lymph nodes of these MS mice. 13 The opportunity to unravel such immunological host–microbiota effects is the strongest argument for using animal models, yet the host–microbiota interface is the most questioned feature of the GM humanized mouse concept.
Challenges
Several reports describe an abnormal immunophenotype of mice with a human GM. The number of intraepithelial lymphocytes (IELs) was lower in GF mice with rat or human GM compared with mice colonized with a mouse GM. Furthermore, only the mouse GM was able to change histological characteristics of the IELs and epithelial cells. 14 GF mice with human GM had low levels of CD4+ and CD8+ T cells and antimicrobial peptide in the small intestine. 15 Splenocytes of GF mice with infant microbiota produced lower amounts of cytokines involved in Th1, Th2 and Treg responses. 16 Comparable to GF mice, a human GM could not induce Tregs in the small intestine, whereas Tregs were higher in the colon compared with GF control mice. 17 Hence, human-derived bacteria may have a higher affinity or colonization preference to the murine large intestine compared with the small intestine.
The immunological defects are hypothesized to be caused by lack of evolutionary adaptation to the host. It is well known that only a portion of human GM is successfully transferred to mice. In GF mouse experiments, the colonization efficiency typically varies between 44% and 70%,15,18,19 though in one case it was as successful as 90%. 20 A study using antibiotic-treated mice reached a 57–68% recovery of bacterial genes in recipients compared with the donor sample. 5 Mouse-to-mouse GM transfer is generally more successful, with a colonization efficiency up to 93%. 15 Interestingly, rats may be better suited recipients of a human GM compared with mice. Wos-Oxley et al. transferred the same human GM to GF rats and mice and found that the colonization efficiency was significantly higher in the rats (76% versus 44%). 19 Another study demonstrated that 75% of a human GM was established in GF rats. 8
Examples of human GM bacteria associated with human health that are showing variable colonization success in germ-free animal studies.
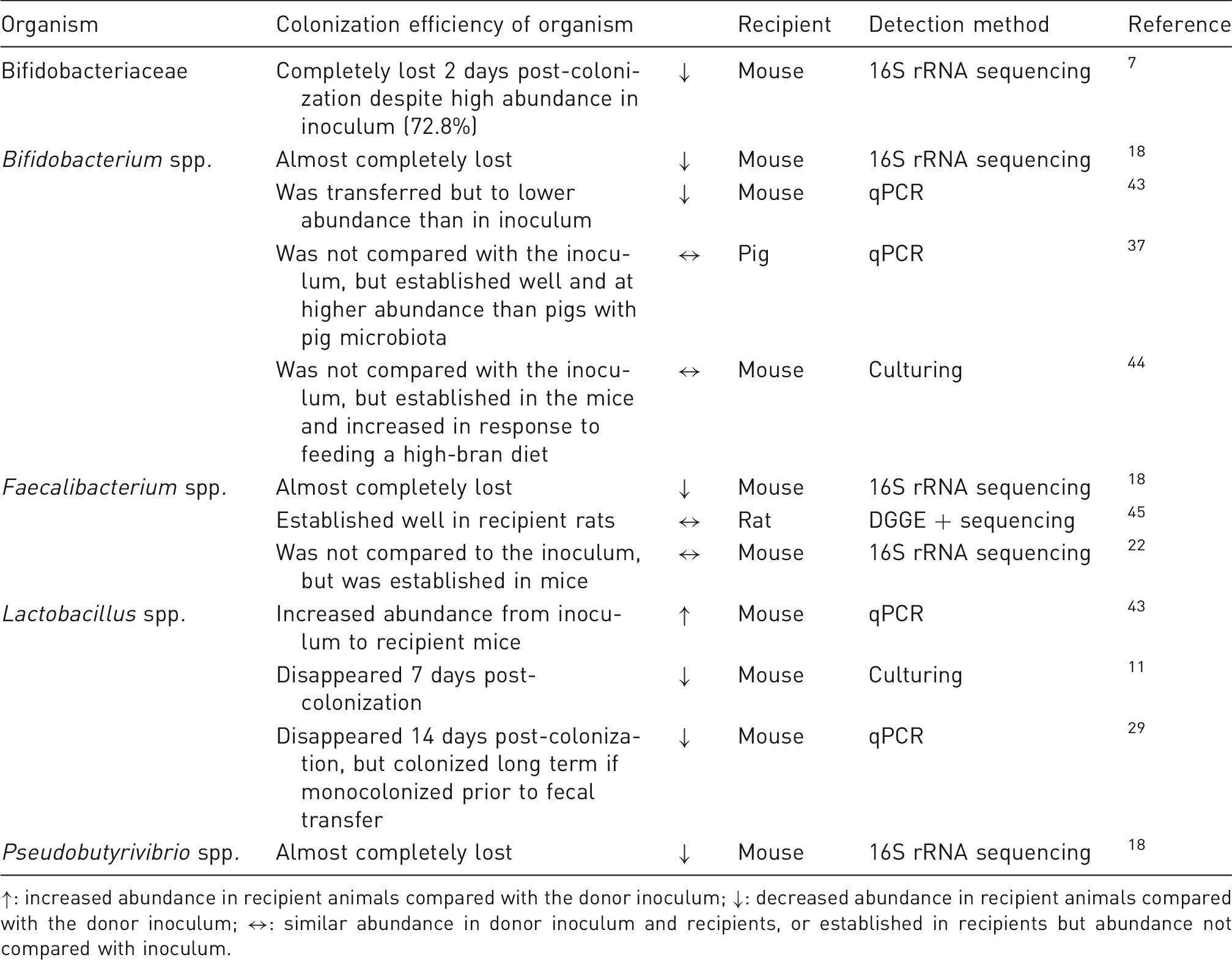
