Abstract
Neonatal mortality is high in laboratory mouse breeding, and causes are poorly understood. Post-mortem analysis of pups is an often overlooked source of information and insight. We present a necropsy protocol for neonatal mice designed for easy practical application by animal technicians.
Neonatal mortality is high in laboratory mouse breeding, and underlying causes are poorly understood. The majority of losses occur in the first three days after birth, 1 and reported average rates of mortality can differ between animal facilities. Our own data show a range between 14% and 39% for the C57BL/6J strain. 2 In farm animals, important causes include hypothermia, pregnancy and parturition complications, poor nutrition and injuries. 3 These factors are also likely to affect mouse pups, which have poor thermoregulatory capacity 4 and are often kept in trio breeding systems, where the increase in mortality when an older litter is present suggests that milk competition and trauma may be critical. 1
Post-mortem analysis of dead pups found at cage inspection is a major source of information which is often overlooked, partly due to a lack of proper guidelines, since published protocols about post-mortem analysis of newborn mice are directed to phenotyping pups of genetically modified strains humanely killed at predetermined time points. 5 Furthermore, inspection of breeding cages in the first four days after birth is often avoided due to concerns that inspection may itself increase mortality, which our previous work proved to be unfounded. 6 In addition, the high rate of cannibalism of dead pups seen in this species can result in lost specimens. It is therefore recommended to inspect breeding cages daily, with minimal manipulation during the first four days after birth, as described by Brajon et al. 1
Here, we present a necropsy protocol for 0- to 4-day-old mice designed primarily for immediate use by animal technicians in order to identify possible causes of neonatal death in healthy wild-type neonatal mice. The present paper is based on post-mortem analysis of pups found dead by daily inspection during the first four days after birth in healthy wild-type laboratory mouse breeding colonies (see Brajon et al. 1 ). No living mice were manipulated. Hence, no ethics review was required.
Special attention is given to the presence of external lesions, evaluation of stomach contents, signs of breathing after birth, brown adipose tissue ((BAT) for non-shivering thermogenesis 7 ) evaluation and occurrence of congenital malformations. This systematic method should be complemented by later histopathological analysis performed by a veterinary pathologist, especially in cases where the cause of death could not be identified by gross pathology.
Proof of breathing after birth is obtained through a lung float test, the principle being that after inflation by breathed air, they will float in water. The liver was used as a negative control for the production of gas due to decomposition processes. 8 Evaluation of stomach contents for the presence of amniotic fluid can also be useful to detect stillbirths. Foetuses swallow amniotic fluid during gestation and remove it through gastric emptying. 9 Consequently, a necropsy finding of a stomach filled with amniotic fluid suggests stillbirth or at least that the death occurred soon after birth. These observations should always be correlated with the gross appearance of the lungs (level of inflation and aeration), since processes such as oedema, congestion or meconium aspiration could lead to erroneous conclusions. 10 The appearance of the umbilicus should also be assessed, since tissue reactions such as cicatrisation or inflammation only occur in living organisms. 11
This protocol was developed for C57BL/6 mice to include the most common congenital abnormalities for this strain, such as microphthalmia and hydrocephalus. 12 It is easily adaptable to other mouse strains.
Macroscopic post-mortem analysis is not compromised by refrigerating or freezing pups, but freezing could impair later histological analysis by the production of artefacts. 13
The protocol is composed of an external and internal evaluation. The materials needed are summarized in Table 1.
First, record strain, date of birth, date found dead, cage ID and litter number. During the evaluation, photograph any lesions and abnormalities.