↑: increased abundance in recipient animals compared with the donor inoculum; ↓: decreased abundance in recipient animals compared with the donor inoculum; ↔: similar abundance in donor inoculum and recipients, or established in recipients but abundance not compared with inoculum.
Despite the immunological naivety characterizing mice harboring a human GM, the concept has demonstrated the ability to elicit differential and translationally relevant immune responses. Recently, GF mice with cancer patient GM replicated patient responses to checkpoint inhibitor immunotherapy through mechanisms hypothesized to happen via immunological stimulation by the transplanted microbiota.22–24 Mice with GM from patients responding well to therapy recruited more CD4+ T cells 23 and CD8+ T cells22,24 to the tumor microenvironment than mice with a non-responder GM. However, one of the studies found that only a portion of the GF mice with patient GM recapitulated the human responder phenotype, and these were the same mice whose GM was most different from the donor GM. 24 This may illustrate an element of luck and suggest that the desired outcome may simply be reached by including enough study replicates and hoping for the best. In a model of induced lung inflammation, mice with human GM mounted study-relevant and measurable inflammation, though it was lower than in mice with mouse GM and despite having reduced levels of Th1 and Th2 cytokines before the model was induced. 16 Hence, if the read-out of the model is of a magnitude significant enough to be biologically meaningful, it may be less problematic that the immune baseline is below that of mice with mouse GM.
Can we overcome the challenges?
Physiological, anatomical and immunological abnormalities of GF mice are well documented and summarized elsewhere. 25 Most of these features are normalized after colonization with a complex GM. However, irrespective of whether the GM transplant is murine or human, a GF period in early life prior to colonization alters immunological shaping 26 and ability to confer colonization resistance against pathogens 21 later in life. Hence, to allow for correct immunological development study mice should not be the parent ex-GF generation, but rather the offspring generations born with the GM. 21 Sex also influences the GM profile. GM from a male donor established differently in male and female mice, 27 suggesting that alignment of donor and recipient sex may be necessary.
Few, if any, attempts to optimize the colonization efficiency in human-to-mouse GM transfer studies have been published. Yet, several procedural steps from collection of the donor material, handling, and storage to the transfer itself and the conditions following transfer may affect colonization efficiency. For practical reasons, feces is the most frequently used donor material, but a more truthful representation of the entire gastrointestinal tract in the donor sample might also enhance the diversity of the established microbiota. Transfer of fresh versus frozen material works equally well,6,28 but the exact cryoprotection protocol, anaerobic conditions, single versus repeated gavage or the use of other administration forms may all influence transfer rate. For instance, a model-community of 10 bacterial strains from human feces was more successfully established in GF mice by separate monocolonizations and subsequent co-housing compared with one single administration of all 10 bacteria. 3 Husbandry factors, diet probably being the most important one, are likely to impact the colonization. Genetic humanization, for example, of epithelial colonization factors, or humanization of immune cell populations are also interesting avenues yet to be explored. For obtaining an immunological phenotype similar to specific pathogen-free (SPF) mice, it may be necessary to add indigenous mouse bacteria to the human GM, as demonstrated with segmented filamentous bacteria. 29 Such hybrid approach combines the ecological community of a human GM with a more developed immune system and may thus be a better reflection of the reality.
What are the alternatives?
The major limitation of mice with human GM is the altered immunological shaping compared with mice harboring an SPF GM. Interestingly, SPF mice are criticized for exactly the same compared with their wild counterparts.30–32 In our efforts to exclude unwanted microorganisms according to SPF health standards, the GM of SPF mice seem to have lost important immunomodulatory organisms. However, it complicates the picture that SPF rodents harbor different GM across animal institutions, which in turn elicit widely different phenotypes33,34 and make generalizations impossible. Nevertheless, the question has been raised whether SPF mice are good models for humans at all.30–32 We do not live in clean enclosures, and it is a natural part of our immunological education to be exposed to a variety of both beneficial and pathogenic microorganisms. Laboratory mice with a GM from wild mice have significantly improved disease resistance due to a better poised immunological state, and provide a provocative alternative to the too-clean SPF mice, 32 and maybe also to mice with human GM. A large functional overlap between SPF GM and human GM has been shown, 35 and is an argument in favor of mouse cohorts with a co-evolved GM (wild or traditional SPF) that is functionally comparable to that of humans, yet able to induce immunocompetency in the hosts.
Rats have already been mentioned as potiential superiour recipients of human GM. Pigs are often proposed as more precise models of humans because of the highly comparable physiology. The GM of GF piglets colonized with human GM do indeed seem to have a good conformity with the donor. 36 In the same study, the authors did several rounds of GM administrations to the piglets over 10 days, 36 which may also have enhanced the colonization efficiency compared with rodent studies often relying on a single round of oral gavage. Another study found that a human GM induced immune parameters in GF piglets, that is, they had more CD4+ T cells and MHC class II expressing cells in the gut compared with piglets colonized with pig feces. 37 In this study, Bifidobacterium established well in the porcine hosts and may provide at least a partial explanation of the results. 37 Recently, a dog GM gene catalog was published and demonstrated that compared with the mouse and pig microbiome, the dog GM was most similar to the human GM, 38 implying that dogs may be a highly translationally relevant model system for human microbiome research. However, compared with mice, there are other limitations related to the use of rats and pigs and other animal models. Choice of model is in all cases a balance between practicalities, economics, what is accessible (for example, genetic modifications) and the expected translational value. In some cases, it might even not be an animal model that is best suited. For instance, for nutritional and metabolic studies merely considering the intestinal ecological habitat without regard to host–microbiome interaction and immune system development, in vitro gut simulator systems could be a valid alternative.
Conclusions
All models, including rodents colonized with human GM, have their pros and cons. It is essential to understand these thoroughly in order to obtain valid insights. Careful consideration should be given when immunological pathways are important for the model, whereas the humanized microbiota concept is probably more robust for nutritional studies and phenotype transfer studies. However, as exemplified throughout this review, there are still hypotheses to be tested that may help in optimizing the concept.
Footnotes
Acknowledgement
The author would like to acknowledge conversations with former colleagues at the University of Copenhagen and Taconic Biosciences.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