External evaluation
Measure and record the following naked-eye observations of external appearance:
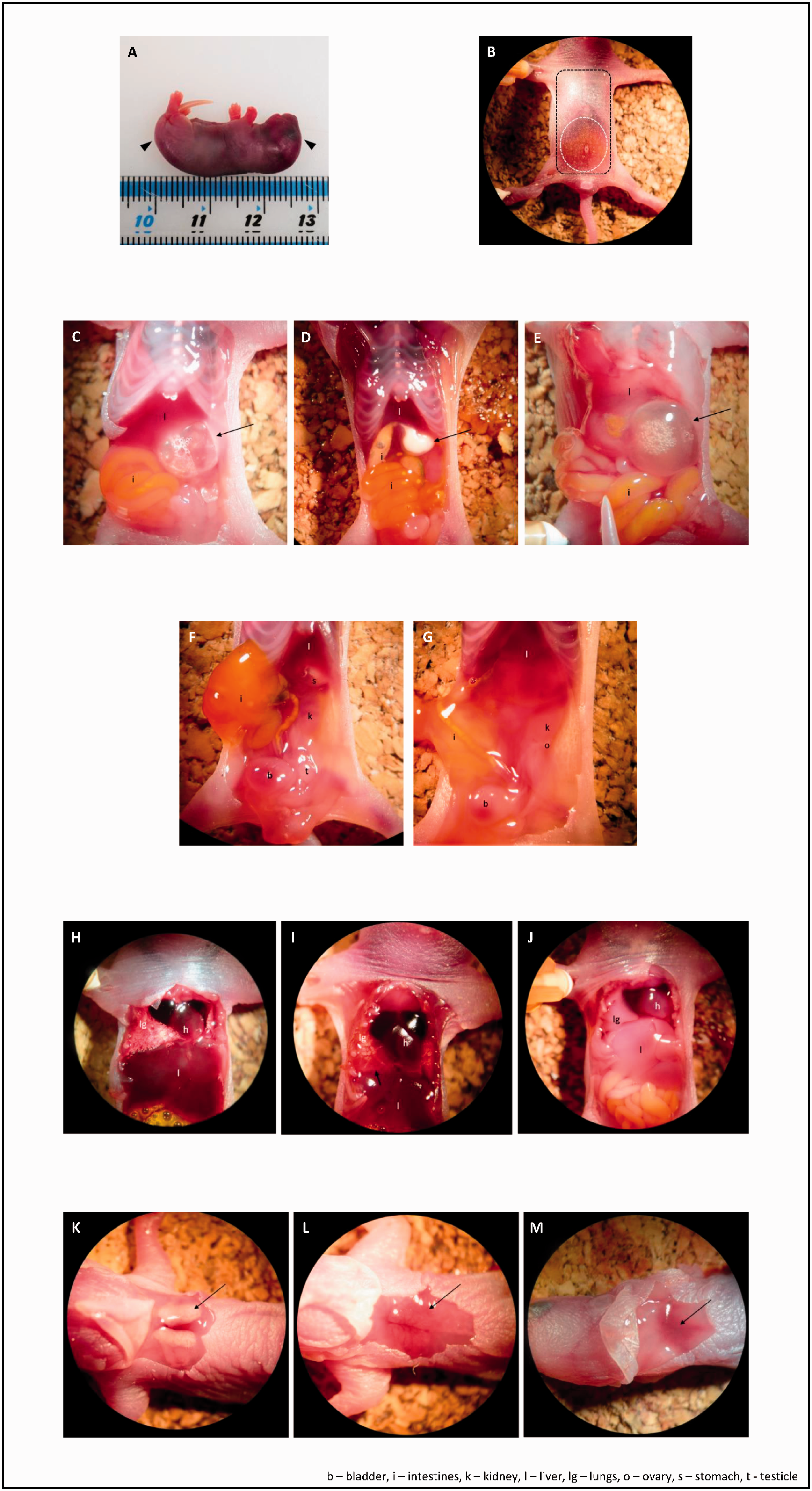
Weight and crown-to-rump length of intact pups (Figure 1(a)) with a digital scale and ruler.
Post-mortem analysis. (a) Crown-to-rump length. (b) Skin (black) and abdominal (white) incision pattern. (c)–(e) Stomachs with air (c), milk (d) and amniotic fluid (e). (f) and (g) Reproductive organs ((f) male; (g) female). (h)–(j) Lungs ((h) aerated/inflated ; (i) arrow indicating congestion due to hypostasis; (j) not inflated/no sign of aeration). (k)–(m) brown-yellow (k) and brown-pink ((l) -(m)) . BAT; different dimensions and amount of visible blood vessels. Haematomas and other lesions (e.g. bite wounds), including partial cannibalism (parts of a pup missing). Animals born without limbs or organs can be distinguished by the presence of intact integument. Limbs, toes and tail for number and integrity. Skin colouration. Presence/absence of both eyes, any abnormalities in size (e.g. microphthalmia) and integrity of the eyelids. Head-shape abnormalities, particularly a domed cranium as a sign of possible hydrocephaly.
Observation under the stereomicroscope:
Sequentially position the animal in right, left and ventral recumbency. Repeat the observations of external appearance. Examine the skin for remnants of foetal membranes. Secure the pup in dorsal recumbency using needles placed through the paws. Examine abdominal colouration and look for signs of internal bleeding (dark colours). Observe the umbilicus for signs of cicatrisation and umbilical-cord remains. Examine the pigmentation of the anogenital area to determine the sex.
14
Open the mouth with a forceps and examine the oral cavity for abnormality such as cleft palate.
Internal evaluation
Start the dissection by pinching the skin caudally to the umbilicus and follow the pattern illustrated in Figure 1(b). Careful blunt dissection of subcutaneous tissues is performed using Vannas scissors until the entire skin patch can be removed with thumb forceps, without puncturing the abdominal or thoracic cavities.
Record the appearance and integrity of the subcutaneous and muscle tissues.
Access the peritoneal cavity by pinching the abdominal wall with thumb forceps in the pubic area, and follow the incision pattern described in Figure 1(b)
If fluid is present, record the colour and viscosity, and collect a sample for later biochemical and cytological analysis using a syringe.
Evaluate the appearance (colour, shape and dimensions) of the abdominal organs and the gut contents.
Using thumb forceps, move the intestines to the left side to expose the stomach. Record its size and level of distension, pinch with watchmaker forceps, make an incision and evaluate its contents (Figure 1(c)–(e)). Continue the incision along the greater curvature of the stomach, evert the walls and observe their appearance.
Observe the reproductive organs (Figure 1(f) and (g)). Record the sex.
Make an incision at the costochondral junction at both sides of the thoracic wall. Remove the sternal fragments and inspect the thoracic contents. Evaluate lung colouration and look for signs of aeration (inflation and the presence of small visible air bubbles; Figure 1(h)–(j)). Photograph the lungs. Remove a lobe of lung and a same size fragment of liver into an Eppendorf tube filled with water. Record whether the fragments float.
Reposition the pup in ventral recumbency. Secure it as previously described.
Make a circular skin incision in the interscapular area. Remove the skin, record the presence and appearance (colour, shape and presence of blood vessels) of the two BAT lobes (Figure 1(k)–(m)).
When no gross abnormality is identified as a cause of death in the internal and external evaluation, the carcass should be retained for further histopathological examination by a veterinary pathologist. For this, immerse the whole pup in 50 mL 10% buffered formalin. The sample should be submitted with a detailed description of the data and observations collected during the internal and external inspection.
Routine application of this protocol following the discovery of dead mouse pups will easily allow the collection of valuable information that might otherwise be discarded, and contribute to the understanding of neonatal mortality. Formulation of strategies to minimise this welfare problem will also impact on improving the efficiency of laboratory mouse breeding colonies and therefore to a reduction of the animals used in research.
A detailed description of this protocol is available from the corresponding author on request.
List of materials.

Footnotes
Acknowledgements
The authors thank all staff at the i3S Animal Facility and the Babraham Institute Biological Support Unit, especially Ana Abreu, Ângela Ribeiro, Urszula Karpinska and Minnie Eve, and the i3S Histology and Electron Microscopy Platform, member of the national infrastructure PPBI–Portuguese Platform of Bioimaging (PPBI-POCI-01-0145-FEDER-022122).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was financed by FEDER – Fundo Europeu de Desenvolvimento Regional funds through the COMPETE 2020 – Operacional Programme for Competitiveness and Internationalisation (POCI), Portugal 2020 and by Portuguese funds through FCT – Fundação para a Ciência e a Tecnologia/Ministério da Ciência, Tecnologia e Ensino Superior in the framework of the project PTDC/CVT-WEL/1202/2014 (POCI-01-0145-FEDER-016591).
